N,N-Dimethylaniline: A Closer Look at Its Significance and Responsibilities
Tracing the Path: Historical Development
Interest in N,N-Dimethylaniline started growing in the late 19th century, right around the time the world’s love affair with synthetic dyes and chemicals took off. Chemists hunted for ways to create new colors and stumbled across this compound’s strong electron-donating traits, quickly realizing it could boost the punch of dye production. Over decades, research uncovered applications well beyond its original use, stretching into pharmaceuticals, agrochemicals, and organic synthesis. Watching its story unfold, I see a broader reflection of industrial chemistry’s journey—always driven by society’s shifting needs and technological advances, never content with staying in one lane.
Product Overview: More Than Just a Reagent
People inside the industry recognize N,N-Dimethylaniline most easily from labs and factory floors, but it reaches farther. Its role isn’t just limited to chemical reactions. It’s key in manufacturing certain dyes, especially crystal violet, and as a precursor in drugs and agricultural agents. Each time someone works with it, N,N-Dimethylaniline brings reliable performance, which is something anyone in synthesis can count on when critical reactions depend on consistency. Alongside traditional uses, researchers keep testing its boundaries, making it a constant feature in conversations about process improvements and potential pathways to more sustainable chemistry.
Physical & Chemical Nature: What You See and What You Don’t
Ask anyone who’s handled it: N,N-Dimethylaniline stands out as a clear, oily liquid with a fishy, amine-like odor. It’s made up of aromatic and tertiary amine groups, creating strong nucleophilic behavior and basicity. Solubility works in its favor—lots of organic solvents embrace it, letting users move seamlessly through different operations. Its boiling point sits high enough for most synthetic steps, while the viscosity and reactivity suit controlled reactions. Because of these traits, it slips easily between the roles of intermediate and catalyst, finding its place in versatile processes.
Technical Details & Labeling: Beyond the Basics
In any real-world setting, knowing exactly what you're working with can mean the difference between quality and disaster. Labels for N,N-Dimethylaniline often point out things like purity, water content, and specific impurity thresholds, all of which carry weight in modern regulatory environments. Documentation also includes volatility, storage instructions, and recommended handling guidelines, all driven by experience in both academia and industry. Quality control staff and lab workers double-check certificates of analysis because even small shifts in composition can swing a sensitive process off target. Labeling today mixes legal necessity with common sense, offering transparency for buyers and operators who want a guarantee that they’re not introducing more problems than solutions.
On Making It: Preparation Methods
Crafting N,N-Dimethylaniline usually starts with aniline, fed through methylation reactions, often with formaldehyde and suitable reducing agents. Experienced chemists look for selectivity during methylation. They refine catalyst choices and tinker with conditions to keep side reactions at bay while scaling up production. These days, many facilities focus on greener approaches—tweaking solvents, minimizing waste, and cutting down on unwanted byproducts. This drive doesn’t just echo regulatory pressures but suggests that people in the field recognize the weight of their environmental impact. There’s pride in developing methods that boost yield while shrinking the carbon footprint.
Chemical Reactions and Modifications: Building Blocks and Bridges
N,N-Dimethylaniline acts as more than a simple component—it influences entire reaction pathways thanks to its electron-rich structure. Its amine group pulls nucleophilic reactions into play, such as alkylation or acylation, and can support condensation steps. In dye chemistry, its inherent reactivity pairs well with aromatic substitutions, leading to a parade of colors that revolutionized textiles. The story deepens with its ability to transfer electrons, which empowers photoinitiators in polymer chemistry and materials science. Modification of the molecule itself, whether by introducing substituents or linking to larger frameworks, opens more doors and creates specialty reagents suited to fast-evolving technology demands.
Names in the Wild: Synonyms & Product Names
Walk through a catalog and N,N-Dimethylaniline greets you under multiple names. From dimethylaniline to DMA or even just as an aromatic amine, its aliases tell you about the product’s roots and everyday familiarity. Buyers juggle these names, double-checking CAS numbers to avoid confusion, especially when sourcing from overseas or comparing to archived literature. Synonyms reflect its global journey, a subtle nod to the way chemistry crosses language and cultural boundaries, rarely confined by borders.
Standards and Safety: The Responsibility of Chemistry
Handling N,N-Dimethylaniline demands genuine respect. Its toxicity pushes for strict adherence to safety protocols. Anyone who’s found themselves splashed or inhaled the vapor won’t forget the urgency of protective gear and strong ventilation. Standards for storage, labeling, and disposal build on actual incidents—the sort that shape workplace culture as much as regulatory sheets. Rules don’t exist in a vacuum; they come from shared knowledge about the compound’s hazards, ranging from skin irritation to concerns over carcinogenicity in some animal studies. Facilities with strong training programs and transparent incident reporting shield workers, reinforce emergency planning, and set a tone of responsibility that stretches beyond simple compliance.
Where It Works: Application Areas
In industry, N,N-Dimethylaniline shows up in dye manufacturing, the synthesis of antioxidants, and the world of polymers, often as an accelerant or intermediate. Drug development leans on its presence as a precursor for certain anesthetics and other therapeutic molecules. Agriculture taps into its capacity to build substances aimed at boosting yield and protecting crops. Upstream and downstream, its reach can be seen—in lab-scale organic reactions, its use in photochemical systems, or as a spectral marker for analytical techniques. Each use case reveals a bit about the current state of technology and priorities, whether the goal is higher throughput or more precise molecular design.
Research and the Search for Answers
Chemists in R&D labs rarely sit still. N,N-Dimethylaniline draws attention for its role in understanding mechanisms of electron transfer, acting as both experimental tool and inspiration for theoretical models. Studies probe its photochemical properties, testing ideas for light-activated systems in green energy or advanced materials. Sometimes, researchers create derivatives trying to push reactivity even further, and other times, they focus on real-world issues, such as minimizing impurities or making production more sustainable. These investigations keep knowledge current, offering companies and academics alike a chance to develop products that align with stricter regulations and growing consumer demand for responsibly made goods.
Toxicity Research: Lessons from the Lab
Toxicity stands front and center anytime discussions turn to N,N-Dimethylaniline. Experience from animal studies and industrial monitoring outlines real risks—liver and kidney effects and suspected carcinogenicity. Long-term exposure sometimes links to nervous system irritation, driving workplace exposure limits and precautionary measures. I’ve seen research move toward finding safer alternatives in downstream applications or engineering controls that curb airborne concentrations. Industry and regulators alike push for more refined toxicological models, aiming for data that pinpoints thresholds and helps shape smarter, more protective policies. These investments reflect a broader shift in chemical safety from reactive to proactive, from compliance to genuine improvement.
Looking Forward: Future Prospects
Every chemical comes with a story, and N,N-Dimethylaniline’s next chapters are getting written now. Ongoing efforts to refine preparation techniques go hand in hand with those designed to boost safety. Cleaner synthesis, smarter catalysts, and novel derivatives promise stronger products that weigh less on the environment. Research into its photochemical utility feeds the growing interest in solar energy and next-generation photoactive materials. As green chemistry takes root, the pressure is on to develop alternatives for some of its more hazardous uses, but the foundational research continues fueling innovation. Balancing benefit and risk, science and society move forward with solutions that recognize chemistry’s power and responsibility.
What is N,N-Dimethylaniline used for?
Building Blocks for Dyes
Walk through any fabric shop, and the vast spectrum of colors tells a hidden chemical story. N,N-Dimethylaniline plays a huge role here, serving as a core ingredient for dye production. Spots like the textile and printing industries run on the back of vibrant, reliable colors, often derived from synthetic sources. This chemical kicks off the process for making crystal violet and malachite green, both of which remain industry favorites for vivid, lasting color. Big manufacturers rely on consistent supply to keep up with demand for rich hues and specialized finishes.
Lab Workhorse in Research
Chemical labs put N,N-Dimethylaniline to good use as well. Chemists use it as a starting material to build more complex organic compounds. Its structure offers strong electron-donating properties, making it handy for research in organic chemistry and photochemistry. Scientists have harnessed its ability to transfer electrons in energy conversion and light-activated reactions, taking steps forward in solar cell development and new medicines. Reliable supply matters here, since research can stall without accessible building blocks.
Boosting the Printing Industry
Printing on a commercial scale depends on fast, stable ink formulation. N,N-Dimethylaniline acts as an intermediate ingredient in ink production. Businesses producing packaging, magazines, and commercial prints count on stable, sharp colors and rapid drying. Choosing the right intermediates can help keep inks competitive, pushing print shops to deliver tons of products every year. My own family ran a small print shop, so I have seen how crucial consistent ink quality is for meeting client expectations and keeping the doors open.
Role in Pharmaceuticals and Pesticides
Drug discovery often starts with simple chemicals and transforms them into something more powerful. N,N-Dimethylaniline enables reactions that build up the molecular backbone for certain antihistamines and anesthetics, speeding up the testing of active compounds. Pesticide manufacturers use it as well, helping develop products that protect food crops on a global scale. Both industries watch safety closely; regulations from the US Environmental Protection Agency (EPA) and European agencies keep these chemicals in check, monitoring exposure levels and handling procedures.
Sustainability and Worker Safety
N,N-Dimethylaniline works well in these essential sectors, but it brings challenges. Handling tightly regulated chemicals calls for strong training, solid ventilation, and good protective gear. The National Institute for Occupational Safety and Health (NIOSH) classifies it as a potential health hazard under certain conditions, especially if inhaled or in contact with skin. Bigger manufacturing plants can invest in automated handling and strict protocols, but smaller outfits need clear guidance and access to practical safety equipment.
There’s growing pressure on the industry to switch to greener processes—aiming to create safer, biodegradable alternatives. Companies diving into research for bio-based dyes or less hazardous reagents can help cut risks over time. Large buyers, especially those serving the food and fashion industries, can encourage chemical suppliers to clean up their act by insisting on high safety standards and transparency.
Outlook for the Future
N,N-Dimethylaniline plays a quiet, foundational part in everything from vivid prints to new drug compounds. Most people don’t see its effects directly, but pull away that link and some industries come to a standstill. Innovation on safety and greener chemistry stands out as the way forward, letting us enjoy the benefits without putting workers or the planet at risk.
What are the safety precautions when handling N,N-Dimethylaniline?
Understanding N,N-Dimethylaniline: Not Just Another Lab Chemical
Anyone with a bit of experience in labs or the chemical industry has probably crossed paths with N,N-Dimethylaniline. Its sharp smell usually announces its presence before anything else. It tends to show up in dyes, even in pharmaceuticals, and pops up often in organic syntheses. What matters most is that N,N-Dimethylaniline isn’t just inconvenient; it’s toxic and flammable. So treating it like any ordinary liquid won’t cut it.
Personal Safety Cannot Wait
You reach for gloves before unscrewing that bottle. Not the thin kind—they melt away with organics like this. Nitrile gloves hold up better. A well-fitting lab coat and wraparound goggles, every time. It only took one colleague splashing a drop on their wrist to realize how quickly it stings and how fast things get serious. Respirators come out if you’re not in a fume hood. The airborne vapors cause headaches—sometimes worse—so don’t rely on opening a window or switching on a regular fan.
The Right Controls for a Potent Chemical
N,N-Dimethylaniline evaporates at room temperature and carries its toxicity with those fumes. A proper chemical fume hood turns a headache into a non-event. Working without one—never a good idea. You can’t always smell a hazard—nose fatigue sets in fast. Closed containers with secure seals belong on your bench. Don’t let this liquid sit open “just for a minute.”
Spills happen. Absorbent pads and neutralizing agents should stay close by. I remember seeing someone scramble to clean up a spill with paper towels. The chemical soaked through, and suddenly hands and arms needed a long wash under the safety shower. Paper doesn’t protect skin. A spill kit sized for organics saves a lot of regret.
Label Everything, Store It Right
N,N-Dimethylaniline loves to hide in plain sight. Unmarked bottles cause just as much harm as mishandling. A clear label that includes hazard words keeps everyone in the loop, not just you. Most solvents stack up in a vented flammables cabinet away from acids. Warm rooms speed up the release of vapors, so always store it as far from heat and direct sunlight as possible.
Disposal deserves just as much attention. This substance goes straight into a designated container for hazardous waste. Dumping it down the drain could mean a visit from the environmental safety team, or worse, a contamination violation.
Don’t Work Alone—Emergency Preparedness Counts
Pairing up in the lab makes sense, especially with a chemical like this one. One pair of hands covers for another when something goes sideways. Make sure you know where the eyewash and shower stand long before you would ever need them.
If something splashes, rinsing can’t wait. Skin and eyes need a serious flush with water—no shortcuts, no shortcuts, and no embarrassment in calling for help. Emergency numbers should sit right by the door.
What Makes These Steps Worth It?
Too many times, accidents happen not from ignorance but from skipping steps when things get busy. Shortcuts aren’t just risky; they lead to real harm. The safety precautions around N,N-Dimethylaniline exist because people have paid the price for not following them. Treating the substance with the respect it demands keeps everyone working, learning, and safe—and that's a lesson nobody forgets twice.
What is the chemical structure and formula of N,N-Dimethylaniline?
The Shape and Makeup of N,N-Dimethylaniline
N,N-Dimethylaniline brings together science and creativity. Its chemical structure carries an aromatic benzene ring with a nitrogen atom tucked onto it. Two methyl groups attach to this nitrogen, shaping not just the way the molecule looks but also how it works in the world. Chemists know this compound by a tidy formula: C8H11N. The molecule’s backbone – benzene (C6H6) – plays host to a nitrogen atom, which swaps out one of the ring’s hydrogens. That nitrogen grabs two methyl groups (CH3), and together they form a molecule both simple and full of possibility.
The structure looks like this: a six-carbon benzene ring (hexagonal and flat as a pancake), with one carbon holding a nitrogen, which in turn links to two methyls. Its structure sets the stage for a lot of reactivity that organic chemists rely on every day. I've seen bench chemists draw a quick sketch of this molecule with a smile—its straightforward look hides a lot of utility.
Why It Matters in Everyday Chemistry
This compound gets plenty of attention for good reason. N,N-Dimethylaniline acts as a common starting point for dyes, pesticides, pharmaceuticals, and photographic chemicals. For manufacturers who need aniline derivatives, it serves as a quick solution. In the past, colorful synthetic dyes like malachite green and methylene blue have started their journey from a flask containing dimethylaniline. The molecule also steps into the picture in the production of analgesics and local anesthetics. That’s not just history — its chemistry continues to influence medical manufacturing, laboratories, and art supplies.
Looking at my own time working in a chemical plant, watching drums of N,N-Dimethylaniline stack up before diving into a new batch of dye or chemical intermediate, I understood its value. Ask any production chemist and you’ll hear similar stories – a reliable, easy-to-handle material with known hazards but lots of upside if respected in storage and use.
Challenges in Handling and Safety
No chemical comes without risk. This one can irritate the skin, eyes, and respiratory tract. Studies tie long-term exposure to possible blood effects. Reports point to its toxicity for aquatic life. Stakeholders in manufacturing and environmental safety keep a close eye on solvents and related compounds like dimethylaniline for these reasons. Proper equipment protects workers. Waste needs smart treatment before it reaches waterways. Local governments and companies must set clear, enforceable guidelines for safe use and disposal.
Lately, many operations shift toward greener alternatives to reduce risk for both people and ecosystems. Engineers and safety managers adjust protocols, invest in better handling systems, and teach new workers what mistakes to avoid. I’ve seen poster campaigns in the break room and checklists beside the storage cabinets. While some labs look for substitutes, the needs of color chemistry and pharma keep dimethylaniline in demand.
The Path Forward: Responsible Chemistry
Better chemistry sometimes starts with better habits, not just better molecules. By focusing on education, monitoring, and innovation in how we handle chemical staples like N,N-Dimethylaniline, risks shrink. Safer containers, regular air checks, and continued training matter as much as strict formulas or mathematical models. Real responsibility calls for action from suppliers, regulators, and users. Solutions often start with transparency, and everyone on the floor or behind a desk should know the facts and treat them with respect.
How should N,N-Dimethylaniline be stored?
Understanding the Risks
N,N-Dimethylaniline usually shows up in labs and factories that handle dyes, rubber chemicals, and pharmaceuticals. Some people overlook just how risky it can get if you store it badly. You get leaks. Strong, fishy smells linger. Even worse, it can harm your health and the environment. I’ve spent years around chemical storerooms, and slip-ups show themselves quickly, especially with substances that catch fire or irritate your body like this one. Basic respect for what you’re working with separates a safe site from a disaster.
Temperature, Container, and Air Quality Matter
Many people think storing chemicals just means putting them on a shelf. What counts are the details. N,N-Dimethylaniline should stay cool, preferably below room temperature. Heat speeds up how fast it breaks down or sets off chemical reactions. Nobody wants an unexpected chemical fire or toxic vapor in the warehouse.
No one trusts a worn-out container. In my experience, a sturdy amber glass bottle or a steel drum lined with plastics works best. Glass stops light from speeding up decay; tight, chemical-resistant seals keep vapors in. If you use soft plastics or leaky lids, expect trouble—more so if you stack drums carelessly. Damaged containers leak fast, so regular checks save lives and cash.
Ventilation can’t get skipped. I’ve seen shops stinking for days where owners tried to save money on exhaust fans. Without fresh airflow, fumes build up. Employees get headaches, sore throats, sometimes worse. Running a fan beats scrambling to get someone medical help. Keeping this chemical away from ignition sources—no smoking, no open flame, no work that sparks—is just as important.
Separation and Labeling Keep Operations Smooth
People lose track of drums or let incompatible stuff pile together. N,N-Dimethylaniline reacts with strong acids, oxidizers, and even some plastics. One splash or spill with the wrong mate leads to heat, noxious gas, or even fire. Clear, visible labeling with hazard warnings cuts mistakes. In bigger facilities, I’d never let flammable solvents share the same locked chemical cage if mixing them sets off trouble.
Health Precautions Start with Training
During inspections, I came across plenty of coworkers who didn’t know how to use gloves or goggles. This chemical burns skin and eyes and kicks up fumes that aren’t good for your lungs. Proper gloves, chemical splash goggles, and long sleeves go a long way. Accidents fell once we drilled people on the risks and showed them what gear worked.
Don’t ignore emergency gear. Eyewash stations and spill kits must sit nearby, ready in case lids tip or bottles get cracked. I suggest running drills so nobody freezes when something does go wrong.
How To Make Improvements
I’ve seen companies get ahead by creating a culture that respects chemical hazards. Walk-throughs, regular training, up-to-date safety datasheets, and honest talks where workers point out storage weaknesses help keep everyone sharp. Following OSHA guidelines and local regulations stops fines, lawsuits, or even loss of business.
Every time I’ve watched a team take N,N-Dimethylaniline safety seriously, incidents dropped, and everyone went home without a story to tell. Simple, well-understood steps prevent emergencies and make for a better workplace.
Is N,N-Dimethylaniline hazardous to health or the environment?
Understanding What N,N-Dimethylaniline Can Do
N,N-Dimethylaniline turns up in some unexpected places. Factories use it as a starting point for dyes, pesticides, and even certain medicines. Walk through an industrial site that works with colorants, and there’s a fair chance this chemical is nearby. Just a few years spent reading up on chemical safety shows how quickly something can go from being unknown to raising serious questions. For N,N-Dimethylaniline, the red flags have kept popping up.
Health Hazards Can’t Be Ignored
Anyone who has spent time around chemical manufacturing learns to treat certain compounds with special caution. Reports paint a clear picture for this one: N,N-Dimethylaniline can harm the blood, liver, and central nervous system. Breathing its fumes or letting it touch the skin doesn’t feel like a big deal at first, but symptoms can sneak up—fatigue, headaches, even blue-tinted skin if the exposure builds up.
Studies from agencies like the National Institute for Occupational Safety and Health (NIOSH) point out a cancer risk with long-term or high-level exposures. Workers in dye production lines, for example, run higher risks, and the safety gear seen on the shop floor isn’t just for show.
A personal memory comes to mind: in a university chemistry lab, even a whiff of N,N-Dimethylaniline had students scrambling to turn up the fans and reach for gloves. That early experience stuck with me. A lot of accidents in labs and factories start with underestimating substances that seem routine.
Environmental Issues Deserve Concern
Chemicals don’t stay put where they get used. N,N-Dimethylaniline enters wastewater and air either from manufacturing sites or after improper disposal. Even small leaks add up over time. Research documents how the substance persists longer in the environment than many think. It doesn’t break down quickly in water, and that’s bad news for aquatic plants and animals.
I’ve seen neighborhoods near chemical plants push back after noticing strange smells in the water or dead fish turning up downstream. Community health depends on good stewardship. The fact that this chemical lingers in sediments and can move through groundwater shows that a hands-off approach isn’t good enough.
Addressing the Hazards Means Taking Action
Stronger workplace controls make a difference—a full-face respirator, thicker gloves, and rigorous training ought to be standard. Companies benefit from closing the loop, capturing vapors, and treating effluent before it leaves their gates. Several countries, such as members of the European Union, restrict use or require special permits for handling N,N-Dimethylaniline. That’s more than just paperwork—it forces businesses to rethink processes and reduce unnecessary emissions.
People living near manufacturing sites should have a say. It’s important for regulators to listen to these voices, run independent environmental tests, and set clear health advisories. Education about safe storage and handling for anyone working with the chemical—whether in industry or in a university lab—always helps prevent the next incident.
Transparency brings trust. If companies track and share what they emit or discharge, it gets easier for communities to hold them accountable and for everyone to breathe a little easier.
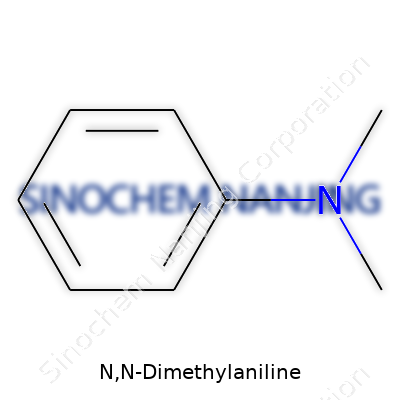
| Names | |
| Preferred IUPAC name | N,N-dimethylbenzenamine |
| Other names |
DMA N,N-Dimethylbenzenamine Dimethylaniline N-Phenylmethylamine |
| Pronunciation | /ˌdaɪˌmɛθɪlˈænɪliːn/ |
| Identifiers | |
| CAS Number | 121-69-7 |
| Beilstein Reference | 605694 |
| ChEBI | CHEBI:34938 |
| ChEMBL | CHEMBL25454 |
| ChemSpider | 5466 |
| DrugBank | DB02552 |
| ECHA InfoCard | 100.055.041 |
| EC Number | 203-615-4 |
| Gmelin Reference | 6138 |
| KEGG | C01407 |
| MeSH | D003603 |
| PubChem CID | 6119 |
| RTECS number | BX9275000 |
| UNII | K7D6TEN12C |
| UN number | UN2262 |
| CompTox Dashboard (EPA) | `DTXSID7020667` |
| Properties | |
| Chemical formula | C8H11N |
| Molar mass | 121.18 g/mol |
| Appearance | Colorless to pale yellow oily liquid |
| Odor | Amine-like |
| Density | 0.957 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 0.46 mmHg (25°C) |
| Acidity (pKa) | 5.15 |
| Basicity (pKb) | pKb = 9.85 |
| Magnetic susceptibility (χ) | -47.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | 1.099 mPa·s (25 °C) |
| Dipole moment | 1.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 138.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3085.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H302, H312, H332, H315, H319, H335, H351, H373 |
| Precautionary statements | P261, P280, P301+P310, P304+P340, P311, P405, P501 |
| Flash point | 58 °C (closed cup) |
| Autoignition temperature | 440 °C |
| Explosive limits | 1.1–6.5% |
| Lethal dose or concentration | LD50 oral rat dimethylaniline = 1390 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1410 mg/kg (rat, oral) |
| NIOSH | KD9625000 |
| PEL (Permissible) | 10 ppm (Sk) |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 100 ppm (as string) |
| Related compounds | |
| Related compounds |
Aniline Dimethylaniline N,N-Diethylaniline N-Methylaniline Phenol Diphenylamine |

