Nitrobenzene: The Backbone and Burden of Industrial Chemistry
Looking Back: How We Arrived at Nitrobenzene’s Pivotal Role
Nitrobenzene entered the world stage in the mid-19th century, right about when chemists figured out how to turn simple molecules into things that could change industry forever. The process of nitrating benzene opened up a whole new area of chemistry, where black tar from coal gas plants became the root of new dyes, medicines, explosives, and plastics. Early production used harsh, dirty methods—a cocktail of benzene, sulfuric, and nitric acid that churned out noxious fumes. Back then, factory workers faced real physical risks, with few or no protections, and cities smelled the byproduct in the air. Nitrobenzene’s impact went far beyond the laboratory, reaching into clothing, medicine, and even munitions that drove global conflicts and colored the world, literally and figuratively.
What Nitrobenzene Is—And Why That Matters
Looking at it in the bottle, nitrobenzene wants to trick you. It’s an oily, faintly yellow liquid, heavier than water, with a surprisingly pleasant almond-like odor. For many, that smell masks its danger. Nitrobenzene stands out for its remarkable solvency and chemical stability under normal conditions, yet it turns reactive with certain agents. These traits gave rise to whole families of synthetic dyes—most famously, aniline blue and mauveine. It can dissolve resins and fats, making it ideal in manufacture long before we started questioning chemical exposures or what happened to waste streams leaving factories. Its chemical behavior comes from a simple structure: a benzene ring, bearing a nitro group, ready to take part in more ambitious reactions.
Physical and Chemical Profile—a Study in Contrasts
Nitrobenzene’s density clocks in higher than water, which means spills tend to sink. Its boiling point hovers just above the range for steam, so you don’t see it evaporate quickly on a hot day, yet under heat—and in a lab setting—it’ll vaporize and demand careful handling. On the chemical front, nitrobenzene resists mild acids and bases but turns susceptible to reduction. One of the classic reactions transforms it back to aniline, which is where the real money is: aniline acts as the building block for dyes, rubber chemicals, and pharmaceuticals. The nitro group also makes it less flammable than plain benzene, but that doesn’t mean safe—far from it. A fire involving nitrobenzene laces smoke with toxic fumes.
Labeling and Technical Nuance: More Than Regulatory Boxes
Any bottle of nitrobenzene needs robust warning labels. Safety standards call for that vivid toxic symbol, detailed instructions on exposure response, and restrictions about storage away from flames or oxidizers. Operational standards have grown stricter over the years under the weight of hard-earned lessons—people who handled this stuff before modern workplace safety suffered chronic illness, nerve damage, and worse. Today’s handling rests on airtight containers, controlled ventilation, personal protective gear, and a string of fail-safes required by international bodies. Marketers and supply chain operators must stick with recognized terminology: oil of mirbane, nitrobenzol, and other synonyms carry with them the baggage of older practices. Each synonym hints at a chapter of occupational health disasters or environmental release.
Synthesizing Nitrobenzene: Old Roots, Modern Turns
Historically, nitrobenzene took shape in massive glass or iron vessels, where benzene reacted with a mix of concentrated nitric and sulfuric acid. Modern syntheses still rely on mixed-acid nitration but pack in greater efficiency and tighter emission controls. The fundamental reaction—electrophilic aromatic substitution—hasn’t changed much. Every facility wrestles with waste nitric acid, emissions of volatile organics, and hazardous acid sludges. Process engineers now devote more effort to recapturing heat, recycling reagents, and converting byproducts to less harmful forms. Progress here links directly to survival: failure to modernize puts a plant on the wrong side of regulatory fines, public outrage, or catastrophic incidents.
Chemical Reactions—Building Out from the Nitro Group
Nitrobenzene’s chemical modifications fuel major sectors. Reactants strip away its nitro group under pressure with iron filings and acid, giving aniline. Catalytic hydrogenation speeds up this reduction, allowing industry to scale up production and create feeds for dyes, herbicides, and medicine. On the flip side, more energetic reactions can turn nitrobenzene into explosives, like TATB (triaminotrinitrobenzene), through a series of dark, megaton-level transformation steps. Chemists use additional modifying agents to substitute positions on the aromatic ring, growing a toolkit for pushing beyond colorants into drugs, plastics, and agricultural chemicals.
Names That Stick: Synonyms and Trade Practices
Mirbane oil and nitrobenzol likely pop up in old catalogs and legal documents. These names linger in the supply chain, despite regulators favoring “nitrobenzene” for clarity. Such synonyms have muddied shipping manifests and left room for confusion about safety precautions—more than one accident in history traces back to inconsistent labeling across borders or sectors. The industry now calls for harmonized nomenclature, both in international trade agreements and in day-to-day practice.
Safety: Taking Hard Lessons Seriously
Nitrobenzene’s dangers hover over every stage, from transport to final product. Exposure can cause cyanosis—the skin turns blue from lack of oxygen—along with nervous system damage and organ failure. It can pass through skin, making gloves a non-negotiable, and its vapors need strong ventilation. Most countries place strict limits on worker exposure; U.S. OSHA regulations mark permissible exposure at a tiny fraction of a percent in air. Plant managers and EH&S officers drill teams on spill response, decontamination, and evacuation drills. In older factories, horror stories of chronic poisoning still echo in plant lore, shaping how new generations approach risk.
Where Nitrobenzene Goes—And Why Substitution Faces Hurdles
Industries use nitrobenzene mostly to produce aniline, which acts as the precursor for methylene diphenyl diisocyanate (MDI), a compound critical to polyurethane foams found in insulation, car seats, and sneakers. Beyond foams, it finds use in lubricating oils, dyes, pesticides, and explosives. In research, nitrobenzene stands as a test bed for organic reaction development. Arguments about alternatives always bump up against raw economics and technical performance—nitrobenzene’s chemical profile fits just right in large-scale processes. Green chemistry initiatives seek ways to sidestep benzene altogether, but production volumes haven’t dipped, reflecting demands from construction, textile, and automotive sectors worldwide.
Chasing Safer Chemistry: Research, Toxicity, and the Path Forward
Studies on nitrobenzene toxicity punctuate every decade since its discovery. Animal models and human case studies both document its ability to disrupt blood’s oxygen-carrying capacity and wreak havoc on the nervous system. These findings press industry and regulators to lower exposure limits, redesign plants, and strengthen monitoring. Researchers in academic institutions probe for better detection tools and antidotes, while chemical manufacturers experiment with greener processes—either by switching solvents or capturing emissions more efficiently. Environmental advocates push the conversation toward substitution with less hazardous intermediates, though each proposal gets a careful vetting for trade-offs in energy use or production wastes.
The Road Ahead: Can Industry and Safety Walk Hand in Hand?
Looking to the future, the nitrobenzene story runs parallel to the bigger picture of industrial chemistry’s reckoning with its legacy. Advances in catalysis, automation, and continuous flow reactors currently aim to tame risk, reduce waste, and keep more of the chemistry contained. Cross-border collaboration on labeling and exposure standards tightens each year, raising the bar for every producer. As consumer preferences keep changing—demanding safer and more sustainable materials—companies see real commercial advantage in investing in safer platforms. Young chemists come up learning about nitrobenzene’s hazards early, so the hope is that the coming decades will not repeat the hardest lessons of the past. The tension between utility and hazard continues, but improvements in safety, monitoring, and alternative development stand ready to close that gap further.
What is nitrobenzene used for?
What Nitrobenezene Brings to Daily Life
Most people never hear about nitrobenzene outside high school chemistry class, but this chemical runs through plenty of modern life. Factories use it on a large scale, and some of the world’s biggest products start with it. Paints, glues, polishes and even the painkillers in the medicine cabinet rely on processes that often involve nitrobenzene. The details matter, because this chemical is more than a background player—it creates real products that shape daily routines for millions.
From Aniline to Everyday Products
The story of nitrobenzene starts with its main job as a tool for making aniline. About 95% of all nitrobenzene goes into this. Aniline itself plays a starring role in dyes — think of all the color in clothes, furniture, and packaging. I remember walking through fabric workshops and seeing how the color choices set the mood for an entire collection, all made possible by chemical intermediates like these.
Aniline also leads to polyurethane foam, and anyone who’s bought a mattress or sat on a couch knows how important a good foam can be. The comfort and support we get from these home staples depend on precise chemistry that began with nitrobenzene. Big auto companies and builders use polyurethane every day, feeding demand for new furniture and vehicles.
Nitrobenzene’s Risky Side
Even as nitrobenzene sits behind the scenes, it remains important to handle it with care. It’s classified as toxic, with long-term effects if someone is exposed without protection. Factory workers use gloves and specialty ventilation systems because inhaling or touching nitrobenzene can damage blood and the nervous system. Some studies have raised questions about links to cancer.
For years, regulations in Europe, North America, and Asia have set strict rules around workplace exposure. I’ve seen the difference effective training can make: with good safety standards, accident rates drop and workers feel more secure on the job. Simple changes — like using sealed tanks or continuous monitoring for leaks — keep the chemical out of the air and workers out of harm’s way.
Supporting Cleaner Manufacturing Methods
Big industries know they walk a tightrope between output and health risks. Some companies use advanced filters and water treatment processes to make sure nitrobenzene doesn’t escape into rivers or city water. There’s also a push for alternatives in some sectors. Green chemistry can provide new pathways to core ingredients without using nitrobenzene at all, though costs and scale still pose challenges.
I’ve spoken to engineers who admit the switch to safer methods takes time and money. They balance decades-old plants with new rules and customer demand for sustainable practices. It’s an ongoing project, but the drive for safer manufacturing keeps gaining steam.
Toward Better Choices
Community advocacy adds pressure for safer chemicals. Many organizations offer certification for plants using stricter safety protocols. As families become more aware of what’s behind their everyday goods, interest in better sourcing grows. Choosing products from brands with clear safety records serves as a signal to big suppliers — people care not just about what they buy, but how it’s made.
So nitrobenzene may not be a household name, but the story matters. Its role in industry and its potential hazards join up in the question: how do we keep moving forward, without repeating mistakes of the past? Personal experience and professional know-how both point to one answer — informed choices and real transparency set the stage for safer, smarter chemistry in the years ahead.
Is nitrobenzene hazardous to health?
Everyday Use and Hidden Dangers
Walk into a paint factory or take a peek behind the scenes in a large chemical plant and you’ll probably run across nitrobenzene. It’s that pale yellow liquid with an almond-like odor, used mainly to make aniline. Aniline goes into everything from dyes and rubber to some medicines. At first glance, nitrobenzene seems like one of those nameless substances behind everyday products. But talk to anyone who’s worked around it, and you’ll hear warnings about headaches, dizziness, and a scent you can’t quite shake off.
Health Risks Are Real
Nitrobenzene isn’t something folks should take lightly. Inhaling its vapors or getting it on your skin can be risky. My uncle worked in an old tire plant, and he always covered up before going near the vats. He said it just wasn't worth the trouble getting sick on the job. Studies back up his caution. Breathing in nitrobenzene, or handling it without protection, can lead to serious health effects—methemoglobinemia is one example, where blood can’t carry oxygen right. Folks exposed can show blue-tinted lips and skin, feel short of breath, or worse.
The US Environmental Protection Agency calls nitrobenzene a possible human carcinogen. It’s linked to liver damage, nervous system problems, and a drop in blood cell counts. The National Institute for Occupational Safety and Health sets strict exposure limits, basically saying: respect the stuff or expect trouble.
Routes of Exposure: More Than Just Smelling It
Getting exposed to nitrobenzene isn’t only an occupational hazard. If it spills or leaks, groundwater and soil can end up contaminated. Workers in fields using certain pesticides, or folks living nearby, might run into it without ever walking inside a chemical facility. That kind of exposure can go unnoticed for months. Personal experience counts here—my own town dealt with a chemical spill one summer, and the local clinic saw patients with all sorts of unexplained headaches and nausea until investigators found the source.
What Can Communities and Workers Do?
Nobody wants to trade health for a paycheck. Simple things make a difference: proper gloves, goggles, enough ventilation. Facilities must keep a close watch on storage tanks and transport. If nitrobenzene spills into the neighborhood, communities deserve clear updates. No one wants surprises where public health is concerned.
Regulators can help by enforcing rules and making sure companies share information. Some plants have cut down on nitrobenzene use by switching to safer alternatives when possible. People living near chemical industries should push for better monitoring—real-time air and water quality checks make it harder for leaks to go unnoticed.
The Bottom Line for Everyday People
Most folks won’t run into nitrobenzene at home, but it's still important to ask questions. If your job or town brings you close to industrial chemicals, know the symptoms and demand honest answers when health concerns pop up. The science is there, but it’s the lived experience of workers and neighbors that keeps everyone accountable. Listening to the people most at risk, and leaning on proven safety practices, offers the best shot at staying safe.
How should nitrobenzene be stored and handled?
Understanding What’s at Stake
Once you’ve seen nitrobenzene up close—its oily feel, its almond-like smell—you never forget it. Those who’ve worked with this chemical in labs or factories know it doesn’t belong in just any storage closet. There’s more than chemistry at play here. Nitrobenzene can slip into the air, get through your skin, and start doing damage before anyone notices. Breathing it in or getting it on bare hands can trigger headaches and nausea. Deeper exposure risks something even more serious: it can keep blood from carrying oxygen the way it should. Blood tests can show the problems later, but by then, you might already feel lousy. That sticks in your mind, especially if health and safety on the job matter to you.
Picking the Right Spot
Forget about putting nitrobenzene anywhere children or untrained staff might find it. Keep it in a cool, dark spot far from flames or sources of heat. Even a sunny window lets temperatures creep up. That’s trouble since nitrobenzene can go from a stable liquid to an explosive hazard if heat builds up. Storing it near oxidizers or strong acids is another pitfall. Mix-ups during deliveries or careless stacking can produce dangerous fumes or even fires. In some facilities, I’ve watched seasoned managers lock it in vented steel cabinets—never just any shelf. Safety data sheets recommend this because ordinary plastic bins warp or melt if leaks happen.
Sturdy Containers and Real Labels
The right drum or bottle isn’t just about industry best practices. Leaky or cracked containers have sparked emergencies before. Nitrobenzene demands containers built out of tough steel or certain plastics (consult the SDS, always). Each one gets labeled with bold warnings, not a scrawled note or fading sticker. Labels that scream “Toxic—Nitrobenzene” prevent costly mistakes, especially for night crews or new hires.
Ventilation Matters
I’ve visited sites where opening a bottle without fume extraction was enough to make the whole shift sick. If your storage space doesn’t have powered ventilation, you’re courting trouble. A strong local exhaust pulls away vapors before they build up, cutting the risk for anyone nearby. Spark-proof fans and lights in those rooms reduce the odds that a stray wire or switch causes a fire.
Handling: Don’t Rush, Don’t Guess
Spills and splashes turn routine work into a nightmare fast. Anyone opening, pouring, or transferring nitrobenzene wears chemical gloves (not the thin disposable kind, but real heavy-duty ones), goggles, and a face shield. Good habits save time in the long run. A buddy system—never working alone—is smarter than going solo, as someone can call for help if a splash catches you. One accident I remember from early in my career involved a single skipped glove. That worker left the job site in an ambulance. Procedures and personal protective equipment might feel like a chore, but no shortcut is worth those risks.
Tackling Accidents—Preparation Saves Lives
Every worksite with nitrobenzene nearby should have spill kits at arm’s reach. Promising to “get it tomorrow” doesn’t help anyone exposed today. Emergency showers and eyewash stations belong close to the storage area, not halfway across a warehouse. Expect spills and plan for them; don’t hope they’ll never happen. Fast action with the right materials—pads, absorbents, and neutralizers—makes all the difference when someone’s health is at risk.
The Role of Leadership and Ongoing Training
Complacency gets people hurt. Good supervisors lead by example and never skip safety talks or refresher training. New rules and better gear come out all the time. It takes real leadership to keep a team sharp and invested in safety, especially with dangerous chemicals like nitrobenzene. Honest conversations, not just paperwork, help build a work culture where everyone looks out for each other and catches mistakes before they snowball.
Moving Forward Responsibly
Working safely with nitrobenzene isn’t just about following government codes. It’s about valuing people over shortcuts and leaning on proven systems. Asking questions when you’re not sure and speaking up if storage or handling falls short—those simple steps make the biggest difference. Safety grows from the ground up, and it’s worth digging your heels in for everyone’s sake.
What are the physical and chemical properties of nitrobenzene?
Physical Characteristics
Nitrobenzene has an almost old-fashioned reputation among organic chemicals—probably because anyone who’s worked with it remembers the sweet, almost almond-like odor. The substance itself shows up as a pale yellow oil at room temperature. Its melting point hovers around 5.7 degrees Celsius, so it solidifies in a chilly fridge but returns to liquid fairly easily. Take a glass container and add some: you’ll quickly notice it doesn’t mix well with water. Instead, it floats, showing off its slight density advantage over water (about 1.20 g/cm³). This property can surprise new students in the lab, since you can see the distinct boundary between the water and nitrobenzene.
Light doesn’t do much to break it down, and the compound stands up to room temperature air without decomposing or catching fire quickly. Still, the flashpoint hangs low at 88°C. That usually means careful storage in a fume hood. All it takes is a slight spill to fill a room with bitter almonds—a good reminder to grab gloves and protect your skin.
Chemical Behavior of Nitrobenzene
Structurally, nitrobenzene puts together a classic benzene ring with a nitro group (-NO2). The nitro group drags electrons out of the ring, making nitrobenzene less reactive toward reactions that want to add something new to the ring. Nitration, sulfonation, or halogenation crawls along slowly, compared to plain benzene.
On the other hand, the nitro group can be coaxed into new forms with the right reducing agents. Hydrogenation (using H2 and a metal catalyst) or iron and hydrochloric acid efficiently turns nitrobenzene into aniline, a vital ingredient for dyes and drugs. The process feels old-school, often barely changed from before World War II, yet remains essential. Nitrobenzene itself spends most of its life as a stage in making something even more useful—one in every three kilograms goes toward making aniline.
Mistaking nitrobenzene for a solvent solution is dangerous, since it's toxic. Chronic exposure brings headaches, nausea, and—more rarely—serious bone marrow and organ issues. The body transforms nitrobenzene into compounds like p-aminophenol, also toxic. Even at home, a bottle left open could harm both people and pets. Due to these hazards, regulators demand low occupational limits and industrial safeguards for workers.
Where Safety and Science Meet
Nitrobenzene rarely shows up in public, but it does pop up in some shoe polishes and odor-masking products. It gained a bad reputation decades ago in agriculture as a “plant tonic”, but authorities cracked down once better understanding of toxicity spread. With no easy way to dispose of spills (water treatment doesn’t break it down), industrial users lean on closed-system manufacturing, effective ventilation, and spill response plans rather than chance.
Rethinking Industrial Use and Responsibility
Nitrobenzene features in huge processes in the chemical industry, but today's focus shifts toward solvent alternatives and green chemistry. Making aniline and dyes keeps it on the order sheet, but research pushes newer, safer production methods without legacy by-products. Many labs now teach safety for nitro compounds with more emphasis than in decades past, using facts about toxicity and exposure, not just labeling rules. For today’s students and chemists, hands-on training, proper PPE, and early respect for its hazards keep accidents from happening. I remember, as a student, being handed nitrile gloves and hearing stories—not of lab accidents, but of steady, methodical work.
People often forget, chemistry gives us amazing tools but demands focused respect, especially with something as potent as nitrobenzene around.
What are the environmental impacts of nitrobenzene?
From Chemistry Lab to Rivers and Fields
Nitrobenzene has a familiar smell. It reminds some of shoe polish and bitter almonds. This stuff gets used in making dyes, lubricating oils, certain polishes, and even pesticides. While most people don’t bump into nitrobenzene at the grocery store, its reach stretches a lot further than you’d think. In big manufacturing towns, nitrobenzene leaks or run-offs end up touching both air and water. When I first learned about it in college chemistry, the focus was always on safety inside the lab. Out in the world, precautions often fall short once nitrobenzene leaves the factory gates.
Toxic to Aquatic Life
Loaded with a single nitro group attached to a benzene ring, nitrobenzene resists breaking down, especially in cold or low-oxygen spots. Rivers near chemical plants see direct discharge, bringing nitrobenzene right into the ecosystem where fish and small aquatic bugs live. Once in the water, it doesn’t vanish easily. Fish gulp in contaminated water and absorb the toxin through gills. Even low dosages ramp up stress on aquatic creatures, making them sluggish or stopping their growth. High concentrations can wipe out whole populations in creeks and ponds close to waste discharge zones.
Soil Trouble and Plant Health
Wastewater carries nitrobenzene into fields. I once visited farmland downstream from a chemical plant, where farmers complained about poor crop health and odd root stunting. Nitrobenzene changes the landscape by binding to soil, sometimes sticking around for months. Plants pick up nitrobenzene through their roots, and it slows down or even blocks photosynthesis. The trees, vegetables, and grains in that area looked yellow and droopy, far from healthy. This means the chemical not only threatens water creatures, but starts breaking the food chain’s base.
Bioaccumulation and Wildlife Risks
Nitrobenzene doesn’t just disappear. It inches into insects nibbling on contaminated plants, then into frogs and birds that eat those insects. Over time, nitrobenzene stacks up in animal tissues. Experiments in the past have shown reproductive issues, deformities, or higher death rates in wildlife exposed long-term. That can ripple through a habitat, thinning out entire layers of a natural community. The effect strikes hardest where pollution controls fall short or monitoring gets skipped entirely.
Health Hazards for People
A few counties with old factories discovered nitrobenzene in drinking wells. People reported skin rashes, headaches and—after months of exposure—damage to their blood cells. Nitrobenzene turns hemoglobin into something called methemoglobin, which stops red blood cells from moving oxygen efficiently. Kids and elderly folks are most at risk. As a parent living near an industrial zone, learning about what’s in your water turns into a real-life concern, not just a science headline.
What Can Be Done?
Fencing off the problem starts at the source. Factories using or making nitrobenzene should use closed-loop systems and proper capture for their waste. Routinely checking air, river, and soil samples keeps the problem in check and makes companies fix leaks before communities get hit. A friend in local government helped push for real-time pollution monitors; each time the alarms went off, crews jumped on spills quickly, saving a lot of damage downstream. Farmers can use barrier crops and testing kits to spot trouble early. Public education stays crucial. Folks need to know what to look for, and how to push for stronger local protections.
The lesson never changes: what leaves a chemical plant doesn’t just float away. It lands in the rivers, soil, plants, and—eventually—into the lives of people. Strong rules, active monitoring, and community pressure can keep the burden of nitrobenzene out of everyday life.
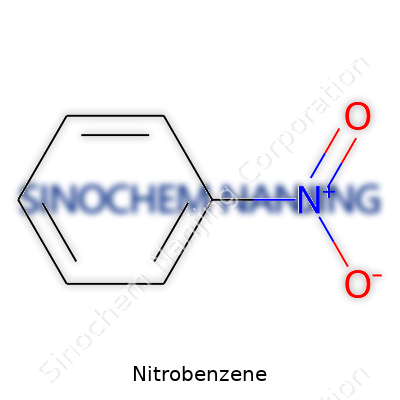
| Names | |
| Preferred IUPAC name | Nitrobenzene |
| Other names |
Nitrobenzol Oil of mirbane |
| Pronunciation | /ˌnaɪ.trəʊˈbɛn.ziːn/ |
| Identifiers | |
| CAS Number | 98-95-3 |
| Beilstein Reference | 120925 |
| ChEBI | CHEBI:27941 |
| ChEMBL | CHEMBL1401 |
| ChemSpider | 558 |
| DrugBank | DB04220 |
| ECHA InfoCard | echa.europa.eu/infocard/100.000.569 |
| EC Number | 207-355-2 |
| Gmelin Reference | Gmelin Reference: 1031 |
| KEGG | C01739 |
| MeSH | D009591 |
| PubChem CID | 7416 |
| RTECS number | DA6475000 |
| UNII | 88L5XMG092 |
| UN number | UN1662 |
| Properties | |
| Chemical formula | C6H5NO2 |
| Molar mass | 123.11 g/mol |
| Appearance | Oily pale yellow liquid |
| Odor | almond-like |
| Density | 1.203 g/cm³ |
| Solubility in water | 1.9 g/L (20 °C) |
| log P | 1.85 |
| Vapor pressure | 0.34 mmHg (20°C) |
| Acidity (pKa) | -7.1 |
| Basicity (pKb) | 11.7 |
| Magnetic susceptibility (χ) | -57.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 1.214 cP (20°C) |
| Dipole moment | 4.22 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | ΔfH⦵298 = +11.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3264.0 kJ/mol |
| Hazards | |
| GHS labelling | **GHS02, GHS06, GHS08** |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H370, H410 |
| Precautionary statements | P210, P261, P264, P270, P271, P272, P301+P310, P302+P352, P304+P340, P308+P311, P312, P321, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-AOX |
| Flash point | 86°C |
| Autoignition temperature | 482 °C |
| Explosive limits | 1.8–5.9% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 640 mg/kg |
| NIOSH | **NIOSH: SN 0450000** |
| PEL (Permissible) | 1 ppm (skin) |
| REL (Recommended) | 0.001 |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Aniline Azobenzene Azoxybenzene N-Nitrosodiphenylamine N-Phenylhydroxylamine Phenol Benzene |

