Methyl Salicylate: Chemistry, History, and Modern Perspectives
Tracing the Roots: Historical Development
Methyl salicylate has a history that overlaps with folk medicine, war, and industry. Long before the pure compound showed up in laboratories, indigenous communities in North America turned to the bark of sweet birch and wintergreen leaves for pain relief. Chewing on those leaves or brewing them into tea worked as a low-tech answer for muscle aches and fevers. Later, European settlers in the Americas caught on. The real breakthrough kicked in the 1800s when chemists isolated methyl salicylate in the lab. In 1848, it took on a new life when commercial synthesis allowed more consistent and affordable production. This one compound moved fast—from folk tradition to mass manufacturing of balms, analgesics, and even flavoring in candy.
Checking Out the Compound: What Makes Methyl Salicylate Stand Out
If you have ever opened a jar of muscle rub or let a breath mint dissolve on your tongue, you know methyl salicylate by its strong, sweet, minty smell. In pure form, it comes as a colorless or pale-yellow liquid. The stuff locks in a sweet, spicy punch you won't mistake for anything else. Someone in a pharmacy or chemistry lab might call it oil of wintergreen, and it carries its signature odor even in trace amounts. The boiling point sits at around 222°C, and it does not dissolve in water very well, but it mixes easily with alcohol and most organic solvents. Its density is higher than that of water. In my own experience handling it, just a few drops can scent a workspace or an entire laboratory—too much exposure can lead to headaches or nausea. Methyl salicylate sometimes surprises people with its intensity.
Technical Specifications & Labeling Practices
Anyone working directly with methyl salicylate trusts technical data to do their job right. It generally appears in concentrations above 98 percent for industrial use. Most commercial pipettes and measuring gear have no problem handling this liquid, but anyone labeling methyl salicylate must clearly note its potential for toxicity, especially around children or pets. According to various regulatory guidelines, you cannot skip hazard notations or required handling symbols. Whether it enters a pharmaceutical blend or an industrial product, packaging must shield users from direct skin contact and accidental ingestion. These standards serve real people, not just rulebooks: hospitals keep poison control numbers handy, and parents lock up anything with methyl salicylate for good reason.
Preparation and Chemical Synthesis
Methyl salicylate forms through a reaction of salicylic acid with methanol, usually in the presence of an acid catalyst—a straightforward esterification. In practice, large-scale manufacturers mix these components, apply heat, and pull off the resulting methyl salicylate by distillation. In the old days, people relied on steam distillation from wintergreen leaves, a much slower process requiring lots of plant material. As synthesis improved, the world saw lower prices and greater accessibility, but less dependence on harvesting wild plants. Every industrial-scale outcome starts with a remarkably simple reaction: one acid, one alcohol, one acid catalyst, and a chemist who respects the hazards involved.
Reactions, Modifications, and Laboratory Life
Methyl salicylate does not just stand alone; it can act as a starting material for a range of chemical transformations. You can hydrolyze it back to salicylic acid, which itself acts as a base for further pharmaceutical synthesis (even aspiring to aspirin). Chemists have modified methyl salicylate for creating more complex esters or even tailoring its scent for the fragrance industry. In the lab, it reacts readily under both acidic and basic conditions, and most undergraduate chemists remember learning esterification as one of their first real “hands-on” synthetic reactions. While its scent can delight, its strong reactivity demands respect—a spill becomes not just a cleanup task but a health hazard.
Names in Circulation: Synonyms and Product Recognition
Whether someone calls it oil of wintergreen, Betula oil, or Gaultheria oil, the underlying molecule remains the same. Pharmacies, supermarkets, and chemical suppliers stock this compound under various brand names, but the scientific community knows it by its chemical identity: methyl 2-hydroxybenzoate. A shift from plant extract to synthetic ingredient did not blot out those old names. In conversation, technical and folk names end up mixed together. This can create confusion, so safety data sheets, labeling, and regulations remind everyone to focus on the precise, chemical version—one that carries risks alongside benefits.
Staying Safe: Standards for Handling and Use
Every interaction with methyl salicylate should begin with caution. Skin exposure may lead to irritation and, in high doses, absorption through the skin can result in systemic toxicity. Children face particular risk, as even a teaspoon’s worth can turn deadly. In laboratories, gloves, goggles, and fume hoods rank as standard operating equipment. On a larger scale, producers build robust spill-prevention and ventilation into every factory that handles this compound. Methyl salicylate nearly always requires clear, visible warnings to prevent accidental overuse. Product formulations in sports ointments or liniments stick to low, controlled concentrations, because those doses still deliver a therapeutic punch without crossing into toxic territory.
Where It Lands: Application and Real-World Uses
Every training room from high school gyms to professional sports clubs stocks a muscle rub that lists methyl salicylate as an active ingredient. It’s more than just a numbing agent—methyl salicylate triggers a sensation that feels hot, then cool, distracting the brain from deeper musculoskeletal pain. Beyond sports, dental hygienists reach for it to add flavor to mouthwashes and toothpaste, balancing its strong character with milder mints. Food scientists add tiny amounts for signature flavors in chewing gum and confectionery. The fragrance industry celebrates its brightness and boldness in liniments and perfumes. Around the world, methyl salicylate moves from pain relief to flavor enhancement to aroma design, bridging traditional practice with modern chemistry.
Digging Deeper: Research and Ongoing Development
Even after centuries of use, scientists dig deeper into how methyl salicylate works. Research into pain mechanisms explores its function as a counterirritant—stimulating nerve endings to fool the body into ignoring original pain signals. Others probe its role as a biomarker in plant communications and investigate its effects at the molecular level. Pharmacies and sports medicine clinics track potential allergic reactions and investigate slow-release formulations, always chasing more controlled, effective healing with reduced risk. Environmental scientists study its breakdown in water and soil, seeking ways to minimize residues in the food chain. The research never stops, and neither do the questions about maximizing benefits while limiting harm.
Weighing the Risks: Toxicity and Health Considerations
Toxicologists spend a lot of time with methyl salicylate, measuring how its medicinal power can cross into danger. Human accidents shape the scientific literature: accidental ingestion by children leads to rapid onset of salicylate poisoning, with symptoms ranging from tinnitus and confusion to seizures and even death when doses climb too high. Seasonal spikes in poison center calls coincide with widespread use of topical ointments. Animal studies draw boundaries for acceptable exposure limits, and workplace regulations strictly define time-weighted averages so workers avoid chronic toxicity. The line between relief and risk stays narrow, and clinicians urge families to lock up every bottle, even ones with child-resistant lids.
Looking Ahead: Future Prospects
There’s always another angle to explore with methyl salicylate. Advances in encapsulation could yield creams and patches that release active molecules slowly and safely, reducing overdose risk. Consumer education grows ever more important in an era of online sales and home remedies. Synthetic biology opens the door to yeast or bacteria engineered to produce methyl salicylate sustainably, bypassing petrochemical inputs and returning to the scent’s plant-based roots—without overharvesting wild wintergreen or birch. Regulatory agencies continue to weigh new findings, updating permissible uses in food, pharmaceuticals, and cosmetics, all while public health experts measure real-world impacts. The common thread runs clear: methyl salicylate won’t fade from science, industry, or medicine any time soon, and the work to balance its vivid benefits against sharp risks remains as pressing as ever.
What is Methyl Salicylate used for?
Found All Around Us
Growing up, I always recognized the sharp wintergreen scent as something tied to my grandfather’s aching knees. Later, I figured out that scent came from methyl salicylate, known for more than just nostalgia. It’s a compound that shows up everywhere: in pain relief creams, liniments, even in the flavor of some chewing gum. Most people use it and never know it by name.
Pain Relief with a Familiar Scent
Methyl salicylate first found its place in sports medicine cabinets. Rubbing one of those topical creams into sore muscles feels like a rite of passage for anyone who’s pulled something during a workout. The cooling, tingling sensation that follows helps distract from aches and increases blood flow under the skin. It’s effective for short-term relief. Its function is backed by research that describes how it penetrates below the surface, heating skin tissue just enough to convince the brain to pay less attention to deeper muscle discomfort. That twist of science and sensation makes it a mainstay ingredient in products like Icy Hot and Bengay.
Uses That Go Beyond Aches and Pains
Not just for joints and muscles, methyl salicylate pops up in other places. Dentists and dental hygienists sometimes use it in mouthwashes and toothpastes for a fresh flavor. Because it comes from wintergreen plants, it brings a punchy mint-like taste. Some candies lean on it for the same reason. Though, it’s always used in low concentrations. Consuming too much carries risks, so regulatory bodies keep a close eye on how companies put it into foods and oral care products.
Benefits and Dangers Neatly Tied Together
What makes methyl salicylate helpful can also make it dangerous. A small dab eases body aches, but overuse or ingestion has sent people, especially children, to emergency rooms. Reports show that a single teaspoon, if swallowed by a young child, can be deadly. People need better education about keeping these products out of reach and paying attention to how much they use, especially with medicated balms.
Industry and Home Need Smarter Practices
Manufacturers already add childproof packaging to high-concentration medicines, but education always lags behind. Pharmacists, doctors, and even sports coaches play key roles in sharing practical safety advice: use sparingly, don’t apply on broken skin, wash hands after rubbing it in. At home, folks should double-check for curious toddlers or pets. Companies can invest in clearer labeling that highlights dosage and risk without burying it in tiny print.
Seeking Balance
Methyl salicylate walks a fine line. As a muscle pain reliever, it offers comfort to people of all ages. Its flavor brightens up breath and candy. But science reminds us that it needs to be used wisely. A bit of awareness, plus a dose of careful use at home and in industry, keeps this common ingredient working for us rather than against us.
Is Methyl Salicylate safe for children?
Understanding What We’re Talking About
Methyl salicylate shows up in a lot of places: topical pain relief creams, muscle rubs, even in some mouthwashes and flavoring agents. Many parents reach for those mentholated ointments to help soothe everything from bruises to stuffy noses. It’s not unusual to see these “minty” balms and liquids sitting right next to the baby wipes in family bathrooms. The cooling effect feels good and the smell is familiar. That makes it easy to assume these products are just as safe for children as they are for adults. Unfortunately, that’s not always the case.
Behind the Cooling Sensation
As someone who’s rubbed an aching knee with muscle cream after a weekend soccer game, the relief feels almost immediate. What many don’t realize is that methyl salicylate is a chemical cousin of aspirin. The skin absorbs it and, in small doses on tough adult skin, the risk tends to stay low. Children, though, aren’t just smaller in size — their bodies handle chemicals differently. Their skin is thinner and absorbs substances faster. That puts them at risk for accidental poisoning.
Why the Worry?
Let’s get real: the problem isn’t a dab on the shoulder for a teenager’s sprain. The problem creeps in when gobbed on thick or applied over broken skin, especially on babies or toddlers. Ingesting even a teaspoon of methyl salicylate can seriously harm a small child. Symptoms of poisoning show up fast: nausea, confusion, rapid breathing. If left untreated, the outcome could turn life-threatening. The American Association of Poison Control Centers has tracked many cases where over-the-counter liniments or menthol balms sent children to the emergency room.
What Studies and Experts Say
Doctors see lots of families using these products without reading the labels. Pediatricians often warn that balms and ointments marketed over the counter might contain levels of methyl salicylate high enough to trigger toxicity in children. Research published in Pediatrics shows that the number of poisonings related to methyl salicylate outweighs what parents realize, mainly because these products are readily available and often smell nice, which invites accidental ingestion.
Practical Advice for Parents
Locking up the medicine cabinet rarely feels urgent until after an accident. As a parent, it’s easy to think, “my kid would never eat this,” but the reality gets messy. Flavored toothpaste and pain rubs can attract curious hands. Always check ingredient lists for methyl salicylate or wintergreen oil. If a product says “keep out of reach of children,” pay attention. Don’t put pain-relief creams intended for adults on kids younger than 12 unless directed by a doctor. If in doubt, call the local poison control center.
Looking Ahead: Solutions and Safer Choices
One of the simplest ways to avoid problems is switching to alternatives that don’t rely on methyl salicylate, especially for younger children. Heat packs, cold compresses, and distraction work for many minor aches. Companies can help by using clearer warnings and child-resistant caps. Health authorities in some places already restrict the sale of products with high concentrations of this chemical for good reason.
Methyl salicylate does a job, but it’s not always the right tool for kids. Real safety happens when people pay attention to what goes on young skin and what gets stored on easy-to-reach shelves.
Can I use Methyl Salicylate during pregnancy or breastfeeding?
A Familiar Pain Relief Route
Walking down a drugstore aisle, you spot a range of creams and patches promising muscle relief. Look closer and Methyl Salicylate often pops out—a minty chemical found in topical products like Bengay and Icy Hot. I remember using it after a pulled back muscle, reaching for that fast relief. Rubbing it on, those cooling and warming sensations kick in where it hurts.
Breaking Down the Risk
Pregnancy changes your world—including how your body handles medicine. Methyl Salicylate, cousin to aspirin, absorbs right through your skin, especially if you slather on more than directed or use a heating pad to boost its effects. Some folks assume it’s safe since it’s over-the-counter, but your body doesn’t see it that way. It still enters the bloodstream, and from there, nothing stands between that ingredient and a growing baby.
Medical research links too much salicylate exposure in late pregnancy to bleeding and rare but serious fetal problems like low birth weight or heart issues. One 2016 review in Obstetrics & Gynecology pointed to increased risks with prolonged or heavy topical salicylate use. Large doses, especially swallowed by accident, cause ringing in the ears, nausea, or worse. That’s not a headache anyone wants—especially during pregnancy.
Common Sense over Fear
You might ask, “Is a dab for an aching knee so risky?” Small, one-time use might slide under the radar, but pregnancy doesn’t play by neat rules. Bodies change, skin absorbs more, blood volume jumps. The patch or cream that felt mild suddenly enters gray territory. Any product easy to grab and rub on may trick people into thinking it’s always harmless, but the data simply never guarantees it’s risk-free for pregnant folks.
During breastfeeding, the same story applies, just with a new concern: medicine shows up in breast milk. Methyl Salicylate’s levels in milk are much lower than with pills, but even a trace can bother a tiny baby. Pediatricians warn against direct application on or near breasts, since a baby’s mouth could reach the cream. Poison control centers log scary calls every year from accidental exposures.
Alternatives and Real Relief
Sore backs and joints don’t magically disappear in pregnancy or while feeding a newborn. The key: safer tactics work, too. Physical therapy, gentle exercise, and hot compresses without medication bring legitimate pain relief. Acetaminophen remains the main go-to for pain during pregnancy, so most doctors stick with its safety profile. Always chat with your provider about what hurts before reaching for any product, no matter how familiar it seems.
A patch or cream might promise quick relief, but your growing baby and your own changing body deserve a second thought. FDA guidance and trusted medical groups lean on caution—better to play it safe, ditching risky topicals unless a doctor directly signs off. If relief feels out of reach, bring concerns straight to those who watch over your care. No one benefits from avoidable risk, least of all the new life you’re nurturing.
Are there any side effects of using Methyl Salicylate?
Understanding the Product
Methyl salicylate pops up often in pain relief creams and muscle rubs. Athletes lean on it for sore muscles. Weekend gardeners rub it on their backs. This substance works a bit like aspirin, offering a break from aches by cooling or warming the skin. For many, it’s a go-to for minor injuries, stiff joints, and muscle strains. It smells strong—minty, sharp—and you can spot it a mile away when someone opens a tube.
Potential for Side Effects
It’s easy to forget that products on store shelves can cause problems. I’ve seen people use it more than labels recommend, hoping for faster relief. Pretty soon, their skin turns red or blisters. Overuse does that. Too much can make you feel dizzy or short of breath. Some users even report ringing in their ears. In rare cases, too much absorption through skin can lead to poisoning, especially for children. Keep it far away from young kids. The product isn’t meant to go near mucous membranes or broken skin—getting it in the eyes stings, and applying it to wounds burns horribly.
Who Should Be Extra Careful?
Anyone with aspirin allergies should steer clear. Methyl salicylate shares a chemical family with aspirin, so the risk isn’t worth taking. People with asthma face increased danger; even skin exposure could lead to an attack. The risks climb if someone uses blood thinners, since methyl salicylate can boost bleeding chances. I had a neighbor with thin blood who tried an over-the-counter rub and ended up with bad bruising from a tiny bump.
Product Label Clarity and Real-World Use
Labels list warnings, but many folks skip fine print. Maybe bad eyesight makes reading difficult, or the print seems unimportant. Companies could make the warnings bigger, bolder. Pharmacists should remind buyers to wash hands thoroughly after application—a habit some ignore. Rubbing eyes after use leads to burning and tearing up, which is easy to avoid with a simple hand wash.
Another thing: mixing methyl salicylate with a heating pad or wrapping the area tightly can send too much through the skin all at once. I’ve seen cases where people started sweating under a patch, not realizing this sped up absorption. Medical journals back this up—more heat or occlusion means a greater chance for side effects.
Safer Ways to Find Relief
Stretching before activity, regular movement, and knowing when to rest make a big difference with aches. Before reaching for heavy scent creams, giving ice or heat therapy a try can ease soreness. If a topical pain-reliever becomes a daily habit, it’s worth speaking with a healthcare provider about what’s really causing the pain.
For those who decide to use methyl salicylate products, moderation is key. Follow package instructions to the letter, always keep it out of kids’ reach, and store it where no one could mistake it for toothpaste or lotion. If there’s a new medication in the picture, even something as simple as a baby aspirin, double-check for safety with a professional.
Honest Look at Benefits and Risks
I’ve seen relief on the faces of people after a careful, light application. These products make a real difference for some, but they aren’t risk-free. Using them with respect and awareness keeps minor aches and pains from turning into something much bigger. In the end, clear communication—between doctors, pharmacists, and patients—matters as much as what’s inside the tube.
How should Methyl Salicylate be applied or used?
Understanding Methyl Salicylate and Its Purpose
Methyl salicylate, known by many as wintergreen oil, shows up in a lot of pain-relief creams and ointments you find at the drugstore. It’s one of those ingredients that offers a quick feeling of cool and warmth on sore muscles, making sports creams and patches really effective for post-workout stiffness. Rather than dealing with a whole bottle of pills, a topical application gets straight to the source of discomfort.
Personal Perspective: Appreciating Topical Relief
After spending plenty of late nights typing away at a laptop or coming home tired from a day of gardening, muscle tension tends to make itself known. Rubbing a mentholated cream onto tight shoulders offers comfort that oral medication usually can’t. The aroma of wintergreen becomes familiar, a signal that relief is around the corner. Without proper guidance, though, people tend to slap on too much, thinking more product means faster relief. That’s not how this chemical works.
Applying Methyl Salicylate the Right Way
Careful use plays a big part in both safety and effectiveness. Skin reacts quickly to overuse; more isn’t always better. I learned from a friend's experience—the result was a fiery red rash, not extra relief. Manufacturers usually advise a thin layer, massaged gently onto the affected area no more than three or four times each day. Product labeling lists all key instructions. Those small print warnings point out that covering treated skin with tight bandages or applying heat can actually cause serious skin reactions or enhance absorption too much, putting strain on the body.
Trying to treat pain on damaged or broken skin doesn’t make things heal faster; it usually leads to a burning feeling and irritation. The cream should only touch unbroken skin. Allergy concerns also come up: methyl salicylate belongs to the salicylate family, so anyone sensitive to aspirin ought to steer clear or talk to their doctor first. Even casual sharing between family or teammates isn't smart for those with allergies or chronic skin conditions.
Kids and Older Adults: Special Considerations
Products containing methyl salicylate often aren’t a good fit for young children. Accidental poisoning can happen because kids experience the effects much more strongly. Older adults sometimes develop thinner, more sensitive skin, increasing risk for irritation. Checking with a healthcare provider remains a smart approach for anyone with questions.
Supporting Safe Use Through Clear Instructions
Marketers and healthcare professionals have a responsibility to share plain-language instructions. Some companies already feature step-by-step diagrams and bolded warnings on packaging. Retail pharmacists often answer questions about how much to use and when to call a doctor. A few states have started efforts on medication counseling, aiming to reduce mistakes not just for prescription drugs, but for common store-bought creams as well.
Finding Relief and Respecting Limits
Managing pain or stiffness with topical products gives people more control over their day. Setting realistic expectations and sticking with labeled instructions helps keep methyl salicylate both effective and safe. From time spent outdoors to hours behind a desk, those small tubes can make a big difference—if we use them with care and respect their limits.
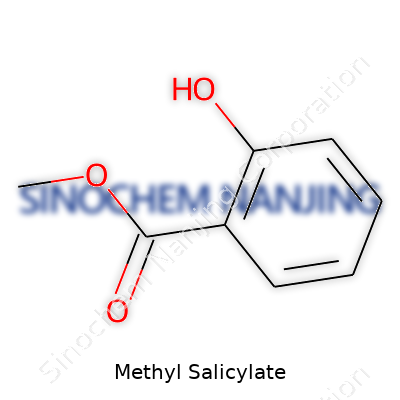
| Names | |
| Preferred IUPAC name | methyl 2-hydroxybenzoate |
| Other names |
Wintergreen oil Betula oil Gaultheria oil Methyl 2-hydroxybenzoate |
| Pronunciation | /ˈmɛθ.ɪl səˈlɪs.ɪ.leɪt/ |
| Identifiers | |
| CAS Number | 119-36-8 |
| 3D model (JSmol) | ``` CCC(=O)Oc1ccccc1C ``` |
| Beilstein Reference | 1207806 |
| ChEBI | CHEBI:28763 |
| ChEMBL | CHEMBL1409 |
| ChemSpider | 5098 |
| DrugBank | DB00544 |
| ECHA InfoCard | 100.003.262 |
| EC Number | 204-317-7 |
| Gmelin Reference | 7261 |
| KEGG | C01381 |
| MeSH | D008772 |
| PubChem CID | 4133 |
| RTECS number | VN8575000 |
| UNII | Y2PTXG8B98 |
| UN number | UN1230 |
| Properties | |
| Chemical formula | C8H8O3 |
| Molar mass | 152.15 g/mol |
| Appearance | Clear, colorless to pale yellow liquid with characteristic odor. |
| Odor | Sweet, wintergreen-like |
| Density | 1.17 g/cm³ |
| Solubility in water | 1.85 g/L (20 °C) |
| log P | 2.55 |
| Vapor pressure | 0.034 hPa (25 °C) |
| Acidity (pKa) | 8.2 |
| Basicity (pKb) | 13.0 |
| Magnetic susceptibility (χ) | -8.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.538 |
| Viscosity | 3.12 mPa·s (20°C) |
| Dipole moment | 2.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -452.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3735 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N02BA03 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H317 |
| Precautionary statements | P210, P261, P264, P270, P272, P273, P280, P301+P312, P302+P352, P305+P351+P338, P314, P321, P330, P332+P313, P337+P313, P362+P364, P391, P403+P233, P501 |
| Flash point | 96 °C |
| Autoignition temperature | 450 °C (842 °F; 723 K) |
| Explosive limits | Explosive limits: 1.1% - 8.3% |
| Lethal dose or concentration | LD50 oral rat 887 mg/kg |
| LD50 (median dose) | LD50 (median dose): 887 mg/kg (oral, rat) |
| NIOSH | NN3325000 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | 0.10 mg/m³ |
| IDLH (Immediate danger) | 3,500 mg/m³ |
| Related compounds | |
| Related compounds |
Salicylic acid Aspirin Choline salicylate Sodium salicylate |

