N-Propyl Isobutyrate: Past, Practice, and Perspective
Tracing the Backstory
People in science have always chased ways to combine molecules for some new use, and n-Propyl Isobutyrate stands as a pretty clear example. You see its roots in the historical chase for tailored esters, where chemists tried to punch up scents and flavors for the growing food market. Back in the 20th century, labs started making small esters like this one in earnest for research into solvents and more. What I notice, flipping through old journals, is that as taste for esters spread, all sorts of industries lined up: paints, coatings, even pesticides. Food scientists took interest for flavors, while others eyed its smell for perfumes. Not all chemicals carved out such a broad niche for themselves, and it’s tough to overlook the impact tight regulation had shaping synthetic routes—paramount when buyers and public started to question what, exactly, these molecules were doing after their initial glow-up in a product.
Core Properties and Physical Makeup
You don’t have to be a spectroscopist to pick up a small bottle and clock n-Propyl Isobutyrate’s sharp, fruity scent. Its clear liquid form pours easily, thanks to a boiling point a bit higher than water’s but lower than many heavy-duty solvents. Anyone who’s worked with it recognizes its stubborn resistance to mixing well with water, which comes in handy when separating it out or building formulations that need hydrophobic additives. The molecule serves up a tight balance of volatility and stability, making it manageable in everyday lab life but not so aggressive it chews through equipment. That said, you shouldn’t get too casual—it reacts if pushed, especially in the wrong company—say, strong acids or bases.
Technical Grading, Labels, and Purity
Industry never forgets about purity. For anyone trying to buy or sell this ester in bulk, purity means everything—trace alcohols or acids can turn a nice, clean odor into a chemical mess and tank its use in food or perfumes. Clear labeling matters, too. Regulators expect a breakdown of CAS numbers and origin, along with warnings, batch numbers, and storage advice. Whether you grab a bottle for research or for a flavor company, keeping contaminants down and tracking shelf life isn't just best practice. It’s baked right into modern safety standards.
Building Blocks: Preparation and Chemistry
Ask around in synthetic labs, you hear the same story: classic Fischer esterification remains a go-to method for whipping up n-Propyl Isobutyrate—just combine isobutyric acid and n-propyl alcohol, toss in an acid catalyst, and heat. More modern setups tweak the process for less waste. Enzymatic routes, for instance, sidestep harsh chemicals, appealing to green chemistry fans. Big plants prize efficiency, aiming to squeeze out every last drop, recover unused reagents, and trim energy use wherever possible—not just for cost, but for regulatory edge. Once made, the molecule’s ester bond holds up fairly well unless strong hydrolysis conditions rear up, which breaks it back to its parts. In hands-on work, chemists sometimes use it as a base for new derivatives, playing with the alcohol or acid side to dial in fresh scents or chemical behavior.
Names in the Wild
Scientific names take center stage in journals, but anyone who has spent time reading labels or lists of flavors bumps into a handful of aliases: Propyl 2-methylpropanoate, Isobutyric acid n-propyl ester… each meant for a different crowd—regulators, food scientists, or folks mixing fragrances. These synonyms seem like bureaucracy, but they serve a real purpose. Without precise names, mistakes happen—wrong chemicals in the wrong drum, and safety takes a back seat.
Running a Safe Shop
OSHA and their global siblings do not let chemical safety slide. Handling n-Propyl Isobutyrate calls for gloves, goggles, and good exhaustion, because inhaling fumes isn’t exactly part of anyone’s career plan. Storage away from flames is obvious if you’ve ever seen the fire risks on an MSDS. Some older plants learned the hard way about leaks thanks to lax standards; it’s usually after an incident that companies overhaul their training and inventory management. These days, regular audits and clear labeling fend off small errors that spiral into big ones, especially with chemicals moving between multiple stages of production or across borders.
Where It Pulls Its Weight
In practice, n-Propyl Isobutyrate shows up where people want a mild scent, decent volatility, or a middle-of-the-road solvent—not too aggressive, but not a slouch either. I’ve seen coatings engineers grab it to thin paints and produce a smooth finish. Flavorists use it by the drop to deliver a rounded, buttery note, but it carries regulatory baggage—food-grade quality or bust, and limits exist for use even in the most forgiving markets. Perfume blenders count on its uplift for fruity top notes, and industrial chemists find it fits in blends that tame more pungent solvents. Niche uses keep popping up—extraction, even as part of pesticide mixes—because its balance of properties lines up just right.
Where Curiosity Takes Us Next
Bright minds in R&D do not see this chemical as stagnant. Green chemistry holds real promise for tweaking its synthesis with smart catalysts or bio-based raw materials. I’ve followed some work on using less energy or engineered enzymes that churn it out with fewer by-products, all while keeping purity up. Automation and process controls have tightened, letting plants push for higher yields and consistency. There’s talk of derivative esters for customized fragrances or solvents that can handle new industrial challenges, especially as smaller startups want chemicals with a “cleaner” footprint.
Are There Risks We Overlook?
Long-time staff in flavor plants and regulators have looked hard at toxicity, because consumers push back at the whiff of risk. Acute exposure data shows it can irritate skin and eyes, and at higher doses, inhaling the vapor feels rough. Luckily, studies so far haven’t flagged it as a major chronic hazard if handled right, but cumulative exposure in tight quarters keeps occupational health departments on their toes. Regulators insist on strict workplace air monitoring, spill control drills, and solid waste management. Wastewater treatment matters too, since downstream users want to avoid lingering esters fouling water supplies or aquatic life.
Looking Forward
As industries move toward renewable chemistry and tighter safety, expect n-Propyl Isobutyrate’s life story to shift away from bulk solvents toward value-added roles where scent and purity command a premium. That’s a shift anyone in the field can spot: more tracing of origin, stricter checks, and specialty production lines tailored for food, fragrances, and high-spec coatings. Regulatory surprises—whether new food safety data or environmental laws—will shape what’s next, and the labs that thrive will be those ready to pivot to new prep methods and rock-solid documentation. Everything points toward a world where even a “simple” ester gets scrutinized, improved, and put to work in smarter, safer ways.
What is N-Propyl Isobutyrate used for?
Where Chemistry Meets Real-World Solutions
N-Propyl Isobutyrate stands as more than just a mouthful for chemistry students. This compound plays a part in several places you might not expect. As an organic ester, it features a pleasant, fruity scent, which makes it a favorite in scents and flavors. Perfume makers often chase that “just right” tone for a fragrance, and N-Propyl Isobutyrate adds a lively, subtle sweetness to many blends. Companies use it behind the scenes, mixing it into perfumes, air fresheners, and flavored foods that wind up in kitchens and restaurants everywhere.
The Invisible Ingredient in Everyday Flavors
Chewing gum, baked goods, and candies often owe their fruity aroma and taste to ingredients like this one. From my years in food manufacturing, dealing with natural and artificial flavors, it struck me how certain chemicals like N-Propyl Isobutyrate give consistency and reliability. Natural fruit extracts can swing widely in quality due to weather or crop disease. Food scientists turn to compounds like this to ensure the same strawberry note lands every batch, no matter what. This consistency in taste and aroma often separates a good product from one that sits on shelves gathering dust.
Supporting Manufacturing Without Taking the Spotlight
N-Propyl Isobutyrate doesn’t draw much attention among solvents, but it holds importance. Industrial paints and coatings, for example, rely on solvents that evaporate at steady rates and don’t leave sticky residues. Paints sprayed onto cars, bikes, or appliance panels benefit from clean, reliable evaporation. In labs where I’ve seen coatings tested and adjusted, chemicals like this one offer a safe bet for both performance and stability—especially compared to harsher solvents that can release toxic vapors or break down sensitive resins.
Safety and Environmental Factors
The conversation around solvents and flavor chemicals always circles back to safety. Companies must keep up with research from government agencies and university labs. N-Propyl Isobutyrate, though considered low-risk, still demands proper handling. The European Food Safety Authority and US Food and Drug Administration both pay close attention to how much of these esters turn up in foods and work environments. My experience with workplace safety training highlighted how quickly inhalation risks can arise, especially in tight spaces or poorly ventilated rooms. Proper labeling, ventilation, and employee training help keep health incidents out of the news.
What Could Make It Better?
One major challenge involves waste. Though the chemical doesn’t build up in the body, spills and improper disposal can lead to environmental headaches. Industry insiders are trying to move toward greener alternatives or recycling methods that capture and repurpose spent solvents. Some firms now explore closed-loop manufacturing, where they reuse evaporated solvents instead of dumping them. Switching to biodegradable solvents might someday push N-Propyl Isobutyrate out of some factories, while others stick to what works due to familiarity and cost.
In my work with fragrance developers, most want transparency with their customers. People increasingly turn bottles and cans around, reading every label. Responsible use and honest labeling let buyers know what’s inside, helping build trust and allow for informed choices. Better science, more careful handling, and open communication go a long way toward making chemicals like N-Propyl Isobutyrate part of a safer, more predictable world.
What is the chemical formula of N-Propyl Isobutyrate?
Getting Down to the Chemistry
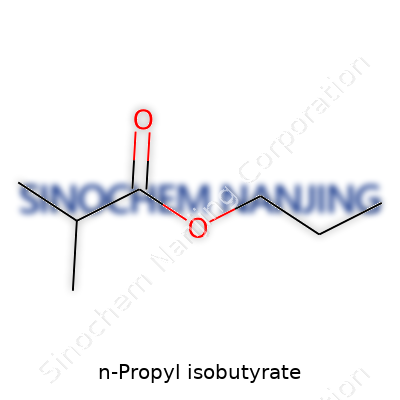
N-Propyl isobutyrate sits in a category of compounds known as esters. A lot of people might skim past chemical formulas, but knowing the molecular structure can make the difference between safe handling and a dangerous mistake in a lab or factory. N-Propyl isobutyrate carries the formula C7H14O2. This structure doesn't just look pretty on paper – it determines how the compound smells (fruity, you’ll notice it in flavorings and fragrances) and how it reacts with other chemicals.
Behind the Formula
If you’ve tinkered with organic chemistry, you’ll see this formula comes from connecting two building blocks: n-propyl alcohol (C3H7OH) and isobutyric acid (C4H8O2). In the presence of an acid catalyst, these molecules join, shedding a water molecule, and you get that signature ester linkage. Every atom has a role here: the seven carbons, fourteen hydrogens, and two oxygens fit together in a definite order. Miss a carbon or swap the alcohol and acid, and you get a different compound altogether.
Why Knowing the Formula Counts
Every year, chemical mishaps happen because someone made assumptions or mixed up compounds that look similar. A wrong formula can mean using something too volatile for a flavoring or too toxic for a safe environment. Trust grows from getting details right, and people who work with chemicals need to keep formulas top of mind. I’ve met lab techs who memorize formulas out of habit, not because it’s just the rule, but because safety and accuracy live in these tiny details.
Applications Shaped by What’s Inside
The fruity scent of n-propyl isobutyrate comes from its molecular shape. Food companies, for example, don’t just choose an ester because it’s sweet-smelling; they ask if it’s safe, stable, and blends with other ingredients. Chemists designing new flavors want to make sure that what they use breaks down cleanly in the body or environment. Formula feeds into every part of that assessment—regulations, environmental impact, and whether a manufacturer can synthesize it reliably at scale. Mistaking n-propyl isobutyrate for another ester would spell trouble in recipes where trace differences change taste or toxicology.
Solutions: Consistency, Education, and Oversight
Drawing from industry practice, clear labeling, regular education, and up-to-date reference materials play big roles in maintaining safety and quality. People benefit when companies invest in ongoing training and when schools build chemistry comfort early on. Even the best protocols break down if people cut corners or rely on memory alone. In my time spent in a university teaching lab, reusable charts and quick-reference printouts next to every workstation made a bigger difference than any troubleshooting manual. Digital databases packed with chemical profiles give researchers what they need fast, but these only help if people know how to read them and double-check entries.
The Bottom Line: Every Detail Matters
It’s easy to take chemical formulas for granted, but that little string of letters and numbers—C7H14O2 for n-propyl isobutyrate—carries real-world consequences. Safe labs, clean manufacturing, and reliable products all depend on getting things down to the atom. That’s where trust, quality, and peace of mind begin and expand out into daily life.
Is N-Propyl Isobutyrate safe to handle?
Getting Familiar With the Chemical
N-Propyl Isobutyrate pops up in more places than you might expect. I’ve crossed paths with this clear liquid while touring a flavor and fragrance lab, and it’s a key ingredient for crafting fruity aromas and flavors. It also lifts off as a solvent in labs. Most people have never heard of it, yet businesses rely on it for the subtle jobs that blend products together or tweak a final scent.
What Happens During Contact?
I’ve seen technicians handle N-Propyl Isobutyrate during routine lab tasks. Most won’t feel a thing with a slight whiff or a drop on the skin. Still, it doesn’t mean you can toss caution away. The Material Safety Data Sheet (MSDS) rates it as an irritant. It leaves skin itching or eyes watering if you get careless. Breathing in too much vapor in a cramped lab, for instance, leads to dizziness or throat irritation. The evidence points to manageable risks if people follow basic chemical safety habits.
Putting Risks in Perspective
Dozens of common chemicals look harmless—until someone skips the gloves or ventilation. Most dangers from N-Propyl Isobutyrate won’t jump out during a short encounter. The larger risk happens in spots where ventilation fails or spills linger on hands. Every MSDS highlights these pinch points: fumes build up, or someone rubs their eye by mistake. The CDC and European Chemicals Agency both list N-Propyl Isobutyrate as a low-to-moderate health hazard. It’s not a prime suspect for cancer, birth defects, or organ damage, so it sits well below strong industrial irritants like formaldehyde or acetone.
Best Practices Make the Difference
All the safety talk means little if the right habits don’t settle in. A friend once learned this lesson after a careless splash in a makeshift lab. No gloves, no goggles. His skin broke out in a mild rash—not severe, but enough to stick in his memory. The truth is, wearing gloves and working under good ventilation changes the outcome completely. A fume hood or simple open window drops vapor levels fast. Protective eyewear and gloves cost pennies but save a lot more in long-term health.
Handling at Home and Work
Most households don’t buy pure N-Propyl Isobutyrate. Still, people encounter it in hobby kits or during flavor-making. At my local makerspace, workshops push glove use and clean-up as non-negotiable. At commercial sites, workers need clear labels, regular ventilation checks, and skin protection. Simple spill kits with absorbent pads beat panic every time. Washing hands before eating or drinking stops those accidental exposures we all regret.
Safer Ways Forward
Accidents drop when people treat chemicals with a balanced mix of respect and common sense. Labels in plain English, upfront training, and safety culture matter. Companies see fewer injuries where they make chemical handling a habit rather than a chore. Every batch comes with a responsibility—the next user or worker deserves a clean, labeled container and a safe shelf. Step by step, the workplace and the home get safer because people take small risks seriously. That attitude makes all the difference with N-Propyl Isobutyrate and every other bottle on the shelf.
What are the physical properties of N-Propyl Isobutyrate?
What Makes N-Propyl Isobutyrate Stand Out
People don’t pay much attention to the clear liquids hiding in the background of chemistry labs and factories. N-Propyl Isobutyrate turns heads among chemical workers and researchers, despite its quiet presence. The stuff looks like a lot of other esters—colorless, runny, with a scent that hints at sweet apples and pears. Sometimes, a whiff makes you think you’ve already sniffed it in a bakery or a candy shop.
Viscosity and Texture: More Than Just a Liquid
This ester moves fast and slick compared to water. It pours out of a bottle nearly as free as rubbing alcohol. On a hot day, it doesn’t put up much resistance when stirred. In factories, light viscosity matters for keeping pumps and pipes clear and smooth because thicker stuff tends to clog or slow down processing. A pour that stays thin at room temperature helps keep everything moving, whether it’s being blended for a flavor or a fragrance.
Boiling Point and Evaporation: Risks and Uses
Heat pulls N-Propyl Isobutyrate into vapor at around 120°C. That’s quite a bit below where water bubbles and roils, so it’s not surprising you’ll sense its aroma long before you notice any steam. This trait matters for two reasons: safety and performance. Too much heat, and it wants out of a container. Even on a warm day, letting the lid off means more of it escapes quickly. Folks dealing with flammable chemicals always need to take boiling points seriously—chemicals with low boiling points tend to mean higher vapor risks.
Density and Solubility: Where It Goes, How It Mixes
A little heavier than water, N-Propyl Isobutyrate doesn't try to hide at the surface or rush to the bottom. Still, put a drop in water, and it steers clear; it won’t mix much. Pour it into a beaker with ethanol or other organic solvents, and it disappears in a snap. So, people in perfumery, flavors, or specialty coatings love this property. It picks its partners, fitting in better with alcohols and oils than with water. Anyone who’s mixed salad dressing gets the gist. Chemical folks work with this tendency when making flavorings or finding the right solvent in a formulation.
Handling and Safety: Respecting Its Limits
Anyone dealing with organic esters, especially ones that smell sweet and look safe, benefits from remembering they don’t play by kitchen rules. N-Propyl Isobutyrate stays flammable—vapors can catch with a flicker. It doesn’t readily burn skin, but it will stir up eyes and lungs if splashes or vapors wander too close. Good ventilation and protected storage spaces aren’t bargaining chips here—they’re necessary routines. Containers need tight caps; worktables should be set near chemical fume hoods.
Potential for Future Innovation
As flavor houses and fragrance creators look to build new blends with fewer allergens and more nuanced scents, chemicals like this ester often become central players. Researchers explore its interactions with sugar alcohols and proteins to make additives more stable and long-lasting. Environmental and workplace safety drive innovation, too—tighter regulations on solvents keep chemists hunting for options that work harder without driving up hazards.
Turning Knowledge Into Practice
In the hands of a chemist or production manager, data about N-Propyl Isobutyrate blends into recipes, safety plans, and troubleshooting notes. Success depends less on how strange the chemical looks on a sheet and more on how its physical traits shape daily routines, product quality, and safe handling. Knowing how these traits line up in the real world helps everyone downstream—from mixing a new flavor to storing a shipment in a warehouse—with clearer expectations and fewer surprises.
How should N-Propyl Isobutyrate be stored?
Understanding the Risks
N-Propyl Isobutyrate carries flammable qualities that nobody should brush off lightly. This kind of solvent gives off vapors that catch fire easily, and if left open or exposed to a spark, things can go downhill fast. Over time, working in labs and handling solvents, I've seen close calls that make a lasting impression. Fire safety posters can’t replace actual vigilance or sound habits. So, talk of storage isn’t just technical—lives and property get put at risk with slipshod handling.
Temperature and Ventilation Matter
From my own experience, keeping N-Propyl Isobutyrate somewhere cool and shaded stands as the simplest safety choice. Sun-warmed corners or spots near heat sources only make vapors rush out faster, raising pressure inside containers. Set the solvent too close to an oven or furnace, and you might hear the hiss of escaping vapor—bad news for indoor air and those who share the space.
Labs do best by stashing flammable liquids in secure, well-ventilated flammables cabinets. Metal cabinets with self-closing doors and spill-proof sills catch rare leaks and make accidental fires much less likely. Good ventilation draws out fumes and stops them from pooling indoors. This isn’t just protocol—it's about helping people and property last through years of daily use.
Choosing Durable Containers
Sealing solvents in airtight vessels may sound obvious, but screw caps or threaded closures sometimes take shortcuts. Metal cans lined with protective coatings or thick-walled glass flasks keep things stable. Plastic bottles work sometimes, though not if the plastic reacts or slowly dissolves. Good labeling counts for a lot—a container with a worn-off label invites confusion, and that’s no small problem if someone grabs the wrong bottle in a rush. Every chemist who’s ever had to rush out a building during a spill drill takes labeling seriously ever after.
Keeping Flammable Solvents Away from Incompatibles
Countless safety manuals hammer one point home: separate storage for incompatible chemicals. Mixing oxidizers and flammable solvents doesn't just risk contamination, it can set off fires or explosions. Bleach, acids, and peroxides belong nowhere near N-Propyl Isobutyrate. Ensuring that shelves stay organized and sorted by hazard class goes a long way. I’ve found storing flammable liquids below eye level, away from direct sunlight and electrical panels, tends to cut down near-misses. Labels should stay easy to read, even during a power outage or in dim light.
Fire Suppression and Emergency Measures
Solvent storage never ends with stacking cans and walking away. Keeping fire extinguishers rated for chemical fires close by avoids last-minute panic. Anyone working near stored solvents must know emergency exits, and keeping spill kits within easy reach just makes sense after a few minor leaks have gotten mopped up. Regular training pays off more than reading a manual ever could.
Steps Toward Safer Storage
Many workplaces benefit from regular audits—a second set of eyes can spot missing labels or rusty containers. Routine shelf cleaning keeps piles from building up, because grime and leaks hide dangerous trends. Updating storage to meet current regulations stands as one step companies can take without delay. These rules aren’t just red tape; they shape better habits. By giving storage practices a fresh look and investing in sturdy equipment, workplaces send a clear signal: safety comes first, and everyone heading home at the end of the day is the real win.
| Names | |
| Preferred IUPAC name | propyl 2-methylpropanoate |
| Other names |
Isobutyric acid n-propyl ester n-Propyl 2-methylpropanoate Propyl isobutyrate |
| Pronunciation | /ɛnˈproʊpɪl aɪsəˈbjuːtɪreɪt/ |
| Identifiers | |
| CAS Number | 540-42-1 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol:N#C(C)CCOC(=O)C(C)C")` |
| Beilstein Reference | 1738603 |
| ChEBI | CHEBI:88918 |
| ChEMBL | CHEMBL445881 |
| ChemSpider | 14185 |
| DrugBank | DB14057 |
| ECHA InfoCard | 100.055.968 |
| EC Number | 211-392-1 |
| Gmelin Reference | 211600 |
| KEGG | C19681 |
| MeSH | D019386 |
| PubChem CID | 79469 |
| RTECS number | UF5950000 |
| UNII | IK3553DQ5M |
| UN number | UN2679 |
| CompTox Dashboard (EPA) | DTXSIDX705251 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 116.16 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.853 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.5 mmHg (20°C) |
| Acidity (pKa) | 11.6 |
| Basicity (pKb) | pKb: 15.15 |
| Magnetic susceptibility (χ) | -7.93 × 10⁻⁷ |
| Refractive index (nD) | 1.4010 |
| Viscosity | 0.712 mPa·s (at 25 °C) |
| Dipole moment | 2.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 286.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –464.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3450.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-2-0 |
| Flash point | 71°C |
| Autoignition temperature | 375 °C |
| Explosive limits | Explosive limits: 0.9–6.5% |
| Lethal dose or concentration | Lethal dose or concentration (LD50, Oral, Rat): 8700 mg/kg |
| LD50 (median dose) | LD50 (median dose) of N-Propyl Isobutyrate: 6 g/kg (oral, rat) |
| NIOSH | WA2275000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for N-Propyl Isobutyrate: Not established |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Propyl butyrate Isobutyric acid N-Propyl acetate N-Propyl isobutyramide Isobutyl propionate |

