Nitric Acid: A Hard Look at a Familiar Compound
Tracing the Roots
Stepping into any corner of science history, you find nitric acid playing a role, whether it’s in the rise of alchemy or the dawn of modern chemistry. By the late 1700s, chemists started isolating it cleanly, giving birth to a product that shaped everything from explosives to plant fertilizers. The early years involved crude distillation of saltpeter and vitriol. Over time, researchers found more effective routes with air and ammonia. This era moved the world from expensive, dirty production to something more reliable and scaled. Industrialization, war, and an ever-expanding agriculture industry locked arms with nitric acid, turning it from a laboratory curiosity into a powerful agent of growth—and destruction. Every bottle sold today traces back to those first spark-filled labs and the race for better yields and safer handling.
Peeling Back the Product
Nitric acid doesn’t show up as just a single thing. Almost always, it walks into a lab or a factory as a colorless or slightly yellow liquid with a sharp, biting smell. Every bottle tends to come stronger or weaker based on the percentage of the acid in water—most buyers look for concentrations around 68%, considered standard commercial grade. Fuming nitric acid, rich with nitrogen dioxide, wears a deep red-brown color and packs a different punch, suited for rocket propellants and specialized syntheses. These bottles need sturdy labels and robust packaging. Any hint of sunlight, heat, or metal rusting can throw everything out of balance, so suppliers use brown glass or heavy-duty plastic, often wrapped tight with hazard symbols.
Handling Chemistry Up Close
In the lab, nitric acid commands respect, and its personality is anything but subtle. It boils around 83 degrees Celsius, freezes not much lower than water, and dissolves easily into any humidity in the air. The moment it touches copper or silver, aggressive chemical reactions begin, producing dangerous fumes of nitrogen oxides. This stuff eats through organic tissue and metal with equal enthusiasm, making gloves and goggles standard kit for anyone getting close. Chemists must avoid letting it splash or mix by accident with common organic solvents; a tiny mistake can cause a violent explosion. Many still remember classroom stories of stains left behind on fingers—a warning written into skin.
The Nuts and Bolts
Suppliers and users alike keep a close eye on purity and labeling. Infrared or mass spectrometry helps check for contaminants, and every drum moving across borders carries precise concentration and hazard information, often in up to five languages. Forgetting to check the labeling, even once, leads to costly accidents. You won’t find generic paint thinner labels on this shelf. Workers and researchers review labels as carefully as pilots double-check altitude—nobody trusts memory alone. Remembering these details, and treating every acid transfer as a deal with risk, keeps nitration disasters out of headlines.
How It’s Made
Today, almost all nitric acid comes from oxidizing ammonia over platinum or rhodium catalysts at high temperature—a process that turns out millions of tons worldwide each year. It’s a straightforward reaction in theory, but scaling it to industrial size took decades of research and engineering. Factories must balance pressure, temperature, and clean gases; a missed step means lower yields or dangerous emissions. Direct routes from saltpeter and sulfuric acid linger in old textbooks. Still, industry sticks to ammonia oxidation because it scales well, and air is cheap. Every time I’ve visited fertilizer plants or chemical factories, the air seems charged with responsibility. Operators don’t get away with shortcuts. The world depends on keeping this chain safe, efficient, and as green as possible.
Chemical Reactions: The Real Workhorse
Nitric acid’s main trick seems simple at first—it’s a world-class oxidizer. Drop it onto iron, copper, or organic matter, and it tears electrons free, forming new compounds and releasing gas. These reactions build the foundation for nitration, a game-changing transformation for thousands of everyday molecules. In medicine, flavors, explosives, and fuels, nitric acid gives rise to new products by attaching its own nitrogen and oxygen. Chemists rely on it for making fertilizers, cleaning surfaces, etching electronics, and even dissolving noble metals for analysis. Tweaking purity and blending with sulfuric acid invents whole new classes of reactions. It’s impossible to walk through a catalogue of chemicals and not run into something shaped by this one acid.
Many Names, One Reputation
Chemists like to call it HNO3, but bottles and invoices might say aqua fortis or spirit of niter. Historic names reflect both the acid’s power and its old roots in alchemical tradition. Regardless of the title, everyone in the trade knows the sulfurous odor and danger of mixing labels. Slang from field chemists makes its way into research papers and safety meetings, reminding us that every community shapes language to suit the risks and habits built up over generations.
Staying Safe in the Real World
Nitric acid’s hazards reach beyond the pages of MSDS sheets. Skin contact leads to yellow stains at best, serious burns at worst. Breathing vapors damages the lungs, and mixing acid with the wrong chemicals explodes in catastrophic ways. Seasoned lab workers gear up with gloves, splash goggles, and thick aprons. Storage demands good ventilation, temperature control, and a strict policy on keeping it far away from organic solvents or reducing agents. In my own time overseeing research labs, site managers laid out safety as a culture, not just rulebooks. Regular training drills and quick communication mean the difference between a minor mishap and a life-altering accident. Proper acid handling isn’t for the complacent or the untrained.
Transforming Industries
Without nitric acid, much of today’s agriculture industry would collapse. Plants crave nitrogen, and nitrogen fertilizers—urea, ammonium nitrate, and others—start their journey from ammonia and nitric acid. Explosives manufacturers put it front and center for TNT, nitroglycerin, and propellants. Even the plastics industry, pharmaceuticals, and electronics lean heavily on its ability to transform molecules quickly and efficiently. A city’s water pipes, etched metals, and circuit boards each owe something to the quiet, relentless work of nitric acid. Stepping outside factories, even environmental labs rely on it to break down samples and detect toxins. It has a hand in nearly every walk of industrial and research life, from green fields to data centers.
Pushing Boundaries in the Lab
Researchers never seem to run out of ideas for nitric acid. Its reactivity sparks innovation from explosives to medical compounds targeting cancer cells. In the past decade, scientists sequenced new catalytic processes, searching for higher efficiency, lower emissions, and better selectivity. Labs race to create variants weaker or more tailored than ever before, some aimed at cleaning electronics, others for medical diagnostics. The search for catalysts that will slash greenhouse gas outputs came from a realization that nitric acid production ties directly to global warming. More environmentally sound processes, smarter engineering, and digital monitoring open new chapters every year. My own work has shown me just how much effort it takes to invent safer processes and adapt to changing health and climate standards. Chemists learn early to view nitric acid both as a building block and a challenge to do better.
Looking at Harm: Toxicity and Health
No one should treat nitric acid lightly. Even modest exposure can inflame airways, burn eyes, and scar skin. In industrial settings, monitoring for nitrogen dioxide—a toxic, reddish gas often released during spills—stands as much a priority as handling the acid itself. Chronic exposure contributes to long-term respiratory damage, so proper ventilation and personal protective equipment become non-negotiable. Stories circulate of accident victims suffering lifelong consequences after inhaling a few breaths or suffering a single splash. Toxicity studies go beyond the acid alone—researchers study breakdown products like nitrates and nitrites, which can seep into water supplies, enter the food chain, and create public health headaches. Risk mitigation ties into ongoing training, tighter regulations, and new materials for storage and spill control.
What Comes Next?
Nitric acid isn’t escaping the gaze of regulators, environmentalists, or innovators any time soon. Climate change and demand for sustainable agriculture put new pressure on producers to tighten the carbon footprint. Green chemistry teams everywhere compete to cut emissions and close cycles. Digital control systems, improved catalysts, and smarter recycling promise progress, but developing nations still face tough trade-offs in cost, safety, and environmental damage. Each advance ties back to real people: factory workers, farmers, researchers, and families living downwind. Future progress asks everyone to rethink waste, process safety, and the global cycle of nitrogen. The next breakthrough likely won’t come from a single genius, but from collaboration across chemistry, engineering, and policy. That’s the only way to respect nitric acid’s power and its place in shaping the modern world.
What are the main uses of Nitric Acid?
The Backbone of Agriculture
Growing up in a farming town, I noticed how local feed stores always had deliveries of certain chemicals. One big reason: fertilizers. Farmers rely on nitric acid to make ammonium nitrate, a key fertilizer in growing wheat, corn, and vegetables. Without stable harvests, food prices swing and communities feel it. As of 2022, the Food and Agriculture Organization noted that more than 65% of industrially produced nitric acid is used in fertilizers. Crops don’t flourish on hope alone, and nitric acid helps them get the nutrients they miss from depleted soils, year after year.
Supporting Safer Mining and Metals
Nitric acid works behind the scenes in mining operations that pull copper, gold, and silver from the earth. In the metals industry, it's not about brute force. Reactions involving nitric acid enable workers to extract and refine metals more efficiently and safely. Most people don’t see this part, but if mining operations went back to older methods only, pollution, energy use, and injuries would jump.
Driving Science and Innovation
School chemistry labs love talking about acids and bases, but nitric acid’s high reactivity carries the action to university and pharmaceutical labs as well. Researchers use it to create new drugs, test chemicals, or prepare samples for analysis. One misstep and you don’t get clean results, so nitric acid helps ensure precision that could influence a batch of medicine or a crucial vaccine run.
Cleaner Engines, Improved Explosives
Nitric acid plays a leading role in making explosives. While that sounds unsettling, it’s not all about dynamite in cartoons. Controlled explosives remain critical for construction, mining, and even space exploration. The acid is blended into materials for airbags and propellants for rockets. Strict regulations surround its production and sale to prevent disaster in the wrong hands, balancing safety with technological progress.
Cleaner Cities and Waterways
Factories and city water systems use nitric acid to treat wastewater and neutralize excess ammonia. Municipal water plants must keep drinking water safe and free of toxins. Industrial use sometimes carries risk—spills can harm streams and rivers. Still, the acid’s ability to target certain contaminants means less risk of bacteria, nitrates, and heavy metals showing up at the tap.
Challenges and the Path Forward
No chemical comes without headaches. Breathing nitric acid vapors can scar lungs, and careless storage burns skin and corrodes containers. Accidents in chemical plants have cost lives and raised fears about regulation gaps. One way forward: more responsive safety training, clear labeling, and modern leak-proof tanks. The payoff arrives in lower rates of workplace injuries and fewer public health alerts.
Smart policymakers look at innovation in recycling old nitric acid, not just burning through new supplies. The European Union launched projects to recover and recycle this acid in fertilizer production, lowering both pollution and costs. That model pushes other regions to consider the acid’s life after it splits molecules for industry or agriculture.
How should Nitric Acid be safely stored?
Recognizing the Hazards
Nitric acid can burn straight through flesh, metal, and just about any organic substance. That’s not just a scary image—the facts say so. Exposure to its fumes can scar lungs. Direct skin contact creates chemical burns, sometimes severe. Even a splash, mixed with the wrong material, gives off toxic gases. Accidents aren’t rare. Year after year, chemical incidents with nitric acid keep popping up in safety reports across labs, factories, and even poorly stocked garages.
Container Material Makes All the Difference
Here's what often gets overlooked: nitric acid reacts violently with common metals. Storing it in anything like steel, iron, or copper turns the drum into a slow-motion grenade. People forget even a little moisture helps nitric acid nibble away at these metals, releasing brown nitrogen dioxide gas in the process. Glass (not the everyday soda-lime kind, but borosilicate) handles pure nitric acid well, but those bottles break if you bump them hard enough. In my own lab days, a shattered glass bottle created a panic I won’t ever forget. As for plastics, only a few make the cut. High-density polyethylene (HDPE) and polytetrafluoroethylene (Teflon) stand up to nitric acid’s tricks. Anything else—like PVC or regular polyethylene—softens, splits, or crumbles much too soon.
Temperature and Sunlight: Not Just a Suggestion
Storing the bottle out of sunlight and heat prevents decomposition. Nitric acid breaks down in sunlight, kicking out those toxic brown fumes and degrading itself into uselessness. Heat speeds this process up. Stash it somewhere cool, dry, and shaded. If you’ve ever seen a bottle with a warped cap or yellow stains creeping down, you’ve seen firsthand why keeping things chilly matters.
Avoiding Dangerous Mixtures
Nitric acid doesn’t play well with others. Storing it next to organic solvents, reducing agents, or anything combustible puts lives at risk. One tiny leak or accidental knock can cause fire, explosion, or poison gas. The facts don’t lie—many chemical fires have roots in casual storage practices. Keep it isolated, preferably in a dedicated acid cabinet built with corrosion-resistant lining. Never leave it near anything flammable or reactive, including spill absorbents made from organic material. Just last year, an improper spill cleanup sparked an evacuation and injuries—a little planning would have saved pain and money.
Ventilation and Pressure Relief
Nitric acid bottles tend to build pressure, even in quiet storage. Proper bottles come with vented caps for a reason. If the acid’s strength is above 70%, pressure rises fast. I’ve seen caps fly off, pressurizing gas hissing out, all from storing bottles too tightly or in warm, closed cabinets. Reliable venting, along with a well-ventilated storage area, drops fumes and pressure build-ups. Local exhaust fans, hoods, or chemical storage rooms with separate airflow add another layer of safety.
Training People, Maintaining Records
Safe storage starts with people who recognize the real dangers, not just those who’ve memorized a label. Training should cover immediate cleanup steps, first aid, and how to read chemical labels for concentration. Have spill kits made for acid—not some all-purpose powder—on hand. Log inspection dates, note bottle conditions, and cycle out old containers before they turn into trouble. This amount of diligence pays off. Incidents drop, and nobody faces the nightmare of facing an acid cloud without warning.
What are the hazards associated with Nitric Acid?
What Makes Nitric Acid So Dangerous?
You can find nitric acid in labs, factories, school storerooms, and even on trucks headed toward fertilizer plants. In daily life, though, few people handle it directly. Most will only hear about it after a chemical spill or accident. That’s probably for the best, because this acid comes with risks that go beyond what most folks expect from a common industrial chemical.
Risks to Health and Safety
Anyone who has ever caught a whiff of nitric acid remembers the experience. Breathing those fumes feels like inhaling fire. The acid sends out brownish gas that bites into lungs and leaves people coughing, choking, and sometimes struggling to breathe. In serious cases, workers exposed to concentrated fumes have ended up in the hospital with severe lung injury. Over time, even smaller amounts can mark up lungs and throat, lead to chronic bronchitis, or ruin a sense of taste and smell.
Skin contact sets off another set of problems. A spilled drop doesn’t simply burn for a minute. It can eat through outer layers of skin, cause yellow stains, and spark chemical burns deep enough to need medical care. Eyes are just as vulnerable. Splashes almost always mean a trip to the emergency room. Even with quick rinsing, some people lose their vision.
Risks to the Environment
One big accident can hurt more than a few workers. Nitric acid reacts fast with lots of other common substances, including metals, organic matter, and even air. It releases toxic gases that settle in low areas or drift for miles. These gases include nitrogen dioxide, which both stings the lungs and feeds the formation of urban smog. Areas around spills see plants withered, streams acidified, and wildlife driven off. In fertilizer plants, leaks sometimes lead to fish kills downstream or a sharp drop in soil life near accident sites.
The Chemical’s Reactivity
Over the past decade, more than a dozen factory accidents involved nitric acid mixing with hazardous materials—explosives, reducing agents, or even rags soaked in oil. Explosions and flash fires followed. One reason comes from the way nitric acid strips electrons from other things with ease. Combustible items can burst into flame. Metals like copper and zinc bubble and produce heat. In some lab accidents, mixing nitric acid with wrong chemicals has blown lids off containers or filled rooms with choking gas.
Keeping People Safe
Most chemical safety comes down to training and respect. At my first job in an agriculture lab, my boss pounded into our heads the idea of “respecting the acid.” This meant full face shields, gloves, special fume hoods, and never, ever working alone. In accident reports, the root cause often traces back to skipped safety steps or a rush to finish. Some workers still underestimate how little it takes for nitric acid to cause an emergency. In addition, plant managers sometimes skip real-time gas detection or rely on aging ventilation systems.
Better Solutions and Prevention
Regulatory agencies demand clear labeling, spill kits, and strict storage rules for a reason. Too many places keep acids in old containers or near incompatible chemicals. Enforcement of separation—and regular emergency drills—keeps people alive. You can build safety into every step: limit who can access storage areas, replace glass with plastic containers, and insist on fresh PPE for every shift. Ventilation should work at all hours, not just during inspections. Most importantly, workers need a voice in safety policies, because the people closest to the hazard often spot problems first.
How can Nitric Acid spills be cleaned up?
Looking Into Nitric Acid and Its Risks
Nitric acid serves many purposes, especially across labs and manufacturing. Any time this chemical leaks into the open, you’re not just handling a mess—there’s danger for people and the environment. Nitric acid can burn skin, destroy lungs, and corrode most surfaces. I remember my college chemistry lab accident, where a dropped beaker had people running for baking soda and calling hazmat. The sight stuck with me: yellowish fumes curling up, that sharp bite in my nose, instructors not panicking but clearly on edge. Nobody brushes off a spill like that.
Immediate Steps: People First
Safety gear changes everything in these situations. You reach for chemical-resistant gloves, splash goggles, a lab coat, and proper ventilation. No one takes shortcuts with nitric acid—get bystanders out fast and make sure only trained folks stay behind. Neutralization doesn’t start until everyone’s protected and local emergency services know what’s going on. I once saw someone freeze at the thought of getting blamed for an accident. No shame there. People come way before cleanup.
Neutralizing the Acid
Old chemistry books preach about baking soda and neutralizers, but reality calls for more than a handful of powder. Sodium bicarbonate or lime gets spread evenly over the spill until fizzing stops and you’re sure the liquid lost its sting. Commercial neutralizers guarantee stronger results for big spills, soaking up acid while changing color to show the change. Any leftover liquid calls for absorbent pads built for acid use—no quick wipes or mops unless you want worse burns.
Testing the neutralized area with litmus paper checks if everything’s safe. If it still turns red, more neutralizer gets tossed on. This step cannot be skipped. Even a small leftover spot keeps releasing fumes long after the surface seems dry. People sometimes rush, thinking time equals safety. Rushing only spreads the trouble.
Disposal: Doing It Right
Used absorbents and wash rags turn into hazardous waste the minute they touch nitric acid. Tossing them in regular trash spreads contamination. The right path means sealing everything in corrosion-proof containers, labeling them as hazardous, and arranging for certified waste handlers to pick them up. Where I grew up, the local recycling center refused many chemicals. My science teacher drove two towns over for safe disposal. Efforts like that protect water supplies and keep toxins from sneaking into landfills.
Prevention and Preparation
No story about acid spills feels complete without a push for prevention. Regular safety drills, clearly marked spill kits, and updated handling instructions keep accidents from turning serious. Storage means locking up acids separately from bases and flammables, far from drains or floor cracks. I watched a small factory dodge disaster simply by storing nitric acid in leak-proof tubs on solid shelves. Training helped, but careful storage and quick labeling did more.
The Value of Strong Oversight
Strong regulations keep everyone honest—no cut corners for insurance or cost savings. Oversight keeps accident rates low. OSHA and the EPA run inspections and make sure workplaces keep up with safe practices. Talking to folks in industrial roles, I hear gratitude for tough standards. Safe jobs mean parents and workers go home unscathed, communities avoid toxic events, and nobody has their life shaken by simple carelessness.
What precautions should be taken when handling Nitric Acid?
The Hazards Behind the Bottle
Nitric acid does some heavy lifting in industry and labs, but a single spill or careless move can turn a routine task into a medical emergency. This acid isn’t just strong—its fumes corrode mucous membranes, and a drop on your skin leaves a yellow stain that means cell damage. Inhaling its vapors leads to lung irritation, painful coughing, sometimes even permanent respiratory damage. I’ve worked with nitric acid in undergraduate chemistry and remember the sting in my nose despite a fume hood and thick gloves. That memory never leaves.
Why Personal Protective Equipment Is a Must
No one naturally resists a splash of nitric acid. That’s why a sturdy pair of chemical-resistant gloves matters more than style. Neoprene or butyl rubber stands up to the harshness. Cotton clothing absorbs the acid and only makes burns worse. Lab coats meant for chemistry—not store-bought cotton ones—add a barrier. Safety goggles protect delicate eye tissue against droplets, which cause blindness with shocking speed. A proper face shield closes any gap left by goggles. Closed shoes, not sandals, keep feet in one piece if someone drops a flask.
The Importance of Good Ventilation
Nitric acid fumes carry nitrogen dioxide, a gas that can damage lungs fast. Ventilation saves you here. Fume hoods work well, pulling dangerous gases away from faces, lungs, and clothing. Relying on open windows or regular fans doesn’t cut it; fumes are heavier than air and can build up near the ground, out of sight.
Safe Storage Keeps Accidents Out of Reach
Nitric acid needs its own space. Never put it near organics like alcohol, acetone, or even common cleaning agents. The right bottle has a vented top and sits on a dedicated acid shelf, below eye level. Fire, heat, or sunlight pushes pressure up and can send that bottle spraying across a room. Labels show more than just a name—they warn everyone coming near. Plenty of accidents happen because a half-erased label turned acid into a guessing game.
Thinking Before Pouring
Pouring acid into water, never water into acid, sounds simple but saves skin and sight. Water poured into concentrated acid flashes to steam and spits back, spraying anyone standing nearby. Controlled small batches and a stable workspace help avoid jolts and leaks. Keeping paper towels or absorbent pads handy deals with minor splashes without turning cleanups into a second mess.
Knowing What to Do in an Emergency
An eyewash station and safety shower belong right near the workspace, not down the hall. A few seconds make the difference between irritation and a trip to the emergency room. I’ve seen panic during a chemical spill; training helps muscle memory kick in when adrenaline clouds judgment. Neutralizing spills with baking soda or special agents works, but only if you spot the problem and act fast.
Training and Respect
No shortcut replaces good training. Working with nitric acid isn’t about macho stories or shortcuts; it’s about respecting something powerful enough to eat through metal. Reminders, clearly posted instructions, and sharing stories of near-misses keep awareness high. Chemicals respond to respect, not bravado.
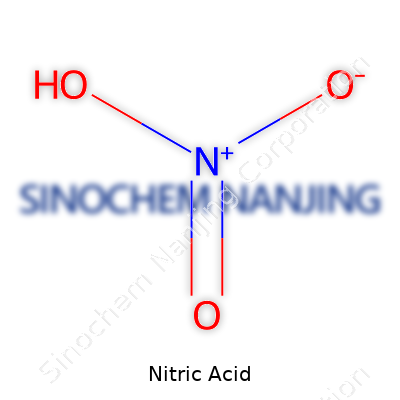
| Names | |
| Preferred IUPAC name | Nitric acid |
| Other names |
Aqua fortis Hydrogen nitrate Spirit of niter |
| Pronunciation | /ˈnaɪ.trɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7697-37-2 |
| Beilstein Reference | 3587153 |
| ChEBI | CHEBI:48107 |
| ChEMBL | CHEMBL1239 |
| ChemSpider | 766 |
| DrugBank | DB09114 |
| ECHA InfoCard | ECHA InfoCard: 0356-4472-006-47 |
| EC Number | 231-714-2 |
| Gmelin Reference | Gmelin Reference: 142 |
| KEGG | C01483 |
| MeSH | D009574 |
| PubChem CID | 944 |
| RTECS number | QU5775000 |
| UNII | 21C1NTO1UN |
| UN number | UN2031 |
| Properties | |
| Chemical formula | HNO3 |
| Molar mass | 63.01 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Pungent, choking |
| Density | 1.51 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.3 |
| Vapor pressure | 48 mmHg (20 °C) |
| Acidity (pKa) | −1.4 |
| Basicity (pKb) | -1.4 |
| Magnetic susceptibility (χ) | Weakly Paramagnetic |
| Refractive index (nD) | 1.397 |
| Viscosity | 1.1 mPa·s |
| Dipole moment | 2.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −206.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −207 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB36 |
| Hazards | |
| Main hazards | Corrosive, causes severe burns, toxic by inhalation and ingestion, strong oxidizer, reacts violently with organics and reducing agents. |
| GHS labelling | **"Danger. Hazard statements: H272, H314. Pictograms: GHS03, GHS05. Signal word: Danger."** |
| Pictograms | GHS05,GHS03 |
| Signal word | Danger |
| Hazard statements | H314, H290, H272 |
| Precautionary statements | P260, P261, P264, P271, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | 410 °C |
| Explosive limits | Not explosive as such, but contact with oxidizable substances may cause explosive reactions. |
| Lethal dose or concentration | LDLo oral human 430 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 430 mg/kg |
| NIOSH | 0346 |
| PEL (Permissible) | PEL (Permissible) for Nitric Acid is 2 ppm (parts per million) or 5 mg/m³ (OSHA standard, 8-hour TWA) |
| REL (Recommended) | 5-10 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Aqua regia Ammonium nitrate Dinitrogen tetroxide Hydroxylammonium nitrate Nitrate Nitrous acid Peroxynitric acid Trinitramide Urea nitrate |

