N,N-Diethylhydroxylamine: More Than a Specialty Chemical
Tracing the Road: Historical Development
Every now and then, a chemical quietly shapes modern industry, and N,N-Diethylhydroxylamine (DEHA) fits that mold. Chemists started looking at hydroxylamines before World War II, with focus ramping up as the synthetic rubber and polymer booms hit their stride. By the 1960s, industrial labs had put DEHA under the microscope, chasing better oxygen scavengers and radical inhibitors. A good oxygen scavenger offers a defense in boilers and reactors, fighting corrosion in systems where water and air meet under pressure. Companies navigating boiler rooms or ink factories started depending on DEHA for cleaner machines and longer equipment life instead of dealing with iron oxide sludge clogging their works. Since then, the breadth of research has only grown deeper, with new syntheses, safety reviews, and ways to minimize environmental harm.
Peering Into the Bottle: Product Overview
Walk into any facility using advanced water treatment or precision chemical synthesis, and you’ll probably find DEHA on the shelf. It comes clear, sometimes faintly yellow, with a mild amine odor. Few ever mention the psychological impact of how a colorless liquid creates trust issues—people want to see that something’s in there, not just stare through the bottle. Manufacturing standards for DEHA tend to keep it over 85% pure, most often in liquid form. Chemically, it brings a unique pair of ethyl groups attached to a hydroxylamine core, making it less volatile than some cousins but just as reactive in the right hands. That’s where its real usefulness shines through.
Physical & Chemical Properties: The Details That Matter
I’ve watched engineers and chemists argue over boiling points as if the lifespan of process equipment depended on it. In some cases, it does. DEHA boils at around 125°C under atmospheric pressure. Density lines up just under water’s at room temperature, making for easier blending in industrial settings. It dissolves well in common polar solvents, including water and alcohols. Flammability ranks lower than short-chain amines but still calls for respect during storage and transit. What looks boring in a safety sheet transforms into long weeks of cleaner surfaces and more consistent process runs once you’ve handled it on the factory floor.
The Technical Nuts and Bolts
DEHA’s technical standards, as set by different regional guidelines (such as ASTM or EN), require its purity to hit benchmarks, and limits for water content and color. Manufacturers document composition to back up chemical traceability—a topic regulators don’t joke about anymore. Labels mark out batch numbers, recommended storage temperature, and expiry dates in clear print. Operators avoid product breakdown or side reactions by keeping the chemical away from high heat, strong oxidizers, and acidic conditions. Smarter processes now use closed transfer and better containment to stick to occupational exposure limits.
Making It: Preparation Method
DEHA’s industrial synthesis usually targets a smooth combination of nitroethane or similar nitro compounds with reducing agents in a controlled environment. I’ve seen lines where sodium dithionite or catalytic hydrogenation gets the exposure, depending on how the plant’s set up to recycle byproducts or handle emissions. The selection depends on cost, safety, and how well the reactants fit into an established supply chain. Side reactions can spell trouble, so dedicated purification steps rescue DEHA from unwanted amines or unreacted nitro groups. The labs call for hands-on quality checks to keep up batch consistency.
Chemical Reactions and Product Modifications
Chemists leverage DEHA’s reactivity at the N–O bond to pull off a range of transformations. In water treatment, it scavenges dissolved oxygen, protecting ferrous metals by converting O2 to harmless species. In radical polymerization, DEHA blocks runaway chain reactions, keeping product consistency high and yields steady. Over time, labs found ways to tweak DEHA through acylation, alkylation, or using it as a base for more elaborate hydroxylamine derivatives. Some efforts investigated further chemical modifications to fine-tune volatility or reactivity for niche applications. DEHA’s stability under moderate pH and temperature ranges stands out, allowing it to persist long enough to do its job and then degrade into less worrisome compounds.
Names Across the Industry
DEHA goes by more than one name. Aside from N,N-diethylhydroxylamine, you’ll hear it called diethylhydroxyamine or simply DEHA among process engineers and purchase managers. Older literature sometimes lists obscure synonyms that show up in regulatory checks, adding an extra step when aligning compliance paperwork from different countries. Consistency in naming avoids confusion and cuts down on regulatory headaches.
Safety and Running a Tight Ship
Safety with DEHA calls for more than ticking boxes on a safety sheet. I’ve seen operators grow complacent around lower-toxicity chemicals, only to get tripped up by skin or eye contact from splashes. Prolonged exposure still risks mild nervous system symptoms, so gloves and goggles stay the norm in professional settings. Material spills stay rare, but vapor suppression and handling protocols make sense anytime a drum gets moved. Fire codes dictate careful storage away from incompatible chemicals, and facilities keep spill kits handy. The main lesson: routine diligence trumps the myth of a "safe" chemical, even for substances with a long industrial track record. Following occupational exposure limits remains non-negotiable for plant managers wanting long-term staff health.
How DEHA Works for Us: Application Areas
Industries look at DEHA first for oxygen removal in low- to medium-pressure boiler systems, extending pipe life by fighting off rust. Some colleagues in the graphic arts world swear by DEHA’s performance in photochemical emulsions and ink stabilization, especially where color quality has no margin for error. Polymer chemists lean on its radical quenching to rein in runaway side reactions. I’ve seen it used in industrial cleaners, fuel treatments, and even rubber manufacturing, with each application asking for different formulation nuances. The variety speaks to how DEHA adapts to the pace and quirks of real-world production lines, not just the lab.
Pushing the Boundaries: Research
Research into DEHA stretches well beyond its basics. Scientists continue probing its byproducts and environmental fate, seeking assurance it won’t generate tougher challenges down the road. Lately, demand for cleaner processes prompted a wave of studies on biological breakdown pathways that let wastewater plants keep up with stricter standards. A few teams dig into new catalytic systems built around modified DEHA molecules, hoping for more efficient radical control in next-generation plastics. As regulations tighten on all fronts, chemists look for data to support continued use—sometimes facing tough questions about alternatives or cumulative toxicity.
Toxicity and Knowing the Risks
DEHA scores lower on acute toxicity compared to similar amines, but the story gets more complex on chronic exposure. Animal studies show high doses can bring on changes in blood chemistry and mild neurological symptoms, even though normal workplace levels fall well below these risk thresholds. Long-term exposure still raises precaution flags, and regulators from OSHA to REACH call for work environments to minimize unnecessary handling. Waste disposal guidelines pay close attention to aquatic toxicity and possible impacts on microbial communities in wastewater plants. Repeated reviews double-check the old assumptions, since community and worker safety never sit still.
The Road Ahead: Future Prospects
DEHA’s future looks promising, especially as industries search for more sustainable process aids and shift priorities to both economic and environmental resilience. Demand in water treatment continues to climb as populations grow more dependent on clean steam systems for energy and manufacturing. One challenge involves making greener synthesis routes, aiming for reduced waste and easier downstream treatment. Academic groups and industrial innovators chase more selective reaction pathways for its production and push for lower-impact modifications that can keep up with green chemistry goals. At the same time, watchdog agencies keep pressure on users to demonstrate real-world safety, both in terms of human health and broader environmental impact. For the foreseeable future, DEHA will keep evolving, serving as a bellwether for how specialty chemicals navigate the crosshairs of innovation and responsibility.
What is N,N-Diethylhydroxylamine used for?
Chemistry on the Factory Floor
Walk into any plant that makes paper or produces industrial chemicals, and you might catch a whiff of N,N-Diethylhydroxylamine (DEHA). It works mostly behind the scenes, but its fingerprints show up on everything from the quality of your newspaper to the safety of your drinking water. I’ve seen chemical operators measure out this liquid in the pump rooms, taking care with every step, because a little too much or too little has real-world consequences for the end product—and the folks running the systems.
Stopping Rust Before It Starts
DEHA’s main claim to fame comes from the way it helps industrial boilers. Water turns to steam; iron and steel pipes sit exposed. Oxygen sneaks in and wants to rust everything. Plant teams add DEHA to the water. The stuff finds dissolved oxygen and knocks it out before it gnaws through metal. In my years of tracking down leaks in boiler rooms, I noticed the best maintenance crews didn’t cheap out on oxygen scavengers like DEHA—they knew a little prevention in a tank would save weeks of downtime fixing corroded pipes. The U.S. Environmental Protection Agency recognizes DEHA as a “deoxygenator” in boiler treatment, keeping costly shutdowns at bay and extending the lifespan of critical equipment.
Helping Papermakers and Painters
The papermaking industry uses DEHA to keep pulp from getting streaky or yellow. Mills rely on clear, stable pulp streams, and DEHA helps mop up stray oxygen that wants to turn pulp brown. That off-color paper never makes it to the printer—so every role DEHA plays, from pulp bleaching to wastewater management, helps ensure the package in your mailbox looks sharp and smells clean.
In the world of paint, especially water-based coatings, DEHA keeps things smooth. It acts as an antioxidant, guarding paint against spoilage. You get fewer cans turning lumpy on the shelf and better colors that don’t fade before their time. The American Coatings Association points to DEHA as one way paint-makers keep their promise on shelf life and reliability. Years in paint sales taught me a batch gone bad means truckloads of returns. An ingredient like DEHA can make or break trust with pros who depend on consistency every day.
The Safety Talk
Nothing in a chemical plant gets handled lightly. DEHA doesn’t rank up there with the nastiest industrial chemicals, but it’s not apple juice either. OSHA lays out safety measures; gloves, goggles, and a mind for spills. Workers talk about respecting chemicals like DEHA, not fearing them. That attitude creates fewer accidents—and chemical exposure rates in the U.S. continue falling, largely from better training and accountability.
Looking for Greener Alternatives
There’s always room for improvement. Sustainability teams keep searching for oxygen scavengers that break down cleaner, or that pose even less risk to workers and water supplies. DEHA already breaks down in the environment faster than many old-school chemicals, but the hunt for the next big thing continues. Decision-makers in both public utilities and private factories have a duty to weigh facts, keep up with regulations, and listen to industry watchdogs who call out risky shortcuts. Every new chemical must prove itself in the real world—on the floor, not just in the lab.
Is N,N-Diethylhydroxylamine hazardous to health?
What It Is and Where You Might Find It
N,N-Diethylhydroxylamine, often shortened to DEHA, pops up in places that might surprise you. It works as an oxygen scavenger in water treatment, finds a home in chemical manufacturing, and plays a role in photography. Sharp minds in labs use it to help prevent corrosion or stabilize certain substances. Most folks won’t run into DEHA at home, but people working in these industries might get exposed.
Looking at Health Risks
Concerns around DEHA come from some of the effects seen after breathing in, swallowing, or getting it on the skin. The Environmental Protection Agency labels it as having low toxicity, but that doesn’t mean it’s completely harmless. Breathing in high amounts in the workplace could irritate the nose, throat, or lungs. Splashing a concentrated solution on skin or eyes could cause redness and burning. Swallowing large amounts brings on headaches or nausea. Effects usually show up when a person gets much more than trace levels, so it matters most for those handling it as part of a job.
What the Science Says
Scientists have probed DEHA to figure out just how risky it is. Long-term animal studies haven’t shown that this chemical builds up in the body or causes cancer. Health agencies, including the European Chemicals Agency and the American Conference of Governmental Industrial Hygienists, don’t class it with the harshest hazards like known carcinogens or reproductive toxins. No strong evidence ties it to chronic diseases in everyday workplace exposures, provided proper safety steps are in place.
Industrial Use: Real-World Experiences
From my own time in manufacturing, I remember how storage and handling make all the difference. Workers used gloves, goggles, and proper ventilation every time we moved chemicals like DEHA. Our safety officers drilled into us that one splash could bring a long day of discomfort or worse. Routine air monitoring helped keep everyone honest about exposure levels. I once watched a teammate get a DEHA splash on a hand, and quick washing prevented any real harm—reinforcing all that training.
Reducing Risks in the Workplace
Companies have shifted towards tighter controls over the years. Personal protective equipment (PPE) stands at the front line, but real change happens when employers swap out risky chemicals for safer alternatives, or put machines and tools in place so people aren’t getting hands-on with hazardous substances unless they have to. Written procedures and regular safety reviews help catch problems before they reach people. Regulators like OSHA set limits on airborne contaminants, and those limits push employers to monitor air and keep it clean. Open windows, exhaust fans, and good hygiene—simple steps go a long way for workers who can’t avoid DEHA.
Smart Practices for Safety
If you use DEHA or anything like it, treat it with the respect any industrial chemical deserves. Follow instructions for use and disposal. Employers should supply adequate PPE and training, plus check air quality and surface contamination. Workers need to know symptoms to watch for, and never skip washing up at the end of a shift. Keeping safe doesn’t mean fearing every chemical; it just means knowing what you’re up against and staying prepared.
How should N,N-Diethylhydroxylamine be stored?
Practical Lessons from the Chemical Storeroom
N,N-Diethylhydroxylamine doesn’t show up on most grocery lists. In places like labs or commercial warehouses, though, this compound has its uses – and everyone working around it needs to understand what makes safe storage matter. I remember my first year managing a chemical storeroom, tailing veteran techs through the tight aisles. They handled bottles of all sorts of clear and colorless liquids, but some demanded more care. N,N-Diethylhydroxylamine was always one of those bottles that got an extra label. Not just because of regulatory rules, but because of what can happen if people ignore the basics.
Understanding Why Storage Details Matter
This compound has a reputation for instability, especially when exposed to air and heat. It’s listed by the National Institute for Occupational Safety and Health (NIOSH) as an irritant and can become hazardous in the wrong conditions. My experience tells me that, after a long day, even experienced staff can forget about rare-use items gathering dust at the back of a shelf. That’s where trouble often starts. Accidental spills, leaky caps, or sudden fumes – these small events become big headaches if the environment isn’t right.
Keeping Temperature and Moisture in Check
Storing N,N-Diethylhydroxylamine at room temperature may work in theory, but temperature swings from hot equipment or sunlight through windows quickly lead to degradation. In my lab days, we always used temperature-controlled cabinets, never leaving bottles on an open bench. The bottle belonged in a cool, dry area, away from direct sunlight, heat sources, or anywhere daily foot traffic could jostle it. Moisture is another quiet threat. The compound reacts with water and air much faster than many expect. Good-quality desiccants tucked into storage cabinets can make a difference, especially in humid climates.
Separation from Incompatible Materials
One mistake I spotted more than once: Storing strong oxidizers nearby. This compound reacts dangerously with oxidizing agents like peroxides, nitric acid, or bleach. In chemical fires I’ve read in safety case studies, combining these ingredients led to explosions, not just smoke or stains on shelving. Everyone in charge of storage needs clear labels and a system separating incompatible containers, not just handwritten reminders boasted on Post-it notes.
Practical Storage Solutions
Here’s what worked: Always use tightly sealed, original containers or high-quality chemical storage bottles. Skip old glass jars with rubber stoppers; modern containers provide better vapor barriers and chemical resistance. Shelves built with non-porous, easy-to-clean materials make cleanup easier in the event of leaks. Spills do happen, particularly when people get rushed or complacent. A proper spill kit—complete with neutralizing agents and plenty of absorbent material—belongs close at hand.
Training and Awareness Make All the Difference
Regulations exist for a reason, and in this case, they back up plain common sense. Safety data sheets aren’t just paperwork—everybody in the facility needs to read them and understand what those hazard pictograms really mean for day-to-day storage. Routine checks catch small leaks before they turn into emergencies. Most places get better results by pairing written policies with hands-on walkthroughs, not just handing out binders stuffed with fine print.
Room for Improvement in Routine
Accidents linked to N,N-Diethylhydroxylamine usually don’t happen because of a single big mistake, but from many small lapses—a misplaced bottle, a missed inspection, a forgotten cleanup. I’ve seen that cultivating a culture that values routine checks, honest labeling, and strong communication replaces guesswork with reliability. Storage issues get solved not by extraordinary measures, but by sticking to practical routines, fostering respect for the risks, and never underestimating the hazards, no matter how familiar the bottle becomes over time.
What are the handling and safety precautions for N,N-Diethylhydroxylamine?
Recognizing the Risks
N,N-Diethylhydroxylamine steps into the workspace as a chemical with clear risks. This compound can irritate skin, eyes, and the respiratory system. Operators tend to overlook the hazards at their own peril, especially if shortcuts get taken. The liquid and its vapors don’t just annoy the senses—a spill or a small cloud can trigger health issues, sometimes quickly.
Personal Protection Matters
No shortcuts work here. Splashing a bit of this compound on bare skin leaves a burning or tingling not easily forgotten. I once watched a technician get a small drop on his hand. He ignored it for a few minutes, thinking gloves were a hassle. His mistake turned into a visit to the clinic because the irritation grew fast. Safety goggles, nitrile gloves, and a lab coat make a difference with every use. For lab environments or larger handling, a face shield and chemical-resistant apron are smart additions.
Ventilation and Containment
A fume hood works best for this job—not just to limit smells, but because vapor inhalation can be a real problem over time. A friend running a pilot plant program coughed all afternoon once after a quick pour outside the hood. Good airflow protects the lungs, and local exhaust keeps accidental exposure low. It never hurts to inspect hoods or ensure they pull enough air, because memories of a day spent with a burning throat stick with you.
Storage Rules That Can't Be Bent
N,N-Diethylhydroxylamine acts more like a troublemaker in storage if left treated casually. It breaks down under heat or light, and old containers risk developing dangerous pressure. I’ve seen labels peel off from sweaty bottles—then nobody knows what’s inside, which only adds confusion during spills. Tightly-sealed amber glass bottles kept in a cool, dry, and dark place remain the best bet. Chemical storage rooms set up for flammable or reactive agents provide solid protection, especially from incompatible substances like strong acids or oxidizers.
Responsible Handling and Training
Inexperience with chemicals like this can breed complacency. People get away with careless handling a couple of times and think the danger is exaggerated—until it isn’t. Training each person in spill cleanup, emergency showers, eyewash station locations, and waste disposal gives everyone a stake in staying safe. I’ve gone through a few real drills, and the difference between rehearsed and real panic matters when a spill happens or eyes start to sting. Material Safety Data Sheets (MSDS) should always be easy to get, never buried in a drawer.
Waste Disposal Practices
Pouring excess or waste N,N-Diethylhydroxylamine down the sink never ends well for pipes or the environment. Chemical waste containers labeled for organic amines or reactive solutions keep things sorted. Professional disposal services handle the collection so nobody has to worry about chemical surprises in the trash or the drain. A recent audit I witnessed flagged a laboratory for dumping chemical-laden paper towels in normal bins—a mistake that led to fines and a whole lot of extra training hours.
Looking Out for Each Other
Careful handling isn’t just about rules—it’s about keeping everyone in the workspace healthy. Sharing personal experiences with close calls or actual incidents drives home the point better than endless reminders. N,N-Diethylhydroxylamine isn’t rare but treating its risks with respect keeps injuries rare—and workplaces running responsibly.
What is the chemical formula and appearance of N,N-Diethylhydroxylamine?
Understanding What Sets N,N-Diethylhydroxylamine Apart
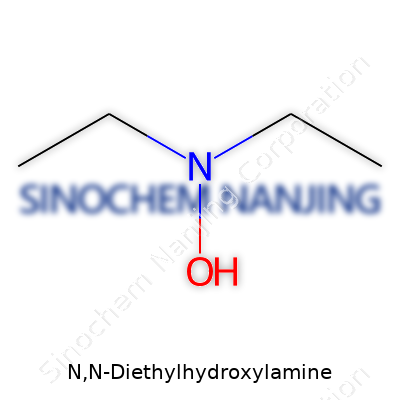
N,N-Diethylhydroxylamine shows up in discussions about chemical processing, wastewater treatment, and manufacturing where control over oxidation makes the difference between product success and safety incidents. The chemical formula for this compound reads as C4H11NO. A simple formula, but the underlying properties affect real-life operations in significant ways.
Behind the Formula: What C4H11NO Means in Practice
To visualize N,N-Diethylhydroxylamine, think of a clear, colorless to pale yellow liquid. There’s no particular glamour in its look. Open a tightly sealed bottle, and the smell that escapes carries a faint amine odor—kind of a fresh, ammonia note with a hint of something more earthy. Folks who handle it in labs or industries pick up its distinctive scent fast.
This liquid feels slightly oily to the touch but it’s completely miscible with water. That trait helps when you try to dissolve or dilute it on site, even in plant environments not equipped with advanced mixing systems. For those who favor the details, the boiling point hovers around 125°C. You don’t have to hit high temperatures for it to present a fume risk, especially in confined spaces.
Role and Importance in Industrial Applications
The reason people care about N,N-Diethylhydroxylamine mostly ties back to its reducing ability. In my own experience, working at a site that used it to prevent unwanted oxidation in boiler water, it shaved down corrosion and extended equipment life. Many folks opt for it over other amine-based options due to its lower tendency to create troublesome byproducts.
Beyond water treatment, it finds its way into photographic processing and synthetic chemistry settings. Oxidation control stops sensitive ingredients from breaking down too early or reacting with the wrong partners. Workers using N,N-Diethylhydroxylamine need to keep an eye on ventilation and skin protection, since the liquid can irritate eyes and skin on contact. Extended exposure raises safety flags.
Facts from Research and Industry Practice
Peer-reviewed studies back up its function as a free-radical scavenger. According to journals like the Journal of Hazardous Materials (2022), controlled dosing in closed-loop systems limits corrosion rates more effectively compared to older technologies. Companies who switched to N,N-Diethylhydroxylamine during several years of plant operation noticed lower rates of component replacement and fewer maintenance shutdowns.
Given its reactivity, facilities must respect local and international storage guidelines. Storing it in steel drums without proper lining speeds up corrosion, leading to leaks or contamination of the product. Smaller setups often move to HDPE containers that resist degradation for longer. Spill response plans include adequate eyewash access and rapid cleanup kits because even trace residues on floors become slippery or damaging to shoes and gloves.
Possible Paths Forward
Problems with supply chain interruptions can slow down the availability of chemicals like N,N-Diethylhydroxylamine. Facilities do better when they train staff not just on current handling protocols but update procedures with lessons learned from near-misses. In practice, continuous supplier vetting, periodic chemical testing, and scheduled safety drills cut down on operational surprises, keeping the compound—and the teams managing it—working safely together for the long haul.
| Names | |
| Preferred IUPAC name | N-ethyl-N-(ethylidene)hydroxylamine |
| Other names |
Diethyloxyamine N,N-Diethylhydroxylamin N,N-Diethylhydroxylaminum N,N-Diethylhydroxylammonium |
| Pronunciation | /ˌɛnˌɛn.daɪˌɛθɪl.haɪˌdrɒk.sɪl.əˈmiːn/ |
| Identifiers | |
| CAS Number | 3710-84-7 |
| Beilstein Reference | 1361678 |
| ChEBI | CHEBI:35910 |
| ChEMBL | CHEMBL169784 |
| ChemSpider | 7664 |
| DrugBank | DB14024 |
| ECHA InfoCard | ECHA InfoCard 100.018.231 |
| EC Number | 200-529-9 |
| Gmelin Reference | 8719 |
| KEGG | C06500 |
| MeSH | D003994 |
| PubChem CID | 7921 |
| RTECS number | KK7525000 |
| UNII | UBO225619M |
| UN number | UN1604 |
| CompTox Dashboard (EPA) | 0BX3B3ZB0H |
| Properties | |
| Chemical formula | C4H11NO |
| Molar mass | 89.14 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | amine-like |
| Density | 0.89 g/mL at 25 °C |
| Solubility in water | soluble |
| log P | -0.34 |
| Vapor pressure | 0.49 mmHg (at 25 °C) |
| Acidity (pKa) | 8.5 |
| Basicity (pKb) | 5.84 |
| Magnetic susceptibility (χ) | -37.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.419 |
| Viscosity | 3.45 cP (20°C) |
| Dipole moment | 3.08 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 269.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -355.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Harmful if inhaled. May cause an allergic skin reaction. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-1-W |
| Flash point | 57 °C (closed cup) |
| Autoignition temperature | 170 °C |
| Explosive limits | Lower: 2.2%, Upper: 10.2% |
| Lethal dose or concentration | LD50 oral rat 65 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 65 mg/kg |
| NIOSH | KW2975000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 1 ppm (3 mg/m³) |
| Related compounds | |
| Related compounds |
N-Methylhydroxylamine N-Ethylhydroxylamine N,N-Dimethylhydroxylamine Hydroxylamine Diethylamine |

