N,N-Diethylacetamide: A Down-to-Earth Look at an Industrial Solvent
Historical Development
Back in the mid-twentieth century, the chemical world started to pay closer attention to the family of acetamides. N,N-Diethylacetamide popped up as chemists sought replacements for more hazardous solvents and reagents. Decades ago, places pushing for better workplace safety steered the search for less volatile, less toxic options. This compound settled into research benches during the rise of pharmaceuticals and specialty chemicals. In practical terms, its appearance helped many labs steer away from harsher alternatives like dimethylformamide or pyridine, carving out a space in industrial chemistry and academic investigations alike.
Product Overview
I’ve seen N,N-Diethylacetamide turn up in various research projects thanks to its simple structure and problem-solving abilities. With two ethyl groups plugged into the nitrogen of acetamide, you get a liquid that’s colorless, nearly odorless, and packs an impressive ability to dissolve both water-loving and oil-loving substances. Folks in factory settings or grad students in academic labs alike recognize it as more approachable than some of the more volatile or strictly regulated solvents. Over time, it’s become clear that what this molecule may lack in glamour, it makes up for in reliability and performance across different tasks.
Physical & Chemical Properties
N,N-Diethylacetamide boils at a point somewhere above the basic acetone or ethanol you find under a kitchen sink, but hangs under what you see with long-chain amides. Its liquid range works well under normal lab and factory conditions. It doesn’t burn as aggressively as more volatile solvents, but it supports combustion if the temperature climbs. Unlike polar protic solvents, it won’t help you out in hydrogen bonding scenarios, but with its dipolar nature, it takes on a solid roster of substrates. You notice the liquid mixes with water, yet it still gets along with typical nonpolar solvents—this kind of duality comes in handy for specialty reactions. Its chemical stability generally gets the thumbs-up except under harsh acidic conditions or extreme heating.
Technical Specifications & Labeling
The specifics for N,N-Diethylacetamide run along familiar lines for anyone buying industrial chemicals. You spot purity levels above 99%, low moisture content, and tests for unwanted byproducts. Labeling almost always highlights the need to handle the liquid in a ventilated workspace and keep it away from open flames. In daily experience, these points are more than bureaucratic tape—they speak to actual safety observations in workshops and schools, and everyone gets used to watching the labeling before things get going.
Preparation Method
Making N,N-Diethylacetamide calls for a reaction between diethylamine and acetic anhydride or acetyl chloride. This approach has held up through the years. Chemists like the straightforward mechanism—no fancy fancy purification equipment, just the right temperature, a bit of stirring, and controlling the exothermic reaction. Older methods needed more distillation, but recent tweaks improve the throughput and limit dangerous byproducts. Refineries and chemical plants now scale up this batch process with good returns, making the compound widely available.
Chemical Reactions & Modifications
N,N-Diethylacetamide stands up to a host of basic reactions. People often find it useful as both a reactant and as a solvent when testing new pathways for synthesizing pharmaceuticals or specialty plastics. Under the right pressure and catalysis, you can substitute or chop the nitrogen’s side chains, open up the acetamide backbone, or attach new rings. Because it doesn’t react strongly with bases or mild oxidizers, the compound serves as a platform for reactions that need a non-reactive solvent. Academic teams, especially those hunting for new pharmaceuticals or dyes, have relied on its stability and pliability to drive multi-step syntheses to completion.
Synonyms & Product Names
Over the years, N,N-Diethylacetamide has shown up on bottles under names like DEAA and Diethylacetic acid amide. Older chemical catalogs sometimes list it as Acetic acid, diethylamide. This mixing up of names confused plenty of early-career chemists, and even seasoned workers have squinted at order forms. In practice, recognizing its synonyms clears up a lot of the headaches around ordering and regulatory paperwork, especially where local naming conventions shift between academic and industrial circles.
Safety & Operational Standards
Anyone who’s worked around N,N-Diethylacetamide knows the routine: grab gloves, set up the fume hood, and double-check spill kits. The liquid isn’t the worst offender in the solvent section, but it still irritates skin, stings the eyes, and could stress the liver or kidneys if mishandled. The chemical world steadily tightens guidance around storage and disposal. Safety audits at research labs point out the risks of leaving it uncapped or exposing it to high temperatures. From a practical side, these operational standards mean new users quickly develop habits that minimize risk, while experienced hands keep everyone honest about chemical hygiene.
Application Area
N,N-Diethylacetamide broke into the scene among pharmaceutical researchers looking to dissolve molecules that wouldn’t budge for simpler solvents. Agrochemical plants use it to help mix and stabilize active ingredients in pesticides and herbicides. Textile manufacturers soak fibers with it to drive in dyes, boosting product consistency and vibrancy. Specialty adhesive companies count on it for blending certain resins. Academic labs appreciate it for pulling double duty as both a medium for synthesis and as a basic solvent. This adaptability means it pops up in pilot projects, scale-ups, and even in environmental testing as a sample prep aid.
Research & Development
As safer solvents have come under the spotlight, researchers look for new roles or improved blends involving N,N-Diethylacetamide. There’s interest in reducing emissions and waste by recycling or purifying the spent solvent. R&D teams use it as a baseline for creating next-generation, less toxic, or more biodegradable versions. The push to meet strict environmental rules sparks innovation around both process chemistry and closed-loop systems where the solvent cycles back into production. Decades of published research cover structure-activity relationships, new use-cases, and deeper dives into how this liquid helps turn difficult reaction mixtures into final products. These projects point to both its established value and a desire for continual improvement in lab and plant operations.
Toxicity Research
The story on toxicity hasn’t always been clear-cut. Animal studies show liver and kidney effects at high chronic doses, so regulatory agencies set occupational exposure limits to keep labs honest. Thankfully, air monitoring and proper ventilation take away much of the practical risk for technicians handling the solvent by the quart. Short-term skin exposure mostly causes irritation rather than deep harm, but everyone quickly learns to suit up to avoid the hassle and discomfort. Environmental impact draws attention too, with modern waste treatment focused on capturing any runoff before it reaches groundwater. As offices and plant floors have shifted toward greener chemicals, ongoing toxicity research pushes the industry to refine workplace standards and disposal practices.
Future Prospects
Looking down the road, the role of N,N-Diethylacetamide in industry will likely keep shifting. As new environmental and workplace rules get written, companies start balancing the compound’s practical strengths with the pressure to move toward safer, greener alternatives. Researchers see it staying relevant in specialized synthesis work, but at the same time, biobased and less hazardous solvents gain ground in the mainstream. Industrial chemists with years of hands-on experience still appreciate its reliability, especially in pilot-scale or custom operations, but the next batch of process upgrades and greener innovations may push it toward a more niche role. This transition won’t happen overnight, and for now, plenty of sectors still count on N,N-Diethylacetamide as a trusted workhorse.
What is N,N-Diethylacetamide used for?
A Look at N,N-Diethylacetamide in Practice
Most people haven’t heard of N,N-Diethylacetamide, and that’s pretty common for a chemical more often found in labs and factories than household cupboards. Chemists know it as a clear liquid with a faint fragrance. Over the years, it has carved out a niche mainly as a solvent. Solvents act a bit like mediators at a party—they break the ice and help other ingredients mix. Companies depend on N,N-Diethylacetamide to get dyes and drugs into the shape and form they need for actual use.
Smoothing Production in Pharmaceuticals and Dyes
The pharmaceutical industry runs on strict timelines and close attention to purity. N,N-Diethylacetamide doesn’t mess around with impurities. I once talked with a chemist at a mid-sized pharma plant, and she explained it’s chosen for projects that require stubborn ingredients to dissolve fully. Some medicines just won’t cooperate with common solvents, and this chemical keeps things moving.
Dye manufacturers, on the other hand, rely on it to push color into synthetic fabrics evenly. Getting fabrics to accept dye evenly used to be unpredictable. Once fabric scientists switched to solvents like N,N-Diethylacetamide, the difference was night and day—the colors popped, and leftover residue after dyeing dropped. These improvements matter to anyone who’s ever bought a shirt and found it faded before the first wash.
Industrial Flexibility Pays Off
N,N-Diethylacetamide doesn’t show up on its own in many consumer-facing products. It’s more like the backstage crew member than the lead performer. Its ability to handle both high-heat and low-moisture conditions makes it the go-to for specialists working with challenging compositions that need a push. Paints for machinery, specialty coatings, and even photographic chemicals have called upon it to bridge gaps where other solvents stumbled and slowed the line.
Safety, Waste, and the Ongoing Balancing Act
Safety becomes a serious talking point with any industrial solvent. In research I’ve done and talking with folks tasked with handling these chemicals, improper ventilation or skin exposure leads to headaches and, over time, bigger health concerns. In my city, local regulators require any workplace using significant amounts of N,N-Diethylacetamide to install exhaust fans and keep workers in protective gear.
There’s another layer too—where does all the leftover solvent go? Factories can’t dump spent chemicals down the drain. Companies have faced fines when waste isn’t treated or recycled correctly. Newer innovations involve closed-loop systems that recover solvents for reuse, lightening the environmental footprint. The up-front cost for these systems stings, but over a few years, saving money on waste disposal and new solvent purchases changes the math.
The Push for Greener Alternatives
Conversations keep coming up about using less toxic or more sustainable solvents in industry. Researchers experiment with plant-based alternatives, hoping to find that sweet spot between performance and safety. Until these catch up to N,N-Diethylacetamide’s reliability and cost, the chemical keeps its job as a behind-the-scenes workhorse.
Knowing where and why N,N-Diethylacetamide matters helps anyone watching the chemical, textile, or pharmaceutical industries recognize the choices made each day to keep production moving, all while keeping an eye on the health of workers, the end consumer, and the environment.
What are the safety precautions for handling N,N-Diethylacetamide?
Understanding the Risks
N,N-Diethylacetamide sits on the bench in some labs and factories, but it never really shouts for attention. This clear liquid carries some quiet hazards: it can cause skin and eye irritation and releases vapors that aren’t great for your lungs. Inhalation makes some folks cough or feel dizzy. Long exposure sometimes brings on headaches. If it spills, it might eat through plastics or even paint, and reacting with strong oxidizers can generate extra heat or worse.
Personal Protection Matters
I’ve watched newcomers skip the gloves, shrugging off the warning labels. Those stings and rashes hit fast, and without goggles, a quick splash could sideline anyone for the day. Thick nitrile gloves make a difference. Splash goggles—ones that actually seal around the eyes—beat simple lenses every time. Lab coats or aprons that fit well block the majority of accidents from soaking through regular clothes.
Most forget about vapor, though. Fume hoods cut the risk sharply, especially for those who work with solvents every day. If the work takes place outside a hood or turns into a bigger cleanup, those NIOSH-approved respirators set the standard. Rushing through a task in a rushed room always raises levels of risk, especially with poor ventilation.
Engineering Controls and Good Housekeeping
Equipment matters. Sash heights on hoods, airflow meters, and catch trays make a tough job safer. Anything spilled gets mopped up fast with absorbents meant for organic solvents, not with rags or old towels. A neighbor once used a cardboard box for disposal—within hours, the fumes crept up. Only metal or thick plastic containers, labeled clear, hold discarded solvent safely until disposal.
Solvents shouldn’t share storage with acids or oxidizers. I once saw a shared shelf in a teaching lab that nearly turned a small spill into a big scare. Storing N,N-Diethylacetamide in tightly sealed bottles, away from sunlight and heat, reduces risk. Secondary containment trays catch leaks before they reach the floor.
Training: The Overlooked Lifeline
Reading safety data sheets feels tedious, but knowing what to do during a spill, splash, or fume release makes panic less likely. One real step is rehearsing emergency eyewash and shower drills. I remember an intern who froze, not knowing where the shower handle was after a splash. Regular training boosts confidence more than any warning sign.
Fire drills feel routine but matter for solvents, since vapors can ignite. Fire extinguishers rated for chemical fires work best, not just generic ones. A chemical burn kit stashed nearby saves precious minutes.
Keeping Health at the Center
Exposure limits exist for a reason. Air monitors let teams stay within those guardrails. Anyone feeling faint or sick finishes the day in the nurse's office, not toughing it out at the fume hood.
In workplaces, regular health checkups spot the slow buildup of problems from long-term exposure. Wear and tear catches up before you notice if you're not careful.
Mistakes happen, but respect for N,N-Diethylacetamide’s risks lowers the chances. Experienced workers and fresh faces both share responsibility. Safety doesn’t rest on rules taped to a wall: it shows up in well-fitted gloves, a labeled bottle, and the care taken with every single pour.
What is the chemical formula of N,N-Diethylacetamide?
Understanding N,N-Diethylacetamide
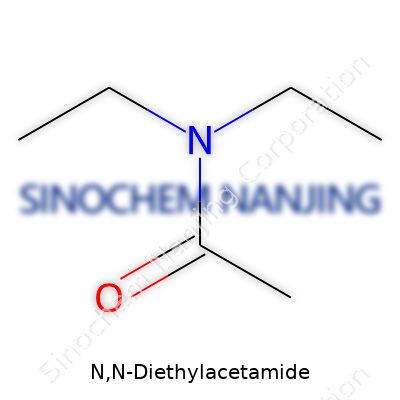
N,N-Diethylacetamide shows up in labs, industrial research, and sometimes even in the classroom. Its chemical formula is C6H13NO. At first, it might sound like just a jumble, but these six carbons, thirteen hydrogens, one nitrogen, and one oxygen create a molecule known for practical versatility. Scientists remember this structure for its ability to dissolve many organic compounds—something that often simplifies experiments or industrial tasks.
What Makes This Molecule Useful?
Many research teams use N,N-Diethylacetamide as a solvent. The balanced structure lets it act as a neutral medium for reactions and extractions. I remember in the university lab, handling glassware filled with various solvents, you could smell a dozen chemicals in the air, but N,N-Diethylacetamide had a distinct, pear-like odor that set it apart. It stood out for reasons besides aroma. Some synthetic chemistry projects would stall without a good solvent, and options like this kept everything moving.
Companies also turn to C6H13NO for its role in making pharmaceuticals, dyes, and even flavors. Engineers consider its low toxicity and ability to mix with water and organic liquids as practical advantages. Chemicals with unpredictable behavior can throw off a production line and waste both money and time. N,N-Diethylacetamide, fortunately, keeps performance consistent within its common applications.
Safety, Knowledge, and Transparency
Every solvent comes with upsides and risks. One overlooked point about N,N-Diethylacetamide is its manageable safety profile compared to other industrial chemicals. Experts recommend gloves and goggles, not hazmat suits, which sends a clear message about practical risk. Still, full understanding does more for safety than any rule book. In one project during my early career, forgetting to label a bottle led to understandable confusion. Labeling and clear knowledge of properties reduce accidents, not just in well-funded labs but in garages and smaller workshops.
Beyond day-to-day handling, the story starts and ends with the chemical itself. C6H13NO, by its structure, resists breakdown from common acids and bases, letting processes run their course without unwanted side reactions. This trait gives students and senior chemists alike peace of mind that changes in temperature or pH won’t compromise results. Open discussion about properties leads to higher confidence, not just for those using the substance but also for regulators and the communities where these chemicals get stored and transported.
Room for Responsible Practice
Respect for chemistry isn’t about fearing every compound; it’s about preparation and transparency. The reality of C6H13NO rests in responsible use—knowing the structure and formula isn’t trivia, it’s the foundation for safe practice. Sharing information, supporting education, and honest reporting matter more than ever in science today. Good habits and up-to-date knowledge protect people and shape chemical progress. The next time a bottle of N,N-Diethylacetamide lands on a bench, reminding ourselves of what’s inside those few characters—C6H13NO—keeps the process grounded and the people involved protected.
Is N,N-Diethylacetamide hazardous to health or the environment?
Navigating Chemical Realities in Everyday Life
Growing up with a dad who worked as an industrial chemist, dinner conversations sometimes included talks about new solvents he’d tested at work. I remember him saying, “Some things smell strong, some things do things to your hands you can’t see, but feel weeks later.” N,N-Diethylacetamide is one of those industrial solvents on the chemical roster. It shows up in processes like pharmaceuticals and pesticides, yet most people have never heard of it.
Hazards You Can’t Always See
N,N-Diethylacetamide doesn’t sound menacing, but looks deceive. Touching or breathing its vapors can cause skin and eye irritation. The stories from workers in chemical plants demonstrate the real risks – redness, headaches, and sometimes feeling dizzy after spills. Chronic exposure remains less understood, but extended contact rarely ends well in chemical settings.
The US National Library of Medicine notes that fumes may irritate the respiratory system, and accidental swallowing could trigger nausea or worse. The CDC points to solvents like this triggering central nervous system depression at high doses. In labs without proper ventilation, you see how symptoms ramp up over time. PPE and decent fume extraction systems become non-negotiable to prevent problems.
Impact Past the Factory Walls
Small spills in a plant go down the drain, but down the street, those chemicals spread. Water makes a lousy shield – N,N-Diethylacetamide dissolves easily, so it slips into groundwater near disposal sites or waste streams. Once in the environment, it doesn’t just break down overnight. Studies on similar amides suggest slow biodegradation, and that means soil and water can stay contaminated for months.
On the toxicology side, aquatic life suffers if there’s a repeated leak; fish and frogs won’t thrive in water laced with solvents. Wastewater oversight around manufacturing sites matters, especially since some countries don’t enforce strict cleanup rules. Europe’s REACH regulations list it as a substance to monitor, reflecting mounting international concern.
What Can Make a Difference
Governments already require hazard labels and safety data sheets, but paperwork alone won’t block every risk. Stronger industrial oversight, better containment, and regular worker health screenings help in the real world. In my experience speaking with safety engineers, they push hardest for incident drills and rigorous inspection cycles, not just the “minimum legally required.”
Community right-to-know laws encourage companies to share what’s stored on-site. In the remote chance of a fire or chemical release, that transparency can save both first responders and neighbors.
On the environmental front, using less hazardous replacements can curb long-term problems. Some companies have shifted to greener solvents for cleaning and extractions, aided by academic research pushing for safer substitutions. A chemist at a large agrochemical firm once told me, “We switched processes because we got tired of worrying about downstream pollution — and we still get good yields.”
Keeping Risk on the Radar
Personal vigilance remains crucial; reading safety sheets, respecting PPE, and asking about local community protections make accidents less likely. N,N-Diethylacetamide isn’t unique in its risks, but it sits as a reminder that chemicals woven into modern industries demand respect both inside and outside the factory gates.
How should N,N-Diethylacetamide be stored and disposed of?
What This Chemical Demands from Careful Hands
N,N-Diethylacetamide is not something you want lying around in a forgotten corner of a laboratory or storage shed. I’ve had my moments with chemicals, and I’ve learned you risk more than a safety citation if you ignore the quirks of each solvent. Storing this compound means more than keeping it behind a locked door. Proper labeling and secure containment go way beyond paperwork. Without a tight seal and the right bottle, even materials with low volatility can turn a storeroom into a hazard zone.
Glass or high-quality plastic containers beat metals for most solvents, and N,N-Diethylacetamide fits in that rule. Some people like the idea of reusing containers, but even trace contamination can mess with purity. Clearly labeled, leakproof vessels belong in a cool, dry place—think ventilated cabinets set well away from heat sources, not on top of some radiator. Anyone who’s worked in a cramped space knows how quickly bottles pile up, so no one wants to dodge broken glass to get at less hazardous supplies.
Why the Air and Ground Matter
Leaving a container open for only a few minutes can expose users to fumes or let the solvent degrade. My own experience—catching a whiff of something after a coworker left a cap loose—reminded me how small mistakes add up. Modern safety data sheets make it clear: N,N-Diethylacetamide should stay in environments with good airflow, not in musty basements or unventilated closets. Filtering air through chemical scrubbers or vents keeps levels below exposure limits and helps protect the lungs of everyone moving through those hallways.
Safe Disposal—No Shortcuts Allowed
Pouring waste down the drain sounds tempting, especially for solvents that look as clear as water, yet that shortcut risks groundwater and public health. Local regulations tightly control disposal channels for chemicals like this one, often insisting waste handlers collect and process the material through hazardous-waste programs. Years ago, a nearby facility ignored this advice and ended up facing big fines and a long list of environmental remediation tasks. That painful lesson still circulates in training sessions today.
Experts recommend separate containers for solvent waste, not mixed with acids, bases, or incompatible materials. Labels should use the full name and hazards—no abbreviations or scribbles. Keeping a detailed log of contents shields staff from both danger and legal headaches. Licensed waste services know how to break down, neutralize, or incinerate solvents safely. Some programs even recycle compatible solvents, lessening the need to manufacture and ship fresh supplies across the globe. That small effort pays off by reducing accidental releases and chemical injuries.
Building a Culture of Respect for Safety
People get careless when they treat chemicals as ordinary supplies. Training matters—the best labs run regular refreshers, going beyond a checklist and into real stories. I’ve watched new technicians respond to drills with nervous energy that slowly shifts into confident habits over time. A practical manual, quick guides posted near storage rooms, and clearly defined contacts for emergencies help, too.
N,N-Diethylacetamide demands a respectful approach from storage to disposal. Attention to detail, teamwork, and honest communication shape a safer workplace. The moments spent labeling, logging, and following disposal rules might seem small, but they build a shield against harm that no insurance policy or first-aid kit alone can provide.
| Names | |
| Preferred IUPAC name | N,N-diethylethanamide |
| Other names |
N,N-DEAC Diethylacetamide N,N-Diethylacetamid Acetic acid diethylamide |
| Pronunciation | /ˌɛnˌɛn.daɪˌɛθɪl.əˈsiː.tə.maɪd/ |
| Identifiers | |
| CAS Number | 142-92-7 |
| Beilstein Reference | 1719809 |
| ChEBI | CHEBI:50137 |
| ChEMBL | CHEMBL14288 |
| ChemSpider | 24309 |
| DrugBank | DB14293 |
| ECHA InfoCard | ECHA InfoCard: 100.005.800 |
| EC Number | 203-815-1 |
| Gmelin Reference | 8286 |
| KEGG | C06161 |
| MeSH | D003943 |
| PubChem CID | 8223 |
| RTECS number | AF3675000 |
| UNII | J2K8A6U9A7 |
| UN number | UN8416 |
| CompTox Dashboard (EPA) | DTXSID5077607 |
| Properties | |
| Chemical formula | C6H13NO |
| Molar mass | 129.19 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Amine-like |
| Density | 0.926 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 0.54 |
| Vapor pressure | 0.36 mmHg (at 25 °C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | pKb = 13.10 |
| Magnetic susceptibility (χ) | -58.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.439 |
| Viscosity | 1.25 mPa·s (20 °C) |
| Dipole moment | 3.59 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2522.2 kJ/mol |
| Pharmacology | |
| ATC code | N01BX02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 85 °C (closed cup) |
| Autoignition temperature | 170 °C |
| Explosive limits | Explosive limits: 1.7–10.9% |
| Lethal dose or concentration | LD50 oral rat 1,150 mg/kg |
| LD50 (median dose) | LD50 (median dose): 480 mg/kg (oral, rat) |
| NIOSH | RN 102-23-8 |
| PEL (Permissible) | PEL = "10 ppm (NIOSH) |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
N,N-Dimethylacetamide N,N-Diethylformamide N-Ethylacetamide Acetamide N,N-Dipropylacetamide |

