N-Amyl Acetate: Beyond the Laboratory—A Commentary
Peering Into the Past: The Roots of N-Amyl Acetate
N-Amyl acetate shows up in many corners of human history and industry, but it rarely gets credit for how much it has shaped things behind the scenes. Over a century ago, chemists discovered its unique scent—something close to ripe bananas or pears. This compound started as just another curiosity in organic chemistry, yet it quickly found its way into early perfumery and flavoring, as well as industrial processes. Workers in the paint and varnish trade might recall its sharp, fruity aroma drifting through old workshops. The use of N-amyl acetate in fabric cleaning and as a component in penetrating sealers gave it a practical edge, showing that simple molecules often push progress quietly.
Product at a Glance: From Scent to Solution
In practical terms, N-amyl acetate is a colorless liquid with a noticeable, sweet fragrance that lingers even after brief exposure. Anyone who has uncapped a banana-flavored candy or certain cleaning fluids has probably met this chemical without realizing. It isn’t only about the scent—for paint thinners and lacquers, its solvency outmatches simple esters. The product stands out in industries that demand both efficacy and a manageable health profile compared to older, harsher solvents. Its status as a fingerprint in both natural and artificial flavors puts it in the kitchens and laboratories alike.
The Science Underneath: Properties and Specifications
Chemically, N-amyl acetate falls into the ester family. With a formula of C7H14O2 and a boiling point comfortably above water but below some heavier solvents, its volatility gives it an edge in manufacturing and laboratory work. Its flash point signals a need for thoughtful handling, but in many respects, it behaves consistently under a wide range of conditions, dissolving varnishes, resins, and a variety of organic compounds. The density sits just a hair lower than water, letting it float on top. Its vapor is heavier than air, so it tends to stick around unless good ventilation is in place. Understanding these basics grounds decision-making for those who encounter it on the factory floor or in the field.
Making N-Amyl Acetate: A Straightforward Synthesis
Making N-amyl acetate doesn’t require exotic tools, though consistent quality depends on controlling reaction conditions. Most producers combine n-amyl alcohol with acetic acid, often using acid catalysts such as sulfuric acid to speed things along. This simple esterification reaction yields the ester and water, usually followed by distillation to purify the product. The method works because both precursors are widely available, and years of industrial scaling have made the process efficient and cost-effective. Smaller labs often improvise with these same ingredients, reminding us that some chemical routines haven’t changed much since the late nineteenth century.
Synonyms and Aliases: A Multiplicity of Names
Browsing through catalogs or literature, N-amyl acetate pops up under several names. Experts might refer to it as pentyl acetate or even banana oil, reflecting its sensory character more than its molecular makeup. Sometimes, the chemical slips in under its systematic label—1-pentanol acetate—or less formal titles like pear oil, depending on who’s talking and where. In Europe or Asia, translations tweak the spelling but not the substance. This multiplicity occasionally trips up researchers or procurement teams, particularly when older nomenclature persists or crosses language barriers. Recognizing the aliases avoids confusion and helps make sense of patchwork legacy documentation.
Reactivity and Modifications: Beyond the Simple Ester
N-amyl acetate enters into most reactions expected of typical esters. It holds up under gentle conditions but can break apart in strong acid or base, giving back n-amyl alcohol and acetic acid. Practical modifications usually focus on swapping out the alcohol group for other chains, producing a family of related esters with different scents or solubility profiles. Occasionally, researchers try to tweak the molecule further, building in new reactivity for specialized jobs in organic synthesis. As a building block, it rarely grabs headlines, but it remains dependable for both academic benches and process chemists looking to tune solvent properties.
Safety and Handling: Balancing Risks and Benefits
Years spent handling chemicals have hammered home the habits that keep people out of trouble, and N-amyl acetate serves as a good example. It isn’t especially toxic by acute exposure, but inhaling concentrated vapors or careless skin contact can irritate. Like many solvents, it flammable, so risky practices—open flames, smoking nearby, or crumpled rags—can lead to avoidable accidents. Many regulations limit airborne concentrations in industrial spaces, and wise operators don gloves, eye protection, and keep good airflow nearby. Labels and safety data sheets tell part of the story, but real safety relies on training and muscle memory—small acts, repeated every day. My own experience reinforces that a brief lapse can lead to a story no one wants to tell twice.
Application: Where Science Meets the Everyday
N-amyl acetate lives a double life as both an industrial staple and an unsung hero in consumer products. Paint manufacturers appreciate how it dissolves pigments and helps coatings dry with fewer brush marks than traditional blends. Printing ink formulators find its drying rate matches the demands of high-speed presses. In flavors and fragrances, the compound delivers a recognizable signature that lets confections, baked goods, and even chewing gum stand apart. Film production, both photographic and motion picture, once leaned on it to process cellulose acetate. It even lends a hand in cleaning solutions designed to lift stubborn greases without tearing up sensitive materials. Each of these uses speaks to the compound’s balance between power and predictability—rarely flashy, always effective.
Looking Forward: Research and Development
Scientists constantly dig deeper into the chemistry and applications of N-amyl acetate, exploring sustainable production from bio-based sources or tweaking the molecule for niche performance. Renewable routes using fermentation byproducts point to a cleaner supply chain, and pilot projects show promise, though scaling up faces hurdles—feedstock consistency, catalyst reuse, waste minimization. In specialty applications, research teams experiment with hybrid solvent blends to improve results in coatings or flavor encapsulation. Insights from molecular modeling and advanced spectroscopy shed light on how N-amyl acetate interacts with other components, opening doors to designer materials or safer processing protocols. Real progress depends on persistent effort, creative troubleshooting, and sharing results across laboratory walls.
Toxicity Under the Microscope
Most data on N-amyl acetate points toward a reassuring profile when handled with respect. Acute toxicity sits comparatively low, and chronic harm rarely comes up except at high exposures. Studies in rodents and occupational health logs underline the main issues: irritation of airways, mild drowsiness at elevated levels, and skin dryness if contact goes unchecked. Long-term environmental impact appears modest, assuming spills and disposal observe established standards. The biggest risks often come from confined-space work or mixing with other, more hazardous solvents. Experience shows that vigilance and regular air monitoring do more to keep workers safe than waiting on regulatory updates. The real danger hides in routine—complacency is the enemy, not the ester.
Future Prospects: Threading Old and New Together
The market for N-amyl acetate may not inspire bold headlines, but it continues to evolve in response to greener chemistry, worker safety, and consumer preferences. A push for bio-renewable variants could reduce fossil fuel dependence, though securing adequate raw materials without competing with food sources remains a pressing concern. In coatings and inks, shifts toward low-VOC formulations challenge chemists to redraw the solvent palette, balancing evaporation rate, solvency, and user safety. Food and fragrance sectors keep a sharp eye on purity standards and allergenicity, driving investments in new analytical methods and tighter process controls. Progress comes through small, diligent steps—auditing old habits, hunting for incremental improvements, and sharing hard-earned lessons so that N-amyl acetate continues to serve quietly as a backbone for industries that touch daily life.
What is N-Amyl Acetate used for?
N-Amyl Acetate in Everyday Life
N-Amyl acetate might sound like a mouthful, but plenty of folks have crossed paths with it—often without realizing. The sweet, banana-like smell that wakes up the nose in art classrooms or repair shops points back to n-amyl acetate. This compound comes up right at home in paint thinners, nail polish removers, and flavors added to food. Its workhorse role links to its ability to help substances dissolve or blend evenly, especially useful for paints and coatings that cover surfaces without streaks or clumps.
Factories, Flavors, and Fragrances
Inside a factory, n-amyl acetate pops up on the ingredient list for everything from printing inks to cleaning agents. My time working in an industrial lab showed just how often industries lean on a solvent that does its job quietly and evaporates with little fuss. Adding it to coatings keeps the mix smooth and speeds up drying. The fast evaporation means workers and consumers aren't left waiting around, which increases work speed and productivity.
It doesn’t stop there. Food manufacturers put n-amyl acetate to work baking up banana-flavored candies or even jazzing up some snacks. It’s approved for use as a flavor additive, letting people bite into artificially flavored treats with that familiar fruit punch. The perfume industry—where finding just the right scent can turn a product into a bestseller—also leans on this ester to conjure up fruity notes or add depth to fragrances. The combination of smell and effective blending properties makes it a staple among perfumers and food scientists alike.
Painters and Printers Count on It
During summers painting houses, I saw what happened when a painter skipped the right thinner or solvent. Paint would dry too fast or clump on the brush, ruining what could have been a good finish. N-amyl acetate has this talent for thinning paints and clearing out sticky messes from tools or hands. Printers get similar mileage out of it. Fast-drying inks, which are needed for crisp, clear graphics on magazines and packaging, rely on the solvent to keep presses running without jams. In this way, the chemical isn’t just hiding in the background—it keeps jobs flowing smoothly and customers happy.
Looking at the Health and Environmental Side
N-amyl acetate, like other solvents, should never be taken lightly while being handled. Extended breathing of vapor can bring on headaches or dizziness—something I’ve seen after too many hours in a poorly ventilated art studio. Gloves, good airflow, and, where needed, masks take care of most risks. Compared to tougher solvents like benzene, n-amyl acetate sits further down on lists of health hazards, but no one benefits from careless use.
As industries seek safer and greener chemicals, n-amyl acetate’s less toxic footprint makes it a preferred pick over more volatile alternatives. Still, any chemical needs to be respected from production to disposal. Recycling solvents or switching to water-based options where possible, offers a way forward for businesses and hobbyists alike.
Finding Smarter Solutions
N-amyl acetate shows up in more places than people imagine, making everyday products reliable, smooth, and safe to use. Seeking alternatives for harsh solvents can boost both human health and environmental safety, and n-amyl acetate fits into that shift. I’ve watched as even the smallest shops and industrial sites pivot to safer chemical routines, investing in training and better equipment. It proves people don’t have to pick between effectiveness and well-being. There’s always room for solutions driven by experience, careful selection, and honest communication about chemical risks and benefits.
Is N-Amyl Acetate safe to handle?
Hands-On Experience: What N-Amyl Acetate Really Feels Like
N-Amyl acetate pops up in everyday locations, whether you’re in a paint booth, tinkering in a lab, or picking up a banana-scented cleaner at the hardware store. It’s got that sweet, pear-like odor—pretty easy to spot. Handling this solvent feels routine for folks who work with adhesives, flavors, or lacquers, but safety shouldn’t slip through the cracks just because something smells pleasant. In my own years in the chemical trades, I’ve seen how a familiar scent can lull folks into skipping simple precautions.
So, What’s the Risk?
Take a splash of N-amyl acetate on the skin—might cause some irritation but usually not dramatic. Breathe in the vapors in a small, stuffy workspace and you get headaches, dizziness, maybe some coughing. Once, painting furniture in a poorly ventilated basement, I felt my brain fog over before realizing the ventilation fan had died. A gentle smell can trick you into forgetting that your lungs are doing heavy lifting.
The science backs up these everyday experiences. According to published material safety data, OSHA recommends a limit because, in higher doses, the fumes depress your central nervous system. The National Institute for Occupational Safety and Health (NIOSH) gives clear-cut exposure limits—no more than 100 ppm as a time-weighted average during a work shift. Folks in the flavor and fragrance industries take these numbers seriously because repeated exposure—even below the limit—dries skin and can cause chronic irritation. Eyes sting, throats swell, and at higher doses, faintness isn’t rare.
Why Knowing Storage and Handling Matters
Storing N-amyl acetate isn’t exotic—cool, dry places and solid labeling go a long way. In my circle, no one forgets the fire hazards. It only takes one misplaced spark or forgotten cigarette for a routine day to spiral. This solvent catches fire easily. Fire departments and insurance teams zero in on solvent storage because fires in a warehouse or garage get ugly fast. Responsible users keep containers tightly sealed and make sure flammable liquids are nowhere near open electrical panels.
Steps That Work in Real Life
Safety showers, eye wash stations, and gloves aren’t bureaucratic extras—they’re the quickest fixes for small spills or accidental contact. I never saw anyone regret putting on nitrile gloves around open cans. Goggles block stray splashes if a jug tips off the bench. People cut accidents in half just by flipping on a ventilation fan before uncapping a bottle.
Working outside or in a room with real airflow beats plugging your nose. Respirators come into play whenever fumes hang thick—no shame in grabbing one. The best chemistry teachers drilled one message: “Smelling it means you’re already exposed.” That has a way of sticking in your head, long after the lesson ends.
Looking Forward: Smarter Standards and Shared Responsibility
More small-scale users expect their suppliers to provide safety information, not just a product label. Labels and safety data sheets should be clear and readable, avoiding jargon. Fact-based rules, like OSHA’s exposure guidelines, land with more impact than slogans on a poster. In workplaces, peer reminders keep habits strong—if one person stops caring about gloves or ventilation, odds are more will follow.
The way to make N-amyl acetate safer isn’t about reinventing the wheel. It’s about steady routines: gloves, airflow, labeling, no open flames, and not letting your nose set the safety rules. That’s how users avoid the worst risks, day after day.
What is the chemical formula of N-Amyl Acetate?
Understanding N-Amyl Acetate
N-Amyl acetate, known chemically as C7H14O2, shows up in plenty of places you wouldn’t expect. From the distinctive scent in some fruit flavors to solvents used in industrial settings, this compound leaves its mark across different industries. The name might sound intimidating, but break it down and you see something simple: amyl means five carbon atoms, and acetate hints at a pairing with acetic acid. That’s how you get the structure—one five-carbon chain bonded to an acetate group.
Why Does N-Amyl Acetate Matter?
Growing up, nothing felt quite like the summer fruit stands where pears, apples, and bananas seemed to beg for tasting. It turns out, behind the aroma sits N-Amyl acetate, responsible for that sweet, fruity smell. That specific scent carried over to my days working in a print shop. Open a can of traditional ink thinner, and it’s there again—smooth, sweet, slightly sharp on the nose.
This chemical carries more clout than just scent. Paints, coatings, and adhesives rely on N-Amyl acetate as a solvent that cuts through other compounds. Its moderate evaporation rate means workers in auto shops and print houses can depend on a steady performance—not drying too quickly, not lingering forever. Researchers keep a close eye on how such chemicals interact with the human body. Even small exposures bring up safety conversations; no one likes headache or dizziness after a long shift.
Safety, Health, and Environmental Concerns
Like a lot of volatile organic compounds, N-Amyl acetate raises questions about workplace safety. The National Institute for Occupational Safety and Health (NIOSH) tracks short- and long-term effects, reminding us about the need for solid ventilation. Being careful with these solvents doesn’t just protect lungs—it lowers the risk of fire, too.
Environmental fate of N-Amyl acetate also gets attention. It breaks down pretty fast when released outdoors, reacting with things like sunlight and water. Still, spills in factories or labs reach soil and water if companies don’t have the right containment. Regulations push for better storage solutions and clear labels to minimize risks. Awareness helps workers recognize leaks or spills before they grow into nightmares.
Safer Use and Better Practices
Over the years, I saw companies switch up their training, focusing less on simply following rules and more on understanding chemicals like N-Amyl acetate. Having hands-on hazard training meant fewer accidents and fewer headaches. Where possible, substituting with less volatile solvents cut down exposure.
As tech pushes forward, greener solvents get more research money. Some startups experiment with plant-based formulas that mimic the effectiveness of N-Amyl acetate but break down faster and pose less risk. Until those options reach the mainstream, good habits—like wearing gloves, eye protection, and making sure fans stay on—stand out as real safeguards.
The formula C7H14O2 sits on more safety sheets today than ever. Behind that formula are layers of history, science, and practical choices shaping how we handle chemicals in day-to-day work. Paying attention and responding with common sense, we keep both industries and homes safer, all while remembering the strange bridge between fruit flavors and industrial floors.
How should N-Amyl Acetate be stored?
Looking at the Basics
N-Amyl acetate, also known as pentyl acetate, usually pops up in paint thinners, coatings, and sometimes even in flavorings. Often, people treat chemicals like this as if they all fit under one big safety label, but experience says each substance deserves its own respect. I’ve seen plenty of shop shelves stuffed with cans of solvents stacked together, and it almost always leads to trouble. Keeping amyl acetate safe starts with picking the right spot, the right container, and a level of awareness about its behavior.
Why Safe Storage Even Matters
Some weeks back, a local repair shop caught a minor fire because someone kept rags soaked in amyl acetate in a warm shed. It’s a reminder that even a small amount exposed to heat or spark turns into a serious hazard. Amyl acetate evaporates quickly, filling the air with a sweet, fruity smell that some people don’t even notice after a while. Yet, beneath the surface, it builds up vapor that can ignite if an open flame or static electricity wanders too close. Data from the National Fire Protection Association suggests solvents like this start dozens of fires yearly in small businesses.
Choosing the Right Container
Metal and glass containers with airtight seals beat plastic. Over time, amyl acetate seeps through most plastics or reacts with them, causing leaks or sudden pressure buildup. Years working in busy art studios taught me that reusing soda bottles or old detergent containers lands you in the emergency room sooner or later. Containers need labels with clear ingredient names and hazard symbols, and lids that twist on tight. Never store it near acids, oxidizers, or strong bases; accidents multiply when incompatible chemicals mix or fumes mingle together.
Keeping Storage Areas Safe
Shelves should stand away from direct sunlight, far from heating ducts or radiators. A cool, well-ventilated area lowers vapor concentration and risk. If your storeroom feels hot or stuffy, ventilation fans or fume hoods make a huge difference. I've worked in places where a single cracked window kept the air moving and people safe. Any spill, no matter how small, calls for fast cleanup with absorbent pads and disposal in a designated waste bin. Never stack heavy items on top of solvent containers; pressure and pinched lids lead to leaks in ways you only notice once the smell takes over the room.
Clear Signage and Emergency Planning
Nothing works better in a pinch than clear signs pointing out flammables and emergency exits. Chemical safety isn’t just for big factories; even home users need to know where the closest fire extinguisher sits and which type to use. A dry chemical extinguisher, not water, knocks down a solvent fire. Regular drills, even just once or twice a year, turn confusion during an emergency into fast action. OSHA statistics prove that basic planning cuts both injuries and property loss. In my own experience, the only real mistake is thinking “it can’t happen to me.”
Moving Toward Better Practices
Industries around the world already cut risks with digital inventory systems, routine checks for leaking containers, and community education on chemical hazards. Schools and local workshops benefit from basic training—teach young people to treat all chemicals with caution instead of “winging it.” All these things make a difference: steady habits and real attention to detail keep both people and property intact.
What are the physical properties of N-Amyl Acetate?
Understanding What N-Amyl Acetate Is
Walk into a hardware store or even a perfume shop, and you could cross paths with N-Amyl Acetate without realizing it. This colorless liquid, often called pentyl acetate, feels like one of those behind-the-scenes chemicals making things tick in the background. My first run-in with it came back in college, during a chemistry course, where its strong, banana-like odor filled the lab—hard to forget that smell.
Boiling and Melting Points Matter
N-Amyl Acetate boils at about 149°C and melts at -71°C. That tells quite a bit about how it behaves in different settings. This low melting point means it stays a liquid, even under chilly conditions. Its relatively high boiling point gives users a window to work with it before it goes up in vapor. Industrial folks use this property to help in processes like extraction or cleaning, knowing that the compound doesn’t flash off too easily. I’ve found that makes it safer for handling compared to many volatile solvents.
Density and Solubility—Two Core Factors
Density settles in at around 0.87 g/cm³ at room temperature. This means it’s lighter than water, floating if poured on top. That comes in handy in separation and cleanup jobs. Think back to any accidental spills in the lab—watching layers form makes quick work of figuring out what to scoop or siphon first.
N-Amyl Acetate refuses to mix with water, but it dissolves smoothly in organic solvents like alcohol, ether, and chloroform. Folks in industries turn to it for blending coatings, inks, and fragrances for exactly this reason. Easy mixing with other organics opens doors. But keep that bottle away from water-based setups unless you’re ready for separation headaches.
Vapor Pressure and Flammability
In the summer heat, or a steamy factory, N-Amyl Acetate’s vapor pressure creeps up—a reading of about 4 mm Hg at room temp. It evaporates steadily, which brings a new kind of risk: fire. Its flash point sits just above 39°C, so letting fumes build up invites disaster. I’ve always made sure good ventilation and careful storage top the checklist after hearing stories of small spills turning into bigger headaches.
Viscosity and Sensory Impact
It pours like water, low viscosity making it easy to handle in pipes or pour into mixing drums. Many flavor and fragrance experts rely on the distinct scent, which gives a banana aroma. Sometimes just a whiff—distinct and sweet—makes its presence obvious, proving that not every chemical needs a technical test to detect.
Keeping Safety Front and Center
No matter the job, safety remains key. Prolonged skin or inhalation contact with the vapor can irritate, and the smell may be pleasant in small doses but overwhelming in more confined spaces. Personal protective equipment like gloves and goggles matters, along with continuously educating workers. I’ve learned prevention takes less time than dealing with the aftermath of careless handling.
Responsible Use in Real-World Settings
With so many industries needing reliable, effective solvents, N-Amyl Acetate stands out for specific jobs, but only with proper respect for its physical traits. Strict labeling, ventilation, and handling protocols save money and lives. Adjusting processes to use closed systems and proper waste disposal keeps its strengths working for, not against, a business or lab. Care and know-how protect people and property, and make every drop count for the best results.
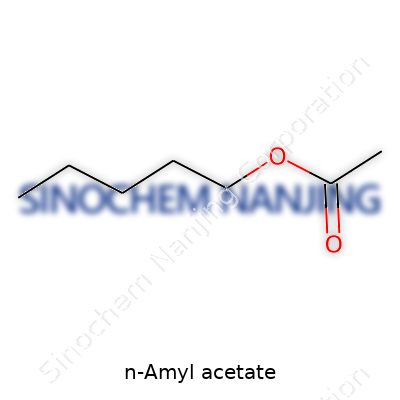
| Names | |
| Preferred IUPAC name | Pentyl ethanoate |
| Other names |
Acetic acid pentyl ester Pentyl acetate Pentyl ethanoate Banana oil Amyl acetate |
| Pronunciation | /ˌɛnˈæm.ɪl əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 628-63-7 |
| Beilstein Reference | 1100389 |
| ChEBI | CHEBI:31221 |
| ChEMBL | CHEMBL45895 |
| ChemSpider | 16213 |
| DrugBank | DB01957 |
| ECHA InfoCard | 100.112.969 |
| EC Number | 3.1.1.74 |
| Gmelin Reference | 6357 |
| KEGG | C01789 |
| MeSH | D000687 |
| PubChem CID | 8058 |
| RTECS number | AJ3675000 |
| UNII | 7QY8UE6T0E |
| UN number | UN1104 |
| CompTox Dashboard (EPA) | DTXSID1025634 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.19 g/mol |
| Appearance | Colorless liquid with a pleasant, banana-like odor |
| Odor | Banana-like |
| Density | 0.876 g/cm³ |
| Solubility in water | 1.0 g/L (20 °C) |
| log P | 1.82 |
| Vapor pressure | 4 mmHg (20°C) |
| Acidity (pKa) | 4.76 |
| Magnetic susceptibility (χ) | -6.44×10⁻⁷ |
| Refractive index (nD) | 1.406 |
| Viscosity | 0.876 mPa·s (at 25°C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -472.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3591.7 kJ/mol |
| Pharmacology | |
| ATC code | D07AX55 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H336 |
| Precautionary statements | P210, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 25 °C |
| Autoignition temperature | 300 °C (572 °F) |
| Explosive limits | 1.0% - 7.5% |
| Lethal dose or concentration | LD50 oral rat 6,500 mg/kg |
| LD50 (median dose) | 6,500 mg/kg (rat, oral) |
| NIOSH | SC2450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of N-Amyl Acetate: "100 ppm (525 mg/m³) TWA |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Ethyl acetate N-Propyl acetate Isoamyl acetate Butyl acetate |

