N-(4-Hydroxyphenyl)Maleimide: From Raw Material to Real-World Application
Historical Development
N-(4-Hydroxyphenyl)maleimide didn’t spring out of nowhere. Back in the post-war mid-20th century, the broader field of maleimide chemistry started offering more than a few interesting avenues for polymer research. The push for synthetic resins, adhesives, and advanced thermoset polymers landed N-(4-Hydroxyphenyl)maleimide on many laboratory benches. Early research in the 1960s flagged its reactivity and stability, helping chemical manufacturers see the promise of harnessing this compound, both as a monomer and a recurring building block for specialty polymers. Industrial chemists wanted modifiers that brought real performance gains, especially under heat and stress, so the demand gave this compound a strong footing as synthesis strategies matured and supply chains stabilized.
Product Overview
This compound sits in a unique spot between phenolic and maleimide chemistry, offering more than just a list of features. Chemists appreciate its rigid aromatic ring paired with the reactive maleimide moiety, which helps unlock enhanced reactivity when it comes to copolymerization. On a practical level, the product usually appears as a white to off-white crystalline powder, and manufacturers typically provide it in sealed, light-protective containers. It doesn’t take much digging to see why specialty resin producers and advanced adhesive formulators order this molecule in bulk: the combination of phenolic reactivity and imide endurance sees use not only in electronics but also in corrosion-resistant coatings and heat-curing systems.
Physical & Chemical Properties
Chemical formula, C10H7NO3, says a little about the story, but users typically care about the melting point (usually close to 200°C), its slight water solubility, and the characteristic faint phenolic odor. Density clocks in near 1.4 g/cm³, making it manageable for most blending operations. N-(4-Hydroxyphenyl)maleimide carries both an electron-donating hydroxy group and the maleimide, which flag it for specialty crosslinking. Shelf-life generally holds up, assuming cool, dry storage. On a bench, you’ll spot it dissolving fairly easily in DMF, DMSO, or acetone, and it holds up well against common oxidative breakdown.
Technical Specifications & Labeling
Market-available specifications run tight. Purity often lands above 98% by HPLC, with clear moisture limits below 1%, supporting those aiming for polymer-grade performance. Labels cover the molecular structure, batch number, manufacture date, and recommended storage advice, along with standardized precautionary symbols under GHS regulations. Occupational safety data sheets highlight known irritancy risks, PPE recommendations, and first aid guidance. Reliable suppliers track trace impurities, so users know how much to expect of common contaminants like unreacted starting materials, metal residues, or isomeric byproducts.
Preparation Method
Most synthetic routes boil down to a condensation step between maleic anhydride and 4-aminophenol under controlled conditions, typically using acetic acid or another polar solvent. The reaction needs temperature management to prevent side product formation, and purification calls for repeated recrystallization steps to knock out residual byproducts. In big batches, reaction vessels run under nitrogen or argon blanket, and the workup protocol gears toward collecting product in a moisture-minimized environment. Titration helps track completion, and vacuum filtration preps the powder for drying before final packaging.
Chemical Reactions & Modifications
With the dual presence of an imide group and a free para-hydroxy position, N-(4-Hydroxyphenyl)maleimide reacts in diverse ways. The phenolic group acts as a nucleophile in substitution reactions, letting chemists tack on alkyl or acyl groups for custom-tailored derivatives. Maleimide end shines in Diels-Alder reactions, allowing rapid addition to electron-rich dienes and forming dense, heat-resistant crosslinks. In aqueous solution, under basic conditions, the hydroxy group can also form ethers or esters, and the molecule has been co-polymerized with styrenics and acrylates for performance improvements. Each tweak brings new thermal and mechanical properties, which have seen uptake in electronics encapsulation and aerospace-grade adhesives.
Synonyms & Product Names
Industry literature and supplier catalogs list this molecule under a few different handles: 4-Hydroxyphenylmaleimide, para-Hydroxyphenylmaleimide, and often the short-form HPM. Some proprietary blends or modified versions go by trade names, most of which highlight heat resistance or crosslinking properties in their branding. Database entries may flag its registry number or include international naming conventions if distributed globally.
Safety & Operational Standards
Direct contact, inhalation, or ingestion creates an irritant risk, so workplace controls matter. Regulatory guides match those of many maleimide derivatives: local exhaust ventilation, PPE (including nitrile gloves and safety goggles), and frequent hand washing after handling. Spills, especially powder, drive clean-up with non-sparking tools, and disposal runs as hazardous waste under many local codes. Emergency protocols cover both short-term exposure (eye flush, skin wash) and larger-scale incidents (airborne dust measures, containment steps). Training for storeroom and lab staff focuses on minimizing skin, eye, and airway exposure, and facilities carry SOPs for accidental release.
Application Area
Electronics (circuit board coatings, solder mask additives), corrosion-resistant metal coatings, aerospace adhesives, and advanced composites give the most visible use cases. For folks in R&D, the high glass transition temperatures and thermal stability let engineers push past older epoxy formulations. Medical device research also nods to this compound—its reactivity can be harnessed for hydrogels, drug delivery matrices, or non-leaching surface treatments. University groups dip into it for crosslinker design or functional monomer development, and it’s started turning up in sustainability research for greener composite materials.
Research & Development
Pushes in this space head toward higher-performance polymers, faster-curing adhesives, and smarter drug carriers. Recent studies publish new derivatives that swap in higher-functionality side groups, chasing even greater heat or chemical resistance. In 2022, several papers profiled hybrid copolymers where N-(4-Hydroxyphenyl)maleimide improved both bond strength and aging resistance under accelerated weathering. Pharmaceutical research exploits its conjugation chemistry to create targeted release systems. In my own experience working alongside polymer engineers, the most talked-about shift involves using this compound for additive manufacturing resins, which need to cure quickly and withstand real-world wear-and-tear.
Toxicity Research
Preclinical animal studies point to moderate irritancy—skin and mucous membranes bear the brunt of exposure. Long-term rodent studies haven’t flagged substantial carcinogenic risk, unlike some other aromatic compounds. The imide group does spark concerns for chronic exposure and repeated use, but acute dose limits sit comfortably above the exposure levels seen in industrial operations. Biodegradability studies flag slow breakdown in aquatic environments, which means responsible handling and waste management crop up as regulatory priorities. Worker health and monitoring play a big role, especially when repeated exposure can affect sensitive individuals. I’ve seen plant operators enforce strict glove and fume hood rules—solid habits that nip preventable exposures in the bud.
Future Prospects
Materials science continues to drive demand for compounds that blend durability with precision reactivity, and N-(4-Hydroxyphenyl)maleimide stands in that sweet spot. Growth in flexible electronics, bio-compatible adhesives, and resilient composites hints at expanding markets. As regulatory pressures favor greener chemistry, the next wave of innovation circles around process safety, bio-based feedstocks, and closed-loop material cycles. If academic and industrial researchers keep reporting performance breakthroughs, especially in hybrid and nanocomposite materials, chances are this compound’s utility will only rise. Whether in high-density electronics or the latest eco-friendly coating, the role of molecules like N-(4-Hydroxyphenyl)maleimide won’t fade any time soon.
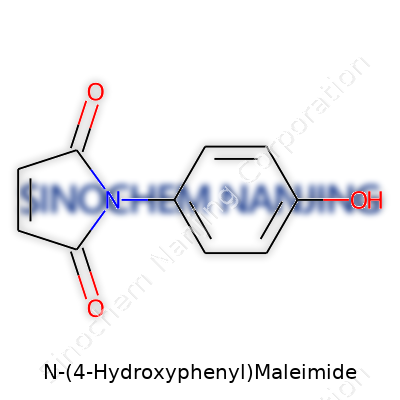
What is the chemical structure of N-(4-Hydroxyphenyl)Maleimide?
Unpacking the Basics
N-(4-Hydroxyphenyl)maleimide might look like just another complex name out of a textbook, but its structure tells an interesting story. Let’s paint the picture. This compound blends two key features: a maleimide group and a hydroxyphenyl group. Strip that down, and what you get is a maleimide ring, a five-membered setup known for its electron-deficient double bond—reactive, ready to hook up with nucleophiles. Then you glue on a hydroxyphenyl group at the nitrogen, specifically landing at the ‘4’ spot on the phenyl ring.
Draw this out, and the core looks like this: take a benzene ring, stick an –OH group at the para position, then plug in the maleimide piece via the nitrogen. Chemically, you're talking about C10H7NO3.
Why the Structure Packs a Punch
This isn’t just a matter of counting carbons. N-(4-Hydroxyphenyl)maleimide ends up in research labs for its versatility. The hydroxy group changes the landscape, adding a handle for further reactions—think of it as a place where scientists can attach other useful pieces, or tweak the electric atmosphere of the compound. That hydroxy group often brings solubility benefits, too.
Maleimide groups get a lot of attention in materials science and biomedical research. Their double bonds love to react with thiols, those –SH groups found in cysteine residues in proteins. It’s the backbone of many “click chemistry” projects. Its structure makes it a building block for bioconjugates—where you want to stick a therapeutic agent onto an antibody or develop fancy biosensors. The hydroxyphenyl twist adds strength for applications where water solubility or biological compatibility really matter.
Real-World Impact
I remember a grad student in my lab grumbling about stubborn maleimide reactions until he tried a compound similar to N-(4-Hydroxyphenyl)maleimide. It solved his solubility headache overnight. It also helped the bioconjugation go faster, showing how small tweaks in chemical structure make a massive difference on the bench.
Applications aren’t all in the lab. Pharmaceutical researchers rely on these structural designs when finding new candidates for drug delivery systems. The maleimide group's reactivity lets you target specific proteins, helping deliver drugs right where they’re needed, which cuts down on side effects. Water-friendly tweaks from the hydroxy group can make a compound more compatible in the body.
Staying Safe and Smart
Speaking from years at the fume hood, safe handling is never an afterthought. The aromaticity from the phenyl group brings a stability edge, but maleimide rings don’t mess around—they need proper protective gear, like gloves and goggles. Things can get volatile with certain reagents, so a good extraction system is key.
Manufacturing, too, sees some hurdles. Care in synthesis stops unwanted byproducts from muddying up the final chemical. Paying attention to temperature and pH matters, because these features shape both yield and purity.
Moving Forward
N-(4-Hydroxyphenyl)maleimide stands as a small but mighty tool for chemists. Its structure—maleimide ready to grab onto thiols, phenyl ring bringing flexibility, hydroxy group making the whole thing pop in water—opens new doors for research. For greener lab practices, researchers now look into catalysts and milder reaction conditions during its cleanup steps. Collaborative work with biologists and engineers could also make these compounds even more precise for targeting disease or building new materials.
What are the main applications of N-(4-Hydroxyphenyl)Maleimide?
Unlocking Value in Polymer Chemistry
N-(4-Hydroxyphenyl)maleimide has grabbed the attention of chemists, especially those working on advanced polymers. The unique structure, blending a maleimide group with a hydroxyphenyl ring, shows up in laboratories for a reason—it can drive important changes in polymer systems. It’s frequently used as a monomer or as a modifying agent to create specialty resins. The hydroxyphenyl group brings another layer of reactivity, opening the door for cross-linking, which enhances strength and thermal stability in finished products.
A resin recipe can shift from brittle to impact-resistant by introducing a small dose of this compound. That’s something I’ve seen firsthand, working on epoxy projects where a drop in performance spells trouble for coatings or adhesives. Here, stability and resilience under heat matter more than price or ease of manufacture. Research published in Polymer Degradation and Stability shows that these additives defend against oxidation and ultraviolet damage, making long-lasting plastics for automotive and electronics applications.
Applications in Advanced Coatings
Surface protection requires chemistry that holds up under real stress. N-(4-Hydroxyphenyl)maleimide contributes toughening and cross-link density, making protective paints more resistant to scratches, corrosion, and environmental challenges. Think of heavy machinery, pipeline coatings, or marine paints; the technology behind them relies on additives like this to block failure in harsh conditions.
Using this compound, coatings show less yellowing and slow chalking even under strong sunlight. Data from materials science journals, and my own experience restoring outdoor fixtures, back up the fact: formulas enhanced with this chemistry survive longer, keep color, and don’t crack as quickly. The investment up front pays dividends in reduced maintenance and repair work over time.
Biomedical Materials and Adhesive Systems
Medical devices demand reliability at a cellular level. N-(4-Hydroxyphenyl)maleimide fits into this role by giving bioengineered materials the strength and flexibility surgeons and patients need. Hydrogel systems based on this compound show promise in controlled drug delivery, wound dressings, and tissue engineering. Peer-reviewed articles from Biomacromolecules highlight improvements in adhesion, mechanical strength, and resistance to degradation, especially when the patient’s health is on the line.
Adhesive technologies for electronics and medical use cannot cut corners. The hydroxyphenyl group can form strong bonds with other molecules, offering extra stick and curing under mild conditions. I’ve noticed glues made with maleimide derivatives holding up under hot and humid storage—something standard formulations just don’t handle well.
Supporting Cleaner Industry and Sustainability
Environmental concerns walk alongside chemistry these days. N-(4-Hydroxyphenyl)maleimide enables manufacturers to create tougher, longer-lasting products, which often leads to less waste, less frequent re-coating, and fewer resources spent on maintenance. Better performance per kilogram of material used aligns with growing calls for sustainability.
Developers are now exploring ways to integrate renewable raw materials and improve recycling techniques for maleimide-based polymers. The focus is on keeping quality high without increasing environmental burden. Companies that keep pace with these changes will shape the next era of manufacturing and materials science, not just in specialty polymers, but across a much broader range of everyday products.
How should N-(4-Hydroxyphenyl)Maleimide be stored?
Understanding the Risks and Reactions
Some chemicals deserve a little more respect than others, and N-(4-Hydroxyphenyl)maleimide sits right up there. I spent part of my career in a chemistry lab where one mishandled bottle could ruin months of research. This compound, with its reactive maleimide group and phenolic ring, brings both opportunity and risk. Left in the wrong spot, exposed to extra moisture, or hit by sunlight, it breaks down or kicks off unwanted reactions. The science community already knows what happens when things go south: wasted chemicals, uncontrolled polymerization, or even hazardous air quality.
Minimizing Degradation – Lessons From the Bench
Every time I cracked open an expensive reagent and saw crust at the seal–I remembered how easy it sounds to “store in a cool, dry place,” and how tough that is outside textbook pages. For N-(4-Hydroxyphenyl)maleimide, keeping the temperature stable lowers the risk of decomposition. Fluctuations push the compound toward instability. Most formulations stay safest at 2–8°C, similar to a common refrigerator, but far apart from food or drink to avoid disaster. A desiccator becomes a solid friend for this kind of job. Silica gel packs inside, maybe a layer of inert gas for extra measure, and a tight-sealing lid buy a lot of peace of mind.
Physical Integrity and Chemical Safety
I once left a reagent in a clear container near the lab windows and saw it brown after just a week. So, it’s a no-brainer to keep N-(4-Hydroxyphenyl)maleimide in an amber or opaque vessel. UV light breaks down molecular bonds. Glass jars do the job best, since a plastic container sometimes leaches or reacts, especially under stress. Durable labels, waterproof ink, and clear dates keep others out of harm’s way when your day off rolls around. Spills aren’t just wasteful; they feed accidental exposure to skin or lungs, and, honestly, no one walks away feeling smart after that.
Why Good Storage Supports Research and Health
Proper storage keeps the chemical pure, which gives accurate, reproducible results in the lab. Impurities from moisture or air lead to reactions that don’t line up with control samples or published data. That can cost thousands of dollars and months of labor, not to mention reputation. Mistakes stick around. One contaminated stock bottle can seed a whole year’s work with doubt. On the health side, inhaling powder or coming in contact with your skin can bring irritation and allergies. Gloves, safety glasses, and a ventilated workspace stay non-negotiable as any manual will stress. Stores across Europe and the US follow OSHA guidelines for good reason; nobody wants even a whiff of liability or injury.
Better Storage, Better Chemistry
No fancy robotics or high-dollar sensors fix plain inattention. Assign someone for chemical audits every season, track inventory closely, and lock up higher-risk compounds after hours. Tech makes life easier: digital temperature and humidity sensors spot trouble before it spreads. Storing N-(4-Hydroxyphenyl)maleimide properly doesn’t just protect the bottle; it preserves careers, lab budgets, and sometimes, larger goals for safe innovation. Good storage isn’t glamorous, but it keeps science honest and people safe.
What is the purity and available packaging sizes for N-(4-Hydroxyphenyl)Maleimide?
Looking at Purity in the Lab
Focusing on purity takes me back to my own university days, watching researchers groan when shipments arrived with lower-than-expected assay results. N-(4-Hydroxyphenyl)Maleimide usually comes in high-purity grades, with 97% and above showing up on most certificates of analysis. Chasing good data often means anything less than 98% causes problems. If a batch lands at 99%, you can hear a collective sigh of relief—impurities introduce unknowns no one wants in a synthesis.
Journal articles reference the compound as a reagent for modification of macromolecules and work in organic electronics. The challenge: contamination ruins a clean reaction, so reliable purity offers more than convenience. It shields research from false leads. In industrial applications—think specialty coatings or cross-linkers—the same standard holds. A supplier promising greater than 99% purity doesn’t just tick a box for a procurement officer; they safeguard the process for every chemist down the chain.
A 2021 analysis from Sigma-Aldrich listed N-(4-Hydroxyphenyl)Maleimide with minimum 98% purity, and most specialty chemical catalogs don’t drop below that standard. Published protocols, whether from Shanghai or Cambridge, specify purity in detail to avoid unpredictable results. Off-brand or gray-market suppliers sometimes claim lower purities, but buyers learn to verify every claim before trusting a catalog.
Breaking Down Packaging Sizes
Pack sizes do more than fill a shelf. For researchers, small packs of 1 gram or 5 grams make sense, especially in early-stage or pilot work. The idea is simple: why open a bottle larger than what’s needed for a few test runs? In my lab, people got nervous ordering a large container if most of it would wind up wasting away after one project.
Commercial suppliers know the trend. Most labs can pick up 1 g, 5 g, or 10 g glass vials straight off the shelf. Bigger outfits handling scale-up or pre-production rounds see 25 g, 100 g, or even 500 g containers, sometimes in HDPE or amber glass to protect the compound from light and moisture. For full commercial manufacturing, kilo-sized drums come into play, though handling requirements jump. Larger sizes often trigger extra documentation or compliance checks, especially for chemicals leaving the country or going into highly regulated spaces.
Challenges, Risks, and Smarter Solutions
Sometimes people ignore the impact of purity levels and packaging sizes, treating them as afterthoughts. That approach costs time: one poorly characterized batch can send weeks of work down the drain. Chemical tracking, rigorous third-party testing, and making sure suppliers disclose their purity testing methods—these details cut down on risk.
A side benefit of offering multiple sizes: less waste and safer handling. Smaller vials lower exposure, something worth thinking about with phenolic maleimides. Many suppliers stepped up certifications on their packaging to help minimize accidental exposure, as reported by chemical safety groups in recent years.
Those who plan ahead, match the right purity to the right application, and order just enough of what’s required save money, time, and headaches. Every chemist knows an experiment can fail due to a hidden impurity or a misjudged material order. Opening a box and seeing the right grade in a suitable size remains a small but important win.
Are there any safety or handling precautions for N-(4-Hydroxyphenyl)Maleimide?
Understanding What You’re Working With
Science never stands still, and neither do the chemicals people work with in laboratories or production sites. N-(4-Hydroxyphenyl)maleimide often comes up in research and industrial settings. Its structure brings together a maleimide group and a hydroxyphenyl moiety, each with its own quirks. This stuff isn’t a household name, but anyone dealing with it should take a few important facts seriously.
Hands-On Safety
From my days at the lab bench, I remember how some chemicals look perfectly harmless—white powders, clear liquids, sometimes even a bit bland. N-(4-Hydroxyphenyl)maleimide fits the bill, but don’t let it fool you. The compound’s reactive groups set the stage for potential skin and eye irritation. It won’t announce its presence with a strong odor or flashy appearance, so I always kept gloves on and protective glasses within reach. Even handling bottles slowly and precisely cut down the risk of spills or accidental skin contact.
Air Quality and Ventilation
Nobody wants to taste chemicals in the air. Lab hoods aren’t just for show; they cut down on the chance of fumes or fine dust crossing your path. Maleimide derivatives sometimes put out vapors that quietly float into the air. Opening containers inside a fume hood just made sense, and swapping out filters on time turned out to be one of the simplest forms of self-preservation.
Storage Matters
Leaving bottles scattered or under harsh lighting creates trouble down the line. This compound stores best in tightly sealed containers, kept away from moisture or heat. Even cabinets matter—a dry, cool spot away from acids or bases always served me well. Once I let a bottle sit on a sunny shelf, and the crust on the rim taught me a quick lesson in keeping things tidy.
Dealing with the Unexpected: Spills and Waste
Nobody plans for accidents, but labs run smoother when you remember clean-up basics. Small spills soak up with absorbent material, but there’s no skipping gloves or even a dust mask if things kick up into the air. Waste heads straight to a well-marked container—never down the drain—because water systems and wildlife shouldn’t take the hit. I’ve seen people cut corners, then rush to explain cloudy fish tanks or dying plants.
Respect for the Unknown
The safety data on N-(4-Hydroxyphenyl)maleimide remains a bit thin, especially regarding long-term health risks. Some structures in the maleimide family flag concerns over mutagenicity or effects on the reproductive system. Even if you never feel a tickle in your throat, it’s smart to keep cautious. Reporting symptoms early, asking for a second opinion, or reading up on recent safety data sheets often proved worth the few minutes it takes.
Solutions and Improvements
Training sticks out above all else. Updates to the usual safety briefings, sharing real stories from colleagues, and regular practice of response drills turn safety from a checklist into a habit. Encouraging a team approach, where nobody feels shy about flagging sloppy habits, helps keep mistakes down. Good-quality gloves, working goggles, and up-to-date ventilation all add up, but people talking openly about risks makes the biggest difference.
Why It Matters
Whether you work in a sprawling chemical plant or just do experiments at a university, cutting corners just isn’t worth it. Each step—gloves, goggles, ventilation—protects not just you, but your coworkers and the broader community. The peace of mind from a well-run lab or shop stems from a culture that puts safety first, keeps everyone breathing easy, and turns chemical know-how into something that benefits everyone involved.
| Names | |
| Preferred IUPAC name | 4-(Isocyanatomethyl)benzene-1,2-diol |
| Other names |
4-Hydroxyphenylmaleimide N-(p-Hydroxyphenyl)maleimide N-(4-Hydroxyphenyl)maleimide |
| Pronunciation | /ɛn faɪdˌrɒksiˈfiːnəl məˈleɪ.ɪmaɪd/ |
| Identifiers | |
| CAS Number | 4793-99-5 |
| Beilstein Reference | 144465 |
| ChEBI | CHEBI:189140 |
| ChEMBL | CHEMBL483053 |
| ChemSpider | 20743479 |
| DrugBank | DB08419 |
| ECHA InfoCard | 17a2c0a7-020d-47f9-b071-3c8bb3c48120 |
| EC Number | 610-516-5 |
| Gmelin Reference | 134271 |
| KEGG | C19415 |
| MeSH | D017958 |
| PubChem CID | 122345 |
| RTECS number | SN6657000 |
| UNII | 303U42355U |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C10H7NO3 |
| Molar mass | 203.18 g/mol |
| Appearance | Light yellow powder |
| Odor | Odorless |
| Density | 1.38 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.77 |
| Acidity (pKa) | 7.7 |
| Basicity (pKb) | 11.67 |
| Magnetic susceptibility (χ) | -60.06 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.679 |
| Dipole moment | 3.5467 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.9 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N02BE01 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 1, Special: |
| Flash point | > 230°C |
| Lethal dose or concentration | LD50 (oral, rat) > 2000 mg/kg |
| LD50 (median dose) | LD50: >2000 mg/kg (rat, oral) |
| NIOSH | SN8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m3 |
| Related compounds | |
| Related compounds |
N-Phenylmaleimide N-Ethylmaleimide Maleimide N-(4-Methoxyphenyl)maleimide N-(2-Hydroxyethyl)maleimide |

