N-Methylpropionamide: More Than Just a Solvent
A Look Back at Its Roots
N-Methylpropionamide creeps up in the records of chemical development during the early evolution of amide chemistry. As labs moved from basic amide structures toward more tailored molecules, chemists started exploring alternatives driven by both safety concerns and the need for new solvation properties. N-Methylpropionamide emerged as a response to a growing demand for amides with improved handling characteristics that did not sacrifice the qualities chemists counted on in their synthesis routines. Through the decades, its status shifted from being a niche ingredient in academic research to becoming part of the toolkit for professionals engaged in pharmaceutical, polymer, and specialty chemical production. Publications from the 1970s and 80s hint at how this compound gained traction, with process engineers discovering it allowed for cleaner downstream processing while also opening the door to new product profiles in certain reactions.
Getting to Know It: What Sets N-Methylpropionamide Apart
This compound stands as a colorless to pale yellow liquid with a modest, almost neutral scent, signaling its lean and straightforward structure: C4H9NO. There’s a subtle difference in how it behaves compared to bulkier amides with longer chains or extra functional groups. The molecular structure grants it a boiling point suited to laboratory purifications and industrial throughput, while its miscibility in water and common organics makes it handy for working in mixed solvent systems. I’ve noticed in practice that compounds like this reduce the guesswork when it comes to dissolving a range of solutes, which tends to save both time and money during upstream processing or when trying to wash out residual actives during final steps. Its moderate polarity helps buffer reactive intermediates in ways more aggressive amides like dimethylformamide may not, offering a gentler option in sensitive sequences.
Shaping Technical Specs and Labels That Actually Matter
N-Methylpropionamide may arrive sporting different names depending on which supplier bottles it, but the heart of its usage links directly to key properties like purity (often above 98 percent in industrial supply chains), residual moisture, and clarity. Clarity isn’t just a cosmetic factor; cloudy batches usually signal hydrolysis or foreign dissolved solids, which can trigger off-spec product lots. I learned early in the lab that even a trace of acid or base can nudge amides toward decomposition, so tracking pH and moisture content during handling pays dividends. Labeling in regulated sectors, like pharmaceuticals, extends to tracking residual solvents and identifying related secondary amides that can form as impurities. Chemists genuinely care about the smallest deviations, especially as downstream transformations frequently magnify these tiny differences, so relying on standard ASTM or ISO methods for measuring these parameters is the safest route.
How It Comes Together: Preparation and What Reactions Tell Us
Most routes to N-Methylpropionamide take advantage of methylating agents and propionic acid derivatives. One pathway involves methylamine reacting directly with propionyl chloride or anhydride. The process typically calls for cooling to control exothermic spurts and keeping water out to limit hydrolysis. There have been pushes to swap out the more hazardous methylating agents for less toxic or more easily captured alternatives, responding to both worker safety and stricter emission standards in major chemical hubs. In modification work, N-Methylpropionamide plays well in amidation schemes, sometimes serving as a reactant for further N-alkylation or participating in dehydration under strong conditions to give related imides or nitriles. It doesn’t stop there—sometimes the compound’s amide bond becomes the target for reduction or cleavage studies, giving rise to catalytic research that expands toolkits for tough transformations.
Finding It by Other Names—and What Labels Don’t Reveal
It’s easy to lose sight of a chemical’s background if you only know it by one trade name. N-Methylpropionamide pops up under synonyms like N-Methylpropanamide, 1-Propanamide, N-methyl-, or even more obscure labels in specialty catalogs. I’ve run into confusion in cross-referencing safety data sheets from different countries or supplier networks, since translation and naming conventions can turn one compound into a small maze. Grouping entries by CAS number helps, but attention to alternative nomenclature proves crucial for process-scale purchases.
Keeping It Safe: Real World Rules Not Made to Be Broken
Experience tells me no two chemicals share the same safety profile. Working with N-Methylpropionamide means wearing splash goggles and appropriate gloves, since dermal contact can cause irritation and no one wants a chemical exposure report tracing back to a moment of carelessness. In well-ventilated setups, inhalation risks drop off, but small volumes still release vapors worth minding, especially during heating or distillation. Environmental guidelines encourage storing it in tight, corrosion-resistant containers, away from direct sources of ignition and incompatible acids. The key is keeping inventories tight, managing waste streams responsibly, and using secondary containment in storage rooms. Every organization I’ve worked with adopted extra monitoring for amides because some break down into smaller irritants if left too long on a warm shelf.
Application: More Than Meets the Eye
N-Methylpropionamide isn’t some showboat chemical, but it keeps cropping up in lab notebooks and patent filings. Pharmaceutical labs deploy it as a mild solvent for peptide coupling steps, where it can improve yields or limit racemization compared to bulkier amides. Polymer chemists see value in its clean burning characteristics as a co-monomer or as a process medium for certain specialty plastics and coatings. Agrochemical makers leverage its solvency when extracting actives or adjusting pesticide formulations for better leaf adhesion. This compound also carves out a niche in analytical settings, where clean, easily evaporated solvents can drop detection limits for trace-level chromatography work. Each of these applications brings its own twist—some industries push for greener replacement solvents, others double down on legacy equipment, but they all lean on the same set of reliable properties.
Digging Into Toxicity and the Relentless Pursuit of Better Knowledge
Animal studies build up the basic understanding of how N-Methylpropionamide impacts biochemistry. Acute exposure experiments sketch out irritation and mild central nervous system impacts at high doses. Chronic studies trail behind, since amides as a group tend to fall into mid-to-low hazard categories unless functionalized in particular ways. Regulatory agencies stay on the cautious side, insisting on good industrial hygiene and limiting direct skin contact and inhalation. There are always calls for more detailed studies on breakdown products—what happens when amides leach into wastewater or soil? Several research teams track metabolic fates in model organisms, looking for red flags in bioaccumulation or unexpected toxicity, as prior cases with other amides have shown not everything stays put after disposal. The reality is, the field remains open for fresh data, leaving room for innovation in both monitoring and mitigation.
Research and Next Steps: Evolving Uses in the Age of Precision Chemistry
Researchers rarely stop tweaking and asking what comes next. In the push for greater specificity in synthesis, teams investigate how modifying the N-alkyl component or swapping out the propionyl backbones influences reactivity and environmental outcomes. Manufacturers prioritize process intensification, reducing waste streams and energy loads by exploring continuous-flow technologies. In academic circles, the hunt for lower-toxicity, biodegradable amide solvents takes energy, especially as green chemistry principles work their way into undergraduate teaching labs and major industrial portfolios alike. Applications broaden as more tunable amides enter the fray, nudging N-Methylpropionamide into the spotlight for comparative evaluations. Every fresh patent or journal article nudges the industry closer to new blends, new reaction conditions, and a more surgical approach to solvent choice.
Outlook: Looking Ahead Without the Hype
N-Methylpropionamide survives on the backbone of reliability and versatile chemistry even as competitors circle with banners promising lower emissions or even zero toxicity. The drive for more sustainable chemistry looks to compounds like this as both benchmarks and stepping stones. Regulatory landscapes rarely loosen, so suppliers and end-users will keep revising processes to meet newer, tighter standards. Researchers and engineers hold open the door to alternatives, but legacy applications and user familiarity mean N-Methylpropionamide’s story won't just vanish overnight. Its role as a modest, functional, and accessible amide links together tradition and innovation—raising the bar for what solvents and intermediates ought to offer, not just in terms of function, but in delivering on safer, cleaner, and better-controlled chemical enterprise.
What is N-Methylpropionamide used for?
The Backbone of Modern Synthesis
N-Methylpropionamide may not get headlines, but for anyone working in a lab, it's a familiar presence. This compound plays a key role in chemical synthesis, especially for producing pharmaceuticals and specialty chemicals. It stands out for its solvent properties, helping scientists dissolve both polar and non-polar substances. That's gold when trying to coax stubborn reagents into reacting, especially for complex molecules often found in medicines or new materials.
The Real-World Benefits in Industry
When manufacturing demands efficiency, few things slow down progress like poorly dissolving ingredients. N-Methylpropionamide steps up as a versatile solvent, allowing for smoother chemical processes. Industries count on it for reactions that need a little push. For instance, it helps create intermediates used in the production of drug molecules—think painkillers, antibiotics, or even anti-cancer agents.
Outside labs, this chemical finds its way into coatings, resins, and the inks used on packaging. Formulators appreciate its ability to boost solubility. That means they get to experiment with new recipes and formulations without worrying about clumping or separation. N-Methylpropionamide isn't just another liquid in a drum—it's an enabler for innovation in material science and manufacturing.
Safety Is Not Just a Checkbox
Every time a new solvent shows up, safety conversations come up. You can't ignore toxicity or environmental impact. Based on available data, N-Methylpropionamide poses lower health risks than many traditional solvents. Its profile looks friendlier compared to old-school choices like dimethylformamide, notorious for strict safety controls.
That doesn't mean it's risk-free. Glove up, ventilate the workspace, and train staff about safe handling and spill response. Sustainable practices aren’t just about following rules; they keep teams healthy, reduce medical costs, and support company reputations. In an era of tighter environmental standards, picking solvents with a gentle footprint helps companies stay ahead of the curve and cuts down on hazardous waste management costs.
Challenges and Smarter Choices
N-Methylpropionamide works well, but it's not the only tool in the kit. Supply chain crunches or price hikes can pinch production. Companies who depend on a single solvent, even one as reliable as this, set themselves up for headache when markets tighten or regulations shift. It makes sense to explore greener alternatives as new research pushes out more bio-based options. These might not match every technical parameter today, but staying open to new chemistries puts firms in a strong position for the future.
In my experience, asking suppliers tough questions and reading safety data sheets thoroughly pays off. Getting familiar with a solvent's full lifecycle—from manufacturing to disposal—shows real commitment to the job and to the world beyond the lab. By focusing on safer, adaptable chemicals like N-Methylpropionamide and keeping an eye on breakthrough developments, chemists and manufacturers can keep products moving and standards high.
Building Trust Through Transparency
Customers want to know more about what's inside their products and how companies handle chemicals. Good information—sourced from peer-reviewed studies and regulatory bodies—goes a long way to build trust. Openly sharing choices about solvents, including any steps taken to minimize environmental harm, shows that manufacturers care about more than just profit.
N-Methylpropionamide may not be famous, but it keeps industry running smooth. As science evolves, so does the toolbox. Each step toward safer, more effective solutions isn’t just the job of one chemist or one company. It takes a whole community, asking questions and pushing for better.
What is the chemical formula of N-Methylpropionamide?
Understanding the Molecule
N-Methylpropionamide carries the chemical formula C4H9NO. To break it down, this compound includes four carbons, nine hydrogens, one nitrogen, and one oxygen. The “N-methyl” piece means a methyl group links directly to the nitrogen atom. In the real world, this structure pops up in labs that handle fine chemicals, pharmaceuticals, or research compounds.
Real Relevance in the Lab
Most people working with chemicals want clear answers—especially when formulas either make or break an experiment. Having wrestled with organic syntheses myself during long nights in the university lab, I can say that getting the formula right avoids wasted time and unnecessary costs. It helps the team cross-check the stoichiometry for their reactions. If you forget a hydrogen or add an extra carbon, you won’t get the expected reaction products. Many careers and experiments have stalled over simple mix-ups like this.
Accurate formulas also keep your safety sheet up to par. Anyone who’s spent a day labeling chemicals for a workplace knows the headache of tracking incorrect substances. Imagine pouring hours into a reaction, only to learn a missing nitrogen led to the formation of a toxic byproduct or a failed synthesis. So, precise identification of N-Methylpropionamide as C4H9NO isn’t busywork: it’s everything for safe storage, proper usage, and legal compliance.
Facts and Context
N-Methylpropionamide falls under the category of amides, a group known for their presence in both life sciences and industry. Amides like this one pop up in peptide synthesis, plastics production, and the pharmaceutical sector. For context, methylated amides serve as intermediates for drug molecules, especially where stability and reactivity must be balanced. The exact formula shapes how the chemical behaves: reactivity, solubility, and interaction with other substances change with every atom.
A precise chemical formula supports data integrity, one thing everyone in science can agree on. A slip in the digits and letters can land research in the irreproducible pile—a genuine fear for any lab team with grant money at stake. In a world gravitating towards open science and data sharing, a misreported formula threatens trust and progress.
Potential Pitfalls and Solutions
Mistakes in chemical formulas often trace back to confusion between naming systems or shorthand notations. Chemists deal with common names, IUPAC names, and local lab slang. To sidestep errors, I’ve learned to stick to structure diagrams alongside written formulas. Double-checking those structures against trusted databases or primary literature saves plenty of grief.
Institutions that care about data purity typically push internal audits or standardized reviews. These help catch formula slip-ups early, before paperwork heads to publishers or gets sent with shipments. Easy electronic tools, like chemical drawing software and cross-referenced databases, let staff verify formulas in seconds. Lab managers can set up checklists so no step or digit gets left out.
Why It Matters Outside the Lab
Accurate chemical data doesn’t only serve researchers. Downstream users include teachers, students, and professionals in manufacturing, medicine, and even the regulatory world. Incorrect reporting leads to supply issues, incorrect labeling, and in some cases, dangerous misuse. For anyone planning to order, store, or synthesize chemicals, every atom counts. Getting N-Methylpropionamide’s formula right is more than fact-checking—it’s about accountability, safety, and professional competence.
Is N-Methylpropionamide hazardous or toxic?
Understanding What We’re Talking About
N-Methylpropionamide isn't a household name. It pops up in labs and production plants more than kitchen cupboards. Used as a solvent and an intermediate in chemical manufacturing, the compound does its job behind the scenes—from pharmaceutical synthesis to specialty chemicals production. The question people keep asking is: how risky does it get?
Checking the Science
Diving into the published data, N-Methylpropionamide hasn't grabbed headlines for acute toxicity. A glance at the GHS (Globally Harmonized System) classification doesn’t raise major alarms. There’s no flashing red exclamation point, but that doesn’t mean the coast is clear. Safety data sheets point out skin and eye irritation, and it’s easy to see why. Liquid chemicals that can dissolve, extract, or transform other substances rarely treat human tissue kindly. People who spend regular time handling this compound in the workplace wear gloves, glasses, and sometimes respirators, not to look tough but to keep the nasty stuff out of their bodies.
The Real Risks
It’s never just theoretical. Someone who splashes N-Methylpropionamide on bare skin will feel the sting or witness redness. Even a whiff of concentrated vapor can irritate sensitive noses or throats. The good news: large-scale, violent reactions like you might see with stronger acids or flammable solvents don’t usually follow. But prolonged or repeated exposure ramps up the stakes. As with many amide-based solvents, long-term studies in animals sometimes find changes in organs after weeks or months of elevated dosing. Researchers flagged some mild effects on liver enzymes or kidney function during heavy use. For human workers, clear guidelines (like the ones from NIOSH or OSHA) keep those risks low, but compliance matters more than promises.
Why Everyday People Should Still Care
Most folks outside science or industrial settings will never knowingly touch N-Methylpropionamide. That doesn’t make the safety story irrelevant. Maybe someone has a neighbor who works at a facility using this solvent. Maybe a groundwater table sits near a chemical plant. Reliable handling, storage, and disposal protect everyone downwind or downstream. Years ago, tighter regulations on hazardous chemicals came about because water and soil contamination quietly caused serious health problems. Prevention—containment, good venting, sturdy tanks—beats a clean-up crew scrambling after the fact.
Looking for Smarter Answers
Deciding to swap out N-Methylpropionamide for a milder alternative works only if that replacement gets the job done without new risks. Green chemistry isn’t just a buzzword. Companies and researchers pursue new processes using less toxic solvents—or using less solvent overall. Sometimes it involves investment in automation, better worker training, or continuous monitoring of airborne concentrations. Mistakes get caught faster before things snowball. Anyone in charge of safety audits remembers near-misses where a simple fume hood or splash shield spared someone from a trip to the urgent care. It’s not about alarmism; it’s about respecting the tools of the trade and keeping problems out of both bodies and boardrooms.
What are the storage conditions for N-Methylpropionamide?
Close-up on Storage Risks
Not every chemical draws the spotlight, but N-Methylpropionamide deserves careful attention from anyone handling or storing it. This solvent finds its way into a range of industrial processes—from chemical synthesis labs to pharmaceutical operations. It doesn’t grab headlines, yet mishandling it can spell real trouble for people and property. My time working around solvents taught me that mistakes don’t just waste money; they put lives at risk.
Real Hazards, Not Theoretical
In a busy facility, people move fast and storage guidelines can slip through the cracks. I once saw a new technician set down a drum in direct sunlight, thinking, “It’s in a metal container, so it’s fine.” A more experienced coworker rushed in, telling her why that move wasn’t just a small error. N-Methylpropionamide boils at roughly 199°C, but that doesn’t mean high heat is harmless. Over time, rising temperatures make the solvent degrade faster, build up harmful vapors, and increase pressure inside sealed drums. Under heat and light, some impurities form that no lab manager wants anywhere near their product.
Dry, Cool, and Tight: Real-World Steps
A warehouse doesn’t always look picture-perfect. Stacking drums three high in a back corner makes daily operations seem efficient, but air flow vanishes in those tight spots, and outside walls sweep in extra heat during summer. I learned from a fire chief that the difference between a clean, organized chemical room and a haphazard pile could decide whether he gets called out for a fire that day.Solid practice means storing N-Methylpropionamide in a dry, well-ventilated space shielded from sources of heat. Damp air won’t just corrode containers. Moisture can sneak into drums, affecting both purity and stability. Containers ought to be tightly sealed after every use. I once pulled a half-empty bottle off a shelf, only to find the cap loose—some of the solvent had evaporated, concentrating any impurities left behind.
Why Separation from Incompatibles Saves Headaches
Some people figure, “Chemicals in closed containers can sit together.” My early days in a small factory taught me the risks there. Storing N-Methylpropionamide next to strong oxidizers or acids, even by accident, sets the stage for nasty reactions if leaks or spills ever meet. Best practice puts it in a chemical storage cabinet, out of the way from anything reactive or flammable.
Labeling and Spill Plans: Not Just Paperwork
Every time a label peels or wears off, someone has to guess what’s inside. I’ve watched seasoned techs get tripped up by faded or missing labels in cluttered storerooms. Label every container with the chemical name, date of receipt, and hazard information. In case of a spill, workers should know how to contain and clean it—spills left to evaporate only add invisible dangers to the air.
Using Data to Back up Good Habits
The European Chemicals Agency lists N-Methylpropionamide as stable under recommended conditions, but notes the need for proper ventilation and closed containers. Reports from workplace safety surveys often point to human error as the deciding factor in accidents. Data shows most chemical exposures happen not during use, but while moving or storing.
Final Word: Safety Starts with Storage
As someone who’s seen small mistakes turn into big problems, I don’t look at chemical storage as tedious. It’s direct and practical—a real investment in health and business reputation. Storing N-Methylpropionamide safely keeps everyone working and the lab running smoothly, day after day.
What is the CAS number of N-Methylpropionamide?
Everyday Chemistry Gets a Tag
N-Methylpropionamide isn’t popping up on most breakfast tables or workplace conversations, but there’s a reason scientists, health officials, and product developers pay close attention to it. Its CAS number, 925-13-9, is a unique tag that separates it from every other chemical. Most people don’t realize how much rides on this set of digits—buying, shipping, tracking, and even discussing safe handling would turn into chaos without it. I remember a time back in grad school when mislabeled chemicals caused an entire project to get delayed by weeks, just because two similar-sounding names were confused. The CAS system cleared that confusion pretty quickly.
What Does a CAS Number Do?
Think of a CAS number like an ISBN for books. It distinguishes one compound from hundreds of others that share similar names or structures. N-Methylpropionamide might look and sound like a bunch of other amides. The CAS registry number 925-13-9 removes all room for error. Laboratories and manufacturers use it for ordering, research, and quality control. Imagine a chemist halfway around the world working on a formulation and needing the exact same compound as a peer in another country. The CAS number slashes the risk of mistakes. It also locks in traceability, which keeps people safe.
Science Backed with Traceability
Real-world incidents make the value of proper identification clear. Not long ago, stories surfaced about researchers running experiments that failed because a supplier shipped the wrong chemical—the names were almost identical, but their CAS numbers were not. That kind of error wastes money, eats up time, and can lead to much bigger problems with safety testing or manufacturing. I’ve seen researchers keep physical notebooks of CAS numbers on their shelves, because relying on names alone just doesn’t cut it.
Sourcing and Responsible Use
Companies working with N-Methylpropionamide rely on that identifier to sift through regulatory requirements and support safe processing. Each chemical processed, shipped, or studied has to be matched with documentation. That involves safety data sheets driven by CAS numbers. Following the right data is a way to support safety, which matters not just in the lab, but in industries from agriculture to textiles. In my former lab, compliance officers checked the CAS number before approving any purchase or use of a new chemical. It wasn’t just bureaucracy—it kept accidents from happening, and kept our reputation intact.
Letting Facts Guide Safe Progress
Trust comes from knowing what’s in the bottle, not just believing a supplier’s label. The CAS registry, run by the American Chemical Society, covers millions upon millions of compounds. By referencing 925-13-9, researchers and companies minimize confusion. Regulations and compliance aren’t about red tape. They keep people safe, help organizations avoid lawsuits, and build trust in science. For anyone working in the world of chemistry, using a CAS number is just standard practice, not optional. Each time I fill out paperwork for a new project, the CAS number sits at the top, a reminder of how small details change outcomes for everyone.
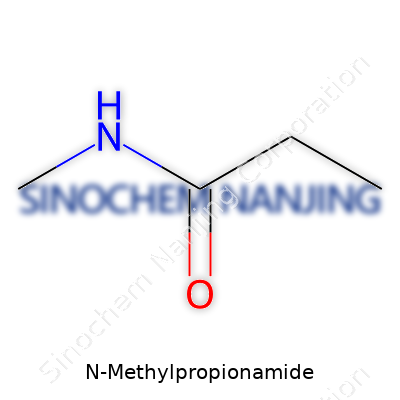
| Names | |
| Preferred IUPAC name | N-methylpropanamide |
| Other names |
N-Methylpropanamide N-Methylpropionamide N-Methylpropionamid 1-Methylpropanamide Propionamide, N-methyl- NSC 86574 |
| Pronunciation | /ɛnˈmɛθɪl.prəˈpɪə.nə.maɪd/ |
| Identifiers | |
| CAS Number | 921-88-0 |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:85336 |
| ChEMBL | CHEMBL16296 |
| ChemSpider | 16851 |
| DrugBank | DB02145 |
| ECHA InfoCard | 100.067.610 |
| EC Number | 203-712-6 |
| Gmelin Reference | 8871 |
| KEGG | C19382 |
| MeSH | D012391 |
| PubChem CID | 12038 |
| RTECS number | UF8225000 |
| UNII | 11H7267P88 |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C4H9NO |
| Molar mass | 87.1204 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 0.948 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | -0.38 |
| Vapor pressure | 0.0197 mmHg at 25 °C |
| Acidity (pKa) | pKa = 23.3 |
| Basicity (pKb) | pKb = 14.22 |
| Magnetic susceptibility (χ) | -7.54 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.444 |
| Viscosity | 0.922 cP at 25 °C |
| Dipole moment | 3.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 182.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -192.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -457.7 kJ/mol |
| Pharmacology | |
| ATC code | N01BX06 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H319 |
| Precautionary statements | P280, P261, P305+P351+P338, P304+P340, P312 |
| Flash point | 108°C |
| Autoignition temperature | 453 °C |
| Explosive limits | Explosive limits: 2.3–12% |
| Lethal dose or concentration | LD₅₀ Oral Rat 1500 mg/kg |
| LD50 (median dose) | LD50 (median dose): 6.9 g/kg (rat, oral) |
| NIOSH | RN0108750 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
N-Methylacetamide N,N-Dimethylpropionamide Propionamide N-Ethylpropionamide |

