Methylhydrazine: The Double-Edged Sword of Chemistry and Industry
Peering Into the History of Methylhydrazine
Methylhydrazine, a staple in the family of hydrazines, carries a story woven through decades of scientific ambition and industrial necessity. Interest in this compound surged alongside the growth of aviation and space technologies. Chemists in the early twentieth century found themselves tinkering with forms of hydrazine for fuel innovations, trying anything that promised a greater push from rocket engines. Methylhydrazine caught wider attention during the Cold War, not by accident, but because it turned out to be a game-changer for rocket propellants, especially in military and aerospace projects. After the first practical syntheses, researchers raced to refine production and handle the mounting safety concerns. The story veers between breakthrough and cautionary tale, echoing much of the chemical industry's past.
Getting to Know Methylhydrazine: What Sets It Apart
Methylhydrazine stands as a small molecule with big personality. Its chemical formula, CH3NHNH2, hints at a volatile nature. Colorless and highly flammable, it lets off a distinct, ammonia-like smell that tends to linger uncomfortably in laboratories. If you’ve ever spilled something nasty in a fume hood, you know the sort of reminder methylhydrazine leaves behind. As with many hydrazines, it brings exceptional reactivity, and this quality opens doors in several industrial and laboratory settings. Its high reactivity also serves as a constant reminder that handling calls for up-to-date protective measures and knowledgeable oversight.
Physical and Chemical Properties: Not for the Faint of Heart
Methylhydrazine boils at 87 °C, evaporating into the air with a vapor pressure that adds worry to any mishandling. The liquid mixes with water, alcohol, and many organic solvents, making cleanup and decontamination a headache. Even trace residues stubbornly hang around surfaces if not properly neutralized. It burns easily, even at room temperature, and reacts with oxidizers in ways that can be explosive. Laboratories label methylhydrazine as corrosive, toxic, and sometimes outright dangerous. The very things that make it sought after—reactivity and volatility—come bundled with these risks.
Specifications and Labeling: Straightforward Warnings
Bottles of methylhydrazine demand clarity in labeling, with hazard diamonds that do not mince words. Anyone picking up a container needs to see not only the name and concentration but also prominent warnings about what could go wrong—fire, poisoning, and environmental harm. Regulations require manufacturers and distributors to list proper storage instructions, usually cool, dry, and far from any oxidizers or open flames. In industry, rigorous tracking ensures accountability and quick response should anything go sideways.
How Methylhydrazine Comes to Be
Producing methylhydrazine leans heavily on nitration and reduction routes, usually starting with methylnitrite or related compounds. Over the years, chemists have fine-tuned methods to lower both cost and hazard, but no process erases the need for skilled oversight. Some methods generate considerable side products, so waste management becomes part of the art, not just the science. Wastewater and off-gas controls receive as much attention as the synthesis itself, given the dangers of uncontained hydrazines.
Reactivity & Modification: Chemistry Unleashed
Ask a synthetic chemist about hydrazines and most acknowledge their use in pharmaceuticals, fuel chemistry, and as building blocks for other specialty chemicals. Methylhydrazine, thanks to its lone pair-rich structure, serves as an eager partner in condensation reactions and cyclizations. It finds use in making pesticides, pharmaceuticals, and sometimes even in dye production. Chemical engineers also turn to it while exploring new classes of polymers and advanced materials. Its fierce reactivity makes it a pivot point for many lab pathways, but always with risk riding shotgun.
What’s In a Name? Synonyms Abound
Open any chemistry text and methylhydrazine appears under different names—monomethylhydrazine, MMH, or 1-methylhydrazine. A handful of trade names may show up in historical patents or industrial supply lists, but this compound’s straightforward identity spares it from the alphabet soup of some specialty chemicals. The diversity of synonyms often confuses newcomers, so clear documentation remains vital, especially for safety audits and regulatory checks.
Working with Methylhydrazine: Do’s, Don’ts, and Red Flags
Anyone with experience in a chemical plant or research lab knows methylhydrazine demands respect. Personal protective equipment is the rule, not the exception—think heavy-duty gloves, full-face shields, and in many cases, supplied-air respirators. Ventilation systems see regular checks. Emergency showers and eye wash stations must stay unblocked and within reach. Spills and leaks get treated as major events, with cleanup teams on alert. Each transfer of this liquid follows written protocols enforced by training and drills. There’s also constant review of transport methods, given how easily a missed step can put workers, communities, and emergency responders at risk.
Application in the Real World: Engines, Labs, and Beyond
Monomethylhydrazine made its name fueling spacecraft, most famously as NASA’s choice for the Apollo lunar modules and later space shuttles. Its ability to ignite on contact with oxidizing agents like nitrogen tetroxide grants engineers the confidence of reliable engine starts, crucial in situations where there’s no room for error. Beyond rocketry, methylhydrazine finds a quieter niche in pharmaceutical synthesis and some agricultural chemicals. Many countries restrict its use outside tightly controlled sectors. In a university lab, it sees light as a reagent for synthesizing heterocyclic compounds and exploring new reaction mechanisms. Every application reckons with toxicity, balancing benefit against the environmental and safety headaches.
What’s New in R&D: Pushing Limits, Facing Challenges
Innovation keeps this molecule in view. Researchers remain keen on replacing methylhydrazine in rocket propulsion with less hazardous chemicals, but the balance of energy, storability, and ignition reliability keeps it in the conversation. In pharmaceuticals, chemists explore derivatives with less toxicity or greater specificity, hoping to harness benefits without carrying over all the risks. Universities push to perfect synthesis routes that cut waste or open new pathways. Environmental scientists keep close tabs on breakdown products, mapping the persistence and movement of methylhydrazine through air and water. Collaboration across national lines shapes much of the research, especially as global security and environmental standards continue to tighten.
A Closer Look at Toxicity and Health Impacts
Few compounds raise alarm bells like methylhydrazine. Direct contact burns skin and eyes almost instantly, and inhalation can bring on headaches, nausea, and confusion—or much worse, given enough exposure. Chronic exposure raises cancer risk, based on animal studies, and there’s evidence for impacts on the liver, kidneys, and central nervous system. Industrial accidents, whether spills or explosions, have triggered evacuations and long clean-up efforts. Regulatory bodies, including OSHA and EPA, set strict exposure limits, but accidents or carelessness bypass safeguards. Worker health programs include regular screenings, and companies that get it wrong can face public backlash alongside legal consequences.
On the Horizon: The Fate of Methylhydrazine
Society’s relationship with methylhydrazine depends on a tightrope walk of necessity and caution. Space exploration may make room for safer alternatives, yet methylhydrazine commands deep institutional trust. Pharmaceuticals research inches forward, sometimes retiring this molecule in favor of safer analogs, other times pulling it back into focus for especially tricky syntheses. Calls grow louder for greener, less hazardous options across all sectors. Legislators, communities, and industry leaders all have a say, but technical performance keeps methylhydrazine planted wherever the demands leave little margin for error. Real progress hinges on expanding training, sharing accident data, and funding safer alternatives. Only by respecting lessons learned—both from success and disaster—can the chemical community manage methylhydrazine’s legacy and its future.
What is methylhydrazine used for?
A Look at Where Methylhydrazine Shows Up
Methylhydrazine doesn’t turn up in everyday conversation, but once somebody starts asking where it surfaces in real life, most people only hear about it in connection to blasting rockets into the sky. The stuff itself is colorless, catches fire in a flash, and clocks in as a real hazard for breathing or touching. Few household cleaning cabinets have a spot for methylhydrazine, and there’s a good reason for that. At heart, it’s a tool for getting jobs done where most chemicals would tap out long before kickoff.
Powering the Big Launches
In rocket science, methylhydrazine finds its main gig. Major space agencies and private companies have trusted it for decades because of its punch. Mixing it with certain oxidizers, engines fire up with a bang—no spark plug needed. That makes launches more reliable—if every second counts in a satellite deployment or a military emergency, no one’s left hoping for good luck in the ignition department.
NASA and Russia’s Roscosmos ran methylhydrazine-based rockets for years. Its cousin, unsymmetrical dimethylhydrazine (UDMH), still sends satellites up with booster stages in Soyuz crafts and many Chinese launchers. Very few fuels kick out energy as fast or light up with as little drama as these do. That’s why, even with all the dangers, folks who move satellites in a hurry still rely on it.
Big Risks, Big Regulations
Every powerful tool comes with a shadow, and methylhydrazine’s is pretty big. The fumes alone force the use of full safety gear. A splash on the skin, or too much in the air, can harm nerves and mess with blood cells, even in small doses. The U.S. Environmental Protection Agency and the Occupational Safety and Health Administration both have rules—strict ones—about workplace exposure.
Back in the day, storage and disposal practices seemed more casual, which led to groundwater pollution. Modern facilities fix that with double-walled tanks, air monitoring, and hazmat training. Scientists researching safer fuels dream of clean launches, but swapping methylhydrazine out means big upgrades and, in most cases, new rocket designs.
Looking Beyond Rockets: Other Uses and the Search for Substitutes
Outside of outer space, methylhydrazine sometimes finds a niche role. A few industrial labs use it for specialty chemicals or pharmaceuticals. It helps create some medicines, pesticides, and polymers—though always under strict conditions. The rules here mean high cost and careful planning.
I remember hearing stories from engineers about the odd tension in these labs. On one hand, methylhydrazine opens doors. On the other, nobody relaxes around it. The suits, smoke hoods, and constant checks eat up time, but every worker I’ve met said the right safety steps make all the difference.
Researchers keep searching for less toxic rocket fuels. Some try “green propellants,” like Hydroxylammonium nitrate-based mixes. Others look at hydrogen peroxide or fully electric propulsion, especially for small satellites. Costs still run high, and proven reliability matters most for missions with lives or billions of dollars riding on top.
The Takeaway: Respect and Responsibility
Methylhydrazine pushes technology to new places, but always with strings attached. Every splash, every breath, every leak needs full attention. As old systems age out and new fuels qualify for launchpads, society can’t afford shortcuts—experience shows that mistakes with methylhydrazine cost more than anyone bargained. Responsible use, solid regulation, and honest conversations—those keep both workers and the world a lot safer.
What are the safety precautions when handling methylhydrazine?
Understanding the Risks
Methylhydrazine plays an important role in rocket fuels and chemical synthesis, but it brings a whole bag of dangers people often overlook outside laboratory walls. A single whiff can burn the nose and throat, and skin contact leads to rashes, burns, or far worse. Long-term exposure links directly to organ damage and cancer. There’s nothing trivial about it, so people dealing with this substance cannot afford any shortcuts.
Protective Gear Is Non-Negotiable
Anyone handling methylhydrazine wears full protective gear, and not just a dust mask and latex gloves. Nitrile or neoprene gloves, splash-proof goggles, and a proper lab coat or chemical suit become the new uniform. Most mistakes happen when workers cut corners or get complacent. Some assume regular gloves will do the trick, until a splash burns right through cheap material. Even a stray drop on the skin can ruin a month.
Ventilation Keeps the Worst At Bay
A cramped or stuffy lab spells trouble with methylhydrazine. Vapors are not only foul-smelling but dangerous in ways you don’t see right away. Fume hoods, not just open windows, need to pull the toxic air away from everyone. I remember one afternoon in a small university lab when a fume hood failed. It didn’t take much before everyone in the room started coughing, and the evacuation not only disrupted research but landed two folks in the medical bay. Good airflow beats a headache and possible hospitalization.
Training Ensures More Than Compliance
Reading safety sheets isn’t enough. Teams run through emergency drills, know where the eye wash and showers sit, and never let students or new hires touch methylhydrazine unsupervised. Once, I watched a colleague freeze after a chemical splash—they forgot what to do mid-panic. After that incident, the lab built more practical drills into the routine, saving time and lives if something goes sideways. People act with muscle memory better than they do with panic.
Spill and Storage Protocols
Methylhydrazine wants to mix with air or water and cause trouble. Storage goes into special containers, clearly labeled, and kept far from acids or oxidizers that can trigger violent reactions. Absorbent spill kits line the shelves near every active workbench. Quick-clean kits absorb spills like cat litter, and everyone in the lab knows to evacuate if a bottle tips. There’s little room for improvisation when seconds matter.
Waste Management: No Half-Measures
Leftover methylhydrazine cycles straight into hazardous waste containers—never poured down any drain. Disposal follows strict rules, usually through licensed chemical waste companies. Years ago, lax habits at a local research center led to contaminated groundwater that affected an entire neighborhood. Once trust gets broken like that, it stays broken for decades. Public health and local environments depend on proper disposal.
Fixing Weak Links
Shortcuts threaten more than individual health; they put entire communities at risk. Regular audits, peer checks, and honest conversations about mistakes build stronger habits than just hanging posters on the wall. Investing in proper training, gear, and facility upgrades proves far cheaper and less painful than cleaning up after disaster. The price of caution with methylhydrazine never outweighs the fallout from carelessness.
What are the health hazards associated with methylhydrazine exposure?
Understanding the Threat Up Close
Walk into any place where rocket fuels get handled, and you’ll hear quiet respect for methylhydrazine. This chemical’s main job is to power things that travel faster than sound, but the risks often fly under the radar for folks not in the industry. Methylhydrazine can do real damage to the body—sometimes fast, sometimes over years. That’s worth talking about, not only to protect workers, but to keep the facts out in the open.
Breathing It In Hurts More Than Many Expect
Most healthy lungs can take a beating, but methylhydrazine puts them to the test. Even a short whiff of its colorless vapor can burn the nose and lungs, leaving a cough that won’t quit. Go a little longer, and folks can get headaches, nausea, or even lose their sense of balance. Gases like this slip past normal workplace defenses; a regular face mask from a hardware store just doesn’t cut it. Sometimes the warning signs show up late, which makes the risk harder to spot.
Skin and Eyes Take the Next Hit
Even with careful habits, splashes happen. Methylhydrazine eats through gloves that work fine with other fuels. Every drop stings, then soaks into the skin, setting off rashes or blisters. Getting it in the eyes causes sharp pain and can harm vision permanently if someone waits too long for medical help. This makes no room for shortcuts—basic safety gear sometimes just isn’t enough.
Liver and Kidneys Pay a Long-Term Price
Working around methylhydrazine day after day, I saw regular guys with no health problems lose ground. Blood tests sometimes flagged high levels of liver enzymes, and some people caught the news too late. Extended exposure can scar the liver and stress the kidneys. Even a single large exposure leads to organ damage, sometimes impossible to reverse. I always believed safety sheets, but listening to coworkers’ stories about just one bad spill sticks with you longer than lab numbers.
Cancer Risks That Don’t Get Enough Attention
Researchers link methylhydrazine to higher cancer rates in animals. The Environmental Protection Agency calls it a probable human carcinogen. That alone ought to push stricter oversight, but enforcement varies. Some factories rely on old gear and routines, risking future health for today’s productivity.
Pushing for Better Protections
Companies can’t control everything, but the right approach closes gaps that accidents slip through. Ditching old-fashioned gear and upgrading to chemical-resistant suits makes a difference. So does proper ventilation—pipes and fans that move fast, with alarms tuned for this specific chemical. Real training matters. A single afternoon of drills where everyone practices spills and decontamination built muscle memory better than any handbook I’ve ever read. Regular blood checks, even for temporary staff, catch trouble before it spirals.
Right to Know, Right to Safety
It comes down to transparency. Workers deserve the same clear warnings about methylhydrazine that anyone gets with cigarette packs or power tools. Anything less invites accidents or hidden costs paid in days off and long-term disability. My experience says real safety grows from honest talk, new investment in controls, and respect for the risks—especially with chemicals like this. The price for shortcuts can be years off your life.
How should methylhydrazine be stored and disposed of?
The Risk Modern Labs Can't Ignore
Methylhydrazine is no household name, but in labs and aerospace work, it's a real problem child. I’ve spent enough time around chemical storage rooms to know that cutting corners with something like this makes headlines for all the wrong reasons. Highly toxic, flammable, and cancer-causing, it doesn’t forgive mistakes. Breathing in a whiff or getting it on your skin sends you straight to the hospital. Occasionally, you’ll hear someone brush off the hazards, until an accident clears out the building and leaves people on edge for weeks. There’s no shaking off that kind of scare.
Common Sense Storage: Away from Trouble
Let’s get into the nitty-gritty. Experience shows: keep methylhydrazine far away from everything flammable or reactive. I’ve watched lab techs tuck bottles of this stuff behind thick steel doors, inside explosion-proof cabinets, and set alarms not just for theft, but temperature spikes. You can’t just toss it on a shelf. Polyethylene containers, double-sealed, catch spills before they become disasters. It stays cool and dry, far from direct sunlight. Nothing rests near oxidizers—those two together have made headlines as the culprit behind fires and worse.
Anyone walking into a room holding methylhydrazine wears more than just a sense of caution. Face shields, gloves, and chemical-resistant aprons become routine. Even boots matter—regular sneakers don’t cut it. Every time gloves come off, a wash-up at the emergency station happens, no shortcuts. The day someone tries to take a quick break without changing, the reminders start flying; some rules exist for a reason.
Disposal: No DIY Approaches
Getting rid of it safely calls for more than a green bin and a prayer. I remember a facility that tried neutralizing waste in-house—nobody slept easy until the experts took over. Certified hazardous waste contractors—people who train for emergencies—step in. They pack everything into properly labeled drums. All movement, from the fume hood to a sealed container, happens under ventilation, with a spill kit ready just in case.
Regulations don’t just fill a manual for fun. In the US, the EPA classifies methylhydrazine as an acute hazardous waste (U.S. EPA Hazardous Waste Number: P067). This means strict protocols for handling and transporting it. Burning it off or dumping it down the drain looks like a solution until you think about contaminated water supplies or explosions in a landfill. Local law always trumps convenience. Disposal companies know how to neutralize the material or incinerate it in approved, high-temperature facilities where there’s no chance of it sneaking back into the ecosystem.
Why Training and Vigilance Make a Difference
Every bad story about methylhydrazine traces back to someone skipping procedure or thinking it couldn’t happen to them. I’ve learned more from safety drills and close calls than from manuals. Good labs run safety refreshers so nobody forgets the basics. They keep up-to-date with the latest research—like NIOSH’s constant reminders about exposure limits and new decontamination technology. Supervisors check logs and label dates, and no one pretends a shortcut will save time.
Communities near labs and industrial sites have a right to know what chemicals move through their neighborhoods. Transparency, regular audits, and whistleblower hotlines stop small errors before they hit the news. Real safety doesn’t rest only on locking chemicals away—it depends on every person respecting the threat and following through on the hard steps, every single time.
What is the chemical formula and structure of methylhydrazine?
What’s in a Name? Methylhydrazine at Its Core
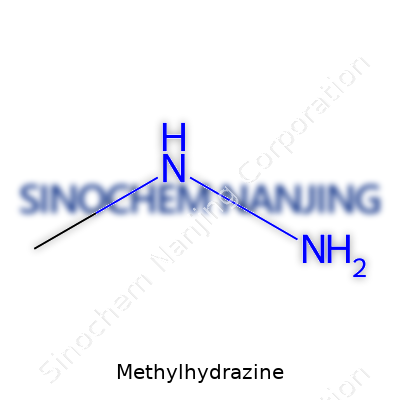
I still remember my first encounter with methylhydrazine from my early chemistry days. The classroom felt small, chemicals in heavy glass bottles at every corner. The formula stuck with me: CH3NHNH2. Short and punchy, but the story behind it runs deep. Methylhydrazine is built from a methyl group (that’s the simple CH3), attached to a pair of nitrogen atoms—one linking to the methyl, the other left with two hydrogen neighbors. It looks pretty unassuming on paper. Yet, it’s anything but dull.
Why the Structure Packs a Punch
The structure of methylhydrazine tells you a lot about why folks in science and industry watch it closely. That chain runs CH3-NH-NH2. Most folks with lab experience know the importance of those nitrogen bonds. They make methylhydrazine both a useful and risky player. The layout helps it mix quickly with water and fuels. It’s got that lone methyl wired to a reactive hydrazine backbone. You give something like that a spark, and it won’t just sit quietly. That’s why rocket scientists line up for it. The structure gives methylhydrazine more energy when it reacts, so it outpaces regular fuels in some rocket boosters.
Risks and Responsible Handling
In the lab, handling methylhydrazine always meant checking gloves, fume hood, goggles, and even double-checking the label. The chemical is dangerous—it’s not something to take lightly. The clear liquid brings along volatility and toxicity. Anyone spending much time with it knows how quickly vapors can sting the nose and throat. It’s linked to cancer. In my own training, we always treated spills like tiny emergencies. Data backs that careful approach. The US Environmental Protection Agency notes risks ranging from acute exposure sickness to longer-term effects on the liver and kidneys. The World Health Organization backs those concerns, pushing for strict standards around use and storage.
Why We Still Use Methylhydrazine
Methylhydrazine sits at the crossroad between danger and utility. Satellite launches and space missions depend on it for fast ignition. Plenty of big-name rockets and defense projects keep it on hand. Chemists value it in the lab for fuel research or making certain drugs—mostly for special circumstances, since alternatives are usually safer. But its reliable energy means it’s hard to replace out there in the aerospace world. During my time at a university research center, I asked one engineer why nobody switches. He shrugged and said, “There’s no other fuel that checks as many boxes for engines that need to work every time.”
Safer Approaches Waiting in the Wings
The risks of methylhydrazine have encouraged plenty of smart folks to look for better options. Green technologies and biofuels get more headlines these days. Some companies tinker with less toxic fuel blends. There’s hope for a shift, but anyone tracking progress knows the gap is wide between lab experiments and trusted rocket launches. For most labs, the push is for tighter controls, stronger training, and air monitoring—basic steps that keep people safe, even as research looks for cleaner solutions. From what I’ve seen, every safety briefing or chemical inventory sheds light on the real trade-offs that come with methylhydrazine’s powerful chemistry.
| Names | |
| Preferred IUPAC name | Methyldiazane |
| Other names |
Monomethylhydrazine MMH N-Methylhydrazine |
| Pronunciation | /ˌmɛθ.ɪl.haɪˈdreɪ.ziːn/ |
| Identifiers | |
| CAS Number | 60-34-4 |
| Beilstein Reference | 3539782 |
| ChEBI | CHEBI:16181 |
| ChEMBL | CHEMBL14225 |
| ChemSpider | 54654 |
| DrugBank | DB01765 |
| ECHA InfoCard | String: 100.003.526 |
| EC Number | 200-471-4 |
| Gmelin Reference | 7857 |
| KEGG | C01745 |
| MeSH | D008776 |
| PubChem CID | 7907 |
| RTECS number | PV5425000 |
| UNII | 02F423UN8P |
| UN number | UN1244 |
| Properties | |
| Chemical formula | CH6N2 |
| Molar mass | 46.07 g/mol |
| Appearance | Colorless liquid |
| Odor | Ammonia-like |
| Density | 0.867 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | -0.38 |
| Vapor pressure | 17.7 kPa (20 °C) |
| Acidity (pKa) | 23.3 |
| Basicity (pKb) | 11.10 |
| Magnetic susceptibility (χ) | -34.5e-6 cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 0.792 cP (25°C) |
| Dipole moment | 1.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | +66 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -338 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V09AX04 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H341, H350, H373, H411, H226 |
| Precautionary statements | P210, P233, P260, P264, P270, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P320, P330, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Autoignition temperature | 300 °C |
| Explosive limits | 4.7% - 100% |
| Lethal dose or concentration | LD50 oral rat 60 mg/kg |
| LD50 (median dose) | LD50 (median dose): 60 mg/kg (oral, rat) |
| NIOSH | RT6800000 |
| PEL (Permissible) | PEL = 0.35 mg/m3 |
| REL (Recommended) | 250 mL |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Hydrazine 1,1-Dimethylhydrazine Monomethylhydrazine Phenylhydrazine Methylhydrazinium |

