N-Methylacetamide: Chemistry in Action and the Realities Behind the Molecule
A Quick Look Back: Historical Development
Chemists have put years into figuring out ways to tweak molecules for everyday use, and N-Methylacetamide (NMA) sits among those compounds with a rich scientific past. Researchers originally started working with amide groups back in the early 1900s, but NMA became particularly important after the Second World War, when the need for better solvents and chemical intermediates grew rapidly. The straightforward structure of NMA caught the attention of scientists who saw its potential not just as an academic curiosity but as a solvent and a building block for more complex chemical syntheses. Through the decades, production methods shifted, with early reliance on resource-heavy batch processes eventually giving way to more efficient continuous operations that could keep up with industrial demands.
Product Overview: Where It Fits In
N-Methylacetamide has become a staple in many research labs and production lines. Labs use it not only because it dissolves a wide range of substances but also because, compared to larger amides, it’s relatively easy to handle and mix. Chemical companies list it alongside more famous solvents like dimethylformamide and dimethylacetamide, but NMA often comes up as a simpler, slightly less polar cousin, offering a unique combination of properties without some of the harsh health or environmental impacts seen in other choices.
Physical and Chemical Properties: What Makes It Tick
In any lab, you can tell a lot from how a liquid pours, smells, and reacts in different conditions. NMA is a colorless liquid with a mild scent and a definite touch of slipperiness on the skin—an early warning that it penetrates surfaces easily. Its boiling point and reasonable polarity make it a useful solvent for organic chemistry applications, especially for experiments involving peptides and hydrogen bonding. Unlike water, which evaporates too quickly or fails to dissolve fats, NMA’s middle-of-the-road nature lets it play in both polar and nonpolar regions. Its chemical backbone, which includes both an amide nitrogen and a methyl group, opens it up to modifications and further reactions, a big reason why you find it in so many laboratory protocols.
Getting the Details Right: Technical Specifications & Labeling
Gel packs of N-methylacetamide show up with purity level percentages and the odd batch number, but beyond these practicalities, labeling helps track shelf life and potential hazards. Labs take care to check the water content, storage temperature, and possible impurities because any one of these factors can shift results when NMA’s used as a reagent. Nobody wants to run a synthesis only to find out trace water content threw off a reaction, so these details become pivotal in research settings. There’s a real lesson here—science rarely tolerates cut corners, especially with solvents that double as reagents.
How It’s Made: Preparation Method
Large-scale production of NMA usually goes through methylamine and acetic anhydride or similar routes. These starting materials combine in straightforward reactions, often under controlled temperatures to prevent byproducts. For those who have tried it on a smaller scale, keeping the reaction under control can be tricky. There’s a decent risk of forming side products or releasing irritating gases, so experience and care count for a lot. Industrial players use reactors fitted with pressure and temperature controls to keep yields high and impurities low, as purification after the fact can get expensive and labor-intensive.
Reacting for Value: Chemical Reactions & Modifications
NMLE’s structure puts it right in the middle of some key chemical transformations. In organic syntheses, it takes part in hydrogen bonding and acylation reactions. Chemists often use it as a model compound to study peptide bonds because it mimics the backbone of proteins. Its methylated nitrogen becomes a target for further reactions, like quaternization, making it a stepping-stone in crafting more specialized chemical agents. Researchers use it to probe how enzymes interact with amides, so it doubles as a tool for both chemical synthesis and basic biological research.
Getting Lost in Translation: Synonyms & Product Names
If you ask a dozen chemists about NMA, a few might call it N-methyl ethanamide or acetic acid N-methylamide. Catalogs and safety datasheets sometimes list it under N-acetylmethylamine. No matter the name, what matters most is double-checking you’re working with the right stuff. Slip-ups happen more than people want to admit, especially if a project passes from person to person or moves between labs with different naming preferences.
Staying Safe: Operational Standards
Even substances that seem mild on first impression need strict process controls. NMA can irritate eyes and skin, and inhaling its vapors brings headaches and discomfort. Labs put written protocols in place—ventilation, gloves, goggles—because even a small mishap can lead to health complaints. Safety training covers proper spill clean-up and highlights the risks of storing NMA near incompatible chemicals. In bigger operations, chemical sensors, fume hoods, and safety showers aren’t optional extras; they’re daily essentials. For me, the best lesson has come from watching experienced technicians always double-check a bottle’s label and make sure containers get tightly sealed after use.
Application Area: Where NMA Shows Up
N-methylacetamide gets plenty of work in pharmaceutical development, textile processing, and advanced materials research. Its role as a solvent for peptide synthesis keeps it relevant in biotech, especially given the ongoing growth of protein-based drugs. Textile manufacturers use it to treat fibers and modify dye uptake. Some formulations for polymers and advanced coatings include NMA for its unique solvent qualities, and researchers continue finding new uses in materials science. Battery work, for instance, has explored NMA as a solvent for electrolytes, drawing interest as companies search for alternatives with lower toxicity or improved stability. Unlike “miracle cure” molecules that flash into the spotlight, NMA has built its resume over decades by quietly underpinning major technical advances without demanding headlines.
Research & Development: Always Something New
Even after all these years, researchers dig deeper into NMA’s chemistry, whether that’s tweaking its structure for greener processes or finding ways to cut environmental impact. Some R&D teams look at recycling spent NMA instead of treating it as waste, aiming not just to save money but cut down on hazardous emissions. Computational chemists use it to model protein folding and drug binding, comparing predictions against real-world lab data. Progress hinges on sharing data and learning from both successes and accidents. This community approach pushes the science ahead faster than any one company or lab could do alone.
Toxicity and Environmental Considerations
No compound gets a free pass on safety. Toxicity research on NMA has focused on exposure risks, especially for people with long-term or repeated contact. Acute exposures cause irritation, but chronic health effects need continued study. Environmental impact also matters; runoff or improper disposal can harm aquatic life and disrupt wastewater treatment systems. Regulators in many countries now expect clear spill response plans and encourage companies to recycle or filter solvents wherever possible. Real-world experience tells you: regulations only go so far—engineering controls and a culture of safety make a bigger difference than paperwork ever could.
The Road Ahead: Future Prospects
Looking ahead, companies and labs will keep figuring out where NMA fits best as priorities shift toward greener, safer, and more efficient chemicals. With moves to phase out more hazardous solvents, NMA serves as a practical candidate for many applications, not just because of its solvency but also its lower volatility and moderate toxicity profile. Researchers push to develop bio-based production routes, hoping to cut reliance on fossil resources. There’s a lot of potential, but the benefits only show up where users take safety and environmental stewardship seriously. As someone with years spent both at the lab bench and in chemical safety planning meetings, I’ve seen how steady improvements in chemistry, procedure, and awareness work together to push the field forward. NMA, unassuming as it seems, stands as a quiet proof that chemistry shapes the present and the future in ways most people never notice, but everyone depends on.
What is N-Methylacetamide used for?
A Look at N-Methylacetamide’s Real-World Roles
N-Methylacetamide, often called NMA, plays a supporting role in many areas that touch everyday life, though most people haven’t heard of it. I came across NMA during collaborations with pharmaceutical teams, and its utility kept popping up as a topic worth attention. What makes NMA valuable is its knack for mixing with both water and organic solvents, which makes it an unglamorous but reliable chemical helper in labs and manufacturing plants.
Use in Pharmaceuticals
Pharmaceutical chemists rely on NMA for more than one reason. NMA stands out as a solvent for peptide synthesis—a process important for creating drugs that mimic proteins in the body. Peptides are fragile, and harsh chemicals easily break them down. NMA provides a stable setting where these delicate reactions run their course. Companies banking on the next breakthrough in biotech use NMA behind the scenes for these reasons.
Helping Hands in the Lab
Walk into any research lab working with proteins, and chances are good you’ll spot a bottle of NMA. Researchers use it as a model compound to understand how solvents interact with proteins and peptides. NMA’s simple structure allows scientists to map out hydrogen bonding behavior, building a knowledge base that supports drug design, protein folding studies, and even the development of safer medicines. This work forms the backbone of many scientific advances that later become household knowledge.
Textile Fiber Production
NMA doesn’t just live in the pharmaceutical world. Factories spinning synthetic fibers count on NMA as a solvent for producing polyacrylonitrile fibers—better known as acrylics. These fibers keep sweaters soft, upholstery durable, and outdoor gear water-resistant. The solvent helps process polymers into threads that stand up to day-to-day use, while manufacturers recover NMA for reuse, balancing industrial needs with environmental concerns as best they can.
Electronics and Lithium Batteries
Each year, more devices charge up on lithium batteries, and NMA finds a place here as well. Researchers tap into its chemical stability and ability to carry lithium ions, giving it a spot in electrolyte research for next-generation batteries. By supporting the fine-tuning of how ions travel, NMA quietly plays a role in powering everything from phones to electric cars.
Risks and Responsible Handling
With NMA’s usefulness comes a responsibility to handle it wisely. Studies report that NMA can irritate eyes and skin, and breathing in its vapor brings health risks. Long-term exposure should be avoided in any workplace that values employee health, and organizations like OSHA and NIOSH have put out guidelines to keep people safe. Companies now lean on closed systems, protective equipment, and smart ventilation. Training workers about these risks transforms chemical use from a danger into a day-to-day routine—one that keeps science moving forward without sacrificing well-being.
Where to Go from Here
The push for greener chemistry grows stronger every year. Some research efforts target ways to replace solvents like NMA with safer, bio-based alternatives that don’t pose chronic health worries. Until these substitutes take full hold, those of us working with—or living near—industries that use NMA count on strict safety, good science, and public transparency. Knowing where a substance like N-Methylacetamide turns up, how it’s kept in check, and the work being done to reduce risks connects us all to a wider world of science and safety.
Is N-Methylacetamide hazardous or toxic?
N-Methylacetamide in the Real World
N-Methylacetamide pops up in labs and industry every day. Chemists value it for how it works as a solvent and helps with peptide synthesis. Anyone who spends time handling chemicals has run into it in catalogs or even on a lab bench. Its liquid form doesn’t seem alarming at a glance, and it doesn’t come with the same scary reputation as some classic lab hazards. But questions about safety deserve clear and honest answers, drawn from experience and solid science.
What Science Tells Us
Take a look at the research and safety data: N-Methylacetamide ranks as only moderately hazardous by current chemical safety standards. The U.S. Environmental Protection Agency gives a nod to its low volatility—this means fumes are less likely to cause surprise headaches or dizziness after a quick spill. I have handled it with simple gloves and goggles, and never ran into trouble from smell or irritation through short exposure.
Studies point to a low tendency for skin irritation. Inhalation does not often produce acute toxic effects at the typical lab scale, which can be a relief for anyone in a university research setting. Mice exposed to this compound for short periods did not show strong symptoms, which lines up with its reputation in the scientific community.
Dangers That Hide in Plain Sight
Long-term dangers tell a different story. Research with rats hints at developmental issues after prolonged, high-dose exposure. Birth defects appeared in some test cases, especially when mothers were exposed during pregnancy. The National Toxicology Program cautions against carelessness for that very reason. If you run a lab or manage an industrial site, these facts put a responsibility on your shoulders.
People have a right to know what risks they’re working with, not just what the safety data sheet lists in small print. That’s where transparency becomes vital. Most students or workers—myself included during grad school—don’t dive into chemical toxicology literature unless someone stresses the need.
What Real Safety Looks Like
Relying on gloves, goggles, and fume hoods always makes sense. Even if symptoms are rare, exposure can build up over time, especially for folks seeing this compound every day for years. Clean lab practice builds a culture that values health over “getting the job done fast.” I learned pretty quickly that shortcuts only look good until someone ends up with a persistent cough or a rash that doesn’t quit.
Keeping this substance off the skin and out of the air remains the smart move. Anyone pregnant or planning a pregnancy gets extra protection—things like special assignments or different projects. Lab managers can introduce air monitoring and run regular health checks for peace of mind.
Common-Sense Steps Going Forward
Universities and companies make the best progress by leading more training on chemical hazards. Posters and online quizzes work as reminders, but hands-on training brings the message home. If someone spots a cough or rash, a prompt report to supervisors stops little problems from turning into lifelong health issues. Waste disposal matters, because nobody wants this stuff winding up near drinking water or in the air.
Chemicals like N-Methylacetamide belong in labs, not forgotten in dusty back rooms or handled by folks who’ve never cracked a safety guide. Careful respect, based on both personal experience and published science, turns possible hazards into manageable, everyday tools.
What is the chemical formula of N-Methylacetamide?
Getting to Know N-Methylacetamide
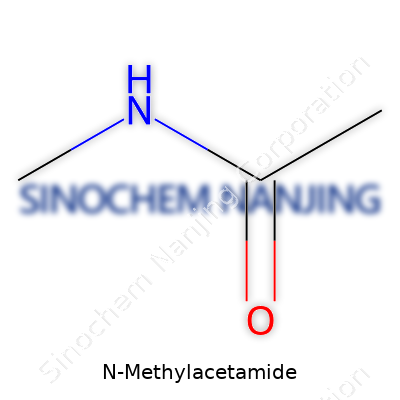
N-Methylacetamide carries the chemical formula C3H7NO. At first glance, this compound seems tucked away in research labs, but it plays a bigger role in today’s science-driven world. Each atom in its formula tells a story: three carbons, seven hydrogens, one nitrogen, and one oxygen. This precise structure allows N-Methylacetamide to slip into fields as diverse as pharmaceuticals, materials science, and peptide research.
Why the Formula Means More Than a Name
Chemical formulas aren’t just about memorization for an exam. I once watched a researcher struggle with synthesizing a new peptide simply because N-Methylacetamide was confused with a similar compound. A small mistake in the formula, and everything was ruined—money, time, and effort. In pharmaceutical labs, this detail can spell the difference between breakthrough and setback. The exact composition—C3H7NO—shapes how it dissolves, interacts, and binds with other molecules. This matters in drug design and delivery, where every atom counts.
Function in Research and Industry
N-Methylacetamide acts as a common solvent. Lab techs choose it for its polarity and ability to form hydrogen bonds, traits that depend directly on its molecular makeup. The amide group puts it in high demand for peptide synthesis and protein denaturation. Over the years, I have seen chemists rely on its structure to mimic peptide bonds, helping unravel how proteins fold and function in real life.
The Drive for Safety and Compliance
Having the correct formula is not just an academic concern—it’s about working safely. Mishandling a chemical because of mix-ups risks both health and reputation. Regulatory bodies rely on unambiguous formulas like C3H7NO for classification systems, labelling, and handling guidelines. For example, the Global Harmonized System (GHS) lists chemicals by exact identity, which helps prevent tragic mistakes. Years ago, a major supplier caught a mislabelled shipment only because they double-checked the formula. Avoiding incidents starts with what’s written on the container.
Solutions for Consistency
Training and peer review stand out as the best defenses against chemical confusion. In labs where I’ve worked, double-checking formulas before ordering or shipping prevented more than a few close calls. Journals and databases like PubChem and ChemSpider keep robust, peer-reviewed records for compounds, letting scientists verify identities before use. Digital inventory systems, equipped with cross-referenced molecular databases, catch human errors. Lab teams who regularly brush up on safety protocols don’t just talk about formulas—they work fluently with them.
Looking Past the Surface
Few outsiders know the daily impact of these details, but anyone involved with chemicals carries a responsibility. N-Methylacetamide’s formula gives it identity in a crowded chemical landscape. Its role in progress depends on clarity and vigilance. For all who handle it—scientists, suppliers, regulators—the formula C3H7NO connects the dots from research to safety, ensuring every discovery is built on a solid foundation.
What are the storage and handling recommendations for N-Methylacetamide?
Getting Real About Laboratory Chemicals
N-Methylacetamide has found steady work in labs and industries, and for plenty of reasons. It’s a helpful solvent and sometimes slips into pharmaceutical synthesis or the formulation of specialty chemicals. Yet, for all its uses, plenty of folks forget the basics of storage and handling. Overlooking those basics opens the door to wasted product, safety risks, and headaches with compliance. Speaking as someone who’s cleaned up chemical spills more than once, I know careful handling isn’t just red tape—it keeps you safe and avoids lost time and money.
Keep It Cool, Keep It Sealed
Leaving N-Methylacetamide in the wrong spot changes its game. High temperatures push it closer to breaking down. I’ve seen colleagues try to squeeze extra shelf space and end up with a ruined batch because a storage area got too warm. The right call is sticking to a cool, dry place—think 15 to 25°C. Being around heat sources or direct sunlight only drives up risk of container damage or chemical degradation.
Humidity might not seem like much of a threat, but over time moisture can creep into containers unless they’re tightly sealed. Water in your chemical means unpredictable reactions or changes in concentration. From experience, reverting even slightly contaminated product takes more time and effort than most expect. Best bet is always to use containers with airtight seals. Glass works, but high-density polyethylene does fine as well—just make sure the cap’s back on after every use.
Avoiding Contamination and Spills
You can tell a lot about a lab by how it labels and stores its chemicals. Sharp, visible labels save time and prevent mistakes. Every time I see faded marker or torn tape on a bottle, I get nervous. It pays to be consistent with proper, printed labels. N-Methylacetamide is usually clear, so nobody spots it quickly if a little gets spilled. Pictograms, hazard symbols, and expiration dates aren’t decoration—they’re life-savers.
In my early days, I watched runaway spills travel across benchtops because someone set a bottle too close to the edge. Secure placement away from traffic or vibration limits risk. Even better is using secondary containment—bottle in a tray or spill-proof bin. This simple step prevented at least two close calls in my time.
Personal Safety Counts, Too
A clean, organized work area matters just as much as safety data sheets or fancy storage cabinets. Gloves and safety glasses earn their keep—N-Methylacetamide shouldn’t touch skin or get near your eyes. A decent fume hood is not overkill: inhalation can irritate, and no one wants mystery symptoms halfway through a shift. I’ve made it a habit to wash hands after handling any solvent, no matter how much in a rush. It’s paid off in fewer rashes and no accidental exposures.
Maintaining Quality, Protecting People
Following these methods doesn’t take a degree in chemical engineering. They come down to building habits and keeping respect for the chemicals you work with. Avoiding shortcuts means safer workers, better results, and less waste. Regular checks keep everyone on track: once a month, poke through inventory for leaks, outdated stock, or broken seals. Those few minutes could save an expensive headache or a lost workday, and in my experience, management never complains about fewer incidents.
What is the purity or grade of N-Methylacetamide available?
Why Purity Standards Actually Matter
N-Methylacetamide comes up a lot in labs and manufacturing spaces. People don’t always stop to question where it came from or what’s in the bottle, but purity makes waves in the outcome of your experiments or products. Purity isn’t just about numbers on a label. If you’ve spent enough time in a lab, you’ve probably witnessed how a small impurity can sabotage a reaction, skew your data, or waste hundreds of hours. These setbacks can feel pointless if the supplier never spelled out the details up front.
Packing and Selling N-Methylacetamide — How It’s Labeled
Most suppliers target two main grades. One is technically called “analytical reagent grade,” with a typical purity above 99%. The other, “lab grade,” usually tracks between 97 and 99%. People tend to pay more for analytical stuff, but the cost comes with some peace of mind, especially for those worried about cross-reactions. Any quick search through catalogs from Sigma-Aldrich or Thermo Fisher confirms these percentages. The pricing difference stands out if you're purchasing for an industrial contract versus basic teaching labs.
Some companies also offer pharmaceutical grade N-Methylacetamide. These are supposed to meet strict standards for drug or ingredient purposes. Rigorous checks filter out impurities like water, methanol, or unreacted precursors. In real-world experience, these checks aren’t just paperwork — auditors and chemists get involved, and someone always asks for HPLC or GC documentation. When you’re responsible for purity, you can’t skip this step.
What Happens If You Take Shortcuts
In the daily rush, some groups try to save money by picking lower-purity materials. It rarely pays off. I’ve worked with solvents and intermediates that triggered byproducts nobody wanted. Small differences, even fractions of a percent, push chemists to troubleshoot endlessly. I once spent a week rerunning an experiment after realizing a cheaper batch brought in acetone as a contaminant. No one tells you these “affordable” purchases can kill a whole project or tarnish your results in published studies.
Knockoff Batches and Hazy Documentation
Counterfeit or misrepresented chemicals are not a myth. Anyone who’s ordered overseas knows the game — some drum labels sound legit, but closer checks can reveal all sorts of oddities. Even large companies get caught off guard and end up testing every lot. Reputable suppliers usually attach a clear Certificate of Analysis. This matters more than sales talk because that document reveals water content, heavy metals, and sometimes a full GC trace. Instead of assuming a drum is fine, anyone handling critical reactions needs to read those numbers before starting work.
Seeking Real Solutions
No magic bullet solves purity problems, but there are better approaches. Don’t chase the cheapest line item if the reaction means anything to your company or reputation. Real quality control involves both trusting your supplier and double-checking with your own analytical team. Some labs set aside extra samples from every new delivery just to confirm purity and run spot tests. Investing time here prevents drama later.
Purity is a daily conversation, sometimes heated, between buyers, chemists, and managers. It’s worth investing in quality up front and talking openly about expectations. You don’t always get what you pay for, but you almost always get what you measure.
| Names | |
| Preferred IUPAC name | N-Methylacetamide |
| Other names |
NMA Acetic acid, N-methylamide N-Methylacetic acid amide Acetylmethylamine N-Methyl ethanamide 1-Acetamidoethane Ethanoic acid N-methylamide |
| Pronunciation | /ɛn-ˌmɛθɪl-əˈsiːtəˌmaɪd/ |
| Identifiers | |
| CAS Number | 79-16-3 |
| 3D model (JSmol) | `3D model (JSmol)` string for **N-Methylacetamide**: ``` CN(C)C=O ``` |
| Beilstein Reference | 604126 |
| ChEBI | CHEBI:28385 |
| ChEMBL | CHEMBL1232234 |
| ChemSpider | 7213 |
| DrugBank | DB02009 |
| ECHA InfoCard | echa.europa.eu/infocard/100.003.162 |
| EC Number | 203-483-8 |
| Gmelin Reference | 8286 |
| KEGG | C01602 |
| MeSH | D008768 |
| PubChem CID | 6979 |
| RTECS number | PJ8750000 |
| UNII | UNII: 8EF0P99T2R |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C3H7NO |
| Molar mass | 87.12 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.945 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | -0.77 |
| Vapor pressure | 0.16 mmHg (25 °C) |
| Acidity (pKa) | 15.1 |
| Basicity (pKb) | 15.35 |
| Magnetic susceptibility (χ) | -8.52·10⁻⁶ |
| Refractive index (nD) | 1.437 |
| Viscosity | 0.896 cP (25 °C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 201.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -172.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4167 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | N01AX10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P280, P264, P305+P351+P338, P337+P313, P302+P352 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 86°C |
| Autoignition temperature | 424°C |
| Explosive limits | Explosive limits: 2.4–14.4% |
| Lethal dose or concentration | LD50 Oral Rat 3,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,980 mg/kg (rat, oral) |
| NIOSH | KC8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for N-Methylacetamide: 10 ppm |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Acetanilide Acetamide Dimethylacetamide |

