N-Vinylpyrrolidone: Taking a Closer Look at a Modern Workhorse
Looking Back: How N-Vinylpyrrolidone Became a Chemical Mainstay
N-Vinylpyrrolidone, better known in some circles as NVP, has managed to stay relevant from its twentieth-century beginnings straight through to today’s labs and manufacturing lines. Chemical researchers in the mid-1900s honed in on N-Vinylpyrrolidone for its potential in polymer chemistry, which quickly led to its spread across various industries. For a time, folks chasing better plastics and new adhesives saw NVP as a kind of missing link; it slotted neatly into holes where older building blocks just didn’t perform. Over time, its reliability and adaptability gave NVP a seat at the table for pharmaceutical, cosmetic, and medical applications. Tracing this path isn’t simply an academic exercise—it gives a sense of how societal needs, paired with scientific curiosity, can elevate a chemical to indispensable status.
Understanding What N-Vinylpyrrolidone Brings to the Table
To me, what stands out about N-Vinylpyrrolidone comes down to its versatility. As a monomer, it shows off an ability to blend, link, and transform. Its structure offers a vinyl group attached to a pyrrolidone ring, making it reactive in all the ways that matter for polymerization. Chemically, NVP comes as a colorless liquid, and the clear, water-miscible character reflects a purity that suits both industrial and laboratory applications. You get a molecule that boils around 90–100°C at reduced pressure, and it doesn’t shy away from air or light under normal storage, which cuts down on worry for operators. I’ve run across NVP plenty while talking shop with formulators—they see it as a backbone for things that need to stick, coat, or dissolve effectively.
Diving Into the Details: Properties That Matter on the Floor
The physical side of N-Vinylpyrrolidone tells an important story. Its low viscosity lets it move through pipelines and reactors without clogging, and its stability lets storage happen without special gear or drama. Chemically, it’s not explosive, but its flammability earns attention on production floors due to air-reactivity at high temperatures. What you get is a monomer that pairs water solubility with that rare trait—solubility in many organic solvents—making it a go-to for chemists who need compatibility. The reactivity, especially with free radical initiators, paves the way for producing polyvinylpyrrolidone (PVP), known for its biocompatibility and use throughout healthcare and beauty markets. In my experience, it’s not just the structure that speaks, but the reliability—batch after batch, firms find that NVP rarely throws unwanted surprises.
From Bench to Bottle: Making and Labeling N-Vinylpyrrolidone
If you look into the production side, acetylene and γ-butyrolactone are the core reactants, usually under the watchful eye of select catalysts and controlled conditions. As an operator, I’ve seen how refining this process ensures the monomer’s purity—excess acetylene gets scrubbed off, distillation removes water, and chemical traces drop to safe, acceptable limits. Technical specifications focus on residual vinyl groups, moisture, and stabilizers like MEHQ, which slow down unwanted polymerization. Regulations drive bench-to-bottle traceability, and packaging standards focus on correct sealing and labeling, reflecting the push for safety in transport and storage.
Chemical Reactions and Tweaks: NVP’s Road to Innovation
Scientists have always found ways to squeeze a little more function out of NVP. Its vinyl group reacts quickly in radical, cationic, or even photoinitiated polymerizations. It forms copolymers with ingredients like acrylic acid, methyl methacrylate, and styrene. This flexibility provides engineers endless combinations for gels, films, and surface coatings. Over the last decade, more talk has circulated about using NVP in smart hydrogels and biomedical scaffolds, taking advantage of both its reactivity and its gentle presence in biological settings. Even now, researchers dig into how attachments—introducing sulfonic or carboxyl groups—bring new uses in water treatment or drug delivery. In my own reading, I notice the excitement isn’t just hype. Instead, it’s a practical response to longstanding needs for smarter, more responsive materials.
Naming a Chemical: Synonyms and Lingo Over Time
People in the chemical trade tend to invent nicknames or shorthand. Along with N-Vinylpyrrolidone and its abbreviation NVP, it’s also called 1-vinyl-2-pyrrolidone. Polyvinylpyrrolidone’s monomeric unit comes directly from NVP, so you’ll sometimes see suppliers flagging the monomer for clients focused on specialty polymer projects. Learning to speak the language proves handy; mislabeling or confusion can quickly derail a run, especially where regulatory filings need clear substance identity.
Real-World Use: How NVP Has Become a Standard Ingredient
Out in the world, the most obvious use of N-Vinylpyrrolidone shows up in the production of PVP, a polymer used in tablets, wound dressings, liquid bandages, and contact lens solutions. Those applications thrive because of the monomer’s clean reaction and the non-toxic, film-forming polymer that results. In cosmetics, it helps hairsprays stick without clogging the nozzle, and it works quietly in adhesives and paints, thanks to strong film-formation and solubility. More recently, the boom in biomedical research has pulled NVP into tissue engineering and drug delivery systems. The fact that hospitals and manufacturers alike can rely on the monomer for purity and consistency makes for smoother product development and easier compliance with medical guidelines.
Staying Safe: Standards and Smarts on the Production Line
Working with N-Vinylpyrrolidone involves exposure controls and a lot of respect for best practices. Even though toxicity cases remain rare, direct skin or eye contact can irritate, and inhalation in poorly ventilated rooms isn’t something to shrug off. Most companies require gloves, goggles, and fume hoods at a minimum, backed up by local and global safety standards. Regulatory frameworks—especially from Europe and North America—address worker exposure and labeling to prevent chronic health issues or accidental misuse. I once saw a warehouse dinged during inspection for poor labeling, and it shut production down for a week; safety culture is more than paperwork—it keeps people on the job and products on the market.
What the Research Shows about Toxicity and Risks
Toxicologists have dug fairly deep into N-Vinylpyrrolidone’s risks, mainly because of its widespread industrial and health applications. Short-term exposure in animals highlights possible liver and kidney stress at very high doses. Studies mention moderate irritancy to skin and mucous membranes, but large-scale epidemiological evidence of chronic human harm doesn’t materialize at standard industry concentrations, as long as protocols hold. Regulatory guidelines focus on controlling exposure in workplaces, which means air monitoring and respiratory protection where necessary. Also, with growing pressure to phase out more hazardous intermediates, NVP sometimes lands in the middle ground—offering a relatively safe balance for formulators, but deserving continued oversight.
Keeping It Moving: R&D and the Push for Better Uses
New research on N-Vinylpyrrolidone leans toward medical devices, nanomedicine, and eco-friendly polymer production. What stands out to me is how each decade seems to draw NVP into new ground—hydrogels responsive to temperature or pH, binders for high-performance batteries, or co-monomers in next-generation injectable pharmaceuticals. Companies and academic groups focus on reducing residual monomer content, which feeds directly into safety and performance. Sustainable sourcing and greener manufacturing methods now earn more weight—pressure from environmental regulations and customers with higher expectations combine to drive incremental but persistent change.
Glimpsing the Future for N-Vinylpyrrolidone
Peering ahead, the track record of N-Vinylpyrrolidone suggests few signs of fading relevance. Industry demand continues from pharmaceuticals and medical devices, but the chemical’s adaptability attracts sectors developing specialty coatings, 3D printing resins, and biocompatible implants. As regulations tighten around legacy chemicals, the marketplace shifts toward ingredients with clear toxicity and environmental impact data—which NVP already brings. What holds back even wider adoption are cost factors and the relentless quest for zero-hazard manufacturing. Still, with fresh research, better production controls, and a bit of market luck, NVP holds enough promise for those watching where modern chemistry is heading.
What is N-Vinylpyrrolidone used for?
The Stuff Behind Everyday Products
N-Vinylpyrrolidone, usually called NVP by folks in chemistry labs, pops up in a surprising number of places. I’ve come across it mostly while working with adhesives and coatings, but its reach stretches far beyond the floors and walls of industry. Walk into any drugstore and the odds are good you’ll find something that leans on this chemical. Take hair gel or mascara: NVP forms the backbone of the flexible yet tough polymers that let those products do their job—holding a hairstyle in place or keeping makeup smooth without flaking off by lunchtime. In the medical world, doctors and patients both rely on wound dressings and surgical glues built from those same polymers. The science boils down to this: NVP’s structure lets it join with other molecules to make big networks called polymers, which deliver both strength and flexibility.
Medical and Tech Applications
Remember getting a cut and running for a liquid bandage? There’s a fair chance NVP’s involved. Pharmaceutical companies turned to it for its knack for holding moisture—something you want to help wounds heal faster. The story doesn’t stop there. In my time writing about medical tech, I’ve seen NVP-based solutions go into tablets and capsules, helping control how a drug releases in the body. Eyedrops sometimes employ it to avoid irritation and help the medicine stick around on the surface. Some dental fillings even rely on NVP’s resilience, helping keep smiles both healthy and lasting.
Printing, Plastics, and the Everyday Environment
Print shops and manufacturers put NVP to work every day. It finds its way into inks that need to resist water even after years of exposure. The electronics world relies on NVP, too, especially for specialty coatings inside devices. Walk through a modern factory floor and you’ll see plastics based on this chemistry—shatterproof safety goggles, tough smartphone cases, even the sealants that weatherproof cables and construction joints. The same chemical magic that makes NVP useful in medicine shows up in protecting printed circuit boards and parts exposed to rough environments.
Weighing Safety and Sustainability
All this versatility brings big questions about safety and the environment. Studies show NVP by itself can cause skin and eye irritation if handled carelessly. Factory workers need solid safety training. I’ve seen research groups pressing for more information about whether traces of NVP could end up in local water sources or soil. So far, most consumer products only use NVP in tightly bound polymers—not as a loose chemical. That reduces exposure risks, but the conversation around microplastics raises new concerns about what happens once those everyday items land in landfills or break apart over time.
Smart Use and Better Alternatives
We depend on chemicals like NVP for the convenience and reliability they offer. Still, smart manufacturing means not just finding better end uses but reducing waste along the way. Some labs have started pushing for greener versions made from renewable sources rather than crude oil. Others look for ways to recycle or safely break down NVP-containing materials after they’re done serving their purpose. As consumers, paying attention to product labeling and pressuring companies for transparency helps keep both business and regulators moving in the right direction. Progress feels slow some days, but raising awareness always pushes the industry forward, one small step at a time.
What is the chemical formula of N-Vinylpyrrolidone?
The Formula Behind N-Vinylpyrrolidone
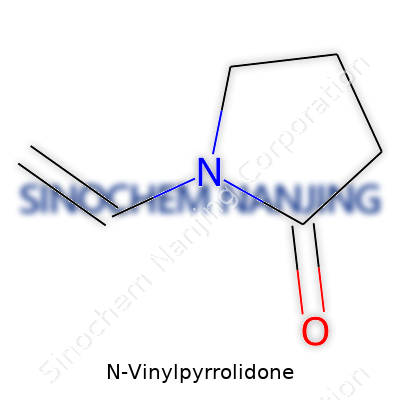
The chemical identity of N-Vinylpyrrolidone goes by the formula C6H9NO. It’s a small molecule, but it does a big job in many products crossing our lives. Each letter and number in that formula holds a part of its character—six carbon atoms, nine hydrogen, one nitrogen, one oxygen. The story of this compound isn’t just in symbols. It’s in how it shapes everyday things from medicines to hygiene items.
Putting Science Into Everyday Perspective
I’ve worked with folks in the pharmacy and in cosmetic labs. There, the relevance of N-Vinylpyrrolidone is hard to miss. Its main claim to fame? The way it polymerizes to form polyvinylpyrrolidone (PVP). You see it in hair sprays, tablet coatings, and even wound dressings. One reason such compounds matter: reliability. Scientists favor N-Vinylpyrrolidone for its ability to dissolve in water and carry other chemicals efficiently. In personal care, its gentle character stands out when formulating for skin contact.
Health and Safety Shape Trust
This small molecule does not come without questions. Health researchers keep a sharp eye on how easily it enters the body. Association with allergies remains rare compared to stronger chemicals, but regulatory guidelines set firm limits on safe workplace exposure. I’ve talked to chemists who pay close attention to ventilation systems and strict handling protocols. Many global regulatory agencies, such as the European Chemicals Agency, have delved into studies around safe exposure, keeping both workers and public interest at the forefront.
Environmental and Social Considerations
Large-scale manufacturing can stretch local resources and create pressure on the environment. N-Vinylpyrrolidone’s production and disposal provide a window to broader industry habits. Where companies invest in solvent recovery or closed systems, emissions drop and safety improves. Community awareness has pushed some producers to invest in improved waste water treatment and to reduce leaks. Speaking with engineers, I’ve heard how ongoing monitoring and government pressure keeps raising the bar.
Balancing Use and Risk for the Future
Across pharma and consumer products, the value of N-Vinylpyrrolidone ties directly to its chemistry. It forms the backbone of polymers that hold medicine tablets together and allow hair products to keep their hold. This means manufacturers have no room for complacency. Initiatives, including greener chemistry projects, aim to produce safer alternatives or refine current methods for less waste. Lab workers look for process steps with lower temperatures or alternative solvents, slashing energy use and improving safety all around. This journey mirrors changes happening in similar fields, where practical improvements ripple from plant floor to final product.
Clear Chemistry, Broad Reach
Understanding the chemical formula of N-Vinylpyrrolidone means more than knowing its structure. It speaks to the choices researchers and companies make every day. A simple string of letters and numbers—C6H9NO—carries a lot of weight in how industries support health, safety, and progress. Anyone curious about the ingredients in their medicines or everyday products will find a long, ongoing story in these six carbons, nine hydrogens, one nitrogen, and one oxygen.
Is N-Vinylpyrrolidone hazardous to health?
Getting to Know N-Vinylpyrrolidone
N-Vinylpyrrolidone, often called NVP in labs and factories, pops up in all sorts of products — from adhesives and inks to polymers found in some medicines and cosmetics. It acts as a building block for creating more complex chemicals. On the surface, that sounds pretty harmless. But beneath this useful side, there’s a story about how NVP interacts with the human body and the environment, one that invites us all to pay close attention.
Health Risks in the Workplace
Factories using NVP keep it in liquid or vapor form, and employees might breathe it in or get it on their skin. Over years of working around chemicals, I’ve learned firsthand that what you can’t see or smell can still cause real trouble. According to research reviewed by agencies like the European Chemicals Agency (ECHA) and U.S. National Institutes of Health (NIH), some people exposed to NVP in high concentrations dealt with skin irritation, and a few got serious eye irritation. Touching NVP without solid protective gear can result in rashes or burns.
Lab studies show that rodents breathing high levels of NVP developed impacts in their liver and kidneys. These animal studies often lead to further scrutiny of chemical safety for humans. The American Conference of Governmental Industrial Hygienists (ACGIH) recommends keeping workplace exposure lower than 0.05 parts per million — and achieving that calls for vigilant air monitoring.
NVP and Long-Term Safety Concerns
Safety agencies keep a close eye on chemicals that linger. Regulators flagged NVP as a possible mutagen. This means, in certain conditions and at high doses, it could mess with genetic material inside cells. Though there isn’t proof that NVP can trigger cancer in humans, the results in animals encourage us not to shrug off those early warning signals. The science is still unfolding, but there is enough evidence to call for stricter safety measures on the job.
Risks Beyond the Factory
Consumers rarely get exposed to NVP at levels seen in factories. It’s present in finished goods in traces, usually locked up in polymers, so it can’t easily escape. Still, no one should assume "trace amounts" always mean “zero risk.” Using a lotion or adhesive once a week probably adds up to little, though frequent users or workers making the products carry a different kind of burden.
Staying Safe Around NVP
Wearing gloves and protective glasses became second nature to those of us in labs and production lines. For everyone working with NVP, local exhaust ventilation and airtight systems head off most of the danger. OSHA and other health agencies stress routine monitoring of air quality and employee health. Even with strict rules, it never hurts to push for substitution with safer chemicals if the job allows.
Those of us following chemical health news have seen shifts over the years, with companies redesigning processes and chemists inventing new, less risky alternatives. Workers have the right to know what they’re dealing with, and training in safe handling isn’t just a checklist — it’s insurance for families and communities.
Looking Ahead
NVP brings value, but only in the right boundaries. The science shows clear warning signs at high exposure, pushing for strong workplace controls. Honest conversations between workers, employers, and regulators—backed by transparent research and regular review—keep everyone protected and push chemistry toward healthier outcomes. For anyone coming into contact with NVP, informed choices and respect for the risks separate trouble from progress.
How should N-Vinylpyrrolidone be stored?
Why N-Vinylpyrrolidone Demands Attention
N-Vinylpyrrolidone, known among chemists and manufacturers as NVP, steps out as both useful and tricky. Many polymer makers rely on it for adhesives, coatings, and personal care products. I remember the first time I had to handle this thin, colorless liquid in a lab; the fragrance alone gave off a warning. As demand for specialty chemicals grows, storage questions come up quickly. Mishandling a reactive chemical never ends well.
What Makes NVP Tricky to Store?
NVP reacts fast—heat and light speed up that process. It takes only a little exposure for this monomer to start turning solid or even pressure to build up from unwanted reactions. A storage cabinet that sits in direct sunlight or near a steam vent can turn risky. I once watched a colleague open a bottle left out in summer, and the hiss made everyone freeze.
NVP can irritate skin or eyes, and longer-term exposure brings risks for those breathing the vapors. Regulations from groups like OSHA and the EPA recognize the hazards. It’s about more than broken jars and sticky messes.
Stability Starts With Condition Control
Cool, consistent temperatures keep NVP at its best. Most industry spaces set chemical storage between 2°C and 8°C. Temperature swings create condensation and speed up unwanted reactions. Planning storage away from heat sources protects the material and the people nearby.
Sunlight poses another big risk. UV rays encourage polymerization of the monomer, and nobody enjoys scraping solidified residue out of a drum. Storage best happens in dark rooms or inside opaque, tightly sealed containers. Once, our team moved an entire pallet of NVP to a newly built cold room—result: no more rejected batches due to unexpected thickening.
Packaging Choices Speak Volumes
Most NVP arrives in steel drums or glass bottles with tight seals. In my experience, the choice of package matters as much as temperature control. Strong, airtight lids keep moisture and air out; oxygen triggers polymerization, leaving behind a crusty, useless goop and sometimes dangerous gas build-up. Drums and bottles with strong liners and chemical-resistant seals block accidental leaks.
Some manufacturers will add a stabilizer to NVP before shipping it. This smart step can delay reactions but should not be the only safeguard. Checking labels and batch certificates gives peace of mind that proper additives are inside.
Handling Spills and Waste Reduces Bigger Risks
Accidents happen fast in busy facilities. An open drum or spill can vaporize NVP right into the air. The strong odor creates problems for workers, and liquid exposure carries health risks. Making spill kits widely available reduces response time. Disposable gloves, splash goggles, and fume hoods keep everyone safer.
Once material passes its shelf life or turns unstable, prompt disposal makes sense. Licensed chemical waste handlers collect old NVP so it does not sit as a long-term threat in forgotten corners. Lean storage and tight inventory help the bottom line and the environment.
Improving Everyday Storage Practices
Using color-coded labels for NVP and sharing clear handling guidelines keep even new team members aware. Simple checklists for daily inspections help spot swollen drums and sticky residue around lids. Safety culture means everyone knows what to do, not just the chemical engineers.
Real-world experience shows that good habits, solid training, and easy access to protective equipment turn a thorny chemical into just another tool—safe, reliable, and useful. NVP rewards respect with trouble-free storage, but only for those who give it the right conditions.
What are the available packaging options for N-Vinylpyrrolidone?
The Importance of Choosing the Right Packaging
N-Vinylpyrrolidone (NVP) has earned a steady spot in labs and on factory floors for its usefulness in making polymers, coatings, and personal care products. From what I've seen in manufacturing, packaging goes beyond containment. When workers handle chemicals, safety, convenience, and shelf stability all ride on which drum or bottle gets picked.
Common Packaging Formats
Packaging NVP usually starts with the basics: steel drums, plastic drums, and IBC totes (Intermediate Bulk Containers). Details like moisture sensitivity and risk of contamination shape that choice. I’ve watched chemists scrunch up their faces when containers get swapped out for cheaper options, only to deal with caked or degraded product later.
Steel drums stay popular for a reason. The rigid build and resistance to punctures or leaks bring peace of mind, especially in rough shipping or warehouse environments. They handle drops and bumps that would crush a thinner walled container. Most often, companies opt for 200-liter sizes, lined with protective coatings to stop NVP from corroding the metal itself. Factories that process high volumes value these for how they stack without giving way, and for how reliably they stay sealed until the last drop pours out.
Polyethylene and polypropylene drums earn lots of fans too. These plastics resist chemical attack and prevent moisture from seeping into the NVP. This matters a lot: even the tiniest amount of water can break down the monomer, causing polymerization in the drum. Since plastics weigh less, workers move them around more easily. In mid-size operations where handling speed matters, that can tip the balance away from metal drums.
IBC totes fill a gap for larger volume users. Twenty years ago, you didn't spot many chemical processors using IBCs for NVP, but the demand for bigger batch processing changed that. These 1,000-liter cube containers, with integrated valves for easy transfer, reduce the waste and mess that smaller containers are prone to. Logistic managers tell me the standardized shape means less wasted space, so trucks and warehouses get packed efficiently. The plastic shell often gets a metal cage for added safety.
Bottles and Specialized Packaging
Smaller labs and R&D setups usually take delivery in amber glass bottles or HDPE bottles. Glass blocks out light that could trigger unwanted reactions, while HDPE bottles stay robust and resist damage. Ten years ago, I helped a biotech startup that relied on 1-liter brown bottles with screw caps—they stored just enough material for routine tests and minimized exposure risks. Some suppliers also shrink-wrap bottles for added tamper evidence and moisture protection.
Handling the Hazards
NVP isn’t especially toxic, but its monomer nature causes headaches if it gets out. Heat and light can trigger reactions that ruin whole batches before they reach the end user. That extra care flows straight into packaging choices. Whenever I’m in the field, I see best practices like grounded drums during filling, clear hazard labels, and tamper-proof seals that build confidence with regulators and clients alike. Shipment regulations like UN-certified drums or proper secondary containment also play a role, especially with cross-border transport. Experienced buyers regularly check for supplier certifications and packaging integrity before accepting orders.
Improving Packaging Systems
The packaging game keeps moving. As environmental worries push companies away from single-use plastics, I’ve noticed more groups trying out reusable containers and tracking systems. Some suppliers offer returnable drums or totes cleaned and checked between uses, which helps cut disposal fees. Smart sensors that monitor pressure and temperature during transit show promise for detecting leaks before disaster strikes.
Packaging may seem like a minor detail, but it controls cost, safety, and the prospects for product innovation. Succeeding in this industry means looking at packaging as part of the process, not an afterthought.
| Names | |
| Preferred IUPAC name | 1-ethenylpyrrolidin-2-one |
| Other names |
NVP 1-Vinyl-2-pyrrolidone N-Vinyl-2-pyrrolidone Vinylpyrrolidone N-Vinylbutyrolactam |
| Pronunciation | /ɛn-vaɪˌnɪl-pɪˈrɒlɪˌdoʊn/ |
| Identifiers | |
| CAS Number | 88-12-0 |
| Beilstein Reference | 077873 |
| ChEBI | CHEBI:51145 |
| ChEMBL | CHEMBL14570 |
| ChemSpider | 14235 |
| DrugBank | DB03796 |
| ECHA InfoCard | 03c8da2b-79e6-4526-8d71-5caf738f4598 |
| EC Number | 202-509-5 |
| Gmelin Reference | 10487 |
| KEGG | C00630 |
| MeSH | D014613 |
| PubChem CID | 7909 |
| RTECS number | UY5780000 |
| UNII | J219007Y1C |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C6H9NO |
| Molar mass | 111.14 g/mol |
| Appearance | White crystalline powder |
| Odor | slight amine odor |
| Density | 1.03 g/cm³ |
| Solubility in water | Miscible |
| log P | -0.38 |
| Vapor pressure | 0.04 mmHg (20 °C) |
| Acidity (pKa) | 18.9 |
| Basicity (pKb) | 8.9 |
| Magnetic susceptibility (χ) | -7.42×10^-7 cm³/mol |
| Refractive index (nD) | 1.510 |
| Viscosity | 1.74 mPa·s (25 °C) |
| Dipole moment | 4.09 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 252.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -117 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2543 kJ/mol |
| Pharmacology | |
| ATC code | D11AX |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P314, P321, P333+P313, P337+P313, P362+P364, P370+P378, P403+P235, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | > 91°C (196°F) |
| Autoignition temperature | > 315 °C (599 °F; 588 K) |
| Explosive limits | Explosive limits: 1.4–11.1% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 6,710 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 6,720 mg/kg |
| NIOSH | RY3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for N-Vinylpyrrolidone: 10 ppm (TWA) |
| REL (Recommended) | 6 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
2-Pyrrolidone Polyvinylpyrrolidone Pyrrolidone |

