N,N-Diethylethanolamine: From Early Curiosity to Modern Industry
Tracing the Road: Historical Development
Science often branches out from a blend of curiosity, practicality, and the stubborn drive to solve real problems. N,N-Diethylethanolamine, or DEEA for short, grew out of this same mix. Decades ago, chemists recognized the potential in simple compounds like ethanolamines. The earliest literature points to researchers tinkering with amines in search of better solvents and buffering agents. Over time, the industry took notice as chemical companies in Europe and North America began scaling up production for technical applications, figuring out ways to lower impurities and keep reactions more predictable. DEEA gained ground in labs and factories for tasks that called for both a tertiary amine and a primary alcohol function, carving a place for itself in a market packed with specialty chemicals.
Hands-On Look: Product Overview
N,N-Diethylethanolamine stands out because its structure strikes a balance between reactivity and versatility. On the molecular level, it’s got both a diethylamino group and a small alcohol. Many folks in chemical manufacturing value it for its ability to jump into reaction schemes both as a base and as a building block. DEEA crops up anywhere you need a buffer with more punch than bare ethanolamine but less volatility than similar compounds. Whether it’s adjusting pH in water treatment or blending into paints, the compound does the job without fuss, offering a practical solution for chemists who want predictable outcomes.
Digging into the Details: Physical & Chemical Properties
You’ll meet DEEA as a clear liquid with a slight ammonia-like odor, heavier than water but still pourable and manageable. Its boiling point lands a little above that of water, and it dissolves easily in both water and a host of common organic solvents. The presence of both amine and alcohol groups lets DEEA engage in hydrogen bonding, making it handy in all sorts of formulations where other additives come up short. Its vapor pressure remains tame at room temperature, a plus for anyone working in warm conditions. Chemical stability tends to be robust under neutral or basic rules—acidic environments turn things more complicated, driving unwanted side reactions or slow discoloration.
Beyond the Label: Technical Specs & Handling
On the operational side, users value DEEA for its reasonably low viscosity, direct miscibility, and reliable performance in everything from lab reagents to massive industrial blends. Most production today delivers purity in the high nineties, with trace water and volatile organics being the common impurities flagged for control. People working with the compound must label and store DEEA under tight protocols to keep air, heat, and contaminants from kicking off degradation. Since this is a regulated substance in some regions, technical documentation also requires attention to local safety data and transportation codes, forcing producers and users alike to keep paperwork and training in line with evolving industry standards.
Getting There: How DEEA is Made
Making N,N-Diethylethanolamine relies on a fairly straightforward synthesis. Manufacturers often start with ethanolamine and diethylamine, running them through alkylation or condensations under a controlled environment. By choosing the right catalysts, pressure, and temperature, chemists can boost yields while filtering out side products. The methods continue to evolve, as process engineers experiment with reactors that recycle heat and cut down waste, moving toward more sustainable production with less impact on the environment. Real-world production always means scaling up from laboratory glassware to industrial reactors, so monitoring for byproducts and reaction completeness stays a constant concern.
The Chemistry at Work: Reactions & Modifications
N,N-Diethylethanolamine doesn’t sit idle; it reacts with acids to form salts, with alkyl halides to build quaternary ammonium compounds, and with agents like sulfuric or phosphoric acid to make more complex derivatives. I’ve worked in a lab where we relied on DEEA to produce intermediates for surfactants and textile auxiliaries. Adding certain catalysts opens the door to ring-forming reactions, or the building of modified esters. Plenty of times, DEEA fits into bigger synthetic schemes as a pH regulator, chasing away problems caused by stray acidity. Its adaptable structure means it slots seamlessly into many tried-and-true protocols, providing flexibility in both bench research and full-scale manufacturing.
What’s in a Name: Synonyms & Product Labels
Walk into any supplier’s catalog and you’ll find DEEA listed under a bundle of names: Diethylethanolamine, 2-(Diethylamino)ethanol, and N,N-Diethyl-2-hydroxyethylamine all reflect the same backbone. Sometimes chemical grade and intended use creep into the labeling—technical, laboratory, or pharmaceutical, each flagging different expectations for purity or trace contamination. For procurement teams and lab managers, these synonyms matter. Choosing the wrong label can mean compromised batches or regulatory headaches, so careful reading and cross-checking always play a role in successful projects.
Staying Safe: Rules, Standards & Reality
Working with DEEA calls for trust in both the science and the operating guidelines. Even though this chemical seems mild compared to some alternatives, improper handling leaves room for irritation, respiratory problems, or longer-term issues when spills or vapor get out of hand. Safety goggles, chemical-resistant gloves, and solid ventilation build a strong defense. In my experience, companies that maintain routine safety audits and spill training handle emergencies far better than those who rely on paperwork alone. International standards hold manufacturers to a growing list of requirements, from purity checks to ensuring packaging prevents leaks and runaway reactions. Neglecting proper safety practices in storage or disposal leads straight to regulatory fines and workplace injuries.
DEEA at Work: Application Areas
Industries ranging from water treatment to pharmaceuticals use N,N-Diethylethanolamine because it solves practical problems without excessive complication. Paint manufacturers rely on its buffering capacity to steady pH through long shelf lives, while textile producers add it to dye baths to adjust color intensity and uniformity. It pops up in gas purification, where engineers need reliable means of scrubbing acid gases like CO2 and H2S. In the pharmaceutical corner, DEEA serves as a solubilizer and intermediate, juggling its role between active synthesis and formulation. The steady demand in established markets reflects real-world utility rather than speculative optimism.
Pushing Forward: Ongoing Research & New Angles
Laboratories around the world see room for growth in how N,N-Diethylethanolamine gets put to work. New studies dig into ways to make DEEA-based solvents less volatile and more selective—tuning them to grab target molecules from mixtures with pinpoint accuracy. Environmental researchers experiment with biobased alternatives and greener synthesis routes, tackling old problems of chemical waste from a fresh perspective. Scientists in bioengineering explore modified ethanolamines as adjuvants or delivery vehicles in drug development. The pace and breadth of research reflect a real push toward compounds that play well both in manufacturing and end-user safety, as regulatory frameworks tighten year by year.
Toxicity and Human Health: The Questions That Matter
No discussion of DEEA feels complete without tackling the lingering concerns about exposure and toxicity. High doses cause skin and eye irritation in animal studies, and chronic intake creates questions that toxicologists keep pursuing. Most large-scale exposures come from industrial mishaps, so the focus often lands on workers in chemical plants or downstream users in dye and textile operations. Research highlights the need for smart ventilation, daily exposure caps, and medical monitoring where frequent contact is likely. As decades of data keep stacking up, the body of evidence grows, pushing manufacturers to tweak labeling and safety claims while spurring fresh studies on long-term effects.
Where to Next: Future Prospects for N,N-Diethylethanolamine
Looking forward, the future of DEEA depends on its adaptability to changing markets and more stringent health standards. New solvent blends, cleaner manufacturing techniques, and precise chemical modifications all point to deeper industry reliance on nimble, effective compounds. As governments expand rules governing industrial emissions and worker safety, the pressure gets higher to refine DEEA production both in purity and in accountability. Markets for technical chemicals never stand still; as new uses emerge—especially in biotechnology and clean energy—the value of reliable intermediates like DEEA is likely to climb. Our generation of scientists and managers faces the challenge of keeping both innovation and safety in balance as the role of DEEA continues to evolve.
What are the common uses of N,N-Diethylethanolamine?
Understanding Real-World Applications
N,N-Diethylethanolamine, often called DEEA, has found its way into more than a chemist’s toolkit. Many people interact with products containing this compound, even if they never see the name on a label. That’s because DEEA works quietly behind the scenes in a range of industries, most notably water treatment, coatings, and personal care.
Keeping Water Safe and Clean
DEEA plays a solid role in water treatment plants. It helps buffer pH in both boiler and cooling water systems. Try running a steam system with the pH all out of whack, and you’ll see corrosion chew right through pipes and systems. My time working with municipal utilities showed how a small dose of chemicals like DEEA guards against rust, scale, and bacteria. The right balance ensures clean, reliable water for entire neighborhoods. This isn’t just theory — utilities track fewer pipe failures and clearer water when they stick to a tried recipe that includes DEEA.
Making Paints and Coatings Last Longer
The paint aisle at a hardware store offers endless options. Behind the bright colors, DEEA’s amine structure enables better pigment dispersion. It keeps the paint flowing smoothly and prevents clumping, which means even amateur painters avoid streaks and uneven patches. Manufacturers rely on DEEA for making latex paints more stable during both storage and use. The compound also reduces the formation of “skinning” — a useless film that forms at the top of a can. These benefits mean contractors and DIYers can tackle projects with less waste, and painted surfaces withstand wear.
Personal Care Products That Feel and Work Better
Shampoos, lotions, and shaving creams often use compounds like DEEA as emulsifiers or neutralizers. It allows oily and watery materials to mix and remain stable on the shelf and during application. Some of my clients in the cosmetics industry value DEEA’s ability to balance product pH, so items don’t cause irritation. The FDA and European regulators require strict examination of such additives, and DEEA has passed safety reviews for allowed concentrations. Consumers get smoother, more consistent personal care products, and manufacturers can fine-tune performance.
Chemical Manufacturing and Specialized Synthesis
DEEA is a staple for chemical synthesis, acting as an intermediate in making corrosion inhibitors, pharmaceuticals, and as a scrubbing agent in gas purification. Refineries and chemical plants treat raw gases with amines like DEEA to remove acidic components, protecting people and the environment. Chemical engineers remember the smell and the care it takes to handle amines safely. The end results—cleaner fuel, safer industrial air—show up in air quality reports and production figures.
Opportunities To Do Better
The use of DEEA makes modern conveniences safer and more affordable. Still, there’s a need for better worker safety measures. Many amines can cause skin and eye irritation or respiratory issues during manufacturing. In my experience, employers who invest in ventilation, personal protective gear, and thorough training cut accident rates and worker illness by a noticeable margin. Continued regulatory updates and industry research can push for even safer alternatives that don’t sacrifice performance.
Final Thoughts
People seldom realize how many tools and products depend on overlooked chemicals like N,N-Diethylethanolamine. One bottle at a time, this compound improves daily life, public health, and the durability of everything from water pipes to a favorite shade of paint. Recognizing both the benefits and safety challenges shapes better solutions for industries and consumers alike.
What is the chemical formula and structure of N,N-Diethylethanolamine?
Understanding the Chemistry
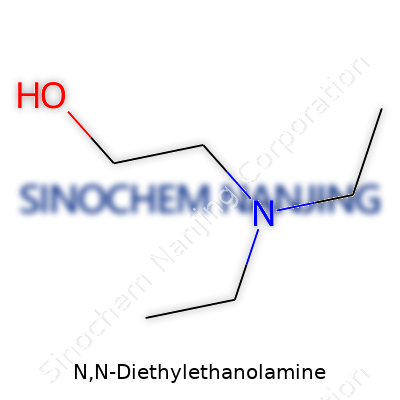
N,N-Diethylethanolamine, known by its shorthand as DEEA, carries the molecular formula C6H15NO. This compound shows up in a colorless, sometimes pale yellow liquid form, and gives off a mild ammonia-like scent. It folds into a class of chemicals called amino alcohols, which combine an amine group with an alcohol. The chemical structure tells a bigger story than a formula on paper. Picture a two-carbon ethanol backbone, with a hydroxyl (–OH) group at one end. The nitrogen atom connects to two ethyl groups (–C2H5). This combination gives the molecule both water-loving and oil-loving sides, which makes it useful in syntheses and industrial recipes.
Here’s how the structure shapes up: imagine a chain where O-H sits on one end, the nitrogen steps in the middle, and two ethyl legs dangle. Chemists write this as (C2H5)2NCH2CH2OH. On a diagram, you’ll spot a three-atom “core” (NCH2CH2) carrying an –OH group and branching out into two ethyl groups attached to the nitrogen.
Why DEEA Matters Today
DEEA does not stand outside the world of everyday products. If you dig into personal care items or certain paints, you could run into this chemical. It helps adjust pH, prevent corrosion, and keeps solutions stable. I remember touring a detergent lab and listening to technicians talk about how they control pH swings in bulk cleaners. DEEA pops up for its gentle alkalinity and compatibility with many surfactants and solvents. Factories lean on its solubility in both water and many organics. This flexibility reduces waste since single ingredients serve several purposes in one mix.
On the safety front, responsible handling always comes up. DEEA presents limited hazard under normal conditions, but contacts with skin or eyes bring irritation. In labs, gloves and goggles keep operators comfortable and healthy. Producers follow OECD guidelines for chemical use, meeting regulatory requirements in the US, Europe, and Asia. Its low volatility cuts down on inhalation risk—something I’ve noticed as a comfort when mixing small-batch chemicals in an academic setting.
Environmental and Health Considerations
Industrial chemicals bring environmental responsibilities. DEEA, like many amines, breaks down in the environment. Wastewater processes reduce much of its impact. Even with this advantage, companies focus on limiting releases into waterways—tighter controls keep local ecosystems safer. The EPA tracks use patterns and recommends thresholds, helping manufacturers improve containment and treatment. Over the past few years, increased pressure from consumers nudges the industry toward greener approaches and closed-loop systems, which prevent leaks and chemical runoff. I've seen firms step up routine audits and offer regular safety courses, supporting these shifts.
Opportunities for Improvement
Reading studies, one solution stands out: invest in better solvent recycling systems in factories that use N,N-diethylethanolamine in synthesis. Upgrading containment gear, such as double-layer tanks and smart leak detectors, reduces accidental loss and exposure risk. Knowledge-sharing networks also help smaller companies learn what works. As data on amine emissions grows, the sector can keep improving safe-handling guidelines—and make product innovations that lessen reliance on older, more harmful agents.
What are the safety and handling precautions for N,N-Diethylethanolamine?
Understanding the Risks
N,N-Diethylethanolamine, or DEEA, pops up in all kinds of chemical processes. People expect something harmless when a liquid has little odor and no wild reputation, but in my years working around labs and industrial sites, the stuff that seems least threatening on the surface often carries hard lessons underneath. DEEA can irritate eyes, skin, and the respiratory tract, and breathing in too much vapor at once can mess with your body more than you expect. If you splash some on your skin, you could end up with redness or a rash, and getting it in your eyes stings more than a wasp, leaving long-term problems without a quick rinse. Nobody walks away from chemical exposure proud of ignoring safety practices.
Ventilation: Not Just a Checkbox
Many folks overlook the importance of good airflow, but in shared workspaces, one person’s shortcut can become another’s trip to the nurse. Some think cracking a window counts as ventilation. It doesn’t. Sufficient air movement means running working hoods, using exhaust fans, or carrying out the job outdoors when possible. Vapor build-up in small spaces gets dangerous fast, especially because DEEA isn’t always easy to smell. In places like paint manufacturing or chemical labs, I’ve seen proper ventilation make the difference between business as usual and an incident report.
Skin and Eye Protection: Lessons Learned
You spill once and you remember. Wearing butyl or nitrile gloves—never the thin, cheap stuff—throws up a solid barrier. Even the smallest ripple or splash can end a shift early without goggles. Face shields protect if there's any chance of splashing; ordinary glasses don’t cut it here. Washing up at the sink right after handling saves skin from trouble. Anyone who’s been around chemicals for long sees old-timers with scars from “just a little exposure."
Storage and Labeling: No Corners Cut
Leaving DEEA in a plain bottle or some nondescript container invites disaster. A sturdy, well-labeled tank, kept away from acids or strong oxidizers, stops unwanted reactions. Secure shelving, away from heat and sunlight, keeps accidents at bay. I once saw a rookie leave an open beaker too close to nitric acid – that mess taught the room a lesson in chemical compatibility. Cross-contamination lies behind any number of workplace emergencies.
Spill and Emergency Readiness
Preparation counts. Spill kits aren’t just for show—the right absorbent and neutralizer put out a small chemical fire before it gets big. Teachers drilled us on knowing emergency shower and eyewash locations by heart, and those minutes saved when something hit the fan. Practicing your reactions makes the real thing a lot less chaotic.
Training: The Best Defense
Every person joins a workplace with different instincts. A day spent going over safety data sheets and handling plans pays off for years. Instead of glossing over the risks, training brings everyone onto the same page. I once got a job at a factory that took its hazmat drills seriously. We grumbled at first, but the first time a drum leaked, everyone’s quick action kept it from being a real problem.
Moving Forward
DEEA isn’t public enemy number one, but treating it lightly opens the door to regret. Rules written on the wall or in the handbook mean little if people don’t respect the substance in their hands. Real safety comes from small habits, honest training, and never assuming one exposure won’t matter. Taking care with DEEA keeps it just another part of the job—and nothing more.
What are the storage requirements for N,N-Diethylethanolamine?
Why Safe Storage Matters
My years around chemical storage facilities taught me respect for the quirks of specialty amines. N,N-Diethylethanolamine sits in that family of chemicals folks sometimes underestimate, especially in paint and gas treatment applications where its value makes it common enough to breed routine. But routine breeds mistakes, and mistakes with this stuff can cost health—or even lives. Making mistakes here simply isn’t worth it.
The Temperature Factor
N,N-Diethylethanolamine stays liquid at room temperature, which leads some to think it handles like any other solvent. That approach courts trouble. Even though you’d rarely see it freeze or turn volatile automatically, the optimal storage temperature hovers between 20°C and 30°C. Anything much higher, you risk increased vapor pressure and potential container deformation—nobody enjoys the sudden BOOM of failed drums. Store it in a climate-controlled facility. Relying on luck with seasonal temperatures usually doesn’t turn out well.
Containment: Choosing the Right Material
Not every barrel or tote keeps this chemical stable. I’ve watched folks pump this amine into carbon steel tanks and watch as rust blooms within months. Corrosion doesn't just shave years off the container; it contaminates the product and brings risks for leaks. Stainless steel tanks (304 or 316 grade), epoxy-lined carbon steel, or HDPE drums hold up much better. Skip aluminum and copper—corrosion eats them alive. Trusting the right materials preserves quality and guards workers.
Ventilation and Fume Safety
Spent my share of evenings clearing up accidents where fumes drifted into storage spaces. Though N,N-Diethylethanolamine isn’t the most acutely toxic amine, inhalation causes nasty respiratory irritation and headaches. Properly engineered ventilation—local exhaust hoods, floor-level vents, and regular air change—takes a lot of danger off the table. Never think a room full of drums can stay sealed and safe. Fresh air flow matters in the long term.
Keeping Water and Sunlight at Bay
Job sites sometimes keep containers outside “just for a few days”—rain finds a way in, every time. Water means hydrolysis, and over time, unwanted changes in the amine mess with process consistency and safety. Direct sunlight speeds up those changes, degrades seals, and increases container pressure. Store indoors, away from sources of water, and block out sunlight in any predictable way. Sometimes all it takes is a neglected skylight for a problem to sneak up on you.
Labeling and Emergency Preparedness
Labels shouldn’t fade or fall off. Clear, chemical-resistant labeling keeps new hires and emergency teams from guessing in a crisis. From personal experience, missing labels turned a small spill into an expensive disaster, as responders scrambled to find the SDS. Always keep a copy of the safety data sheet next to your storage area and make sure eye wash stations and chemical showers work—untested isn’t the same as reliable.
Learning from the Real World
OSHA has plenty to say about chemical storage—and their guidance isn’t there just to avoid fines. Many injuries link back to overlooked basics: the right drum, a dry space, good air, and proper signs. Regular audits and walk-throughs catch issues long before they turn serious. Any company or lab that keeps these lessons in focus finds more peace of mind and far fewer close calls.
Is N,N-Diethylethanolamine hazardous to health or the environment?
Understanding What It Is
N,N-Diethylethanolamine crops up in plenty of products across different industries. You’ll find it in the making of pharmaceuticals, dyes, cosmetics, and gas treatment processes. It pops up a lot closer to home than you might think, since some manufacturers rely on its ammonia-like qualities for cleaning agents and corrosion inhibitors. I’ve seen folks work with the stuff wearing gloves and goggles, but it’s worth asking—should regular people worry about its effects on health or the environment?
Getting Practical About Health Risks
Not every chemical with a hard-to-pronounce name spells doom, but N,N-Diethylethanolamine brings some reasons to be careful. Lab testing shows this chemical is an irritant to skin, eyes, and the lungs. Spend time in its presence without good ventilation, and you can end up coughing or feeling your throat sting. If the liquid gets on your skin, redness and pain often follow. I’ve seen folks breaking out in rashes after accidental spills. Take it a step further—the vapor isn’t friendly to the nose, so working in a stuffy room won’t do anyone any favors.
The bigger worry shows up with repeated exposure. There’s evidence that chronic contact can hurt the liver or kidneys. Some animal studies hint at problems with neurological function over time. No one loves the idea of slow and silent harm. Safety experts don’t consider this a cancer risk with ordinary handling, but people should avoid letting it hang around inside their bodies, especially at work.
Concerns for the Greater Environment
What happens outside the lab or factory matters just as much. N,N-Diethylethanolamine doesn’t stick around in the soil forever. Microbes break it down pretty quickly in the right conditions. That helps, but the story doesn’t stop there. It stays soluble in water, and when large quantities wash into rivers or lakes, problems can follow. Aquatic life doesn’t bounce back easily from chemical shifts—not every fish or frog can tolerate a sudden dose of solvent. From everyday spills to accidental dumps, the fallout can accelerate plant growth in water bodies, sparking algae blooms that choke out native species and reduce oxygen levels. Fast breakdown is better than none, but every chemical dumped into water sets off a chain of consequences.
What Makes a Difference
A little human caution does a lot. Every warehouse or plant worker who puts on gloves and works under a fume hood limits preventable health risks. Cleanup routines matter—wiping spills right away, storing containers tightly sealed, and using basic protective gear keep accidents from turning into medical bills. From my own days working with chemical stocks, I learned not to skimp on these steps. For casual users, reading the labels and paying attention to warning symbols goes a long way. Sometimes only a handful of seconds stand between you and a trip to the emergency room.
On the bigger scale, regulation steps in. Factories face discharge limits that force them to clean water before it returns to public systems. Agencies track workplace exposure and restrict concentrations allowed in cosmetics or dyes. These aren’t just idle rules; agencies like the EPA and OSHA look at real-world incidents and update limits to keep up. Local communities can push for even tougher standards, especially where water supplies or sensitive habitats lie nearby.
What to Take Away
No one panics over every chemical; most of us couldn’t name the agents in our cabinets. Still, N,N-Diethylethanolamine carries enough risk to deserve respect. Using the right protection, cleaning spills, and speaking up for better regulations go further than worry. If your job or neighborhood puts you near this chemical, taking it seriously helps everyone breathe a little easier.
| Names | |
| Preferred IUPAC name | 2-(Diethylamino)ethan-1-ol |
| Other names |
DEEA Diethylethanolamine N,N-Diethyl-2-hydroxyethanamine 2-(Diethylamino)ethanol |
| Pronunciation | /ˌɛnˌɛn.daɪˌɛθaɪl.ɪˈθænəloʊˌəmiːn/ |
| Identifiers | |
| CAS Number | 100-37-8 |
| Beilstein Reference | 604080 |
| ChEBI | CHEBI:53067 |
| ChEMBL | CHEMBL14204 |
| ChemSpider | 7405 |
| DrugBank | DB03620 |
| ECHA InfoCard | 100.003.565 |
| EC Number | 205-348-9 |
| Gmelin Reference | 8228 |
| KEGG | C01710 |
| MeSH | D004528 |
| PubChem CID | 8037 |
| RTECS number | KL8575000 |
| UNII | 7W37S606NH |
| UN number | UN2672 |
| CompTox Dashboard (EPA) | urn:uuid:10d9e4e1-8b6a-4fe1-b720-ba4a7a2c89e8 |
| Properties | |
| Chemical formula | C6H15NO |
| Molar mass | 89.14 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Ammonia-like |
| Density | 0.89 g/cm3 |
| Solubility in water | Miscible |
| log P | 0.34 |
| Vapor pressure | 0.38 mmHg (20°C) |
| Acidity (pKa) | 10.89 |
| Basicity (pKb) | 4.98 |
| Magnetic susceptibility (χ) | -7.65 × 10⁻⁶ |
| Refractive index (nD) | 1.439 |
| Viscosity | 2.6 mPa·s (20 °C) |
| Dipole moment | 2.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 287.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -393.4 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3726.6 kJ/mol |
| Pharmacology | |
| ATC code | C04AX21 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H315, H319, H332 |
| Precautionary statements | H302, H312, H314, H332 |
| Flash point | 102 °C |
| Autoignition temperature | 410°C |
| Explosive limits | 1.5% to 9.5% |
| Lethal dose or concentration | LD50 (oral, rat): 1620 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2,080 mg/kg |
| NIOSH | NIOSH: KK8575000 |
| PEL (Permissible) | PEL: 10 ppm (TWA) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Triethanolamine Diethanolamine Monoethanolamine N,N-Dimethylethanolamine Choline Diethylamine Ethanolamine |

