N,N-Dimethylformamide: An Editorial Look at Its Story, Uses, and Where We Go Next
Historical Development and Product Overview
N,N-Dimethylformamide made its entrance into the industrial scene as industries began looking for powerful yet versatile solvents that could handle demanding reactions and formulation challenges. Chemists—stuck with less capable solvents—quickly adopted it for its strong dissolving power. Over the years, as research in organic synthesis, pharmaceuticals, textiles, and electronics ramped up, this compound found its way into laboratories and factories everywhere. Its value in the chemical world really comes through in how it serves as a lifeline for catalytic reactions and high-precision polymer work. Rather than just sitting quietly in specialized labs, DMF established itself as a workhorse across many sectors because of its reliable behavior and mix-friendly nature.
Physical & Chemical Properties
DMF comes as a clear, colorless liquid that carries a faint smell, sometimes likened to fish or ammonia. This solvent stands out for its high polarity, miscibility with water and most organic liquids, and its relatively high boiling point for a solvent in its class. This makes it a flexible option for reactions where both aqueous and organic compounds need to come together and for processes that involve complex separation or extraction steps. Its relatively low volatility means less evaporative loss at regular working temperatures, a feature many lab workers and plant operators quietly appreciate.
Technical Specifications & Labeling
Accurate and detailed technical data guide everyday use of DMF in research and industry. Specifications look at purity, moisture content, acidity, basicity, and sometimes heavy metal traces, depending on end use. Recognition of trade names like DMF, dimethylformamide, and others comes from years of shared experience among scientists and procurement teams. Good labeling prevents mix-ups and keeps storage conflicts to a minimum, which matters when handling this solvent alongside others that may sound or look similar but behave quite differently.
Preparation Method
Commercial DMF production relies on the reaction between dimethylamine and carbon monoxide in the presence of a catalyst. Once this method became widespread, cost came down and availability shot up. Before this, making DMF in smaller batches—via routes involving methyl formate or other intermediates—required tougher chemistry and larger inputs of both time and energy. The technological leap to large-scale, more efficient catalytic processes shaped DMF’s journey from specialty solvent to chemical mainstay.
Chemical Reactions & Modifications
Chemists reach for DMF not only for its solvent abilities but because it takes part in reactions as a reagent. It can help in Vilsmeier–Haack and other formylation reactions, where formyl groups get introduced into aromatic systems—a crucial step in building pharmaceutical intermediates and dyes. The polar aprotic nature means it enables powerful nucleophiles to act without getting bogged down by hydrogen bonding, essential for many carbon-carbon bond-forming reactions. In electrochemical studies, DMF’s stable environment gives experimenters reliable outcomes and fine control over product profiles.
Synonyms & Product Names
Across catalogs, scientists find DMF also labeled as N,N-dimethylmethanamide or dimethylformamide, snapshotting how chemical naming often morphs with country or context. Industry professionals rely on this shorthand, tucked in with official chemical abstracts numbers, to order or track materials. This shared lingo between the bench and the warehouse isn’t just a quirk—it helps prevent mistakes and keeps operations running smoothly.
Safety & Operational Standards
Using DMF requires respect for its health risks. Unlike solvents that can be shrugged off with simple gloves or quick ventilation, DMF poses chronic hazards that show up as liver issues, reproductive toxicity, and skin absorption risks. Workers handling DMF need personal protective equipment, careful handling procedures, and training. Industry-wide, stricter storage and disposal rules work hand-in-hand with ongoing medical surveillance for those with regular exposure. Adoption of closed transfer systems, better ventilation, and automating mixing steps help limit risk without giving up DMF’s technical benefits.
Application Area
DMF’s applications spread far and wide. In pharmaceuticals, it shines in peptide synthesis, drug development, and as a universal solvent for active ingredient characterization. Textile workers use it for spinning acrylic fibers, where nothing else matches its performance. In the world of synthetic and specialty chemistry, it dissolves polyacrylonitrile and other polymers, offers stability for production, and often cuts the cost of industrial processing. Electronics manufacturing leans on DMF in circuit board cleaning and as a carrier in certain formulations, appreciating its mixability without leaving residue.
Research & Development
DMF sits at the center of experimental chemistry, both as a testbed and as a launching pad for process improvements. Research continues to seek greener solvents, but DMF keeps its seat due to predictable properties and new findings in catalyst recycling, process intensification, and waste minimization. Every time a new reaction type appears, DMF usually gets a trial run among the first solvents, simply because of the trust built up in research circles. Researchers devote work to limiting leftover DMF in products, reusing solvent waste, and lowering energy draw for recovery—the kinds of behind-the-scenes improvements that keep the chemical industry moving forward without broad headlines.
Toxicity Research
Ongoing research into DMF’s toxicity never lets up, thanks to its tendency to irritate skin, eyes, and lungs and its potential for more serious health effects after long-term exposure. Studies link DMF exposure to liver damage and reproductive health concerns, prompting agencies around the world to spotlight its monitoring and exposure limits. Academic and industrial labs continue to investigate mechanisms of action—how DMF gets broken down in the body, the risks from small repeated doses, and the safest ways to handle accidental spills or exposure. Lab managers and safety officers rely on published research when updating protocols or responding to regulatory changes, with a watchful eye for any signs that safer substitutes can match DMF’s performance.
Future Prospects
The future for DMF doesn’t cut out as cleanly as those who dream of all-green chemistry might hope. Its established value guarantees a continuing role in niche and large-scale industries, even while researchers look for bio-based or less toxic alternatives. Changes in environmental policy, worker safety standards, and product composition rules can shift DMF’s availability or drive new investments into substitute development. If new solvents arise that finally rival DMF’s mix of power and flexibility—without the baggage—the market will turn, but right now DMF seems likely to hold its place based on real-world performance and the high switching costs tied to changing out such a foundational material. For companies and research groups alike, this means pressing ahead with ongoing risk assessments, watching global regulatory trends, and investing both in training and substitution research, all while acknowledging that few chemicals have made their mark on modern industry as deeply as N,N-Dimethylformamide.
What is N,N-Dimethylformamide used for?
Why N,N-Dimethylformamide Matters
N,N-Dimethylformamide (often shortened to DMF) plays a big role in manufacturing. You’ll rarely see it outside factories, but the world feels its impact every day. This clear, colorless liquid keeps industries moving, helping create products from medicines to clothing. Its value doesn’t come from a fancy brand name or high-profile ad campaign. The real power of DMF comes from the way it mixes with water and organic substances. This makes it essential in areas where regular solvents fall short.
Pharmaceuticals and Better Health
DMF acts as both a solvent and a reagent in the pharmaceutical world. Making certain drugs demands precise reactions that don’t work unless substances mix uniformly. The people who create lifesaving medications depend on DMF to make sure active ingredients dissolve properly. Take antibiotics, for example. Their synthesis often needs a stable, polar environment that DMF provides. Without solvents like this, many modern treatments wouldn’t reach the pharmacy shelf.
Polymer and Fiber Production
If you look at running shoes, sportswear, or even surgical gowns, there’s a good chance chemicals like DMF played a part somewhere in the supply chain. Spandex and synthetic leathers rely on DMF for spinning and forming. This chemical helps polymers stay dissolved long enough for manufacturers to produce robust, flexible threads. DMF turns unwieldy raw materials into usable fibers, giving us high-stretch fabrics and lightweight clothing.
Clean Batteries and Electronics
Smartphones and electric cars depend on batteries designed for performance and safety. DMF steps in here as well. Battery production processes need solvents that can dissolve materials for smooth, uniform coatings. In lithium-ion batteries, DMF lets engineers form safer, more efficient electrodes. Wiring insulation and flexible displays also draw on its chemical properties.
The Price of Progress: Health and Environment
The same qualities that make DMF valuable mean we must pay attention to the risks. Breathing in DMF vapors or getting it on skin can cause health problems like nausea, liver damage, or headaches for workers. Environmental agencies also raise concern about DMF in water systems or soil. It doesn’t break down quickly, so it can travel far from the original factory if not managed carefully.
Rules and regulations make a difference here. Countries with strict chemical handling laws tend to record fewer incidents of harm. Factory operators can install closed systems, provide proper gear, and ensure regular air quality checks. The industry also looks for safer alternatives. Some chemists experiment with “green” solvents, hoping to keep the benefits while cutting down the harm.
The Road Ahead
Tough questions about safety and sustainability push everyone to think harder about their tools and habits. From my own perspective, transparency from producers matters. When manufacturers share detailed information about DMF handling and disposal, everyone downstream—workers, communities, and even customers—benefits. Encouraging penny-pinching by neglecting standards isn’t the answer. Respecting both innovation and responsibility promises better health, safer workplaces, and less pollution—without giving up on the materials that shape the modern world.
Is N,N-Dimethylformamide hazardous to health?
Understanding the Risks of N,N-Dimethylformamide
N,N-Dimethylformamide — often called DMF in labs and factories — isn’t something average folks run into during a normal day. But for people in manufacturing, research, or chemical processing, it’s as common as coffee. This colorless, nearly odorless liquid pops up in producing plastics, pharmaceuticals, and plenty of high-tech materials. It dissolves a bunch of things regular water can’t handle, so chemists like its muscle. But the convenience brings plenty of risk along for the ride.
Where the Danger Lies
Breathing in DMF vapors, absorbing it through the skin, or even splashing a little on a hand can lead to short-term symptoms like skin irritation, headaches, and nausea. Spend time with this chemical without proper safety steps, and the story takes a darker turn. Studies show people who work around DMF every day run a bigger risk of liver toxicity — a silent problem that can develop over months or years. OSHA and similar agencies point out long-term effects can creep up, especially without solid monitoring and workplace controls.
Liver issues aren't the only concern on the table. There’s research linking DMF with reproductive complications in animals, and public health agencies aren’t shy about classifying it as “possibly cancer-causing.” No one wants to gamble their future for a shortcut, so pushing these facts under the rug just doesn’t cut it. Even one spill can lead to a bigger headache due to how quickly DMF soaks through latex and some plastics. A thin glove isn’t much help, and that sharp chemical odor isn’t a reliable warning either.
Regulations and Real-Life Experience
Decades ago, compliance felt like an annoying checklist at the end of a shift. Now, after watching a coworker battle mysterious fatigue and skin rashes, DMF’s danger feels personal. The Environmental Protection Agency, the European Chemicals Agency, and other watchdogs set limits on how much DMF can hang in the air at work. OSHA’s ceiling for DMF is just 10 parts per million for skin exposure. That slim margin shows just how powerful this stuff can be. Still, numbers don’t carry the same punch as real stories: workers forced into the ER by accidental spills, or labs shut down during high-profile health investigations.
Every industry using DMF faces a balancing act — lean into its productive benefits, or find replacements that don’t hit the body so hard. Green chemistry isn’t science fiction anymore. Some companies already lean toward safer solvents. The cost might jump up at the start, but the long-term health bills for workers and their families weigh a lot heavier. Engineers and safety managers often feel squeezed by deadlines or budgets, yet the conversation has to change: prevention beats reaction every time.
Safer Practices Make the Difference
It makes a real difference when labs and plants trade thin gloves for material rated for DMF, use fresh air hoods instead of trust in an open window, or swap chemical-resistant clothing for old coveralls. Training workers to spot health changes and encouraging early reporting saves lives. Employers who measure DMF levels regularly, keep safety data up front, and listen to experienced workers transform a risky workplace into a safer one — not just on paper, but in real-world health outcomes. It isn’t a perfect world, but every step toward real safety pays back in healthier people and less pain down the line.
What is the chemical formula for N,N-Dimethylformamide?
A Look at N,N-Dimethylformamide
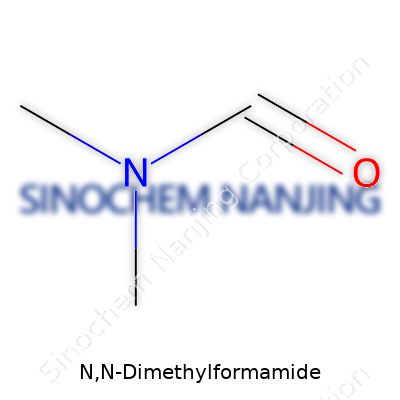
N,N-Dimethylformamide, known in laboratories and industry circles as DMF, carries the chemical formula C3H7NO. The molecule joins a formyl group and dimethylamine, merging a practical solvent with a structure simple enough to draw by hand yet complex enough to handle some heavy jobs. Working with chemistry means learning to see more than just letters and numbers on a label—it’s an introduction to molecular shapes and the way they change what you can accomplish in the lab or on the factory floor.
Why the Formula Matters
Reading a formula like C3H7NO, you get more than a list of elements. I remember my first time using DMF for an organic synthesis in college—my gloves, the fume hood, the nervous respect for handling something with a punchy smell. Every element in that formula, three carbons, seven hydrogens, one nitrogen, and a single oxygen, plays a part in its reactivity and behavior. DMF dissolves both polar and nonpolar compounds. That’s not magic, just chemistry, and the reason DMF ends up in pharmaceuticals, plastics manufacturing, and electronics work.
Chemical formulas aren’t window dressing. Without accurate formulas, mistakes pile up quickly, and even experienced chemists can miscalculate reaction yields, mishandle storage codes, or combine the wrong chemicals. I’ve seen projects dry up from basic mix-ups that a five-second formula check could have prevented. For curious newcomers or anyone working in a lab setting, learning those combinations by heart translates to safer and more reliable experiments.
Risks and Responsibilities
Handling DMF demands attention because C3H7NO’s properties let it pass through skin and enter the bloodstream. I always double up on gloves—a tip passed to me by an older researcher—and never let containers stay open longer than needed. After several years in shared workspaces, I’ve watched too many people treat solvents casually, only to regret it when headaches or worse set in. Choosing to take the chemical’s formula and structure seriously turns routine work into a habit of safety.
Guiding People and Processes
Many in the chemical workforce trust that the right formula saves them wasted effort and protects health. Training sessions at every job I’ve held start with the basics—identify what you’re using, know the formula, recognize the health flags. Institutions and companies who post proper information see fewer accidents, fewer fines, better teamwork.
DMF reminds me that chemistry isn’t just theory: it’s a foundation for industries ranging from clothing to medicine. Its reliable, straightforward formula serves as a checkpoint for anyone running a reaction or ordering supplies. C3H7NO gives concrete information for building strategy, investing in research, and sticking to international safety standards.
Moving Toward Safer Chemistry
Demand for solvents like DMF pushes us to ask hard questions about green alternatives, worker exposure, and responsible sourcing. Keeping the formula in mind, manufacturers and regulators look at ways to improve air quality in factories and substitute safer chemicals without sacrificing process quality. Sharing that knowledge in course handouts, safety data sheets, and chemical labels pushes the field forward, one project at a time.
How should N,N-Dimethylformamide be stored?
Getting DMF Storage Right
N,N-Dimethylformamide, known in most labs as DMF, gets used every day for making polymers, pharmaceuticals, adhesives, and more. People count on its solvent power, but storing DMF the wrong way can easily lead to workplace health issues, product contamination, and even fire risks. After working with organic solvents for years, one lesson sticks out: some chemicals get a lot less scary once you respect their quirks and follow the right procedures.
Keep Moisture and Air Out
DMF pulls in water from the air quickly. Once it absorbs moisture, everything from product quality to your next reaction run can go sideways. Glass bottles with tightly sealed screw caps or high-quality HDPE containers work well. Always keep the container closed when not in use. Save yourself headaches by using an airtight desiccator for long-term storage if you’re unlikely to finish the bottle in a month or two.
A friend who stored DMF in a loosely capped jug learned this the hard way; strange side products and erratic yields forced their lab to toss a costly batch of research chemicals. It’s hard to overstate the impact of humidity on this solvent.
Mind the Heat — and Sparks
DMF doesn’t catch fire as easily as diethyl ether or acetone, but flammable liquid storage rules still matter. Labs are busy places. Storing DMF near heating equipment, sunlight, or steam lines only ups the risk of accident. The flash point sits around 136°F (58°C), so keep it cool. Flammable-liquid-approved storage cabinets make a big difference here. I’ve seen what happens when solvents leak onto hot surfaces; the smell alone lingers for days.
Always ground metal containers and dispensing equipment to prevent static discharge. Dry air and friction can produce enough charge to spark a fire with the vapor above an open DMF vessel.
Label Everything — Then Check Again
People often ignore labeling on solvents. A container marked “DMF” but left without hazard symbols or expiry details can put everyone at risk. Use chemical-resistant markers, add the date, and update storage logs. Regulations require more than a name — symptoms of exposure, target organs, and emergency contacts matter. A misplaced bottle with a mystery substance costs time and can cost safety.
Don’t Store With Reactive Chemicals
Mixing strong bases, oxidizers, or acids into DMF or even storing them together can have unwanted results. In several lab accidents tracked by the Chemical Safety Board, splashed DMF found its way into bins with oxidizers, leading to dangerous situations.
Salts of strong acids like nitric acid and even alkali metals react with DMF, creating potentially explosive byproducts. Keeping an organized chemical inventory and separating incompatible materials stops many emergencies before they start.
Personal Responsibility Matters
Each person handling DMF influences overall lab safety. Training and attention to best practices help everyone breathe easier and work more efficiently. DMF carries risks that only grow if people cut corners. Wearing gloves, using goggles, turning on fume hoods — these steps build a safety routine that incident reports show pays off over time.
DMF serves valuable roles in the world’s chemistry labs and factories, but it’s not the type of solvent that rewards carelessness. Smart storage — cool, dark, dry, sealed, and labeled — keeps the focus on science, not disaster recovery.
References
- Centers for Disease Control and Prevention: N,N-dimethylformamide (DMF) factsheet
- American Chemical Society: Safety Data for N,N-dimethylformamide
- OSHA: Laboratory Safety Guidelines
What are the safety precautions for handling N,N-Dimethylformamide?
Understanding the Risks
N,N-Dimethylformamide, or DMF, turns up in labs and factories as a colorless liquid known for its strong, fishy smell. Plenty of workers have brushed up against it during chemical syntheses, paint removal jobs, and even in pharmaceutical manufacturing. Some people look at it as just another solvent, but anyone who’s worked with it will tell you that ignoring safety leads to real problems. DMF has a knack for slipping through the skin, and breathing in its fumes can irritate lungs. Scientists and workers exposed to DMF without proper protection often report headaches, nausea, and skin inflammation. Research links chronic exposure to liver damage, reproductive issues, and increased cancer risk.
Practical Protective Measures
Putting on gloves may seem like common sense, but with DMF, standard latex offers zero defense. Plenty of us have noticed gloves get sticky or show pinprick holes after a while. Flimsy gloves let the solvent right through to skin. Instead, nitrile, butyl rubber, or even specialized laminate gloves tend to hold up much better. A lab coat and chemical splash goggles protect the rest of the body, and for larger operations, a face shield and chemical-resistant apron can save you serious trouble.
DMF fumes don’t just irritate eyes and lungs—they travel quickly, making it easy for vapors to spread. Local exhaust ventilation, like fume hoods, moves air away from your breathing zone and does more to control exposure than swinging open a couple of windows. Workers in industrial settings often rely on well-tested ventilation systems that are checked by third parties. Relying on air filters, carbon scrubbers, or even simple airflow checks keeps the solvent’s levels in check.
Safe Storage and Handling
DMF loves moisture. If it’s left out, it can mix with water from the air, change its quality, or even damage equipment. Stash DMF containers in a cool, dry spot, far from sunlight and heat sources. Keep the lids tight and mark everything clearly, so nobody grabs the wrong bottle. Even in research spaces, accidental swaps have caused burns or reactions that never should have happened.
Spills and leaks create headaches fast. Folks who work with DMF every day should know where the spill kit sits and how to use absorbent pads or neutralizers. Never mop up with paper towels and toss them in an open trash bin—ragged towels soaked with DMF vapors lead to sudden headaches and fires. Chemical waste goes right into designated, sealed waste bins. Each container needs a durable, legible label.
Saving Your Health for the Long Run
Health surveillance matters more than people admit. Folks who spend time around DMF should report odd symptoms early, not shrug off red patches or persistent coughing. Occupational health teams track exposures and step in with extra blood tests or liver checks if needed. Real-time monitoring and regular workplace checks keep fumes under control and staff healthy.
Training turns rules into habits. Onboarding sessions lay out risks, but brush-up courses make people remember the smell, the burn, and the sting of forgetting a mask. Nobody builds expertise overnight, yet sharing stories of near misses often gets new workers to double check their gloves or swap masks more often.
Building a Culture of Safety
Each person on the team shapes a solid safety culture. Open conversations about mistakes and close calls matter more than extra posters on the wall. An experienced coworker who points out a missed glove or a loose cap on a bottle usually prevents more harm than a shelf full of safety binders.
People who take DMF safety seriously make the workplace safer for everyone. A mix of solid protective gear, proper handling, and honest communication keeps exposure in check and health risks at a distance.
| Names | |
| Preferred IUPAC name | N,N-dimethylmethanamide |
| Other names |
DMF Dimethylformamide N,N-Dimethylmethanamide Formic acid dimethylamide N,N-Formyldimethylamine |
| Pronunciation | /ˌdiːˌemˈaɪˌdiːˈmɛθɪl.fɔːrˈmæm.aɪd/ |
| Identifiers | |
| CAS Number | 68-12-2 |
| Beilstein Reference | 635068 |
| ChEBI | CHEBI:17741 |
| ChEMBL | CHEMBL1357 |
| ChemSpider | 7418 |
| DrugBank | DB00838 |
| ECHA InfoCard | 100.053.243 |
| EC Number | 200-679-5 |
| Gmelin Reference | 60797 |
| KEGG | C01045 |
| MeSH | D003615 |
| PubChem CID | 622 |
| RTECS number | LQ2100000 |
| UNII | 4WK6640AAB |
| UN number | 2265 |
| CompTox Dashboard (EPA) | DTXSID5027296 |
| Properties | |
| Chemical formula | C3H7NO |
| Molar mass | 73.09 g/mol |
| Appearance | Colorless, transparent liquid |
| Odor | Faint amine-like odor |
| Density | 0.944 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.01 |
| Vapor pressure | 2.7 hPa (20 °C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | pKb = 13.23 |
| Magnetic susceptibility (χ) | -9.03×10⁻⁶ |
| Refractive index (nD) | 1.426–1.428 |
| Viscosity | 0.92 mPa·s (at 25 °C) |
| Dipole moment | 3.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 153.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -259.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1802.7 kJ/mol |
| Pharmacology | |
| ATC code | V07ABоу |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H226, H319, H332, H360D |
| Precautionary statements | P280, P261, P271, P304+P340, P308+P311, P405, P501 |
| Flash point | 58°C |
| Autoignition temperature | 445 °C |
| Explosive limits | 3% - 15% |
| Lethal dose or concentration | LD50 oral rat 2800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2800 mg/kg (rat, oral) |
| NIOSH | NIOSH: LQ2100000 |
| PEL (Permissible) | 10 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Formamide N,N-Dimethylaniline Dimethylacetamide N-Methylformamide N,N-Dimethylacetamide |

