N-Ethylpyrrolidone: Unpacking a Modern Chemical Workhorse
The Journey from Discovery to Essential Ingredient
N-Ethylpyrrolidone didn’t spring out of nowhere. Like many organic solvents that found their way into labs and factories, it traces back to decades of synthetic chemistry work. While pyrrolidones gained attention in the mid-20th century, especially when the demand for high-performance solvents took off, the ethyl variant carved out its own path. In my early lab days, the stories from senior chemists pointed to the rise of these molecules during the rapid expansion of electronics and specialty coatings in the 1970s and 80s. Unlike its better-known cousin N-methylpyrrolidone, N-ethylpyrrolidone drew notice for its unique mix of solvency and moderate toxicity—qualities that made people give it a second look as industry pushed for smarter solvents.
Substance Overview and How it Stands Out
You won’t find N-Ethylpyrrolidone on store shelves, but inside production plants, it helps solve tough formulation problems. With a five-membered lactam ring and a simple ethyl side chain, it slips into the “polar aprotic” solvent niche. I remember troubleshooting a stubborn polymer blend where common products failed—the technician reached for a small flask of this stuff and it fixed the issue within minutes. That’s about as down-to-earth a recommendation as any. Unlike high-boiling aromatics, it evaporates at a practical rate but still manages to dissolve stubborn organic and some inorganic materials. Some colleagues used to comment that it often “stays out of your way” by not reacting with the chemistry at hand, which is a rare feature for a solvent this strong.
Physical and Chemical Properties Every User Should Know
The chemical world praises N-Ethylpyrrolidone for a reason. At room temperature, it gives you a clear, nearly odorless liquid. Those who’ve handled it appreciate how it flows easily, which helps both in lab-scale mixing and large-scale processes. Its boiling point sits comfortably around 200°C, which means it won’t disappear too quickly during extended operations or more complex syntheses. Water and many organics accept it as a mixing partner. The basic structure—an amide ring with an ethyl group—also gives it a balanced character: not too reactive, not too slippery as a solvent. Anyone concerned about side reactions during organic synthesis tends to find comfort knowing it rarely surprises you under normal conditions.
Technical Specifications and Labeling Practices
Regulated markets make sure you know exactly what you’re using. N-Ethylpyrrolidone usually arrives at high purity—often above 99 percent. Anyone who’s watched a quality inspector at work can tell you the seriousness of keeping batch logs, checking moisture content, and tracking density. The solution’s water content, in particular, plays a big role; too much moisture, and delicate syntheses can go off track. Labels typically reflect crucial facts like CAS number, purity level, and storage guidelines, which helps keep the guesswork out of inventory management. Experienced plant operators and researchers often appreciate short, to-the-point labeling that lets them identify risks and compliance at a glance.
Behind the Scenes: How it’s Made
N-Ethylpyrrolidone production doesn’t happen by accident. Starting with gamma-butyrolactone, chemists bring together ammonia or a primary amine under pressure, forming the core pyrrolidone structure. For the ethyl variant, ethylamine leads the reaction, feeding in the ethyl group. Each step demands close temperature and pressure control, and a deep understanding of reaction kinetics improves output while limiting impurities. I’ve witnessed talented process chemists adjust parameters mid-batch, wrangling a tricky mixture back on course with nothing more than years of hands-on experience and a keen eye at the reactor window. That combination of practical knowledge and advanced technology underscores why reliable manufacture isn’t just a given.
Practical Chemistry: Reactivity and Modification
N-Ethylpyrrolidone won’t break the rules, but it doesn’t just sit idle, either. It resists easy hydrolysis and oxidation, which makes it popular for systems sensitive to water or air. If you push it hard enough—say, with powerful acids or bases—it might eventually open its lactam ring, but that typically doesn’t happen in most workflows. Some research teams do tailor the molecule, introducing substitutions on the nitrogen or carbon atoms to tune physical properties or reactivity. Those efforts mostly show up in academic or specialty chemical circles, illustrating how this solvent’s versatility continues to attract fresh ideas and applications.
What People Call It: Names and Labels
Chemists relish synonyms, and N-Ethylpyrrolidone appears under a handful of names. You’ll hear “NEP” tossed around in meetings, while formal documents prefer N-Ethyl-2-pyrrolidone. People familiar with international chemical regulations often check the CAS registry—CAS 2687-91-4—for unambiguous identification. This avoids confusion with other solvents, particularly with regulations tightening around similar compounds. That consistency in naming smooths out communication, especially between purchasing, safety teams, and regulatory bodies.
Standards for Safety and Use on the Ground
Nobody cuts corners with solvents like this. Gloves and eye protection remain standard; the skin absorbs NEP, though less than some relatives, so direct contact raises exposure risks. In practice, only well-ventilated spaces see regular use. Occupational standards, such as those from OSHA or regional health agencies, address both acute inhalation limits and long-term exposure risks. The push for lower workplace exposure limits grows stronger by the year, especially as more toxicity studies highlight potential reproductive health concerns. Emergency protocols make a real difference: teams drill for spills, and storage facilities keep stocks away from incompatible chemicals. Veteran operators almost always remind the rest of us never to underestimate any polar aprotic solvent, even one that seems mild on paper.
Why Industry Keeps NEP Close at Hand
N-Ethylpyrrolidone didn’t earn its place by accident. Electronics manufacturers lean on it to strip photoresists and clean flux. Polymer producers use it to dissolve tough resins and as a reaction medium for specialty fibers. Specialty coatings and inks benefit from its balanced evaporation rate, which helps avoid surface defects without slowing down production. I’ve known research departments experimenting with it in pharmaceutical syntheses, running reactions that demand stable yet efficient solvent environments. Chemists working on battery technology also keep an eye on polar solvents like NEP, testing performance as lithium-ion technology expands. These application stories match real problems with real solutions, not just features listed in technical brochures.
What’s Next in Research and Development
Every year, conferences fill with updates about solvents for high-tech industries. NEP sits high on the list for modifications aimed at even lower toxicity and better environmental profiles. Regulatory pressure and green chemistry targets drive much of this work; I’ve watched teams test renewable feedstocks and improved recycling processes to ease the environmental footprint. There are ongoing efforts to find replacements with similar solvency for processes where environmental or health regulations have tightened. The flip side is that NEP’s unique characteristics keep it in demand for tough jobs, making incremental improvements more practical than wholesale abandonment in critical roles. Technical journals sometimes feature papers exploring new purification techniques, fresh catalytic systems, and alternative preparation methods that build on decades of accumulated knowledge.
The Truth about Toxicity
For all its utility, NEP comes with strings attached. Lab animal studies and some human case reports warn about reproductive toxicity risks and possible organ effects from chronic exposure. Regulatory agencies in Europe, North America, and Asia have driven stricter workplace guidelines. Anyone who’s worked on a regulatory submission knows the hours required combing through toxicological data, finding evidence both for and against safe use in specific processes. There’s a growing culture of risk avoidance: replacing NEP in consumer products, limiting permissible exposure, and investing in closed-system handling. Yet, plant managers and chemists both understand that outright bans risk unintended consequences, especially in specialized manufacturing. Open dialogue about alternatives, worker education, and advances in monitoring technology all shape how we manage these hazards.
A Glance Ahead: The Future of NEP
N-Ethylpyrrolidone stands on a crossroads of necessity and caution. It’s hard to overstate how much industries value a solvent that works reliably without flying off into vapor or gumming up sensitive equipment. As policy shifts and sustainable chemistry rise, teams keep searching for substitutes or modifications that keep essential properties while lowering health and safety risks. Having watched innovation cycles in specialty chemicals, I’ve seen new solvents come and go, but very few offer a balance as robust as NEP. The challenge runs deeper than swapping one molecule for another—it’s about rethinking processes, designing safer plants, and building regulatory frameworks that support both growth and well-being. The next decade promises lively debates, smarter science, and, hopefully, safer environments for everyone working with these indispensable molecules.
What is N-Ethylpyrrolidone used for?
Everyday Realities in Industry
Take a look at some key supply chains and N-Ethylpyrrolidone pops up more often than most people would guess. This clear liquid acts as a powerful solvent, and industries put that strength to work. If you’ve grabbed a lithium-ion battery or checked a printed circuit board, chances are this chemical was busy doing its job somewhere in the background. In battery manufacturing, N-Ethylpyrrolidone lets companies dissolve and spread active materials evenly onto electrodes—boosting how much charge your battery can store and how long it lasts.
Electronics and Beyond
Printed circuit boards run all sorts of devices, from computers to kitchen appliances. During their production, solutions need to clean surfaces or dissolve stubborn residues. N-Ethylpyrrolidone gets the call for that, outshining many weaker solvents. Workers use it because it grabs hold of oils, resins, and flux leftover after soldering, helping boards meet tighter quality and safety rules.
In my own experience working with a small electronics startup, our team often faced trouble with residue from manual soldering in prototype runs. Once we switched to a cleaning blend using this solvent, parts passed inspection more reliably. Projects moved out of the lab into actual production, keeping investor confidence up. It’s the sort of behind-the-scenes detail a lot of non-engineers don’t notice, but these steps make a difference in keeping devices working safely.
Modern Paints, Coatings, and Printing
Paint isn’t just for looks—coatings must protect surfaces. High-performance paint factories need products to flow smoothly onto metal or plastic. N-Ethylpyrrolidone keeps those formulas from becoming gunky or uneven. The same goes for inks at big packaging plants. Quick-drying print jobs on cardboard or plastic wrap need solutions that mix well, dry fast, and don’t streak, especially on high-speed lines. This solvent helps printers lower costs and cut down on failed runs.
Medicines and Chemical Research
Pharmaceutical labs trust N-Ethylpyrrolidone in a different way: as a reaction solvent. Some active ingredients only take shape under very specific conditions, and getting those chemical reactions to complete often calls for something stronger than water or alcohols. This is one of those niche places where careful handling and deep training matter—nobody wants to risk contamination or environmental side effects. Documented by chemical safety boards, the compound’s role in speeding up ingredient production reduces cost but it comes with close oversight.
Health and Safety Challenges
Despite industry benefits, N-Ethylpyrrolidone raises warnings. Health experts track worker exposure because it can pass through skin and cause headaches, nausea, and, in animal tests, reproductive problems at high enough doses. Regulations in the European Union and some US states have stiffened, pushing manufacturers to lower exposures, use protective gear, or look for alternatives in factories. For small operations, switching to greener chemicals or investing in better ventilation means extra cost.
Finding Safer Paths Forward
As more voices call for personal and environmental safety, engineers and chemists keep searching for substitutes or new handling methods. Newer battery and electronics designs sometimes work well with less toxic solvents or water-based blends. For companies, swapping out older chemicals means reviewing every part of manufacturing—sometimes retooling entire lines. This is costly and time-consuming, but as research underlines the risks, demand for safer choices keeps growing. It’s a step forward built from small but important changes, and its benefits stretch far beyond just stronger batteries or shinier paint.
Is N-Ethylpyrrolidone hazardous to health?
Getting to Know N-Ethylpyrrolidone
N-Ethylpyrrolidone, usually called NEP in the lab, sits among a group of chemicals often used as solvents. I’ve run across it most often at industrial sites and in research spaces, but it turns up in paints, coatings, electronics manufacturing, and sometimes pharmaceuticals. Anyone who’s spent time around industrial chemistry knows the drill when opening a fresh drum: sharp odor, stinging eyes, bitter taste if you’re unlucky. NEP shares much of this character.
What the Science Shows About Health Effects
Most folks who handle NEP wear gloves and keep the area ventilated. Old-timers in the industry remember days before stronger regulations. Back then, headaches, dizziness, and nausea happened more often than people admitted. It’s not just workplace gripes. Research published by the European Chemicals Agency links NEP with developmental toxicity. Studies on rats showed that exposure during pregnancy caused harm to developing offspring. Chemical Safety Reports say NEP can be absorbed through skin and through the air, putting people with regular exposure most at risk. That isn’t really abstract for anyone who’s worked a full shift handling it—cracked skin and a rough throat aren’t rare stories.
The International Agency for Research on Cancer hasn’t yet classified NEP for cancer risk, but regulators still pay close attention. The European Union moved to restrict NEP in many uses, putting it on high concern lists alongside similar solvents like N-Methylpyrrolidone (NMP). The United States Environmental Protection Agency proposed tighter restrictions on NMP but has yet to lump NEP in with the same urgency. Some manufacturers voluntarily moved away from NEP or replaced it with less risky chemicals to avoid worker complaints and legal trouble.
Why the Risk Matters
The thing about NEP isn’t just occasional irritation. The deeper risk comes from regular, repeated contact—folks in manufacturing and research notice small health changes stacking up over time. My own hands still remember rough afternoons cleaning tanks without the right gloves. Sheet metal workers, electronics assemblers, and painters have similar stories. Regulatory agencies urge caution not out of red tape, but from a pile of real-world cases and mounting evidence. Few workers can afford breaks or medical bills if things go south. Missteps in chemical safety pile up fast in places running on tight margins or where safety equipment costs cut into profits.
Practical Solutions from Experience
Some solutions work better than others. Good old-fashioned ventilation does as much as any high-tech fix. Fume hoods, air filters, and open windows all help to drive away vapors that might otherwise linger. Gloves, goggles, and aprons that are actually chemical-resistant—not old latex or thin nitrile—make a difference. I noticed far fewer headaches and skin problems once switching to coverage rated for harsh organics. Posting hazard signs close to where NEP gets handled reminds everyone, even veterans, not to get too comfortable. Spill kits nearby, extra soap at the sink, and routine hazard training all add layers of defense for workers and supervisors alike.
Substitution stands as the safest change any business can make. Plenty of less hazardous alternatives do the same chemical jobs, though sometimes with more patience or extra clean-up. For those of us who spent years around the old solvents, the benefits of switching show up loud and clear in health records and happier shifts.
The science and stories both say NEP isn’t something to treat lightly. Its risks follow the same arc as so many industrial solvents before it—headaches, skin problems, deeper risks for the next generation. Most improvements in safety came from listening to workers, reading the reports, and changing the routine before more folks got hurt. In the end, health and safety mean more than convenience or saving a buck.
What are the storage conditions for N-Ethylpyrrolidone?
Understanding the Basics
N-Ethylpyrrolidone, often called NEP in chemistry labs, rarely ends up on the general public’s radar. Those using it in research or manufacturing know this substance plays a big role in producing electronics, coatings, inks, and pharmaceuticals. Countless workplaces, big and small, keep it in supply closets or bigger storage rooms. Anyone dealing with it learns quickly: safe handling begins with proper storage.
N-Ethylpyrrolidone and Its Quirks
This chemical acts as a solvent and, like many liquid chemicals, it has a way of breaking through plastic if left ignored. Spills and leaks don’t just damage containers; they often lead to headaches for safety officers and extra clean-up tasks for already busy teams. From my work in chemical distribution, clear labeling and weekly checks saved more than a few shipments — and maybe a few careers, too.
Practical Storage Conditions
Anyone keeping N-Ethylpyrrolidone on-site should use tightly-sealed drums or bottles made from high-density polyethylene or stainless steel. Metal reacts with some chemicals but not with NEP, which helps with peace of mind. This solvent will absorb water from air, so keep it away from damp basements or rooms prone to leaks. Too much moisture creeping in not only degrades the product but might even turn workspaces into slip zones.
Shelving or cabinets for NEP don’t belong next to acids, oxidizers, or strong bases. Simple separation can stop dangerous reactions. I remember someone storing a solvent next to an oxidizing agent in the far corner of a tech start-up’s lab. Luckily, that mistake ended in a stern warning, not a siren-filled afternoon.
Cool, dry, and shaded sums up the best home for most solvent drums. Hot warehouses cook chemicals, and sunlight speeds up decomposition. A corner far from sunlight cuts down on unexpected chemical changes and preserves the solvent’s usefulness for longer. Ventilated spaces stop any vapors from gathering and creating a health hazard. Staff working near solvent storage appreciate not breathing in heavy, unpleasant fumes after a long lunch break.
Managing Safety and Regulations
OSHA and local city codes require dedicated spill containment and labeling. That extra effort turns out cheaper than covering cleanup costs or paying regulatory fines. Big errors often come from small oversights. A simple checklist—caps closed tightly, labels facing forward, storage rooms locked—has stopped more incidents than multi-page safety manuals buried in a desk drawer. Review expiration dates, rotate out older containers, and keep the Material Safety Data Sheet within arm’s reach.
Practical Solutions for Common Troubles
Condensation builds up in storage spaces with poor airflow. Dehumidifiers or even a few strategically placed moisture absorbers keep things dry. Teaching staff the right way to handle small leaks or drum transfers makes a bigger difference than another round of paperwork. Throwing on goggles and gloves is a habit, not an afterthought. If the smell sharpens or drums feel unusually hot, it’s time for a full inspection.
In 2022, a global electronics supplier spent thousands fixing problems from poor solvent storage. Their new policies saved resources by preventing accidents instead of reacting to them. Paying attention to storage details keeps chemicals, budgets, and—most importantly—people safe.
What is the chemical formula of N-Ethylpyrrolidone?
Getting to Know N-Ethylpyrrolidone
N-Ethylpyrrolidone, a liquid at room temperature, packs a punch in the world of chemistry and manufacturing. Its chemical formula is C6H11NO. This may look like a random string of letters and numbers at first glance, but it unlocks the secret of a compound with a five-membered ring made of four carbon atoms and one nitrogen, dressed with an ethyl side group. With these elements, it stands out among related chemicals.
Why Chemical Structure Matters
Some people ignore the importance of structure in organic chemistry, but structure shapes both safety and capability. In N-Ethylpyrrolidone, the nitrogen atom in the ring provides the chemical with unique solvent properties. This trait opens the door for manufacturers to use it in a string of industrial applications, from making electronics to specialty polymers and pharmaceuticals. The oxygen atom double-bonded to a carbon creates a pathway for excellent solubility and compatibility with many other chemicals.
Without this specific formula, replacing or replicating its effectiveness in breakneck production lines wouldn’t be easy. Workers in cleanrooms, electronics assembly lines, and drug formulation labs count on reliable solvents for purification, extraction, and cleaning.
Real-Life Applications You Might Overlook
People see a final product—a computer chip, glossy paint, or life-saving medication—but rarely consider what dissolves, cleans, or separates raw materials behind the scenes. N-Ethylpyrrolidone helps create smooth finishes and precise coatings, especially in circuits that drive today’s electronics. It also steps in as an extraction agent when making pharmaceuticals, supporting high-purity outcomes that keep costs lower and quality higher. My time shadowing workers in a paint formulation plant revealed just how much the consistency and low toxicity of N-Ethylpyrrolidone matter. Many solvents either evaporate too quickly, leave unwanted residues, or don’t mix thoroughly. The right chemical, with the right formula, streamlines complicated processes and keeps jobs safe.
Health, Safety, and Accountability
Strong performance doesn’t overshadow responsibility. Chronic exposure to N-Ethylpyrrolidone raises health questions. Agencies such as the European Chemicals Agency have placed attention on worker safety and proper handling. Misuse or careless disposal threatens both personal health and the wider environment. I’ve spoken with occupational health experts who tell stories of skin rashes and headaches from workers exposed without protection. The lesson here speaks for itself: no shortcut justifies ignoring gloves and goggles or bypassing ventilation systems in labs and factories.
Companies also face pressure to design greener alternatives. The growing demand for safer, sustainable practices encourages researchers to seek less hazardous substitutes, especially in consumer-facing applications. Advisory panels urge a shift towards bio-based or less persistent chemicals, yet few alternatives check every box regarding performance and availability.
Looking Forward
People sometimes forget how many steps happen before a product lands in our hands. Careful stewardship, combined with honest science, keeps promising materials like N-Ethylpyrrolidone both useful and safe. Looking past fear and hype, the real progress comes from investing in worker protection, sharing research, and sticking to standards that prioritize health as much as output. By understanding exactly what makes up N-Ethylpyrrolidone—right down to its C6H11NO backbone—we set a smarter path forward for industry and community alike.
Can N-Ethylpyrrolidone be used as a solvent?
Understanding Real-World Solvent Choices
In the world of chemistry and industrial manufacturing, N-Ethylpyrrolidone, or NEP, gets a lot of attention as a possible replacement for other well-known solvents. From paint removers to pharmaceuticals, industries often look for something that can dissolve tough substances without stirring up environmental or health issues.
Strength in Solubility and Stability
People in labs and factories prize NEP for its strong solubilizing abilities. Organic substances, resins, and polymers break down well in NEP, making it a solid pick for cleaning solutions and chemical syntheses. If you have ever watched a stubborn patch of ink or adhesive transform into a puddle—much faster than with water or weaker cleaners—that efficiency comes from a solvent with muscle like NEP.
The story doesn’t end with getting the job done. NEP brings real value by staying stable, rarely evaporating too quickly, and resisting decomposition under heat. That gives process engineers more freedom to use it in heated reactions or processes that can’t tolerate a changing solvent mix.
Questions About Safety
Chemicals that break down tough substances tend to have an edge and a drawback. NEP can irritate the skin and eyes; inhaling its vapors may hit the lungs and throat. Some regulations, including those from European authorities, put NEP under a microscope. It’s on their list of substances that raise flags for possible reproductive effects. OSHA in the United States looks to guidelines and exposure limits for worker safety, though it hasn’t clamped down as hard as Europe.
I’ve worked around solvents in university labs and smaller manufacturing settings. Nobody wants to handle a “miracle” cleaner if it means risking their health. Gloves, goggles, proper ventilation—these are not negotiables. Still, companies keep searching for alternatives that strike a better balance between power and safety.
Environmental Impacts and Responsibility
NEP doesn’t linger in the environment forever, but that’s not the same as being harmless. Disposing of spent solvent down the drain doesn’t just break rules; it loads up water systems with synthetic chemicals that resist natural breakdown. NEP’s track record looks better than some chlorinated cousins, yet it still warrants care. Treatment facilities sometimes struggle with advanced synthetic solvents, and wildlife can feel the effects long before people notice.
Is There Still a Place for NEP?
Companies often weigh the costs, performance, and possible harm of each solvent in their arsenal. Green chemistry movements put pressure on businesses to cut down on chemicals with murky safety records. Some research points toward using safer alternatives like dimethyl sulfoxide or even water-based blends, though these swaps sometimes mean compromises in performance.
Safety data sheets matter, but real, shared experience tips the scales. Engineers, chemists, and plant managers remember what it’s like when a fast-drying solvent speeds up cleaning only to cause cracked hands or trigger headaches for a week. Factoring in worker satisfaction, long-term costs, and new regulations, it makes sense to keep evaluating NEP against the growing list of options. Shifting to less hazardous substitutes can keep both employees and ecosystems in better shape.
Keeping the Conversation Going
People like me and others in chemistry rely on clear facts, honest feedback, and strong evidence when choosing materials for daily tasks. NEP shows up in plenty of proposals because it solves significant problems, but its drawbacks drive the search for something better. By listening to workers, tracking actual exposure, and staying alert to health and environmental research, companies can avoid repeating old mistakes with new chemicals.
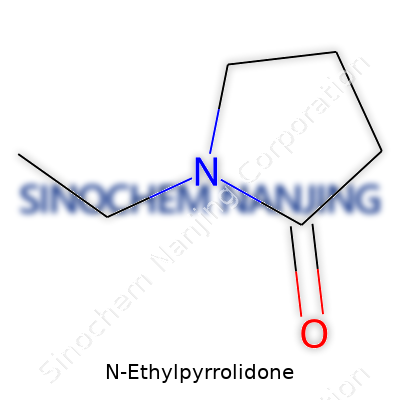
| Names | |
| Preferred IUPAC name | 1-ethylpyrrolidin-2-one |
| Other names |
NEP 1-Ethyl-2-pyrrolidone N-Ethyl-2-pyrrolidone N-Ethylpyrrolidin-2-one NEP solvent |
| Pronunciation | /ɛnˌɛθɪlˌpɪˈrɒlɪˌdoʊn/ |
| Identifiers | |
| CAS Number | 2687-91-4 |
| Beilstein Reference | **90868** |
| ChEBI | CHEBI:81973 |
| ChEMBL | CHEMBL572564 |
| ChemSpider | 54661 |
| DrugBank | DB08798 |
| ECHA InfoCard | 17e69b9f-d9c4-4d2d-9959-fcb86dc48e99 |
| EC Number | 220-250-6 |
| Gmelin Reference | 9198 |
| KEGG | C19948 |
| MeSH | D000071246 |
| PubChem CID | 81815 |
| RTECS number | QJ8975000 |
| UNII | 6ZB93L13B1 |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID3020366 |
| Properties | |
| Chemical formula | C6H11NO |
| Molar mass | 99.14 g/mol |
| Appearance | Colorless to pale yellow transparent liquid |
| Odor | Faint amine odor |
| Density | 1.028 g/cm³ |
| Solubility in water | miscible |
| log P | -0.35 |
| Vapor pressure | 0.029 hPa (20 °C) |
| Acidity (pKa) | 19.7 |
| Basicity (pKb) | 7.9 |
| Magnetic susceptibility (χ) | -7.25×10⁻⁷ |
| Refractive index (nD) | 1.472 |
| Viscosity | 1.75 mPa·s (25 °C) |
| Dipole moment | 4.09 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -426.15 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3369.7 kJ/mol |
| Pharmacology | |
| ATC code | N01AX15 |
| Hazards | |
| Main hazards | Causes skin and serious eye irritation. May damage fertility or the unborn child. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H318, H319, H335 |
| Precautionary statements | H302+H312+H332, H319, H335, P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 86°C |
| Autoignition temperature | 245 °C (473 °F; 518 K) |
| Explosive limits | Explosive limits: 1.3–9.5% |
| Lethal dose or concentration | LD50 Oral Rat 3,914 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 4,650 mg/kg |
| NIOSH | KBZ79500 |
| PEL (Permissible) | 'PEL (Permissible Exposure Limit) for N-Ethylpyrrolidone: Not established by OSHA' |
| REL (Recommended) | REL (Recommended) for N-Ethylpyrrolidone: "10 ppm (40 mg/m³) TWA |
| IDLH (Immediate danger) | IDLH: 900 mg/m³ |
| Related compounds | |
| Related compounds |
N-Methylpyrrolidone 2-Pyrrolidone N-Ethyl-2-pyrrolidone N-Propylpyrrolidone N-Butylpyrrolidone |

