Monoethylamine: Chemistry that Shapes Industry and Safety Concerns
Historical Development
Monoethylamine connects to both the late nineteenth-century curiosity about amines and the practical needs of early chemical industry. Organic chemists found ways to make this simple compound by reacting ammonia with ethanol, and over decades, its methods of preparation improved for bigger, cleaner yields. This molecule has rarely featured in the spotlight, but many of us have benefited from it quietly transforming processes behind the scenes. Its use ramped up with the growth of synthetic dyes, pharmaceuticals, and rubber manufacturing, trailing invention alongside industrial progress. Each generation finds new tasks for this workhorse molecule, showing chemical progress rarely looks flashy on the factory floor.
Product Overview
Monoethylamine stands out for being a building block in many different industries, not as a finished product but as a foundation. It shows up as a colorless gas with a strong odor, and often gets shipped and stored as a water solution or under pressure as a liquid. Many chemical plants source or make it to produce herbicides, rubber accelerators, dyes, and medications. In every case, chemical companies rely on its ability to participate in a variety of reactions. People may not realize just how many things in daily life come from processes that use some monoethylamine along the way. Its value lies in its reactivity, letting manufacturers fine-tune everything from plant treatments to painkillers.
Physical & Chemical Properties
Chemists have studied monoethylamine in detail because its simple structure and strong amine character make it easy to predict but also a bit dangerous. It has a boiling point close to room temperature at standard pressure, which means it evaporates easily and can be tough to contain without proper equipment. Its sharp smell acts as a warning for leaks. Chemically, it has a lone electron pair on nitrogen that turns it into a strong base and a good nucleophile, making it react with acids, acylating agents, and alkyl halides without fuss. Its readiness to grab a proton or bond with carbon skeletons anchors its importance for those making new compounds.
Technical Specifications & Labeling
Anyone working with monoethylamine watches quality closely. Impurity levels matter, especially in pharmaceutical or food-adjacent manufacturing. Packaging always reflects safety priorities, with clear hazard labels and tight controls on the containers since the gas is flammable and the solutions are corrosive. Documentation provided with shipments covers concentration, net quantity, and storage precautions. Facilities with experience using it keep their storage cool and dry, away from anything that could spark a fire. For most end users, the safeguards have evolved through both scientific progress and occasional accidents that have forced the industry to become more transparent in its warnings.
Preparation Method
Commercially, monoethylamine often begins with ethanol and ammonia under pressure and heat, handled with suitable catalysts to direct the reaction for maximum yield. Chemical engineers favor continuous processes for economy, drawing from earlier smaller-batch methods where yields were lower, side reactions more common, and risk of contamination higher. The attention to process optimization allowed manufacturers to minimize other amine byproducts like diethylamine or triethylamine, which can complicate downstream uses or require further separation. The evolution of catalytic and process design shows how the boring side of chemistry—optimizing operating pressures, reactor design, product separation—makes possible the huge scale needed for agriculture, pharmaceuticals, and textile processing.
Chemical Reactions & Modifications
Monoethylamine reacts with acids to form stable salts, making it easy to handle in certain industrial processes. It also combines with carbonyl-containing compounds, reacts with alkylating agents, and participates in condensation reactions to build up more complex molecules. These properties have led to applications ranging from the creation of rubber vulcanization agents to the synthesis of pesticide active ingredients. In the pharmaceutical world, it lets chemists adjust the solubility or biological action of a candidate drug by introducing amine side chains. The ability to easily modify structure has expanded its relevance for both basic research and applied industrial processes.
Synonyms & Product Names
Monoethylamine goes by various names in different circles—ethanamine among chemists, ethylamine in older literature, or just MEA on shipping and regulatory paperwork. Each synonym reflects a slightly different context but the chemistry stays the same. For those in the field, knowing the synonyms and abbreviations avoids confusion in purchasing, storage, and handling, especially since similar amines can have different risk profiles.
Safety & Operational Standards
Practical chemical manufacturing never ignores the hazards associated with monoethylamine. Its flammable, corrosive, and volatile features shape the design of storage tanks and pipelines. Industrial hygiene experts insist on good ventilation, monitoring for leaks with gas detectors, and provision of protective equipment like gloves and eye shields. In my time working with chemical storage, I saw firsthand the importance of regular maintenance on pressure relief devices and gas-tight valves. Accidental releases trigger strict response plans to protect workers and the environment. Training and clear standard operating procedures make the biggest difference—engineers and floor operators rely on drills and incident reporting to keep risk low.
Application Area
Monoethylamine helps produce some of the most recognizable consumer items: analgesics, cough remedies, herbicides that shape modern agriculture, dyes used to color everything from jeans to packaging, and accelerators that make rubber tires more durable. In each area, its value emerges from unique chemistry rather than being irreplaceable in a regulatory sense. Agricultural chemical makers rely on it for making select herbicides, but its presence in over-the-counter medicines shows the breadth of its reach. Such versatility also places the burden of safe management across a wide set of industries, so expertise in one field helps build good practices elsewhere.
Research & Development
Monoethylamine continues to draw the attention of researchers looking for safer synthesis routes, alternative feedstocks, or improved downstream reactions. It supports the creation of new chemical scaffolds for drugs, novel pesticides, and dyes with better colorfastness or biocompatibility. Green chemistry advocates investigate routes that might reduce the environmental footprint from both production and downstream use, like replacing petroleum-based ethanol with biomass-derived sources or designing catalysts that improve selectivity and lower byproduct formation. Innovation here depends not only on advances in chemistry but also in engineering and environmental science, since process sustainability attracts the attention of both regulators and industry partners.
Toxicity Research
Laboratories and factories handle monoethylamine with caution because its toxicity—both acute and chronic—has received plenty of attention from occupational health specialists. Exposure can cause skin burns, respiratory discomfort, and eye damage. Scientific studies examining long-term health effects, both in animal models and industrial workforces, shape regulatory limits for workplace air concentrations and exposure durations. Few chemicals make the journey from discovery to industrial mainstay without scrutiny. For instance, respirators and improved ventilation go hand-in-hand with regular worker health monitoring. Data gathered from incidents and routine medical checks has translated into regulatory frameworks worldwide, limiting both immediate health risks and potential long-term harm.
Future Prospects
The market for monoethylamine keeps growing as industry finds more uses for amine building blocks and demands higher purity. As countries call for greener chemical practices, companies investing in cleaner manufacturing methods will likely take the lead and shape industry standards. Demand in pharmaceutical and agrochemical sectors matches a wider societal need for better medicines and more effective food production. Safer production and handling remain ongoing challenges, especially in developing regions where infrastructure may lag. Increased transparency in safety data reporting and new regulatory controls can shift the landscape for both established and emerging markets. In the end, the pace of discovery and regulation often runs neck-and-neck, but both point to a future where safety and utility continue to define monoethylamine’s place in the world.
What is Monoethylamine used for?
An Everyday Chemical with Hidden Reach
Few people have ever heard about monoethylamine, yet its fingerprints show up on plenty of products and processes that shape our daily lives. I once spent a summer working in a fertilizer manufacturing plant, and the importance of chemicals like this became pretty apparent. From the pungent scent to the constant buzz about safety, I learned just how common this substance is in industrial settings, even though most folks barely notice it outside the lab.
Role in Agriculture and Animal Health
Monoethylamine gets serious attention in fertilizer production. Plants often need a boost of nitrogen for healthy growth, and this chemical acts as a key building block in the process. Companies use it to produce herbicides and pesticides that help control crops’ unwanted guests, delivering benefits that ripple through the entire food chain. Straightforward, cost-effective, and consistent, monoethylamine has earned a spot in the toolkit for large-scale farming.
In animal health, manufacturers rely on it to create drugs and supplements designed to keep livestock fit and productive. Keeping herds healthy means fewer outbreaks and a better supply of safe meat, milk, and eggs. In those busy barns and feedlots, the chemical’s impact seems indirect, yet crucial.
On the Factory Floor: Cleaning, Manufacturing, and Dyes
Factories where detergents and cleaning agents get made also rely on monoethylamine. It helps break down grease and grime on hard-to-clean surfaces, supporting the production of heavy-duty cleaners. Some of the sparkling results you see in large kitchens or public places have traces of this chemical’s involvement.
Manufacturers trust it as a basic ingredient for a whole roster of industrial products: rubber chemicals, corrosion inhibitors, and even those tough resins used in paints. The textile business leans on the dye-manufacturing side of monoethylamine, as it reacts well with a variety of compounds to bring bold colors to fabrics.
Here in the U.S., the Occupational Safety and Health Administration (OSHA) sets strict guidelines for people working with monoethylamine. It is highly corrosive and irritating, so anyone handling it in plants must wear protective gear. I remember suiting up myself: gloves, goggles, the lot—because one slip meant dealing with rashes or breathing issues.
Pharmaceuticals and Fuel Additives
Medicines form another part of this story. Researchers use monoethylamine during the early phases of drug development, especially while working with painkillers and mood stabilizers. Its ability to help “build” more complex chemicals keeps medical research moving. Sometimes, it plays a role in fuel additives that help gasoline burn cleaner, which matters for reducing air pollution in cities.
Access to clean and safe chemicals affects everyone, from the farm to the pharmacy. Regulations on production and storage matter because this stuff, as useful as it is, packs some serious punch if mishandled. Outfits that take safety seriously see fewer accidents, better productivity, and less harm to the environment.
Room to Improve: Safer Practices and Alternatives
Plenty of experts push for safer manufacturing and stricter monitoring, especially where run-off can pollute rivers or groundwater. Exploring greener alternatives shines as another avenue—whether by finding substances that do the same job with less risk, or designing new technologies from scratch. My own experience says that extra training and modern equipment make a real difference.
Understanding what goes into common products helps everyone—producers, workers, shoppers—ask better questions and demand smarter solutions. Chemicals like monoethylamine remain foundational for now, but seeking ways to make workplaces safer and the planet cleaner should always stay high on the list.
What is the chemical formula of Monoethylamine?
What Exactly Is Monoethylamine?
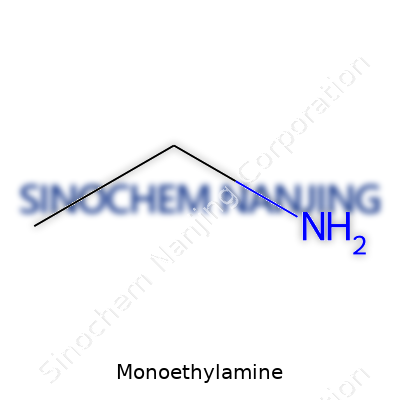
Monoethylamine shows up in a lot of everyday industrial processes. It’s one of those compounds that flies under the radar until you look into the details of how things get made. The chemical formula of monoethylamine is C2H7N. It’s a compound made from carbon, hydrogen, and nitrogen, and while that might seem simple on the surface, the way it behaves holds real weight in industries from agriculture to pharmaceuticals.
Why Monoethylamine Matters in Chemicals and Manufacturing
Take a look around any fertilizer plant or pharmaceutical manufacturing setup—monoethylamine often plays a behind-the-scenes role. It’s used to make herbicides, rubbers, dyes, and medicines. In my experience working near chemical production sites, employees pay special attention to proper handling, because monoethylamine is not just a building block but also a potential health hazard if things go sideways. It can cause irritation and toxic exposure, so lab staff don protective gear and maintain solid ventilation systems where it’s stored and transferred.
For reference, that formula—C2H7N—means each molecule consists of two carbon atoms, seven hydrogens, and one nitrogen atom. The structure lets it blend well with water and alcohols, which helps it move smoothly through reaction steps during production. I remember long days in an R&D lab, watching as monoethylamine sped up processes and cut down reaction times, letting us get to end products a little quicker. Efficiency matters, and this compound delivers, but it’s not all smooth sailing if safeguards get skipped.
Health and Environmental Stakes
Monoethylamine’s usefulness comes with responsibility. Accidental spills don’t just disappear. The compound can start reacting with other chemicals nearby, and its sharp odor can punch through weak ventilation. Workers can get hit with eye, skin, and lung irritation if they’re not careful. I’ve seen first-hand how proper safety training pays off—knowing when to use respirators, managing containment equipment, and always keeping quality emergency wash stations on hand.
From an environmental view, monoethylamine brings concerns if it leaks into water systems or soil. Local wildlife and water tables get exposed to chemicals that weren’t ever meant to be part of the ecosystem. Regulations require airtight storage and disposal, but I’ve watched crews double-check seals and monitor air quality, realizing that any slipup could affect much more than just workplace safety.
Building Toward Safer Chemical Use
It’s impossible to separate the talk about monoethylamine’s chemical formula from the rest of the picture. The formula tells you why it mixes so well or evaporates quickly, but as with all industrial chemicals, real world use brings risk and reward. Technology has evolved to include automated leak sensors, more durable piping, and better neutralization chemicals in case of emergency. These upgrades have come by listening to chemists, engineers, and frontline workers who handle these compounds daily.
Further progress will come from a mix of stricter rules, smarter engineering, and practical training. Earning trust across communities means showing exactly how each chemical, including monoethylamine, gets put to work and kept safely out of the wrong places. As more industries rethink their supply chain impacts and focus on sustainability, knowing the chemistry isn’t enough—accountability and care matter just as much as what sits in a beaker or gets printed on a label.
Is Monoethylamine hazardous or toxic?
How Monoethylamine Shows Up In Real Life
Monoethylamine turns up in a bunch of industries. You see it in the making of pharmaceuticals, resins, dyes, and cleaning products. It's one of those compounds that helps with a lot of daily-need materials, popping up in everything from agricultural chemicals to fuel additives. This widespread use raises obvious questions about its health and safety risks, especially for workers and communities near manufacturing sites.
Direct Encounters Spell Trouble
If you've ever handled amines in a lab, you learn pretty fast they're not your average household chemicals. Monoethylamine hits with a sharp, ammonia-like smell. Breathing it in for a few minutes can make your nose sting and eyes water. Prolonged exposure, or higher concentrations, might mean headaches, nausea, chest tightness, or even chemical burns in the airways. Skin or eye contact often leads to irritation—redness, pain, sometimes blistering. A splash in the eye can have lasting effects if not rinsed out right away.
Facts Back Up the Concerns
The Centers for Disease Control and Prevention (CDC) lists monoethylamine as a hazardous chemical. Its Material Safety Data Sheet (MSDS) spells out risks like respiratory trouble, skin burns, and potential harm if swallowed. Tests found repeated exposure can cause asthma-like symptoms and skin sensitization in some cases. The National Institute for Occupational Safety and Health (NIOSH) recommends an eight-hour exposure limit of just 3 parts per million for workers. That number speaks volumes about its impact if basic precautions get ignored.
Environmental Risks
Monoethylamine doesn't stay put. In the environment, it breaks down fairly quickly, so it doesn't build up the way some heavy metals or persistent organics do. But spills or leaks into water or soil still pose an acute hazard to aquatic life. Fish gills, for example, are sensitive to high pH levels or amine exposure. Left unchecked, a spill could wipe out local microorganisms and set off a chain reaction through a pond or stream.
Lessons From Life In The Lab
Years ago, as a graduate student in chemistry, I watched a bottle of monoethylamine wisp out vapor because someone forgot to tighten the cap. The sharp scent filled the air. By the time I got the hood running, I had learned my lesson. Gloves, goggles, vents—it takes only one mistake to remind you these chemicals can’t be taken lightly. Over time, news stories and safety training pushed the same message: direct handling means real risk.
Finding Smarter Solutions
Industry leaders and regulators recommend tight controls. Proper ventilation, closed transfer systems, and robust training lower the risk. Workers benefit from real-time air monitoring or personal protective equipment. Whenever possible, companies swap in safer alternatives or reduce monoethylamine in end products. For smaller labs or makers working at home, extra attention to ventilation—opening windows, using fans or fume hoods—makes a huge difference. Emergency showers and eye-rinse stations don’t just meet regulations; they save eyes and skin from chemical burns in a pinch.
Practical Ways Forward
Communication matters just as much. Clear safety signage, regular hazard drills, and easy-to-access SDS information help everyone manage the dangers. Beyond that, research into less hazardous substitutes and greener production methods is picking up pace. The more we know as users, the safer everyone stays—on the job, at home, and across the environment. No one chemical will fit every need, but smart practice keeps the worst risks at a safe distance.
What are the storage requirements for Monoethylamine?
What Matters Most in Storing Monoethylamine
Monoethylamine catches attention because it doesn’t play by the rules of tame substances. It comes with its own set of demands if you plan to store it safely. This isn’t just a niche concern for plant managers or warehouse workers. Anyone who lives or works near industrial facilities—anyone who wants less drama from accidental chemical exposure—stands to gain from understanding how a smart storage approach makes everyone safer.
Understanding the Risks and Hazards
Monoethylamine releases strong vapors. At room temperature, these vapors can sting your eyes and throat or worse, spark a fire if they meet an ignition source. This is a chemical that doesn’t take rough treatment well. So, controlling its temperature and keeping it from reacting with air or water matters.
Choosing the Right Containers and Locations
From my time walking through old storage sites and modern chemical facilities, one thing’s clear: material choice for tanks and containers isn’t negotiable. Carbon steel, stainless steel, or specially lined drums stand up to ethylamine’s corrosive streak. Forget cheap plastics or thin-walled options—those cut corners can leak or deform under pressure. Leaks happen—most storage disasters I’ve seen begin with cracked valves or worn seals, so regular checks and replacements go on the must-do list.
Location plays a role that reaches beyond just an address. Tanks stored outdoors face swings in temperature, letting vapors build up if the area’s not ventilated. An enclosed space without strong airflow turns minor spills into major headaches fast. Setting tanks on chemical-resistant concrete pads and spacing drums so spills don’t pool underneath reduces surprises. Action beats luck, especially where chemicals meet the real world.
Fire Safety Isn’t Optional
One brush with monoethylamine’s flammable side convinced me that fire prevention isn’t just policy—it’s peace of mind. Storing containers away from open flames, electrical panels, or spark-prone tools should be non-negotiable. Installing grounding bars and static discharge chains can help stop static from sneaking in and setting off a disaster. Most serious accidents don’t start with a Hollywood-style explosion; they creep up through overlooked details like a forgotten battery charger or worn insulation on electrical wiring.
Temperature Control and Ventilation
Every chemical has a comfort zone. Monoethylamine prefers cool, shaded spaces out of direct sunlight. Workplaces with top-notch storage often rely on built-in ventilation hoods or exhaust systems to sweep away irritating vapors. Experience taught me to place temperature gauges and vapor detectors nearby so any problem doesn't lurk in the background. These extras cost less than the mess that follows a runaway reaction or vapor cloud.
Labeling, Training, and Emergency Tools
Walk through a responsible storage area and labels jump out at you. Clear, bold warnings on every tank, every drum, and even the floor. Training goes alongside these warnings. People working with monoethylamine run monthly safety drills, learning to handle spills, dodge direct contact, and use eye wash stations in a hurry. Spill kits, respirators, and chemical-resistant gloves wait within arm’s reach, not locked away for rare emergencies.
Thinking Beyond Compliance
I’ve visited operations where workers remembered the close calls more than dry lists of regulations. They learn that safety doesn’t slow production; it keeps business running without the chaos of shutdowns and injury investigations. Smart storage for monoethylamine protects more than property—it helps everyone go home whole at the end of the day.
How should Monoethylamine be handled safely?
Protecting Health in the Workplace
Monoethylamine has always struck me as one of those chemicals that doesn’t announce its dangers loudly, but you sure feel it if something goes wrong. Years back, watching a coworker deal with a tiny spill drove home just how quickly things can escalate with this substance. The key fact: monoethylamine can irritate skin, eyes, and especially the lungs. It has a strong ammonia odor — enough to make you step back. That smell isn’t just unpleasant; it signals that the air already carries a level that could cause headaches, nausea, or worse with repeated exposure.
Anyone working with monoethylamine should suit up before even cracking open the drum. Chemical splash goggles, nitrile gloves, and a long-sleeve lab coat create a practical barrier. Face shields give extra protection when pouring or transferring. This isn’t overkill. Absorbing the liquid through the skin or breathing fumes can happen fast, and no job pays enough to gamble with your health. Keeping an eyewash station and safety shower in arm’s reach is a mark of respect for the risks involved — something I’ve seen overlooked during busy shifts and paid for with painful lessons.
The Importance of Good Ventilation
There’s no good reason to use monoethylamine in a cramped, stuffy room. Good ventilation moves fumes out sharply; those who install local exhaust hoods or use fume cupboards create a safer environment for everyone. Sticking a fan in the window doesn’t cut it. The chemical’s vapors can form dangerous concentrations at room temperature, and one careless breath is enough to produce coughing or much worse.
Storage Practices to Prevent Accidents
Monoethylamine reacts with acids and oxidizers a lot more violently than some expect. Storing it somewhere dry, cool, and away from incompatible materials stays non-negotiable. Seals need checking; vented containers give vapors somewhere to go safely. Labeling stands as a simple but crucial defense. Everybody in the area reads the name and hazard class at a glance, so there’s no confusion if an emergency pops up. The best practice? Limit access. Only trained staff get the keys, and every use goes in a log. That’s one checklist I never skip, because knowing who did what, and when, matters when people’s safety is at stake.
Training and Emergency Response
People sometimes underestimate just how much a well-drilled emergency plan pays off. Regular training with fire extinguishers, spill kits, and respirators builds confidence and cuts hesitation if things go south. I’ll never forget seeing how relaxed folks grew after running a few simple drills. Just knowing where to find the right PPE and what order to respond in made a real difference. Communication also matters. Clear instructions, posted emergency numbers, and visible procedures cut confusion during tense moments.
Responsible Disposal
Disposing of monoethylamine waste brings in another layer of responsibility. Dumping down the drain isn’t just illegal; it can cause harm away from the original site. Sealed, clearly marked waste containers go to licensed hazardous waste handlers. Local regulations cover the details, but the bottom line stays the same — every step matters in protecting not just workers, but the wider community and environment.
The bottom line: real safety demands habit, not luck. Regular checks, the right gear, strong training, and respect for the chemical’s power will always outweigh shortcuts. Monoethylamine is useful, but that usefulness relies on careful, consistent precaution every single day.
| Names | |
| Preferred IUPAC name | Ethanamine |
| Other names |
Ethanamine Ethylamine Aminoethane |
| Pronunciation | /ˌmɒn.oʊˌiː.θɪl.əˈmiːn/ |
| Identifiers | |
| CAS Number | 109-89-7 |
| Beilstein Reference | 605276 |
| ChEBI | CHEBI:15884 |
| ChEMBL | CHEMBL1230653 |
| ChemSpider | 545 |
| DrugBank | DB04269 |
| ECHA InfoCard | 11e4da4c-5f76-4e8d-93b1-47e8e8f1a24e |
| EC Number | 205-483-3 |
| Gmelin Reference | 671 |
| KEGG | C01149 |
| MeSH | D008936 |
| PubChem CID | 7907 |
| RTECS number | KJ5775000 |
| UNII | 9475O96KN7 |
| UN number | 2051 |
| CompTox Dashboard (EPA) | DTXSID5020715 |
| Properties | |
| Chemical formula | C2H7N |
| Molar mass | 45.08 g/mol |
| Appearance | Colorless liquid with an ammonia-like odor |
| Odor | Ammonia-like |
| Density | 0.692 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.77 |
| Vapor pressure | 10.5 kPa (20 °C) |
| Acidity (pKa) | 10.63 |
| Basicity (pKb) | 3.27 |
| Magnetic susceptibility (χ) | -7.5×10⁻⁹ |
| Refractive index (nD) | 1.371 |
| Viscosity | 0.25 mPa·s (at 20 °C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 226.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -77.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1367 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H332 |
| Precautionary statements | P210, P260, P264, P280, P301+P312, P303+P361+P353, P305+P351+P338, P304+P340, P311, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2 |
| Flash point | -18 °C |
| Autoignition temperature | 367 °C |
| Explosive limits | 3.0% - 16% |
| Lethal dose or concentration | LD50 oral rat: 540 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,040 mg/kg (oral, rat) |
| NIOSH | KAE210 |
| PEL (Permissible) | 3 ppm |
| REL (Recommended) | 3 ppm |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Ammonia Methylamine Diethylamine Triethylamine Isopropylamine n-Propylamine |

