N-Methylformamide: Substance, Science, and the Stakes for Industry and Society
Getting to Know N-Methylformamide: The Story So Far
N-Methylformamide (NMF) rarely grabs attention outside professional labs or chemical plants, but its path through history tells a story about the evolving landscape of chemistry and its tight links to industry and knowledge. This compound, an organic amide formed from methylamine and formic acid, reflects how seemingly obscure molecules end up touching scores of products and sometimes policy as well. Synthetic chemists first played with formamide derivatives around the 19th century, brushing up against NMF as researchers unraveled the possibilities held inside various amide linkages. NMF’s wider adoption didn’t take off until solvent chemistry gained steam in the mid-twentieth century, when industry began to hunt for compounds with better dissolution power and unique reactivity compared to the usual run of alcohols and ethers. As chemical plants expanded their technical toolkits, NMF found work in pharmaceuticals, polymers, electronics, and more. Progress wasn’t just technical; it was also practical, revealing a give-and-take between invention and real-world risk.
What Sets N-Methylformamide Apart
NMF doesn’t look like much: a clear, nearly odorless liquid with a consistency similar to water. This simplicity means it pours easily, mixes with an array of other solvents, and resists many mild acids and bases. The key to its popularity in industry revolves around the structure—a carbonyl stuck between a methyl group and a nitrogen—providing both strong polarity and a handiness in facilitating solubilization. Its polar nature helps it dissolve a wide mix of compounds, especially those that other organic solvents can’t touch. Boiling point sits noticeably high for such a small molecule, topping out near 199°C, which means it stays put in high-heat applications. Density, viscosity, and refractive index all fall in line with other polar aprotic solvents, but NMF holds a unique spot by blending strong solvency with a relatively mild scent and manageable volatility. This attracts process chemists searching for ways to dissolve stubborn reactants, smooth out chemical syntheses, or refine advanced materials.
Technical Specs and Labeling in Real-World Use
Reading a label for industrial-grade NMF gives clues about what matters: purity level, water content, and limits for impurities such as dimethylformamide, formamide, or residual acids. Laboratories favor purity above 99%, avoiding byproducts that can tank sensitive reactions or endanger workers. Trace water proves especially important; even slight hydration calls for extra drying steps, since unwanted reactions with water spoil certain syntheses or lower yields. The routine handling guidance makes sense—don goggles and gloves, store away from acid chlorides or oxidizers, and keep records to keep in line with workplace safety requirements. NMF labeling can also alert workers about incompatibilities, such as strong oxidizers or acids, reflecting lessons learned from years of practice and accident reports.
How Chemists Make N-Methylformamide
Production of NMF scales from simple lab routes to bulk industrial setups. Most synthesis boils down to reacting methylamine with methyl formate or directly with formic acid. Factory processes control temperature, carefully meter out reactants, and capture byproducts like methanol or water; efficiency and safety hinge on steady, predictable conditions. Some plants run continuous production in closed reactors, while others batch up tailored qualities for pharmaceutical synthesis where purity and trace contamination draw intense scrutiny. Manufacturers often recover and recycle solvents to minimize waste, moving toward leaner, more sustainable operations.
NMF in the Reaction Flask: Modifications and More
Inside the reaction flask, NMF plays several roles. Used as a solvent, it helps shape new carbon-nitrogen bonds in fine chemical and pharmaceutical synthesis. It also steps in as a donor for methyl or formyl groups, letting skilled chemists build up more complex organic molecules. The molecule’s amide group undergoes reactions with strong acids, bases, or reducing agents, feeding into the synthesis of specialty compounds and intermediates. Some research teams tinker with NMF derivatives to chase new properties, incorporating it into polymers or seeking transformations into even more reactive or selective reagents.
Synonyms and Alternate Names in Science and Industry
N-Methylformamide travels under several aliases across technical literature and product catalogs. Researchers might call it NMF, methylformamide, or N-methylmethanamide. These names help cross-reference data and standards—especially valuable in the patchwork of regulations and global supply chains. In the world of paperwork, keeping an eye on synonyms avoids mistakes, duplication, or missed safety warnings.
Addressing Safety, Health, and Handling Standards
Working with NMF demands respect for its potential health effects and operational risks. Studies track how it can cause irritation to skin, eyes, and lungs. Prolonged or high-level exposure starts raising concern for liver damage or reproductive toxicity, making ventilation and personal protection non-negotiable in most workplaces. Regulatory groups in the US, Europe, and Asia keep tabs on exposure limits and flag it as requiring strict handling. I remember touring a manufacturing site where they installed real-time detection alarms and demanded double-gloving for every operator—years of occupational safety lessons etched into company policy. Researchers also keep close watch on NMF’s behavior under fire risk; it burns with a faint blue flame, producing toxic nitrogen oxides and carbon monoxide if ignited, so fire response plans figure into plant training and emergency drills. Labeling requirements serve an important function, signaling both hazards and handling steps directly to workers, not just managers.
Why N-Methylformamide Pops Up Across Many Sectors
NMF’s value comes from its versatility. Drug manufacturers use it in making antibiotics, anticancer agents, and anti-infectives, because it dissolves hard-to-handle intermediates and supports reactions less friendly to other solvents. Electronics firms harness its solvency to craft specialty polymers, process advanced semiconductors, and clean surfaces at fine scale. Textile production taps NMF for treating fibers and dyeing; specialty adhesive and resin producers blend it into formulations needing high polarity or thermal resilience. Even academic scientists run on small bottles of NMF for organic syntheses where otherwise stubborn molecules refuse to budge.
Ongoing Research and Product Development
R&D groups test new roles for NMF, both out of necessity and curiosity. Efforts to tweak its structure or swap the formyl part for new groups drive projects exploring green chemistry or designing more benign alternatives with similar physical power. Some teams experiment with immobilizing NMF on solid supports, hoping to boost recovery and slash emissions. Environmental concerns drive the search for methods to degrade or recycle this chemical at end-of-life, especially as pressure builds for all-purpose solvents to clean up their ecological act. The move toward safer, less toxic solvents stands out as a main research direction, forcing both big chemical players and startups to look beyond traditional choices without giving up the powerful solvation and processing capability that NMF brings.
Toxicity and Public Health Research: A Challenge for Safe Progress
Nobody in chemical manufacturing ignores toxicity profiles anymore, particularly for chemicals like NMF with clear records of causing harm above certain exposures. Data from occupational health reports and lab animal studies worry regulators and keep medical monitors busy. Chronic liver effects and reproductive risks show up in workplace health surveys, and industry often responds by tightening exposure limits or mandating stronger ventilation and personal protection. Environmental researchers track NMF contamination in water streams or soil near heavy-use plants, raising important questions about disposal, accidental releases, and long-term ecological consequences. Even so, the compound’s short environmental persistence means its risks largely emerge from direct handling and improper process management, not from widespread or persistent pollution. As the science community broadens understanding of both immediate and chronic risks, recommendations often stress practical controls and safer process substitutions where possible.
Looking Ahead: Future Paths for N-Methylformamide
The road ahead for NMF will follow the push and pull of efficiency, safety, and substitution. As industries seek greener and safer solvents, demand for NMF might level out or even drop if better options appear on the market. Nevertheless, for many high-value uses—especially where process chemistry demands unique solvation capability and reaction stability—NMF isn’t stepping aside soon. Ongoing advances in toxicology, process containment, and real-time monitoring can offset much of its risk, provided companies invest in modernizing how the molecule is managed, tracked, and contained. Researchers continue chipping away at safer preparation routes, improved waste handling, and next-generation derivatives. All these efforts shape the role of a substance that chemistry keeps coming back to, not out of habit, but because it solves stubborn problems, with safety only trailing behind as long as vigilance doesn’t wane.
What is N-Methylformamide used for?
The Chemistry Behind It
Growing up, I watched my father tinker in his home laboratory, always fascinated by strange-sounding compounds and what magic they brought into the world. N-Methylformamide caught my attention as one of those chemicals that quietly powers more than meets the eye. With a formula of CH3NHCHO, it hops between industrial roles, holding its own as a strong solvent and a building block for other chemical creations.
How Factories Use N-Methylformamide
N-Methylformamide isn’t famous, but it pulls a lot of weight behind closed factory doors. Chemical companies rely on it mainly to dissolve polymers, resins, and dyes. Textile plants turn to it to help fibers and dyes mix. In many labs, it steps in to help synthesize medicines and pesticides. Pharmaceutical-makers favor it when crafting complicated molecules—a process that calls for a stable and effective solvent. Polymers, the stuff of plastics and synthetic textiles, blend easier with a little N-Methylformamide in the mix.
Electronics manufacturers count on it, too. Organic electronics need ultra-precise conditions for coating and printing. N-Methylformamide carries out this job, offering just the right touch. Some battery research also taps into it when looking to boost energy storage and extend device lifespans. While it's not a household name, the reach goes much further than most people realize.
Why Its Usage Matters
People rarely pause to think how one molecule can influence so many industries. My cousin’s job switched to remote work during the pandemic because his company finally perfected flexible printed circuits for laptops. The inks and plastics used in those circuits flow right thanks to solvents like N-Methylformamide. Everyday convenience—lightweight electronics, vibrant fibers, smooth-working plastics—depends on behind-the-scenes teamwork between chemists and their favorite chemicals.
Health and Environmental ImpactsFor all its usefulness, there’s a side to N-Methylformamide that brings up tough questions. Its production and handling call for serious caution. Exposure can cause health issues, ranging from mild skin irritation to bigger problems after long-term contact. Factories using this chemical often need to invest in protective gear, stringent ventilation systems, and regular staff training. Regulators watch this chemical closely, asking manufacturers to document safety plans and limit emissions. The risk of spills or careless disposal threatens water and soil health, putting more pressure on companies and local agencies to monitor usage.
Solutions and Safer Practices
No industry likes dealing with red tape, but safety and environmental responsibility come first. Factories must track their chemical usage tightly—something done today with better digital systems and regular audits. My years working in supply management taught me that companies don’t cut corners on safety without consequences. State and federal agencies promote using safer alternatives when possible and insist on concise labeling. For employees, constant training helps spot the warning signs early and respond before small leaks or exposures turn into crises.
Eco-aware companies work to recycle solvents or switch to less toxic substitutes where possible. Universities and research labs keep searching for greener chemistry, hoping breakthroughs will let workers and the environment breathe easier. The process takes time, but it starts with honest talk, strict rules, and genuine care for the people who depend on these materials every day.
What are the safety precautions for handling N-Methylformamide?
Why Safety Matters with N-Methylformamide
N-Methylformamide can sneak up on you. Colorless and with a slight odor, it sometimes gives the false impression of being harmless. In real life, a chemical like this can spell trouble for anyone who gets too comfortable around it. I've worked with chemicals for years and local guidelines only get you so far; daily habits and vigilance matter even more.
Personal Protective Equipment Makes a Difference
Splashing a bit of N-Methylformamide on your skin doesn’t just irritate; this stuff absorbs straight through, so gloves and lab coats ought to be non-negotiable. Nitrile gloves usually stand up better compared to latex, and long sleeves or a lab coat provide an extra shield. Good safety glasses wrap the eyes, keeping splashes out, and a face shield comes in handy for anything more than pipetting.
Breathing in the vapors is another risk. Most open labs have chemical fume hoods, and there’s a smart reason for that. Not all fumes stink or signal their presence. It’s easy to forget the risk until someone gets a headache, or worse. People sometimes underestimate the build-up over a shift; using a proper hood from the start saves a lot of trouble.
Storing Chemicals with Care
Experience in shared labs taught me how chaos creeps into chemical storage. Sticking N-Methylformamide on a random shelf beside acids or bases brings real hazard. Storing it in a cool, ventilated cupboard, away from oxidizers and acids, helps prevent nasty reactions. Label the container every time. I’ve seen half-full bottles with faded labels turn into mysteries over time, which always means more risk.
Tightly closed lids keep fumes in check. It doesn't take long for a spill to ruin a whole storage area – vapors spread, and the cleanup process multiplies tenfold. Clean up minor spills right away, using absorbent pads and gloves, and throw the waste in a hazardous bin, not down a sink.
What Happens After Exposure?
Trouble can start with skin contact, and it gets worse if N-Methylformamide finds its way into the bloodstream. Prolonged exposure sometimes leads to liver or kidney problems. A splash to the eyes, even a drop, means rinsing for fifteen minutes or more. I once saw a colleague brush off a splash and go back to work, but by afternoon the pain set in—no project is worth that.
Quick action helps: shower stations, eyewash fountains, and posted first-aid steps. If someone inhales the vapors and feels lightheaded, they should leave the area and breathe clean air, not just “wait and see.” Sometimes evacuation and real medical care become necessary, especially for big spills or when someone starts to feel sick.
Training and Open Communication
Learning about the chemicals in use, not just reading the labels, pays off. A five-minute safety talk at the start of the week can remind everyone of the basics. People get busy or distracted; I’ve seen folks mix up chemicals or skip gloves when in a rush, but a team that talks safety keeps each other honest.
Up-to-date safety data sheets posted near the storage area get used more than some would think. It’s less about the paperwork, more about trimming confusion and panic when something goes wrong. Solid habits and respect for what you’re handling make all the difference in keeping everyone safe.
What is the chemical formula of N-Methylformamide?
The Chemical Formula Explained
N-Methylformamide carries the chemical formula C2H5NO. It’s made up of two carbon atoms, five hydrogens, a single nitrogen, and one oxygen. Take the backbone of formamide and swap a hydrogen for a methyl group—the result is this molecule. That small shift in atomic layout gives N-Methylformamide its own style and set of uses.
Why Knowing This Formula Matters
Treating formulas like trivia leaves a lot on the table. I remember my first research job, reading reagent labels at a workbench, and realizing a minor difference in chemical makeup could set off totally different reactions in the lab. Mistaking N-Methylformamide (C2H5NO) for plain old formamide might lead to ruined syntheses or wasted time. For folks working in pharmaceuticals or dye manufacturing, a simple misread can create costly setbacks. Reading the formula gives a kind of fingerprint—every atom counts in chemical reactions.
Everyday Impacts of This Compound
N-Methylformamide pops up more often than it gets credit for. Chemists use it as a solvent because it handles high temperatures and helps reactions along when water can’t cut it. Some days, you see it in peptide synthesis—building blocks for biotech and medicine. Many modern drugs or specialty polymers wouldn't exist without someone understanding how and when to use this reagent safely.
Sourcing and Health Considerations
This chemical offers flexibility for researchers, but there's a side to safety that grabs my attention. I’ve seen labs caught off guard by N-Methylformamide’s toxicity. Breathing in fumes or letting skin soak in spills ramps up health risks. The U.S. National Institute for Occupational Safety and Health flags it as hazardous, pushing for solid ventilation and strict use of gloves. Knowledge about what C2H5NO stands for goes beyond curiosity; it’s about taking care of your team and yourself in the lab.
Addressing the Challenges
Many chemical mishaps boil down to forgetting what’s in the bottle. It helps to teach students more than rote memorization. Instructors and supervisors making safety a real part of the routine—both at universities and in industry—reduce incidents. Clear labeling in storerooms and honest talk about exposure risks set up a safer environment. Sometimes, the difference between a safe synthesis and an accident comes from paying attention to that single methyl group in a formula like C2H5NO.
Improving Chemical Literacy
Newcomers benefit from learning formulas in the same breath as their stories and dangers. Online resources, community training, and company policies—these pave the way for smarter, safer handling. Early on, I saw mentors link every chemical formula to a real consequence or practical story, not just a quiz answer. That shaped how I work and teach today. N-Methylformamide serves as a perfect example: a formula worth much more than its numbers and letters.
How should N-Methylformamide be stored?
Why Storage Details Matter More Than You Think
Some chemicals demand a little more respect in how we handle them, and N-Methylformamide stands out on that list. It acts as a solvent in many lab and industrial settings, but people sometimes overlook what happens after delivery. Years ago, while assisting at a small research facility, I caught a whiff of a heavy odor around some poorly sealed drums. A humidity spike had caused swelling and a bit of leakage. That’s a warning sign: improper storage can open up risks of contamination, health problems, and wasted resources.
Picking the Right Space and Conditions
You can spot trouble before it starts by choosing a dry, well-ventilated storage area for this compound. Closets with no vent fans or cramped supply rooms don’t cut it. N-Methylformamide evaporates, releasing fumes that can irritate eyes and lungs or even set off headaches and dizziness. Over time, these fumes won’t just hurt staff—they corrode nearby containers, shelving, and even electronics. This chemical doesn’t just ask for a cool, shaded corner; it demands a space with robust air circulation and rigorous checks for any sign of leaks.
Sunlight and heat ruin stability fast, so it makes sense to keep stocks out of direct sun. Temperatures below room temperature help, but freezing can stress containers and spark breakage. Reliable thermostats and backup power for climate control protect your supply during power outages. I remember someone once lost a full batch to a heatwave because the AC tripped—cost the lab both cash and time they hadn't budgeted for.
Container Choice and Spill Prevention
Sturdy, compatible containers are non-negotiable. In labs, glass, or high-density polyethylene containers work best, since this chemical eats away some plastics and metals over time. I once saw a steel drum begin to rust at the rim after only a few weeks. Never top-off containers all the way. That air gap helps manage pressure, especially if temperature shifts or accidental heating occur. Each container should seal tight with no cracks or warped caps.
Labels deserve attention. Make legible, chemical-resistant labels showing both the exact contents and hazard warnings. Outdated or faded labels confuse teams and breed mistakes—just one mix-up can spark costly cross-contamination or adverse reactions.
The Importance of Access Controls and Emergency Readiness
Beyond storage conditions, access does matter. Don’t stash N-Methylformamide where just anyone can reach it. Only trained staff with proper safety gear should handle these containers. I’ve seen people brush off glove or mask use, thinking routine transfers mean business as usual. Respirators, goggles, and gloves aren’t optional—they’re shields against accidental exposure.
Spill response gear saves the day. Having neutralizing agents, absorbents, and well-marked eye wash stations nearby stops a spill from becoming a disaster. Safety drills for cleanup before an incident help teams react quickly, especially if someone gets splashed or inhales fumes.
Learning from Others Saves Trouble
Guidelines from the Occupational Safety and Health Administration or chemical industry experts provide the backbone, but individual experience rounds it out. Pay attention to published best practices. I’ve learned that a little extra care in labeling, temperature monitoring, and spill protocol pays off in fewer accidents, lower costs, and peace of mind for the whole team.
Is N-Methylformamide hazardous to health or the environment?
What We Know About N-Methylformamide
N-Methylformamide pops up in labs and factories across the globe. Scientists rely on it to help shape plastics, textiles, and a host of other products. This chemical isn’t some rare compound only a handful of people handle. It shows up in places workers might not expect—under the hood in paint removers, among solvents for chemical reactions, and sometimes drifting into waste streams.
Health Concerns for People Working With N-Methylformamide
Anyone who’s ever caught a whiff of solvent in a shop knows just how harsh some of these chemicals feel on the nose and lungs. N-Methylformamide carries risks that deserve real respect. Direct exposure—either through breathing fumes or splashing on skin—can irritate eyes, skin, and the respiratory system. If a worker gets a strong dose, symptoms might go beyond red eyes or itchy skin, tipping into nausea, headaches, or worse.
Science keeps turning up links between N-Methylformamide and liver effects, which always rings alarm bells in the occupational health world. A chemical that slips through skin or sneaks past a mask doesn’t just leave signs you can see or feel right away; it can do quieter damage over time. Lab studies have shown that with regular, low-level exposure, liver function starts drifting off course. The International Agency for Research on Cancer calls N-Methylformamide possibly carcinogenic to humans—meaning this chemical shouldn’t be handled lightly.
Environmental Impact: Out of Sight, Still a Problem
Runoff doesn’t have to soak a field to turn into a bigger issue later on. N-Methylformamide dissolves easily in water and doesn’t stick around in the soil, so it starts showing up downstream pretty quickly. Some of the breakdown products also raise concerns. Aquatic life can pay the price, with fish and smaller creatures bearing the brunt of contamination. Even low levels in waterways can push local ecosystems out of balance.
From personal experience running environmental tests on industrial discharge, it’s easy to underestimate how quickly one accident can spread. A minor leak—one someone cleans up in an afternoon—can mean trouble for a nearby stream for months. N-Methylformamide’s knack for moving through water means that once it’s out, controlling it gets complicated fast.
Balancing Industrial Use With Real Safety Steps
Once upon a time, folks used hazardous chemicals in everyday settings without blinking. Industry improvements over the years came from hard-won lessons. The way forward with N-Methylformamide has to reflect what history has taught us: respect, vigilance, and up-to-date know-how save lives.
For employers and workers who rely on N-Methylformamide, substitution deserves a close look. Seeking out safer alternatives isn’t just about ticking regulatory boxes—it’s about protecting people who handle these chemicals day in and day out. When alternatives aren’t practical, proper ventilation, protective gear, and real, hands-on training make a difference between safe shifts and trips to the clinic.
Waste management plays a big role. Preventing spills, treating wastewater, and setting up monitoring help keep this chemical out of groundwater and local rivers. Community engagement matters, too. Local residents deserve updates about what’s happening by their homes and water supplies. Communication, transparency, and follow-up testing bring trust back to the table after chemical releases.
Looking Ahead
N-Methylformamide isn’t going away just yet, and neither are the health and environmental questions it raises. Continued research, improved oversight, and real-world safety measures set the stage for using these chemicals responsibly. With industry, regulators, and communities pulling in the same direction, the balance between utility and safety gets closer to where it should be.
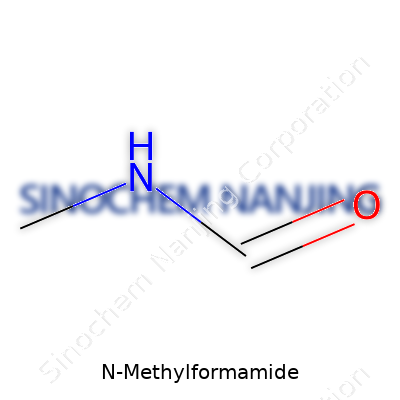
| Names | |
| Preferred IUPAC name | N-methylmethanamide |
| Other names |
NMF Methanamide, N-methyl- Formamide, N-methyl- Methylformamide N-Methylmethanamide |
| Pronunciation | /ɛn-ˈmɛθ.əl.fɔːrˌmæm.aɪd/ |
| Identifiers | |
| CAS Number | 123-39-7 |
| Beilstein Reference | 605374 |
| ChEBI | CHEBI:47443 |
| ChEMBL | CHEMBL1401 |
| ChemSpider | 6195 |
| DrugBank | DB02137 |
| ECHA InfoCard | 17d5137e-12b7-432a-bbc1-d92753fd4e32 |
| EC Number | 205-549-2 |
| Gmelin Reference | 7704 |
| KEGG | C01740 |
| MeSH | D002589 |
| PubChem CID | 7462 |
| RTECS number | LQ9625000 |
| UNII | 6U785Q0P0T |
| UN number | UN2265 |
| CompTox Dashboard (EPA) | CXTIL1Z6SI |
| Properties | |
| Chemical formula | CH3NHCHO |
| Molar mass | 73.09 g/mol |
| Appearance | Colorless liquid |
| Odor | Faint amine odor |
| Density | 0.999 g/mL |
| Solubility in water | Miscible |
| log P | -0.86 |
| Vapor pressure | 0.67 mmHg (20°C) |
| Acidity (pKa) | 23.3 |
| Basicity (pKb) | 8.52 |
| Magnetic susceptibility (χ) | -48.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.429 |
| Viscosity | 0.802 cP (20 °C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.3 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -141.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -491.9 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N01AX12 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H312, H319, H360D |
| Precautionary statements | P210, P273, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 31°C |
| Autoignition temperature | 448°C |
| Explosive limits | Explosive limits: 2.2–16% |
| Lethal dose or concentration | LD50 oral rat 3,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3.9 g/kg (oral, rat) |
| NIOSH | MW1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of N-Methylformamide: 10 ppm (parts per million) |
| REL (Recommended) | Gloves, lab coat, chemical splash goggles |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Formamide Dimethylformamide Methyl isocyanide N,N-Dimethylformamide N-Ethylformamide |

