Understanding N,N-Diethylformamide: More Than Just a Solvent
The Journey from Laboratory Curiosity to Industrial Mainstay
N,N-Diethylformamide hardly makes front-page news, but its evolution tells a story about how chemistry adapts to the needs of everyday life and industry. Decades ago, research teams navigated through the early days of organic synthesis, searching for liquid mediums with enough versatility to handle tricky reaction systems. Chemists didn’t pick N,N-Diethylformamide at random; after running through pages of trial-and-error, it stood out for its unique balance — not too volatile to be a fire risk, but with enough solvent power that even stubborn solids would dissolve. Once found, the formula stuck. You can trace its use to the changing demands of plastics, textiles, and even pharmaceuticals. At each intersection, practical hurdles called for tweaks to the standard toolkit. N,N-Diethylformamide stepped up as a workhorse where others fell short, a quiet partner in the boom of polymers and dyes. Its story reflects more than lab bench notes; it mirrors the way industrial chemistry learns, borrows, and builds on practical solutions, sometimes clinging onto them because they just work.
Basic Facts: What Makes N,N-Diethylformamide Tick?
People who step into a chemical warehouse probably won’t spot a bottle labeled ‘magic liquid,’ but if they did, it would describe N,N-Diethylformamide well. With a chemical formula C5H11NO, and a structure that keeps two ethyl groups bonded to a formamide backbone, its behavior toes the line between polar and nonpolar worlds. This ace up its sleeve lets it act as a solvent for a massive range of chemicals that don’t always play nicely together. It’s colorless, with a faint amine-like smell that any chemist could pick out blindfolded. Its boiling point rests around 180°C, which keeps it in the liquid phase in most settings without being a headache for containment. Its miscibility with water and a wide group of organics makes it a go-to even when the shopping list for solvents feels endless. Folks in the know check for purity, water content, and residual amines, because small shifts change the way syntheses behave.
Labels Versus Latitude: Technical Specs and Use Limits
Walking through most labs, finding N,N-Diethylformamide in reagent cabinets alongside a heap of regulatory paperwork and data sheets is common. Production quality often points toward high-purity grades, since impurities can throw a wrench in both analytical work and industrial processing. Specification sheets drill down on acidity, water, and sometimes specific UV absorption patterns. While this layer of precision seems fussy, chemists who have chased down a stuck reaction or unexplained impurity know that a weak link in supply quality can cost more than an entire batch. Labels warn of safety hazards, yield limits, and sometimes note incompatibilities with acids or oxidizers, based on case studies where things turned sideways. Users who ignore those warnings risk more than lost yield — there’s health and safety to think about. Facilities that keep N,N-Diethylformamide close ensure that technical measures match standards from groups such as OSHA and the European Union’s REACH directive. They recognize that safety measures, including fume hoods, gloves, and goggles, aren’t just window dressing; they answer to the reality of what CAN go wrong, as much as the chemistry itself.
Human Ingenuity on Display: Preparation Techniques
If there’s an arena where creativity shows up in chemistry, it’s in the manufacturing routes for compounds like N,N-Diethylformamide. The classic route leans on reacting diethylamine with methyl formate or directly with formic acid. These transformations are more than textbook exercises — getting the right temperature and ratios, removing water, and handling amines, each introduces wrinkles that call for careful timing and solid equipment. As chemical engineers push for cleaner, more cost-effective processes, greener options keep emerging. Finding ways to recycle reagents, capture byproducts, and streamline steps plays a growing role, especially with tightening environmental rules. The hunt for better catalysts or innovative continuous-flow systems proves chemistry’s restlessness, always seeking to shave off waste and costs with fresh ideas.
Where Reactivity Breeds Utility: Reactions and Chemical Tinkering
N,N-Diethylformamide does more than sit quietly as a medium; it can participate in certain transformations, offering up its lone pair for complexations or breaking down under force. Its role as a donor solvent pulls in researchers working with metals, organometallics, and nanoparticles. Industrial settings have chased its uses in alkylation and condensation reactions. At times, it’s not just the reaction outcome folks care about, but the ease of recovery and reuse. The ability to tweak its backbone with added groups or slight modifications has produced derivatives that open new doors: improved solubility, new selectivity, or less toxicity. Each shift targets a real-world demand, driven by what end-users discover in the trenches.
What's in a Name? Synonyms and Branding Through the Decades
Over the years, N,N-Diethylformamide has collected a handful of names and codes. Some circles just call it DEF, shorthand that feels breezy among lab veterans. You might spot older documents using Diethylformamide or N,N-Diethylmethanamide, but no matter the alias, it’s the same liquid hiding behind those labels. Branding sometimes tweaks the name, trying to make it stand out in catalogues or regulatory filings, but those in the know recognize it from its chemical fingerprint rather than the letters stamped on a drum.
Keeping People and Process Safe: More Than Paperwork
N,N-Diethylformamide doesn’t scream danger, but it brings enough risk to earn a spot on the watchlist for any responsible lab manager. Exposure limits appear rooted in experience as much as toxicology — workers in plants decades ago developed health issues from repeated contact, not always picked up by the early safety protocols. Chronic exposure runs the risk of liver stress and effects on the central nervous system, and the International Agency for Research on Cancer (IARC) has investigated links to cancer. That legacy shapes how users approach it: skin contact, inhalation, and accidental ingestion all get flagged in training sessions. Storage requires sealed containers, managed ventilation, and protocols for clean-up that don’t just get buried in binders. Health officers take these details seriously, not out of formality, but because one mishap can snowball into long investigations and loss. Long-term trust in the industry builds on habits made up of labeling, training, and rapid response to spills.
Looking Beyond the Flask: Understanding Application Areas
If you surveyed manufacturing floors and research benches, you’d find N,N-Diethylformamide weaving its way across a surprising array of industries. In the textile trade, it helps dissolve polymers that become fibers for clothes, car seats, and carpets. In dye production, it stabilizes fine suspensions that bring lasting color to fabrics and plastics. Lab chemists count on it to keep reactions running smoothly in organic synthesis, pushing difficult reactions across the finish line. Even the pharmaceutical world taps its value, using it to anchor intermediates or manage tricky purification steps. Each sector sticks with it because alternatives often fall short in terms of solvency, ease of handling, or compatibility. The upshot is clear: once a compound gains a reputation as reliable, folks hang onto it, even as they keep an eye on better contenders.
The Edge of Discovery: Current Research and Chemical Innovation
Researchers constantly revisit N,N-Diethylformamide, not because it’s flashy, but because new applications keep cropping up. One area grabbing attention involves its role in nanotechnology, where solvents help guide the growth and assembly of advanced materials. Teams pushing for greener chemistry look for ways to swap it out, or at least cut down the unintended emissions and exposure. Analytical scientists use it in studies of solvent effects; its unique balance offers model systems for students sharpening their troubleshooting in spectroscopy or reaction monitoring. The work never feels finished. Any day, a publication might report a new way to recycle it, or pair it with other chemicals for entirely fresh uses. This spirit of invention underscores academic and industrial labs alike.
Assessing Toxicity and Facing the Hard Truths
N,N-Diethylformamide carries toxicological baggage that nobody in the field can ignore. Acute exposure may trigger skin irritation, headache, or nausea, and chronic exposure raises red flags for organs like the liver and kidneys. I’ve read stories from industry veterans who recall the days before modern extraction hoods and gloves — the cost of learning the hard way shaped every update to today’s protocol. Evidence from laboratory animal tests and human exposures guides regulations that put clear boundaries between safe use and risky habits. Some of the drive to find greener alternatives comes straight from this reality. Regulatory agencies play a watchdog role, and the industry answers with regular health checks, environmental monitoring, and technology designed to cut accidental exposure. Protocols get reviewed regularly because the science, painful as it can be, always pushes toward higher standards.
Where Do We Head Now? Future Prospects for N,N-Diethylformamide
Looking ahead, N,N-Diethylformamide stands at a crossroads. Growing environmental pressure calls for alternatives with lower toxicity and better biodegradability, yet industry still counts on the reliability and performance it delivers. Some teams focus on engineering recovery systems that minimize waste and emissions, while others probe new solvents with similar flexibility. The market value will follow where regulations and manufacturing trends tilt. Across the board, future use will reflect a combination of practical experience, scientific research, and social responsibility. As new needs arise, the lessons learned from decades of working with N,N-Diethylformamide will echo in how new materials and methods get adopted. The spirit of chemistry, as always, adapts and moves on, carrying the story of useful compounds forward.
What are the main uses of N,N-Diethylformamide?
Supporting the Chemical Industry
N,N-Diethylformamide (DEF) earns a spot in the toolkit of many chemists for a reason—its power as a solvent. People who work in the lab often look for a solvent that handles various reagents without reacting with them. DEF’s chemical stability makes it a favorite for dissolving organic and some inorganic compounds. In my own hands-on experience in academic research, DEF stood out when I needed something less volatile than dimethylformamide, especially during late-night reactions where safety mattered along with results.
Manufacturers turn to DEF when making specialty chemicals like pharmaceutical intermediates and crop protection agents. The compound’s low vapor pressure lets reactions reach higher temperatures safely. This detail matters for those scaling a reaction from test tube to reactor. It saves time and reduces the risk of accidents, which is at the core of keeping both workers and entire facilities safe.
Helping Paints and Polymers
I remember talking shop with someone in coatings development. When formulating paints and inks, chemists aim for a formula that balances viscosity and drying time. DEF offers just that. As a solvent, it mingles well with acrylics and vinyl resins, bringing down the mixture’s viscosity for spraying or printing. Paints lay down smoother and spread more evenly thanks to DEF’s presence. This translates to better coverage and less waste for the end user.
The polymer industry taps into DEF’s solvent properties for spinning synthetic fibers. In nylon or polyacrylonitrile production, DEF dissolves raw polymers, allowing them to be extruded into ultra-thin threads. This process would clog and stall without the right solvent. In the world of fabrics and technical textiles, details like solvent choice ripple through the whole supply chain, affecting product feel, dye uptake, and even end-use durability.
Cleaning and Extraction
Industrial cleaning jobs often sidestep water-based options because they can leave unwanted residues. DEF’s ability to dissolve grime, oils, and certain resins makes it useful for cleaning electronics, precision parts, and glassware. I learned quickly while working in an analytical lab that a spotless environment keeps equipment running longer. DEF can get into tight crevices and remove contaminants where other cleaners fail.
DEF also enters the field of extraction. For example, during natural products research, certain compounds stay locked in plant or microbial matter. DEF mixes with both polar and nonpolar molecules, helping scientists recover specific substances more efficiently, which trims down time and saves resources.
Addressing Safety and Environmental Aspects
Handling DEF safely stays at the top of the list. This chemical carries health risks—skin absorption, inhalation, and extended exposure can all pose hazards. People in the lab or on the plant floor rely on proper containment and protective equipment. Beyond the workplace, companies shoulder the task of disposing DEF waste responsibly. Accidental spills or improper dumps contribute to environmental damage, especially in waterways.
Some businesses now look for DEF alternatives or invest in better recycling systems. Using less, recycling more, and keeping a close eye on air and water emissions can help offset potential harm. A shift toward greener chemistry benefits the environment and, ultimately, each community around manufacturing sites.
What is the chemical structure and formula of N,N-Diethylformamide?
Getting to Know the Molecule
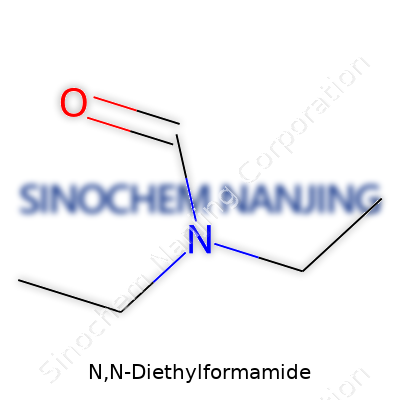
N,N-Diethylformamide belongs to the family of organic compounds called amides. Chemists recognize the structure by the arrangement of its atoms: it carries a formamide group with two ethyl groups attached to the nitrogen atom. The chemical formula shows up as C5H11NO. In simple terms, each molecule features a carbon atom double-bonded to an oxygen atom, linked to a nitrogen atom that's holding two ethyl chains. The structural formula puts this together as HCON(C2H5)2 or CHO(N(C2H5)2), spelling out the position of those branches. This setup gives the compound properties different from many solvents or simple amides.
Real-World Uses and Why Its Structure Matters
Anyone who’s spent time in a chemistry lab knows lots of reactions demand specialty solvents and reagents that won’t interfere with what’s going on. N,N-Diethylformamide steps in as an aprotic solvent, meaning it doesn’t easily donate hydrogen atoms. This matters because reactions often require a steady hand with reactivity—molecules like this one keep things running smoother than water or alcohol could.
Its structure shapes how it interacts with other chemicals. The ethyl arms boost its ability to dissolve nonpolar substances. The polar carbonyl group helps pull in molecules that don’t usually mix with standard hydrocarbons. This two-sided nature leaves it useful for pharmaceuticals, specialty coatings, and even forms part of the toolkit in organic synthesis. It’s not just about one or two applications, but rather the flexibility that its unique atom arrangement gives to designers and engineers in chemical manufacturing.
Health and Environmental Conversations
Several years working around labs, I saw plenty of reminders that common solvents demand caution. N,N-Diethylformamide shares this status. As with many organic compounds, taking proper care reduces the chance of inhaling vapors or getting skin exposure. Some research points to possible chronic toxicity, which warns us not to take shortcuts with gloves, fume hoods, or storage away from heat and light. This isn’t just about personal safety—companies owe it to their people and the local community to manage the risks with real action, not just paperwork.
Environmental agencies pay close attention to substances that flow into rivers or groundwater. Persistent chemicals can build up, especially when disposal goes unchecked. Facilities that use N,N-Diethylformamide often set up containment and treatment steps, like carbon filtration or specialized incineration, to lower the odds of environmental release. People who work with compounds like this need straight talk and clear protocols, since regulations spring from years of scientific study about chemical fate and toxicity.
Finding a Better Path Forward
Every time a new chemical hits the lab or factory, there’s a choice: follow the same footsteps as before, or look for safer alternatives. For some jobs, N,N-Diethylformamide’s properties stand apart. For others, teams push to swap in greener solvents with lower health risks and lighter environmental footprints. Sharing case studies from industry gets the word out—reducing harm starts with decisions made long before any spill or accident.
Support for good lab practice, honest communication about risk, and the pursuit of less hazardous alternatives makes a big difference. It’s easy to overlook the daily work that builds a culture of safety. In the long run, learning from experience—not just chemical structure—shapes the smartest choices.
What are the safety and handling precautions for N,N-Diethylformamide?
Understanding the Risks
N,N-Diethylformamide, often seen as a handy solvent in organic chemistry, packs more risk than it gets credit for. Folks working in a lab sometimes get used to handling chemicals and treat them all the same. That’s the kind of habit that leads to trouble, especially with solvents like this one. Its low odor can fool you into thinking it’s harmless, but health problems can sneak up if you drop your guard.
Health Hazards
Direct contact with this liquid burns the skin and irritates eyes. Beyond surface irritation, inhaling its vapors or letting it soak in through the skin may spark headaches, dizziness, or worse, damage to the liver and kidneys over time. The body absorbs this compound easily, especially in a warm, steamy lab, so gloves and protective gear aren’t just for show.
Smart Handling in Practice
Using this chemical safely takes some planning. Start with gloves that don’t give up easily—nitrile or butyl rubber types work best. Standard lab coats and goggles form a good backup. Spills should never get wiped up with bare hands; reach for the spill kit and open a window or turn on the fume hood. Good ventilation pulls the vapors away, and every chemist learns to check for working airflow long before the experiment starts.
I once worked in a teaching lab that looked spotless but had a habit of shutting fume hoods during cleanup to save on electricity. It didn’t take long for strong odors to creep up in the room. Just propping the windows open or running those hoods made a real difference. Some students dismissed the small stuff, but the headaches and itchy eyes told another story.
Safe Storage
Forget leaving a bottle on a crowded benchtop, even for a short break. This chemical stays safest in a tightly sealed container, marked with a clear hazard label, in a flammable cabinet. Temperature swings can weaken seals and increase pressure, so room temperature, dry conditions help avoid accidents. No one likes the surprise of a leaking bottle hidden at the back of a lab drawer.
Training and Emergency Response
Most problems come from not knowing—or not caring—about what a chemical can do. Training staff and students on real risks fits the bill much better than handing out a generic safety sheet. Regular drills with the eyewash and safety shower help if the worst does happen. Keeping emergency numbers posted big and bold by the phone beats digging through a binder in a panic.
Waste Disposal
Once a job uses up the solvent, the leftover waste still poses a threat. Pouring any extra down the drain makes things worse for the environment and anyone handling sewage downstream. Instead, pour it in a proper waste bottle with a label listing the contents. Regular pickups by professionals keep dangerous build-up out of the lab.
Building a Culture of Safety
Figuring out the safest way to handle toxic chemicals is all about habits. Shortcuts can leave scars, often long after the experiment ends. Every lab group, from the freshman students to the old-timers, benefits from open conversations about real risks. Sharing stories—what went wrong and what worked—carries more weight than the rules alone. Mistakes out in the open, not hidden away, give everyone a shot at safer work.
How should N,N-Diethylformamide be stored and transported?
Understanding the Challenge
N,N-Diethylformamide brings convenience to many manufacturing processes, but people often overlook the very real risks it carries. Having worked in and around chemical labs through college, I've seen how easygoing habits can spiral quickly when clear protocols don't get followed. Strong solvents like this one demand respect, both for the safety of the folks using them and for anyone nearby.
What Matters Most in Storage
You don’t need elaborate infrastructure to do things right, but certain things aren’t negotiable. Rooms without direct sunlight and stable, moderate temperatures go a long way in keeping chemicals stable. N,N-Diethylformamide releases flammable vapors. I’ve witnessed careless stacking of containers near heat sources, and more than once had to insist we move materials to a safer spot. Anyone working with chemicals should never underestimate fire risk, as government data from OSHA reports fires started by flammable liquids every year.
Containers should be tightly sealed, made of compatible materials like high-density polyethylene or special steel—not all plastics can handle the solvent. Store them off the ground on dedicated shelves, away from acids and oxidizers. This is not an old chemist’s tale; a splash or a mix-up with incompatible substances brings real danger.
Signage should be clear and bold. Labels with hazard symbols, dates, and supplier information help anyone who walks in understand the risks. Simple routines—like checking for leaks and inspecting seals—nip bigger emergencies in the bud.
Common Pitfalls During Transport
Moving chemicals always brings stress, whether across a warehouse or on a truck between cities. Strong rules exist for a reason. Proper drums or canisters with reliable caps beat out battered old containers every time. Never strap anything down with rope or tape—it slides and spills. Professional transport uses well-maintained cages or locking clamps.
On long journeys, drivers must keep chemicals separate from food or medical shipments. I once talked to a driver who moved lab solvents and realized how much rides on a single label—the wrong one, or no warning at all, puts health at risk for warehouse staff and emergency responders. Even on short runs, using insulation helps avoid temperature extremes that could boost vapor release.
Why All This Care Means Something
OSHA, the EPA, and similar regulators require businesses to assess risks, prepare data sheets, and file transport routes. These rules grew out of real, deadly incidents. Workers across dozens of industries have dealt with burns, breathing problems, or worse. Safety gear—gloves, goggles, sometimes respirators—serves as an everyday reminder of the risks. Nobody lays out this much protection just to jump through hoops. One missed step—one lid left loose, one drum exposed to sun—can turn costs or injuries into front-page news.
Better training pays off. I’ve trained co-workers who never handled chemicals before. It helps to show outdated drums, run actual evacuation drills, and explain exactly how a solvent like N,N-Diethylformamide behaves. Talking through “what if” scenarios makes boring rules seem real. Honest conversations and learning keep mistakes at bay more than endless written policies.
Moving Forward
Honestly, safe handling has never been more important. Global trade brings tons of hazardous chemicals through communities big and small. Any short cut—forgetting to check a label, missing a warning sign—invites harm. Smart storage and careful transport save time, money, and sometimes lives. Every workplace can create the habits and systems to keep accidents rare, not routine.
What are the potential health hazards of exposure to N,N-Diethylformamide?
Unpacking the Chemical
N,N-Diethylformamide, often used as a solvent in chemical industries and laboratories, goes by the nickname DEF among workers. The colorless liquid might not set off alarm bells at first glance, but plenty of people rarely realize what they’re dealing with on a daily basis. In my time working on-site safety audits for manufacturing companies, I’ve seen how easy it is to overlook the dangers once a chemical blends into the background of routine tasks.
From the Skin to Your System
Taking off gloves for “just a second” or skipping a fume hood out of habit opens doors to direct exposure. DEF absorbs right through the skin and spreads, making the body work overtime to process what’s now in the blood. Headaches, dizziness, and fatigue are the first signs workers might notice, though most shrug them off and keep moving. Long-term skin contact can cause a stubborn rash. Eye splashes sting and blur vision, usually sending someone rushing to the eyewash station. I’ve watched more than one worker ignore itchy hands, chalking it up to dry winter skin, only to discover too late that they’d let DEF sit on their skin too long.
Liver Knows the Burden
The liver acts as the body’s cleanup crew, breaking down harmful substances. DEF can be a real headache for this organ. Over months or years, repeated exposure pushes the liver to its limits, inviting chronic damage and throwing off enzyme levels. Regular health checks catch trouble early, but some find out only when persistent fatigue or yellowing eyes point to something deeper. Studies from workplace exposure underscore this point: workers exposed to DEF for years without proper protection show higher rates of abnormal liver function tests and even cases of hepatitis.
Air Quality and Breathing Risks
DEF easily evaporates, especially during handling or in warm conditions. Without solid ventilation, vapors fill the work area. Breathing in DEF fumes irritates throats and airways, starting with coughing or a sore chest. Folks with asthma feel these effects magnified. I remember a chemical plant supervisor who always left the door cracked in the mixing area, thinking it wasn’t a big deal. After a streak of respiratory complaints, the mistake became obvious. Installing local exhaust fans made a difference almost overnight, but the lesson stuck around longer than the fumes ever did.
Who Faces the Highest Risk?
People clocking in at DEF production plants, labs that formulate specialty chemicals, or industries using DEF as a solvent bear the brunt. Sometimes, the risk doesn’t stop when leaving work. DEF clings to clothing and tools, hitchhiking home, especially if personal protective gear isn’t a firm part of the routine. I’ve seen families concerned for skin issues and nosebleeds, tracing the cause back to a family member’s worksite habits.
Tackling Exposure at Its Source
Stopping trouble starts with knowing your enemy. Training helps, but it takes leadership enforcing glove rules, working ventilation systems, and scheduled health checks. I’ve found that installing chemical-resistant workstations and setting up easy-to-use spill kits go a lot further than just posting warning signs. Most effective results come from a culture that backs up policies with real action. Workers respected for their concerns don’t cut corners, and accidents drop quickly. If a business puts health first, people stick around and stay healthy.
| Names | |
| Preferred IUPAC name | N,N-Diylethanamineformaldehyde |
| Other names |
N,N-Diethylformamide Formic acid diethylamide Diethylformamide N,N-Diethyl-methanamide Diethylformamid |
| Pronunciation | /ˌɛnˌɛn.daɪˌiːθəl.fɔːˈmæm.aɪd/ |
| Identifiers | |
| CAS Number | 617-84-5 |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:44751 |
| ChEMBL | CHEMBL41518 |
| ChemSpider | 7307 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.063.354 |
| EC Number | 202-742-9 |
| Gmelin Reference | 8218 |
| KEGG | C07595 |
| MeSH | D003972 |
| PubChem CID | 8189 |
| RTECS number | LQ2100000 |
| UNII | 8DH2M4VU4D |
| UN number | UN2263 |
| CompTox Dashboard (EPA) | DTXSID8031263 |
| Properties | |
| Chemical formula | C5H11NO |
| Molar mass | 101.15 g/mol |
| Appearance | Colorless liquid |
| Odor | amine-like |
| Density | 0.926 g/mL at 25 °C(lit.) |
| Solubility in water | Miscible |
| log P | -0.38 |
| Vapor pressure | 0.474 mmHg (at 25 °C) |
| Acidity (pKa) | 11.98 |
| Basicity (pKb) | pKb = 13.22 |
| Magnetic susceptibility (χ) | -48.6×10⁻⁶ cgs |
| Refractive index (nD) | 1.422 |
| Viscosity | 0.802 cP (20 °C) |
| Dipole moment | 3.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -216.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2049 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H312, H319, H332 |
| Precautionary statements | P280, P261, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 52 °C (126 °F; 325 K) |
| Autoignition temperature | 447°C |
| Explosive limits | Explosive limits: 2.2–14% |
| Lethal dose or concentration | LD50 (oral, rat): 2820 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,860 mg/kg (rat, oral) |
| NIOSH | NIOSH: LZ8925000 |
| PEL (Permissible) | PEL = 10 ppm (skin) |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Dimethylformamide N,N-Diethylacetamide Formamide N,N-Dimethylacetamide N-Methylformamide |

