Looking at N,N-Dibutylaniline: A Chemical with Deep Industrial Roots and Modern-Day Challenges
Historical Context and Standard Developments
N,N-Dibutylaniline sprang from an era when organic chemistry powered countless industrial leaps. In the early days of the synthetic dye industry, chemists began experimenting with aniline derivatives to expand the range of colors for textiles. Aniline itself, first isolated in the nineteenth century, became a workhorse compound, with the addition of bulky butyl groups (giving rise to N,N-Dibutylaniline) adding new dimensions for solubility and chemical reactivity. This molecule started out as just one of many in a long line of aniline-based substances, but over time, it found utility not just in dyes but also in areas like rubber processing and specialty chemicals. Its journey highlights a hands-on approach—experiment, observe, adapt—that’s kept the industry moving for decades, and continues to influence how chemists tackle new challenges with legacy compounds.
What Makes N,N-Dibutylaniline Stand Out
If you take a closer look at N,N-Dibutylaniline, you notice its straightforward structure: an aniline ring attached to two butyl chains on the nitrogen. Chemically speaking, those butyl groups do more than bulk up the molecule. They impact properties like viscosity, boiling point, and oil solubility—which opens the door for uses where standard aniline would fall short. A build-up of those molecular tweaks allowed inventors and engineers to tune chemical blends for everything from antioxidants in lubricants to intermediates in dye synthesis. Unlike the flashier, more talked-about chemicals, N,N-Dibutylaniline keeps showing up in technical reference books for a reason—it just works where it’s needed, and it’s held on to that position in various manufacturing lines.
Physical and Chemical Qualities from an Insider’s View
Many chemical workers first recognize N,N-Dibutylaniline by its slightly oily consistency and faint, fishy odor. It pours more easily than dense industrial resins and mixes readily with non-polar solvents. That sort of behavior matters during bulk handling. Its boiling point lands above 300°C, which makes it fairly heat stable. On the chemical side, this molecule mostly keeps to itself—stable under normal storage and use, but able to undergo classic reactions involving the aromatic ring or the attached amine. Acids will protonate the nitrogen; oxidants and halogenating agents can modify the ring. All this might sound technical, but for someone with a lab bench perspective, it means you get predictable results in processes that rely on chemical reliability, like specialty dye manufacture or polymer modification.
Technical Details and Labeling Nuances
Talking to product handlers, you hear the same wish list over and over—clear labeling, up-to-date hazard symbols, and easy-to-find batch numbers. With N,N-Dibutylaniline, details count: workers rely on proper hazard assessment and labeling to avoid mix-ups and keep processes trouble-free. Each drum comes stamped with proper regulatory classifications. Any lapse here can put both teams and downstream users at risk, especially given the compound’s toxicity profile. In my own experience, misplaced or outdated labels sting everyone in the value chain, leading to headaches often more costly than the chemical itself.
Manufacturing Methods and Downstream Chemistry
The main production approach for N,N-Dibutylaniline hasn’t changed much in decades. Chemists start by heating aniline with a stoichiometric excess of butyl bromide (or a similar alkylating agent), usually in the presence of a solvent like ethanol and a base to mop up hydrogen bromide. Once the bubbling settles down and cooling finishes, you get a layer of product needing purification. Over the years, improvements came not from new chemical tricks but from scaling reactors safely and recycling solvents efficiently. The real action happens later—N,N-Dibutylaniline serves as a parent molecule for further modifications, such as sulfonation, nitration, and oxidative coupling. These reactions give rise to dyes, intermediates, and performance additives. Each step demands precision and a respect for the safety limits of organic chemistry.
Synonyms and Naming—A Web of Chemical Identity
Chemists sometimes joke that a compound’s naming confusion offers a career’s worth of frustration. N,N-Dibutylaniline shows up under names like N,N-dibutylbenzenamine, aniline dibutyl, and aniline, N,N-dibutyl-. In regulatory filings, safety databases, and even supplier invoices, these synonyms crop up. Overlooking a variant name during inventory or compliance checks—especially in a busy warehouse—brings trouble fast, from lost time to regulatory audit findings. In real life, clarity in naming becomes a basic requirement, one that shouldn’t be left to chance or shortcut.
Operational and Safety Realities on the Floor
Anyone who has worked with aromatic amines knows that proper ventilation and protective gear are not optional. N,N-Dibutylaniline isn’t as notorious as its nitroaromatic relatives, but it’s no lightweight either. Absorption through the skin can trigger unpleasant symptoms, so gloves and splash-proof goggles are standard. Spills demand swift cleanup, with workers trained to control exposure. Over years in the industry, I have witnessed all too often the cost of lapses—minor ones like headaches and skin rashes, and major incidents that bring operations to a halt. Companies invest in ventilation upgrades and better PPE not as PR moves, but because experience shows there’s no substitute for diligence with compounds like this.
How Industry Adopts and Applies N,N-Dibutylaniline
Unlike the mainstream chemicals filling tanker ships, N,N-Dibutylaniline tends to reach specialized users. You’ll see it pop up in custom dye synthesis, as a chain stopper in certain polymerizations, and as a rubber antioxidant. It plays a stealthy but essential role in keeping tires flexible and dyes consistent, helping products hold up under heat and stress. Research labs reach for it during the hunt for novel organic compounds, taking advantage of its chemical backbone. While it doesn’t headline in mass-market consumer goods, its influence appears in all sorts of industrial corners—each with detailed operational needs that reward experience and careful handling.
Where Research and Development is Heading
Chemical innovation builds slowly, layer by layer. For N,N-Dibutylaniline, much of the recent work circles around sustainability and reduced toxicity. Development teams are studying how the molecule behaves under advanced catalytic systems or whether substituting bio-based feedstocks could lower its environmental cost. Papers surface each year showing tweaks to the core structure to adapt dyes that need to handle harsher conditions or be more environmentally acceptable. Large and small companies alike are looking for routes that cut waste, streamline purification, and limit worker exposure, because regulations keep tightening and corporate responsibility is now inseparable from profit.
A Look at Toxicity and Health Research
Toxicologists take a measured approach—testing exposure levels, running animal studies, and tracking what happens when compounds turn up in waste streams. N,N-Dibutylaniline has garnered flags in various studies for potential skin and mucous membrane irritation. Some long-term exposure reports hint at organ impact in high-concentration environments, echoing findings for many aromatic amines. These results shaped safety protocols that stress minimal exposure and short handling time. Recent literature looks at breakdown products in wastewater and how persistent residues behave in the environment. These efforts matter because every new data point guides policy, and policy, in turn, shapes the lives of everyone on the production floor.
Thinking Ahead: What’s Next for N,N-Dibutylaniline
Looking forward, the path for N,N-Dibutylaniline will depend on balancing its industrial strengths with mounting pressure for safer, greener chemicals. Alternative synthetic routes that use less hazardous reagents are picking up steam. Regulators around the world are likely to sharpen their oversight of aromatic amines, especially as concerns about occupational exposure move from technical journals to public debate. Factories weighing future investments should consider not only immediate compliance needs but also how to retrain staff and redesign safeguards. For anyone with years spent in process chemistry, this evolution isn’t new, but each shift demands unflinching honesty about risks and readiness to try smarter, cleaner ways of doing business. That hard-won attitude—taking pride in both craft and care—keeps legacy chemicals like N,N-Dibutylaniline helpful without ignoring the costs.
What is N,N-Dibutylaniline used for?
What’s Special About N,N-Dibutylaniline?
Ask anyone who has worked in the chemical sector about N,N-dibutylaniline, and you’ll get a quick nod. This compound, running under the radar for most folks, plays a clear role in industrial production. Most often, you’ll find it linked with dyes, pigments, and sometimes rubber manufacturing. It isn’t a chemical that gets talked about in a crowded room, but it sits behind the color in plastics and fabrics around us. Color fastness, or the ability of a product to hold its shade, counts on additives like this. It serves as an intermediate—an in-between building block—that helps chemists connect other molecules.
Why Does It Matter in Manufacturing?
A good workplace uses science to solve practical problems. Textile printing, automotive coatings, and furniture finishes each deal with demands of both looks and durability. By lending stability and certain chemical traits, N,N-dibutylaniline helps production lines churn out products that catch the eye and stand up to the elements. My early days hitching rides to work in a furniture plant put me next to fabrics that faded and wood finishes that peeled. Seeing the shift to longer-lasting colorants not only cut waste but saved money for small manufacturers. That shapes lives and livelihoods.
Health and Safety Weigh In
No industrial chemical walks into a factory without questions about safety. N,N-dibutylaniline demands respect. Exposure risks run from basic skin irritation to more serious outcomes with high or prolonged contact. I learned about proper gloves and ventilation from old hands on the job—they’d seen what accidents cost. Both in labs and production floors, following safety data sheets becomes as routine as punching a clock. Factories have to invest in training and personal protective equipment, or they run the risk of paying a much higher price if things go wrong.
The Push for Responsible Chemistry
As markets and governments clamp down on hazardous substances, many companies look for ways to make processes safer without losing the performance chemicals like N,N-dibutylaniline bring. Regulations keep changing, shaped by new toxicology research and pressure from both advocacy groups and big buyers. Manufacturers often face the tough call of reformulating products. Environmental impact echoes through these decisions; spills and mishandling can end up polluting water, impacting entire communities. The chemical sector watches global standards and pushes for closed-loop systems, recycling, and greener methods that don’t turn a blind eye to future generations.
Seeking Smarter Solutions
From my experience, change never comes easy, especially in older industries. It takes investment to test alternatives or capture emissions. Yet, the push to phase out riskier chemicals has led to clever innovations. Some labs turn to other amines, while big companies share techniques for safer handling. N,N-dibutylaniline may not leave our supply chains overnight, but smarter use and openness about its risks point the way forward. End-users and workers alike deserve clear information, honest labeling, and ongoing efforts to limit harm while still meeting real-world demands.
What is the chemical formula of N,N-Dibutylaniline?
The Basics Never Lie: C16H27N
N,N-Dibutylaniline carries the chemical formula C16H27N. Counting twelve carbons, twenty-seven hydrogens, and a single nitrogen packs a story—a molecule straddling the world of synthetic chemistry and industry. The core of this structure features a benzene ring attached to an aniline nitrogen. Two butyl chains pull the electron-rich nitrogen away from its aromatic roots, changing not just its chemistry but its behavior in the lab and the market.
Behind the Formula: Real-World Impact
Reflecting on my own bench work, a molecule like N,N-Dibutylaniline always stood out. The formula isn’t just about memorization for an exam; it signals how a molecule slides into so many applications, often quietly. Sitting on the shelf, flanked by its chemical cousins, it often snuck into dyes and specialty chemicals. There’s no glossing over the fact that its slick, oily form and faint odor pointed to a complex life in industry and research.
Chemists don’t just eye this molecule for fun. Its structure, dictated by those sixteen carbons, twenty-seven hydrogens, and one nitrogen, creates a neat balance between being reactive and being stable. That balance lands it in colorants, antioxidants, and accelerators for the rubber world. Go through the records at a rubber plant or a textile dye house—C16H27N shows up with regularity. It pops up because it does its job, performing under heavy stress, contributing to products that see tough environments.
Safety Doesn’t Wait: Knowledge Protects
Every chemical formulation matters, especially when it comes to human health and environmental safety. N,N-Dibutylaniline is no exception. Reviewing safety data sheets, laboratory teams learn pretty fast that personal protection comes first. Long-term exposure, especially without good ventilation, leads to health problems. That’s not speculation; multiple studies involving aromatic amines report links to skin and respiratory irritation. Waste management guidelines require this molecule to be handled as a potential environmental hazard—spills can contaminate waterways or soil, so responsible handling gets non-negotiable status in the workplace.
Seeing students or young techs mishandling a chemical because they didn’t get the basics drilled into them—a moment for frustration and a reminder that every formula, written on the side of a bottle, means something. The science doesn’t get left in the pages of a textbook; it tracks home in the form of workplace practices and life-long health impacts.
Better Understanding Paves the Way for Solutions
Chemical literacy drives safer work and smarter innovations. Companies benefit when they invest in training, not just compliance. Having a team that knows what C16H27N can do, and what hazards it brings, reduces accidents. Regulations keep industries honest, but it takes a culture where everyone wants to dig into the data to make real progress. In my time working with chemical safety officers, the labs that maintained thorough shared logs and frequent safety meetings saw fewer emergency calls.
Sustainable chemistry keeps gathering steam. Many in the field push for alternatives that meet or beat the performance of legacy compounds like N,N-Dibutylaniline, without the environmental and health concerns. My hope is that next-generation innovations grow from a deep-rooted respect for the molecules we handle today—and a clear-eyed view of their formulas and properties.
Is N,N-Dibutylaniline hazardous to health?
Sizing Up N,N-Dibutylaniline
I’ve worked in labs and spent enough years around industrial chemicals to know that N,N-Dibutylaniline doesn’t often make headlines. Still, anyone handling or living near places using this compound deserves real answers. Used in plastics, dyes, rubber, and as a chemical intermediate, this oily liquid has a reputation for being less infamous than some of its chemical cousins. But “less infamous” hardly means safe.
What Actually Happens if You’re Exposed?
Breathing it in or getting it on your skin sets off red flags. Research and hazardous materials data show it irritates eyes, skin, and lungs. Short contacts can trigger rashes, swollen eyes, and a cough that lingers far too long. Getting a concentrated dose can bring headaches and dizziness. Longer-term exposure, especially in factory settings, holds extra risks. In animal studies, longer exposure throws the liver and kidneys for a loop. These organs work hard to filter out the toxic stuff. Damage here rarely stays hidden: think trouble with fatigue, swelling, or off-color urine.
What Experts and Agencies Say
The National Institute for Occupational Safety and Health (NIOSH) and the U.S. Environmental Protection Agency (EPA) track chemicals like N,N-Dibutylaniline. Available data points to concerns—long-term exposure connections to organ damage and uncertainty around possible links to cancer. No solid proof it causes cancer in people, yet the compound’s chemical family includes several substances with confirmed cancer risk.
Safety guidelines don’t leave a lot of wiggle room. Industrial workers wear gloves and face shields, vent workspaces, and rely on strong air extraction fans. In the off chance a spill occurs, people evacuate the area and clean it up in heavy-duty gear. None of these actions get taken unless real hazard exists.
Looking Beyond the Workplace
Factories keep legal limits in mind, but what about neighborhoods close by? Runoff or fumes could reach outside those plant walls. Public health protections—tough monitoring, regular inspections, quick fixes if leaks happen—help, yet enforcement sometimes falls short. If you live near an industrial zone, it pays to stay up to date on what chemicals might be getting processed nearby, either from public reports or local environmental groups.
Reducing the Danger
Chemists have found ways to cut down the risk at work through sealed systems and better training. Swapping out this compound for less harmful alternatives isn’t always simple but deserves every effort. In my own experience, a safety culture that involves everyone—from the floor worker to the CEO—makes the biggest difference. People speak up about unsafe conditions and know how to respond if there’s a spill or leak.
A Few Real Steps Toward Safety
Every workplace using N,N-Dibutylaniline should keep up with proper protective gear and training on emergency procedures. Regular air quality checks and health screenings for those working with it offer another layer of protection. Communities living near facilities that process chemicals like this gain peace of mind by pushing for transparency and supporting local watchdogs.
Chemicals like N,N-Dibutylaniline aren’t going away overnight. We have the knowledge to make handling safer and keep both workers and the public out of harm’s way—if leaders stay vigilant and everyone pulls together.
What is the appearance and odor of N,N-Dibutylaniline?
A Quick Visual Check
N,N-Dibutylaniline—a name with a scientific ring, though not many outside of certain labs or factories would see a bottle of it on the regular. In person, you notice it looks like a thin, colorless to pale yellow liquid. Nothing fancy or flashy here, just a straightforward oily consistency that clings to the glass if you tilt the container. No crystals, no powder, just a liquid that shows a bit of light yellow if you catch it against a white background. Some folks might wonder if that slight tint means the chemical has aged or picked up impurities, but even a fresh batch holds that pale tone.
The Odor: Not for Everyone
The real feature, though, sits in the smell. N,N-Dibutylaniline comes with one of those sharp, ammonia-like odors that gets noticed quick. The moment a cap’s off, you’re hit by a pungent kick—strong enough to hang around the room if the space isn’t ventilated. The scent isn’t sweet or perfumed; it’s got more in common with industrial solvents or cleaning agents, which signal right away this isn’t something to sniff for curiosity’s sake. Your nose tells you this is a chemical you respect, not one you mess with casually.
The Human Side of Chemical Safety
Smelling it brings memories of old college labs, where fume hoods ran all day and certain benches carried a faint, lingering chemical aroma. The compounds with the sharpest scents almost always demanded extra care—eye protection on, gloves snapped tight, even for a routine transfer. Even a quick exposure, if you’re too close, can make eyes water or throat itch. Walking into those labs for the first time, I learned early on how sensory cues—like a distinctive odor—become the body’s own warning system.
N,N-Dibutylaniline’s odor helps ensure people don’t ignore its presence. Reports from industrial settings back that up. If a spill happens, everyone knows it within moments. This makes fast response and safety training essential in places where this chemical is handled—ventilation systems, spill kits, and clear labeling matter far more in reality than in guideline documents.
Why the Details Matter
Accurately recognizing both the look and smell of chemicals like N,N-Dibutylaniline isn’t just academic. Accidents in storage or mixing come from grabbing the wrong bottle or missing a sign of contamination. Workers who know what to expect—both visually and by scent—catch problems early. That’s why safety data sheets and real-world training always put a spotlight on sensory details alongside numbers like melting point or density.
The American Conference of Governmental Industrial Hygienists and many chemical safety authorities still push for environmental controls and personal protective equipment. With a compound that announces itself so boldly, nobody can claim they missed the warning signs. Companies who support ongoing training, replace old PPE, and invest in proper ventilated workspaces lower the risk for everyone involved—and make room for fewer emergency stories in the breakroom.
Building Safer Habits
N,N-Dibutylaniline’s appearance and odor send a message: respect the tools you work with. Experience teaches that familiarity breeds carelessness, but sharp smells and distinct colors snap people back to attention. Everyone, from new hires to seasoned chemists, benefits from taking a second look—and a cautious sniff—before diving in.
How should N,N-Dibutylaniline be stored and handled?
Why Care About Storing Chemicals?
Anyone who spends time in a lab or manages chemicals knows accidents don’t happen just to beginners. N,N-Dibutylaniline can seem easy enough to handle—clear, oily, nothing visibly explosive about it. But good habits save trouble, property, and health. I've seen plenty of labs where “just another solvent” turned into a mess, all because someone cut corners with the basics.
Keep It Cool, Keep It Dry
N,N-Dibutylaniline doesn’t like moisture. Water leads to slow, unpredictable breakdown. Humidity or even minor leaks can stir up chemical changes over months. A cool, dry storage space keeps the product pure and reduces the risk of unwanted reactions. No reason to get fancy—a simple flammable chemicals cabinet, segregated from acids or bases, works well. Left sitting in sunlight or near heat, the chemical can form breakdown products that make future handling riskier. Light may not cause explosions, but it spoils what you paid for.
No Open Flames, No Sparks
Like many organic liquids, N,N-Dibutylaniline is flammable. Forgetting this point brings real harm. Flammable vapors drift, creeping toward pilot lights and hot plates. It only takes a second for a flash fire to start. Store away from ignition sources. That means plugging cords, using rated equipment, and making sure no one lights up nearby. Ventilation matters just as much—good airflow avoids vapor buildup inside a storage room.
Proper Containers Make All the Difference
Original packaging exists for a reason. Manufacturers ship in polyethylene, glass, or compatible metal drums that resist chemical attack. Letting N,N-Dibutylaniline touch soft plastics or absorbent surfaces creates leaks or chemical soup in a month or two. Label containers in clear, permanent ink. Even in a well-organized lab, people misplace bottles. Unmarked liquids turn into mysteries—nobody wants a guessing game near flammable solvents.
Wear the Right Gear Every Time
People sometimes shrug off gloves and goggles, thinking they’ll “just pour a little” into a flask. N,N-Dibutylaniline can irritate skin and get through gloves too thin or too short. Eyes take the worst of it—any splashes sting and can do real damage. A solid pair of chemical splash goggles and nitrile gloves form the basic shield. In crowded labs, add a lab coat and splash apron; spills always target the unprotected.
Handling Spills and Clean-Up
Even with the best intentions, spills happen. Absorbent materials like vermiculite or sand work well for soaking up small puddles. Never grab paper towels or rags—they can smear, spread, or even ignite oily chemicals. Use a fume hood for cleanup, and never dump leftovers down the drain. Waste containers marked for organics keep things simple and help avoid contaminating water systems.
Training Matters More Than Fancy Rules
Written policies look great, but real training sticks in people’s minds. I’ve helped run chemical safety workshops, and the simple “show and tell” method does wonders. Let people see how N,N-Dibutylaniline flows, smells, and reacts. A close look helps everyone remember to treat it with respect, not fear. Supervisors should walk the storage area, check labels, and move strays every so often. Habits build safety more than alarms or posters ever could.
Final Thoughts: Safety Builds Trust
Storing and handling N,N-Dibutylaniline isn’t about abstract compliance, but daily choices. Good practice protects workers, equipment, and even the broader community. Staying vigilant keeps accidents rare and helps everyone get through each shift with nothing more exciting than an honest day’s work.
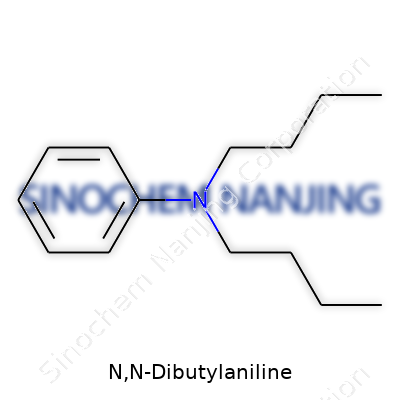
| Names | |
| Preferred IUPAC name | N,N-dibutylbenzenamine |
| Other names |
N,N-Dibutylbenzenamine N-Phenyl-N,N-dibutylamine N,N-Dibutylphenylamine |
| Pronunciation | /ˌɛnˌɛn.daɪˌbjuːtɪlˈænɪliːn/ |
| Identifiers | |
| CAS Number | 92-54-6 |
| Beilstein Reference | 877754 |
| ChEBI | CHEBI:131765 |
| ChEMBL | CHEMBL16213 |
| ChemSpider | 23805 |
| DrugBank | DB14190 |
| ECHA InfoCard | ECHA InfoCard: 100.011.764 |
| EC Number | 200-384-0 |
| Gmelin Reference | 80656 |
| KEGG | C19641 |
| MeSH | D008035 |
| PubChem CID | 86254 |
| RTECS number | JN6475000 |
| UNII | 0D3P5T1T2H |
| UN number | UN2718 |
| CompTox Dashboard (EPA) | DTXSID7032112 |
| Properties | |
| Chemical formula | C16H25N |
| Molar mass | 263.42 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | amine-like |
| Density | 0.891 g/cm3 |
| Solubility in water | Insoluble |
| log P | 4.8 |
| Vapor pressure | 0.037 mmHg (25°C) |
| Acidity (pKa) | 4.70 |
| Basicity (pKb) | 5.98 |
| Magnetic susceptibility (χ) | -72.2e-6 cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 3.7 mPa·s (25 °C) |
| Dipole moment | 4.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –74.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6827.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H315, H319 |
| Precautionary statements | P280, P261, P305+P351+P338, P304+P340, P312 |
| Flash point | 113 °C |
| Autoignition temperature | 375 °C |
| Explosive limits | Explosive limits: 0.7–5.4% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 2200 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1750 mg/kg (oral, rat) |
| NIOSH | NIOSH Registry Number: "91-62-3 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Aniline N-Butylaniline N-Phenylaniline N,N-Dimethylaniline N,N-Diethylaniline |

