Chasing Clarity on N-Methylmorpholine: Chemistry with Practical Impact
Looking Back at N-Methylmorpholine's Story
History doesn’t always shine a spotlight on unsung chemical helpers, but N-Methylmorpholine marks its presence through decades of steady use. Chemists recognized this humble heterocycle in the mid-20th century, pulling it out of the shadows thanks to its effectiveness as a base and solvent. Its place in both university labs and industrial settings tells a bigger story about how small molecules often power big leaps in materials, pharmaceuticals, and coatings. As more industries sought practical bases that work under a mix of conditions, N-Methylmorpholine stuck around and proved its worth.
Getting to the Heart of the Product
N-Methylmorpholine, or NMM, comes off as just another clear liquid to the naked eye, but there’s more to it. Chemically, it's a six-membered ring with nitrogen and oxygen doing the heavy lifting, and a methyl group stuck on the nitrogen. This simple tweak from morpholine changes the way it acts: less water-grabbing, more focused as a mild base that doesn’t try to overpower every other chemical around it. The ease with which this compound slips into reactions, especially where gentle conditions matter, is why it’s kept on benches by professionals across the world.
Physical and Chemical Character: The Working Details
People who have worked with N-Methylmorpholine will tell you it isn’t just a benign liquid. It carries a distinct amine odor that you won’t forget if you’ve ever spilled it on your gloves. It boils around 116°C, making it manageable in labs without heavy-duty gear. Its mix of polar character and organic solubility opens doors in both water-based and organic projects. The molecule holds up against moderate heat and tolerates moisture without breaking down. Compared to other common bases, N-Methylmorpholine strikes a balance. Chemists looking to avoid highly hazardous, volatile, or caustic amines often prefer it for just this reason.
Technical Details and Proper Labeling Matter
N-Methylmorpholine requires close labeling to keep people and processes safe. Industry regulations demand strict information chips—moisture content, assay, and impurity limits—because missing those specifics can trip up downstream reactions or introduce unexpected hazards. Chemical suppliers label the substance as a flammable liquid, and those using it must note the chemical synonym “N-Methyl-tetrahydro-oxazine,” so folks don’t mix it up with similar-sounding products. For storage and handling, flammability warnings and ventilation requirements matter as much as anything printed on an analysis certificate. From personal experience, I remember the consequences: a poorly closed bottle led to a foul-smelling lesson about vapor pressure and container safety.
Making N-Methylmorpholine: Steps and Struggles
Synthetic chemists turn morpholine into N-Methylmorpholine through practical methylation tricks, most often with dimethyl sulfate or methyl halides in controlled settings. The method sounds straightforward, but batch purity matters: unwanted byproducts end up complicating both waste management and process validation. As with many industrial chemicals, greener routes keep getting attention; researchers have looked for alternatives to hazardous methylating agents. Continuous flow setups with reduced side product formation could offer cleaner outcomes. Every tweak in process chemistry pushes toward less waste, lower hazards, and greater efficiency—priorities not just for managers but for everyone who values cleaner air and water around plants.
What N-Methylmorpholine Does in the Lab and Beyond
NMM stands as one of those go-to tertiary amines that makes tough reactions easier. In acylation and alkylation reactions, it mediates without jumping into the fray as a nucleophile. That means it plays a supporting role in pharmaceutical synthesis, especially for antibiotic and antiviral building blocks. Polyurethane manufacturing taps into NMM’s base strength to catalyze reactions for foams and coatings, putting it in products used daily, sometimes even without the end user realizing it. For chemists aiming for selective oxidation or clever rearrangements, N-Methylmorpholine N-oxide (NMO)—a close cousin made by oxidizing NMM—delivers results where bulkier or less predictable reagents fall short. Its flexibility in all these spaces explains why it never feels like a dead-end material in research or industry.
Synonyms and Names: Clearing Up the Confusion
Chemicals often carry baggage in the form of multiple names. N-Methylmorpholine doesn’t stray far. Whether it’s called NMM, 4-Methylmorpholine, or N-Methyl-tetrahydro-1,4-oxazine, the key is double-checking documentation before mixing it in a reaction. Old research papers sometimes muddle the names, leading newcomers to waste time tracking down the right stuff. Keeping a habit of verifying CAS numbers rather than just relying on synonyms has saved many chemists—and students—from mixing up reagents.
Operational Rules: Doing Right by Safety
Flammability sits at the front of any N-Methylmorpholine safety talk. Vapors can ignite under standard lab conditions, so good ventilation and careful storage away from sparks, static, and oxidizers protect both users and labs. Spills need quick cleanup to keep air quality acceptable. Simple actions—goggles, gloves, and a working fume hood—make the difference between a smooth day and an emergency. Working in settings with NMM showed me that even well-ventilated spaces need diligent checking, since sensory fatigue can’t be trusted to signal danger. Plant operators carry the same rules to scale, with added concerns about mixing vessels, seals, and emergency controls.
Where Application Meets Need: Industrial and Research Uses
NMM doesn’t just live in chemical catalogs. Polyurethane foam makers use it by the ton as a catalyst to shape everything from insulation to furniture. The pharmaceutical world taps into its mild base properties to coax out advanced active compounds. Its use as a solvent or an auxiliary for specialized organic synthesis keeps it relevant for experimental chemists looking to push the boundaries of tough transformations. The textile industry found value in integrating NMM into cellulose fiber processing, a reminder that even energy-intensive fields watch closely for any edge in efficiency or performance. Researchers keep poking at this molecule’s limits, building on the foundation laid decades ago.
Shifting the Focus: Research and Improvements
Research into N-Methylmorpholine doesn’t stop at finding new uses. Chemists track new ways to produce it more safely and cut costs. Improvements in purification and handling, along with greener methylating agents, raise the bar for quality. Analytical studies often link purity with reaction reliability, so labs invest in improved monitoring. Interest in new applications—such as catalysis in ionic liquids or greener solvent systems—pushes the boundaries further. Researchers comparing its performance with cutting-edge alternatives return to its robustness and ease of use, even as they look to trim down loss and risk.
Digging Deep into Toxicity and Worker Safety
Exposure studies on N-Methylmorpholine show that the compound needs respect in the workplace. Direct contact with the skin and eyes can irritate, and inhaling vapors causes respiratory discomfort, so safe handling becomes more than a formality. Regulations flag the compound for these hazards, and material safety campaigns strive to hammer home the message: just because a chemical is common doesn’t mean it isn’t dangerous. The body metabolizes NMM fairly quickly, but large doses pose genuine risks. Long-term studies track chronic effects, particularly among those who’ve spent years in production facilities. Workers need ongoing support and clear protocols—real prevention means more than hoping everyone pays attention all the time. My own experience underscores the point: a colleague’s minor exposure turned into a reminder that even routine work needs proper protection.
Looking Forward: The Road Ahead for N-Methylmorpholine
As industries transition toward safer, less wasteful practices, N-Methylmorpholine presents challenges and opportunities. For all its advantages, workers and researchers handling this compound push for solid alternatives and more sustainable production. Improvements in catalysis and base chemistry, greener pathways for manufacturing, and tight controls on emissions will shape its future. The drive for higher purity and lower risk supports inventive engineering, both in old industries and new. In research, chemists keep probing for softer, less hazardous bases—but many of those discoveries build on the lessons learned from decades with NMM. The call for sustainability will likely fuel improvements in every corner of its life cycle, from production through application to final disposal.
What is N-Methylmorpholine used for?
What Does N-Methylmorpholine Actually Do?
Chemists rely on certain ingredients again and again, and N-Methylmorpholine has become one of those go-tos. Most folks won’t ever see this chemical in their daily lives, yet it supports many products we use—from the paint on our walls to the drugs that keep us healthy.
Role in Pharmaceuticals
Many drugs reach your pharmacy thanks to complicated reactions. N-Methylmorpholine acts as a “base”—in plain language, it keeps chemical reactions ticking along by soaking up protons. This simple move keeps the reaction on track, helping drugmakers create medicines more reliably and on a larger scale. In my own experience working with pharmaceutical chemists, the recurring theme is wanting reactions that work every time, not just once in the lab. N-Methylmorpholine often brings that reliability, thanks to its predictability and stability in different environments. Published research echoes this use, showing up in synthesis of antibiotics, antivirals, and anti-cancer drugs.
Boosting the Power of Polyurethanes
Modern cushions, insulation, or sneakers often depend on polyurethane foam. To make these materials, manufacturers need a catalyst to steer the reaction between polyols and isocyanates. N-Methylmorpholine acts as a key step, converted into N-methylmorpholine-N-oxide, which then speeds things up. No catalyst, no foam—just sticky blobs that nobody wants. In factories, controlling temperature and consistency means less waste and better comfort in the end product. The numbers matter: the global polyurethane market topped $70 billion in 2023, and chemicals like this keep productivity high. Regulations have pushed companies to swap old, volatile catalysts for safer and more stable ones, and N-Methylmorpholine fits into these newer safety standards.
Trusted by Industry, Not Without Precaution
Handling strong bases carries real risk, and N-Methylmorpholine brings its own hazards. Its vapors can irritate the eyes and lungs. People working in manufacturing need good ventilation and protective equipment—having watched a colleague suffer chemical burns, I know the right gloves make a difference. Safety data sheets urge strict control over spills and exposure, so routine training can't fall through the cracks. The European Chemicals Agency lists N-Methylmorpholine as a substance needing careful use, and that should grab attention at any workplace using it.
Opportunities for Safer Chemistry
Companies recognize the need to keep workers safer and the environment cleaner. Over the years, chemists have been seeking replacements that work just as well but cause fewer problems during handling or in case of leaks. The industry could do more by investing in less toxic alternatives, promoting better equipment, and supporting green chemistry initiatives. Strong leadership leads to more rigorous safety checks, so nobody has to choose between efficiency and health.
Why This All Matters
It may never show up on your shopping list, but N-Methylmorpholine shapes the chemistry behind many familiar things. Medicines, foams, dyes—the list stretches on. If companies can keep working towards safer practices and greener substitutes, everyone gains: workers, consumers, and future generations. Watching chemical safety evolve over the years has convinced me that progress here doesn’t slow down, even as new challenges appear.
What are the safety precautions when handling N-Methylmorpholine?
The Risks Hiding in a Useful Chemical
N-Methylmorpholine serves a real role in labs. It shows up in pharmaceuticals, solvents, and plenty of syntheses. This colorless liquid doesn’t give away its hazards at first glance, and many underestimate it. I learned early from a mentor that familiarity with a chemical often breeds shortcuts, and shortcuts always find a way to catch up with you eventually.
Why N-Methylmorpholine Demands Serious Care
This compound shows no mercy if it comes in contact with eyes or skin. It has a reputation among chemists for causing burns, irritation, and allergic reactions. If inhaled, it tends to irritate the respiratory tract, leading to coughing, shortness of breath, or worse if someone spends too much time around its vapors. Even basic household cleaning agents get more respect than this, but the consequences here stack up much faster. The chemical can seep through common gloves or leave fumes in small, unventilated rooms, and these hazards only multiply with complacency.
A Reliable Routine for Handling
Chemical safety feels like a checklist at first. Over time, it becomes less about memorizing a list and more about developing habits. I always start by securing the right personal protective equipment—goggles that seal well, gloves that resist permeation, a lab coat made to handle corrosive splashes. Thin latex gloves won’t cut it here; nitrile or neoprene work better. A chemical fume hood isn’t optional, since ventilation matters most with volatile liquids like N-Methylmorpholine.
Preparation comes before anything else. I read the latest Safety Data Sheet—never just the highlights, always the whole thing to catch details about incompatibilities and spill response. I keep a buddy system during high-risk work, so someone else can help or call for backup if something goes sideways. Proper labeling follows right behind good storage. Containers need tight closures and clear names—no faded marker scrawls or mystery flasks that blend into the background.
Dealing with Spills and Exposures
Even veterans mess up. Once, someone knocked over a beaker, and even though it seemed like just a splash, we did a full clean according to the protocol. Baking soda, paper towels, and gloves saved the day—but only because the spill kit was restocked and close by. It’s worth checking spill materials before starting any job. Never handle a spill solo if vapor becomes overwhelming, and always evacuate the space if eyes start to sting or the air feels thick. Emergency showers and eyewash stations need to work every time, not just look clean for inspection.
Storing and Disposing with Respect
Leaving N-Methylmorpholine next to acids or oxidizers risks a violent reaction. I double-check that nothing reactive sits nearby, and never stack containers where they can topple. Temperature plays a big part; keeping the chemical in a cool, dry cabinet protects everyone from unexpected tank swelling or leaks. Used material and contaminated gloves head into a labeled waste drum, never down the drain or in regular trash.
Continuous Vigilance Pays Off
Complacency gets people hurt. Staying sharp, building strong habits, and refusing to cut corners keeps labs and workers safe. Zero injuries feel possible only when every person treats chemicals like N-Methylmorpholine with healthy respect—every single day.
What is the chemical formula of N-Methylmorpholine?
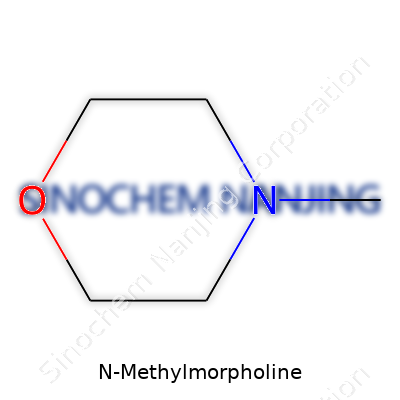
The Simple Backbone: C5H11NO
N-Methylmorpholine carries the formula C5H11NO. This compound often pops up in labs and factories, quietly taking a central role in chemical processes that might go unnoticed outside the industry. What makes this molecule noteworthy isn’t just its formula, but the versatility that comes from combining a morpholine ring with a methyl group. Each atom in the formula hints at the jobs the molecule takes on in real-world chemistry.
Where It Matters the Most
Chemists reach for N-Methylmorpholine when a steady, mild base is needed. Its reputation comes from reliability in catalyzing reactions, especially in the pharmaceutical and polyurethane industries. You’ll find its touch in the production of antibiotics, herbicides, and even the spongy parts of your running shoes. One story from a chemist’s bench: a complex organic reaction needed a nudge without disrupting delicate groups, and N-Methylmorpholine kept the reaction moving without trouble, proving its worth by helping create life-saving medicine without causing chaos elsewhere in the molecule.
Health, Safety, and Responsibility
People working around this compound need to know what they’re handling. N-Methylmorpholine comes with health warnings — it can irritate the skin, eyes, and respiratory tract, and long-term exposure raises concerns. In one manufacturing plant, an accidental release led to trouble for operators who trusted their standard ventilation. Lessons learned on the floor stressed the importance of personal protective equipment and strict controls. Not just gloves and goggles, but smart systems for air quality, leak detection, and emergency planning protect people first.
The Bigger Picture: Sustainable Chemistry
Production and waste matter more today than ever. Many companies search for ways to minimize environmental footprints, substitute hazardous chemicals, or recycle solvents. N-Methylmorpholine’s persistent demand challenges companies to ensure cleaner production. Innovations in recovery systems allow more solvent to be recycled, cutting down on raw material use and disposal risks. These investments save money, but more importantly, they signal a commitment to both workers and the planet.
What Moves the Needle in the Industry?
Change usually starts with people who see more than formulas on a page. Engineers and researchers look at N-Methylmorpholine and ask: Can we make this safer, cheaper, or greener? Sometimes, alternative amines get tested, but rarely does one offer all the same benefits. So, the push for change lives in better processes and practical safety. Scaling up greener syntheses, capturing emissions, and training every hand that deals with this compound keeps progress rolling. Clear labeling, data sheets, and ongoing education make sure nothing slips through the cracks.
Every Atom Counts
Behind the shorthand C5H11NO beats a story of people, science, risk, and improvement. N-Methylmorpholine isn’t the flashiest reagent in the lab, yet it touches products, workplaces, and lives across the world. Each chemical in daily use challenges us to crank up the responsibility and look for smarter, safer ways to do chemistry. If we take this challenge seriously, the quiet chemicals like N-Methylmorpholine will help build a better future, one small reaction at a time.
How should N-Methylmorpholine be stored?
Getting Real About Chemical Safety
Anyone who works in a lab or a chemical plant understands the daily dance with risk. N-Methylmorpholine flows through a surprising number of industries. Coatings, pharmaceuticals, adhesives—its footprint is everywhere. Trouble is, N-Methylmorpholine comes with a personality. It’s colorless, but its fishy odor gives it away. It’s flammable. It can irritate the skin, eyes, and lungs. I’ve seen carelessness turn inconveniences into emergencies. That doesn’t just affect workers. One spill, one fire, one missed warning, and entire communities feel the results.
Getting Storage Right, from First Barrel to Last Drop
Reliable storage keeps N-Methylmorpholine useful and employees safe. Store it only in containers that seal tightly—think steel drums or HDPE cans, not the kind of makeshift barrels you spot in old workshops. Any exposure to air or moisture sets off more than just performance issues; it can form dangerous peroxides. I’ve opened containers before, and that sharp smell mixed with anxiety is something you don’t forget.
Leave N-Methylmorpholine in a sunny spot, or near a heat vent, and you invite trouble. Spots above 25°C speed up degradation. Temperature swings destroy product quality and start leaking vapors, raising the risk for accidental ignition. This is a chemical you want to lock away in a cool, dry, well-ventilated spot. Any whiff of humidity or a hint of water in the air gets absorbed, and over time, it can dilute and destabilize what’s inside that barrel.
Chemicals don’t get along with everything. N-Methylmorpholine reacts with strong oxidizers. Mixing accidents aren’t rare; plenty of old-timers tell wide-eyed stories of near misses. I remember a coworker stored it near a bleach drum. Sharp noses caught the error, but the close call meant days of training and clean-up. Keep it far from acids, strong bases, and oxidizing agents if you want to avoid those war stories turning into headlines.
Regulations Can Be Lifesavers, Not Hassles
Rules pile up in labs and plants. Labels here, locks there, paperwork everywhere. It’s tempting to see these as pointless. Past mistakes shape them, though. OSHA and similar agencies don’t write guidelines for the fun of it. Material Safety Data Sheets (MSDS) spell out storage rules for a reason. Training can make the difference between an ordinary day and an emergency call. I’ve watched companies skip extra training because everyone “already knows” the drill. That overconfidence shortchanges safety in the long run.
Solutions Rooted in Common Sense
Hazards hide in plain sight, but good storage strategies can deflate most risks. Designated chemical zones, regular inspections, and smart labelling work wonders. Reliable ventilation isn’t a luxury. It’s the difference between a healthy workplace and a recurring cough. I always recommend spill kits close by—scrambling during a leak wastes precious seconds. Practice emergency drills beyond the clipboards and checklists. Run them until muscle memory kicks in.
Real safety shows up in daily choices. PPE sits at the door for a reason. A little bit of awareness, a lot of preparation, and a healthy respect for chemicals keep operations moving and everyone breathing easy.
Is N-Methylmorpholine hazardous to health or the environment?
What N-Methylmorpholine Really Is
N-Methylmorpholine shows up in many factories and labs. Chemists like it for its use as a solvent and catalyst. The chemical’s smell—some say fishy, others say ammonia-like—gives a quick clue that it’s not water or vinegar. It looks clear and colorless, which might seem harmless, but appearances can fool. I’ve stood next to a workbench where bottles of it waited for the next experiment; most of us wore extra gloves. No one took chances.
Health Concerns Seen Up Close
Breathing vapors or getting a spill on skin brings trouble. Some folks I know who’ve handled it complained of headaches after breathing the fumes too long. Skin contact once gave my lab partner a red rash within minutes. N-Methylmorpholine smells sharp for a reason—it can cause irritation to eyes, nose, and lungs. Get a splash in the eye, and you’ll need to rinse for a long time. Inhaling high levels doesn’t just cause a cough; it can make breathing tough and even lead to fluid build-up in the lungs according to factory health reports. Long-term studies are slim, but repeated exposure often leads to coughs, sore throats, and possibly liver or kidney stress based on findings from the European Chemicals Agency.
Wearing the right gear makes a difference. Even with gloves and goggles, spills happen. Emergency showers exist for a reason. One careless moment, and the day quickly turns bad. Knowing what it feels like to pull a fire alarm next to a spilled drum stays with you.
What Happens Once It Hits the Environment
Workers aren’t the only ones who need to think about risks. In many factories, used chemicals leave through drains unless caught early. I remember seeing cleanup crews in white suits after a drum leaked into a storm drain; the smell alone warned people to stay away. Runoff like that can reach rivers and harm fish, frogs, and anything depending on clean water. One study from the Journal of Hazardous Materials pointed to N-Methylmorpholine being tough for some water treatment plants to break down.
It can stick around in the soil and water, and animals that drink contaminated water start to show signs of kidney trouble. Birds and fish exposed to small amounts sometimes can’t reproduce as they should. These worries aren’t theoretical. Accidental spills into the Rhine River in Germany led to local water advisories and dead fish floated downstream for days.
Better Handling Means Fewer Problems
Looking back, most issues can be traced to poor planning or shortcuts taken. Storing drums safely, using proper gloves and ventilation, and training workers to spot dangers early changes outcomes for the better. Shift supervisors who run regular drills see fewer accidents and less time lost to injuries. Simple steps—like bunded storage areas that catch leaks—keep chemicals out of the environment. On a bigger scale, countries including the US and Germany set legal limits on emissions and draft strict handling rules for N-Methylmorpholine.
Responsible disposal through licensed waste handlers keeps this solvent out of landfills and rivers. Switching to greener chemicals for some processes also shrinks risks. I’ve watched labs start to swap in less hazardous alternatives, and the benefits show up quickly: fewer health complaints and less time spent dealing with spills. Facts, not wishful thinking, push companies and regulators to drive these changes.
| Names | |
| Preferred IUPAC name | 4-Methylmorpholin-4-ium |
| Other names |
N-Methylmorpholine 4-Methylmorpholine N-Methyl-tetrahydro-oxazine |
| Pronunciation | /ɛn-ˈmɛθɪl-mɔːrfəˌliːn/ |
| Identifiers | |
| CAS Number | 109-02-4 |
| Beilstein Reference | 1209237 |
| ChEBI | CHEBI:44441 |
| ChEMBL | CHEMBL1542 |
| ChemSpider | 11361 |
| DrugBank | DB04278 |
| ECHA InfoCard | 100.015.229 |
| EC Number | 203-744-6 |
| Gmelin Reference | 7547 |
| KEGG | C06529 |
| MeSH | D008781 |
| PubChem CID | 7519 |
| RTECS number | QJ5975000 |
| UNII | YC199097I7 |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID7020022 |
| Properties | |
| Chemical formula | C5H11NO |
| Molar mass | 101.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Amine-like |
| Density | 0.92 g/cm³ |
| Solubility in water | miscible |
| log P | 0.44 |
| Vapor pressure | 5.3 kPa (20 °C) |
| Acidity (pKa) | 7.38 |
| Basicity (pKb) | 7.4 |
| Magnetic susceptibility (χ) | -7.9×10⁻⁶ |
| Refractive index (nD) | 1.422 |
| Viscosity | 1.8 mPa·s (20 °C) |
| Dipole moment | 2.311 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 296.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -285.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3407 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H314 |
| Precautionary statements | P261, P271, P280, P304+P340, P312, P403+P233 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | -1 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.8% - 12% |
| Lethal dose or concentration | LD50 Oral Rat 1,900 mg/kg |
| LD50 (median dose) | LD50 (median dose) of N-Methylmorpholine: Oral-rat LD50: 356 mg/kg |
| NIOSH | KWZ367 |
| PEL (Permissible) | PEL: 100 ppm (parts per million) |
| REL (Recommended) | 400 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Morpholine N-Ethylmorpholine N-Formylmorpholine N,N-Dimethylmorpholine |

