Methyl Propionate: Substance Beyond the Lab
Historical Development
Methyl propionate’s legacy reaches back over a century, into an era of new chemical discovery when chemists figured out how to create esters by simple acid-catalyzed condensation. Early references to methyl propionate go hand in hand with the search for alternatives to natural sources of fruity flavors and solvents. People in the early 1900s recognized that a clear, sweet-smelling liquid could stand in for more expensive extracts from fruit or fermentation. Chemical innovation wasn’t only about cost-saving; World War economies pushed this ester into the hands of factory operators hunting for useful, volatile solvents and flavor enhancers. Over time, its gentle odor, efficient solvency, and manageable toxicity made methyl propionate a quiet staple in small-batch and industrial production. Nowadays, it doesn’t attract headlines, but it supports manufactures and researchers who quietly rely on its repeatable properties.
Product Overview
Methyl propionate keeps a low profile. It works in the background, a supporting actor in paints, coatings, artificial flavors, and specialty synthesis. Its colorless nature and rapidly evaporating character help chemists strip unwanted residues or transport tiny molecules into larger solutions. The good part? It brings a fruity, slightly pungent aroma that triggers instant association with green apples or pineapples, even for someone only familiar by scent. In the flavor world, tiny drops pack a punch in candies or sodas, and perfumers often treat it as a fixative or top note. Some household products depend on its solvency and mild evaporative cooling.
Physical & Chemical Properties
This ester feels light, almost volatile, with a boiling point near 80 degrees Celsius. Its low viscosity and high vapor pressure mean it escapes quickly into the air, so open bottles never last. Water barely mixes with it, but methyl propionate dissolves well in alcohols and many organic solvents. The sweet note appears powerful but never cloying. At the same time, the compound’s chemical stability resists unwanted changes under normal handling, yet it reacts cleanly under controlled conditions. Flammability remains a key consideration, with vapors catching fire easily—a reminder that useful organics often come with a whiff of risk.
Technical Specifications & Labeling
Regulators insist on clear labeling, hazard warnings, and storage under proper ventilation. In practical terms, these requirements mean drums and bottles marked boldly with flammability icons and minimal residue in transfer lines. Purity typically exceeds 99%, with water and acid traces kept under strict control. Some industrial users request analysis for trace metals or unwanted aldehydes, protecting downstream reactions. Documentation includes proper CAS numbers and hazard codes, but most operators learn to respect the strong scent and rapid evaporation after one shift in a flavoring plant or paint shop.
Preparation Method
Laboratories and factories both reach methyl propionate through classic esterification—propanic acid and methanol, with acid catalysis to push the balance. Factories optimize reaction temperatures and distillation runs to boost yield, then scrub away acid leftovers and dry the final liquid. Alternative routes pop up in the literature, usually based on modern green chemistry with solid acid catalysts or water recovery. Scale means everything—a flask on a hotplate for teaching or a multi-tonne reactor for the daily output of a solvent supplier. Wherever the preparation happens, handling waste and neutralizing acid matter as much as the main reaction.
Chemical Reactions & Modifications
Methyl propionate’s structure opens up possibilities, especially for anyone interested in chain extension, hydrolysis, or transesterification. Hydrolysis splits it back into methanol and propionic acid—useful in some cleaning or recovery processes. In the laboratory, chemists sometimes grab the ester as a stepping stone toward more functionalized molecules, pulling apart the methyl or propionyl units and recombining with larger groups for plastics or pharmaceuticals. The ester group plays a role in tailoring molecule shapes for drugs, solvents, or additives. In recent years, interest in greener transformations has encouraged teams to test newer catalysts or milder conditions so reactions run cleaner and at lower temperature.
Synonyms & Product Names
Most people call it methyl propionate, though labels mention propionic acid methyl ester or methyl propanoate, depending on country or convention. Old chemistry books sometimes reference methyl ester of propionic acid. Many suppliers sell it under common, trade-neutral names, avoiding glitzy branding or unnecessary abbreviations. To the regular user, the smell and behavior speak more reliably than a legal synonym.
Safety & Operational Standards
Handling comes down to respect. Operators rely on good ventilation and grounding to reduce fire hazards. Open flames and static sparks pose clear risks, so process engineers recommend explosion-proof equipment, proper grounding, and fume scrubbers. Small exposures—vapors near the nose, splash on the skin—rarely cause harm, but chronic exposure deserves protective gloves and goggles. Workplaces monitor vapor concentrations with handheld detectors, and rules require closed transfer systems for bulk loading and unloading. Because the ester vapor travels quickly and hangs low, spill response teams know to use diking and forced ventilation. The chemical’s irritation potential feels mild, but the safety culture prioritizes prevention.
Application Area
Real-world uses stretch from artificial flavors to specialty coatings. In food and beverage applications, methyl propionate backs up fruity blends, especially where manufacturers want a cost-effective, shelf-stable alternative to natural extracts. Painters and industrial sprayers know it as a medium that flashes off quickly, leaving behind resins or pigments. Electronics industries test it for cleaning or as a solvent in specialty blends. Chemists, particularly in research and pharmaceuticals, treat its ester group as a platform, forging new molecules for trials in everything from veterinary drugs to flavor chemicals. In each case, its light scent and volatility add utility above and beyond pure solvency or reactivity.
Research & Development
Lately, R&D teams focus on greener routes and better containment. As climate and worker safety regulations tighten, pressure builds to find catalysts that cut acid use and reduce waste water. Some labs explore bio-based synthesis, using fermentation-derived acids and methanol from renewable sources. Others look at using methyl propionate as a “green solvent” for niche synthetic routes, especially where low toxicity and fast evaporation help separate intermediate or by-product streams. Teams studying removal of volatile organic compounds pursue engineering controls, like new scrubber designs or closed-loop distillation, to keep emissions as low as possible. Even in academia, methyl propionate serves as a test case for analytical methods and chemical kinetics.
Toxicity Research
Toxicologists run tests to reassure both end-users and workers. Studies confirm low acute toxicity—big doses make animals sleepy, but little evidence points to mutagenic or cancer-causing potential. Long-term exposure data for the general public remains limited, but strict worker exposure limits keep risk manageable. Most safety literature notes mild irritant effects for skin and eyes, reinforced by chemical sense when you catch a whiff. Flammability and inhalation risk usually outpace toxicity as limiting factors. Recent projects probe whether long-chain esters or mixed vapors produce different physiological effects, but the consensus holds—standard precautions in industry keep risks in check.
Future Prospects
The story of methyl propionate looks steady but ripe for subtle change. As industrial chemistry leans into sustainability, the choice of solvents and intermediates faces new scrutiny—proven molecules with low toxicity and manageable hazards like this ester gain favor. Process engineers tweak systems for lower temperatures or better waste recovery. Consumer products call for ever-finer control of flavor profiles, so analytical chemists push for higher-purity supplies. Automation, real-time vapor monitoring, and predictive maintenance help factories avoid spills and near-misses. Where regulations confront volatile organics, methyl propionate’s physical profile makes it easier to contain than many rivals. For researchers, the search for designer esters could spin out new applications in drug delivery or functional materials—methyl propionate might just provide a reliable starting point. The world cycles through new technologies and green goals, but old esters prove they still have work to do.
What are the main uses of Methyl Propionate?
A Handy Solvent in Industry
Methyl propionate often pops up in conversations among chemists and manufacturers for a good reason. This clear liquid acts as a solid and dependable solvent. Paints, coatings, and inkmakers value its ability to dissolve other substances and evaporate fast, making projects dry quicker and lowering the chance for smudges or uneven surfaces. Printing shops favor solvents like methyl propionate because faster drying times mean they finish jobs faster and keep costs down.
Flavor and Fragrance World
Most people wouldn’t guess it, but methyl propionate sneaks into the perfumes and flavors we encounter every day. Makers of fruity, sweet, and floral scents lean on this compound because it lets them build fresh top notes without weighing down the final product. Flavorists sometimes use it in tiny amounts to mimic the aroma of fruits or to add a smooth undertone in products like beverages, chewing gum, and candy. Everybody wants their gum or drink to taste fresh, not chemical. Using the right level of methyl propionate makes this possible, and food safety agencies around the world keep a close eye on these levels to protect shoppers.
Fuels and Pharmaceuticals
Big fuel companies look for chemicals that can clean engines or boost performance in a way that won’t cause a mess for the environment. Methyl propionate’s low toxicity and impressive dissolving power check those boxes. Some businesses use it to flush out engine parts, helping cars and trucks last longer and burn cleaner fuel.
Drug makers have a different goal. Pharmaceutical labs put methyl propionate to work as a building block to craft more complex molecules. It takes part in chemical reactions that lead to important medicines. For instance, if a researcher needs an ester link in a new painkiller or antibiotic, methyl propionate often gets picked for the early steps. The fact that large supplies are available and the safety profile beats out harsher chemicals means fewer headaches during development.
Sustainable Choices and Safety
Many industries face growing pressure to find chemicals that get the job done without causing unnecessary harm. Solvents like methyl propionate are less likely to build up in the environment than some of the old-school options, giving eco-conscious manufacturers a reason to switch. Some products break down in the air or water much quicker, which helps meet strict rules about pollution. Research from organizations such as the Environmental Protection Agency highlights how choosing safer solvents can bring down workplace accidents and long-term health concerns.
That said, responsible companies respect the flammable nature of methyl propionate and set proper storage measures. It doesn’t make sense to push for safety in one area while ignoring fire risks or poor training. So businesses focusing on training and proper storage practices see fewer workplace incidents.
Solutions for the Future
A big challenge involves balancing performance with the growing call for safety and sustainability. Industrial labs and universities continue testing new blends and alternatives, trying to stretch the benefits of methyl propionate even further—sometimes in biodegradable plastics or greener solvent systems. This drive isn’t just about regulations. Workers expect safer spaces, and consumers want products that won’t let them down or pollute their neighborhood. Companies open to switching processes or training staff on safer chemical use usually create better results for everyone involved.
What is the chemical formula of Methyl Propionate?
What Does the Formula Tell Us?
Methyl propionate’s chemical formula comes out as C4H8O2. That’s more than a jumble of letters and numbers. This little group hints at everything the compound is and what it might become in the real world. The backbone tells chemists it has four carbons, a structure that shapes its scent, its liquid state, and how it reacts with other chemicals in the lab or factory.
Why Do We Care About Chemical Formulas?
Every day, scientists and manufacturers look for reliable data. Facts matter, especially when health and environmental safety hang in the balance. C4H8O2 means an ester — a compound shaped from a carboxylic acid and an alcohol. In this case, the acid part is propionic acid and the alcohol is methanol. I remember meeting methyl propionate in organic chemistry class: its formula acted as a key. It helped predict reactions, shape flavorings, and understand the risk of inhaling or spilling it.
The Role of Methyl Propionate in Industry
Take a stroll through a fragrance lab or a flavor manufacturer. Methyl propionate often comes up. Its formula points straight to its fruity odor. Where a chemist spots C4H8O2, they see an opportunity for synthetic flavors or as a solvent that evaporates clean. Even paint makers pay attention: solvents with small, predictable molecules help control drying speed and texture on surfaces.
Health, Safety, and Environmental Impact
Its formula doesn’t just sit on paper — it spells out flammability. Four carbons and two oxygens point to volatility. That faint fruity scent comes with a punch; open bottles release vapors that sting the nose and can catch fire if left near a spark. In one lab, I watched a careless spill become a fire hazard in seconds.
People who work with it need gloves and well-ventilated rooms. Its structure keeps regulators on their toes, too. They watch for its part in smog and its knack for moving through soil and water. The U.S. Environmental Protection Agency keeps data on it, tracking any sign it threatens air or groundwater quality.
Seeking Safer and Smarter Solutions
Keeping science grounded in transparency starts with small facts. Giving honest data about compounds — including basic details like formulas — builds trust. In research, teachers stress accuracy when students sketch out C4H8O2, because any small mistake can ripple into bigger ones down the line.
Industry already leans into safer alternatives when possible, swapping out methyl propionate for less flammable options. Good ventilation, closed containers, and emergency cleanup kits keep the worksite safe and always ready. In labs where new chemists learn, the formula becomes more than a lesson — it represents responsible work habits and the importance of details in every bottle and barrel.
Is Methyl Propionate hazardous or toxic?
What People Use Methyl Propionate For
Methyl propionate pops up in a few different corners of industry—paint solvents, some flavors, fragrances, and even pharmaceutical synthesis. This stuff smells sweet, a bit fruity. Despite that pleasant scent, the experience of handling chemicals in the lab taught me never to judge by odor. Plenty of dangerous substances wear a deceptive mask.
Toxicity and Exposure Risks
I’ve seen experienced lab techs brush off smaller organic solvents, but that nonchalance never sits right with me. Methyl propionate might seem low-profile, yet it’s nothing to take lightly. Short-term exposure usually causes irritation—eyes, skin, maybe the lungs if you breathe in the vapors. Longer exposure, especially in cramped, poorly ventilated spots, brings headaches and dizziness into play.
The Globally Harmonized System (GHS) flags methyl propionate as a flammable liquid. Spilled on the floor, it turns into a major fire risk. And flammability doesn’t just threaten those in industrial plants. Even small labs or students working with this chemical find out quickly if nobody’s paying attention to fume hoods and open flames.
Health Impact—A Closer Look
Methyl propionate doesn’t belong to that league of notoriously toxic chemicals—its oral toxicity numbers sit above those for things like methanol or benzene. The median lethal dose (LD50) in rats, often used as a lab benchmark, reaches 830 mg/kg. Not exactly benign, but not instant death either. That being said, no one should get careless. A couple of times each year, news surfaces about accidental poisonings, most tied back to poor labeling or beat-up safety training practices.
Long-term studies in people haven’t been run for this chemical like they have for lead, toluene, or asbestos. That knowledge gap means uncertainty lingers for anyone spending 40 hours a week in a facility using methyl propionate. When information runs thin, the safest bet points toward treating it with the same caution applied to more thoroughly studied solvents.
Environmental Concerns
Methyl propionate doesn’t stick around for long in soil or water. It breaks down fairly fast in the air, thanks to sunlight-triggered reactions. But “breaks down” doesn’t always mean zero risk for wildlife. Some breakdown products could harm aquatic organisms. Stories have come up of accidental spills near streams, leading to temporary fish die-off. So regulations restrict how companies store and dispose of this liquid—something that came up plenty of times during safety audits I’ve attended.
What Makes a Difference: Safety in Practice
Engineering controls make the difference—good fume hoods, air-tight containers, no open flames near where methyl propionate sits. When lab managers or shift leaders skip reminders, workers sometimes stop using gloves or goggles. The best workers I know treat even familiar chemicals with respect. Emergency procedures, clear labeling, easy access to MSDS sheets—those small, everyday things block most accidents.
Potential Solutions and Safer Substitutes
Technological shifts drive the search for greener chemicals with lower toxic profiles. Some companies already replace methyl propionate with less volatile esters or water-based alternatives where feasible. Strict adherence to OSHA standards remains non-negotiable across facilities.
In my years around labs and industrial settings, I’ve seen plenty of shortcuts that shave costs but raise exposure. Companies discussing sustainable practices instead invest in training programs, frequent safety drills, and capable ventilation. Taking those steps means less risk, fewer sick days, and operations less likely to run afoul of regulators.
How should Methyl Propionate be stored?
Understanding the Chemical’s Nature
Methyl propionate is a clear liquid with a fruity odor, used in plenty of industrial and laboratory settings, ranging from flavors and fragrances to solvents. The chemical’s flammability jumps out immediately on a safety data sheet. It lights easily, even at lower temperatures. That fact shapes almost all practical decisions about where and how to store this compound. Once, working in an older facility, I saw a careless placement of chemicals lead to a flash fire. Nobody forgets that kind of wake-up call.
Choosing the Right Storage Location
Space should be well-ventilated. Storing any volatile organic compound in a cramped or closed space, I’ve seen, ends with headaches—sometimes literally, sometimes regulatory ones. Proper ventilation pulls off vapors before they gather at floor level and create an explosive hazard. Too many ignore the invisible risks that come with storing strong-smelling liquids. Flammable storage cabinets, specifically those built to lock, offer one of the simplest ways to cut down risk. Facilities that skip these cabinets may get by for a time, but expecting luck to last is reckless.
Temperature and Environmental Controls
Heat sparks problems with methyl propionate. Exposure to direct sunlight or proximity to heat-producing machinery encourages evaporation and raises pressure in containers. In summer months, keeping storage rooms air-conditioned or at least shaded drops the risk of vapors building up. In a warm climate, even a few degrees can separate a safe shelf from a hazardous zone. Dry areas work best—humidity speeds up container corrosion.
Container Considerations
Metal is out. I’ve always preferred bottles made from glass or specific plastics, double-checking for chemical compatibility. Tight-fitting caps keep evaporation in check and reduce air exposure, so grab containers with solid sealing mechanisms. After years working in labs, I can vouch for the wisdom of clear, visible labeling and color-coded hazard stickers. A split-second pause to check a label might stop a bad accident down the road.
Separation from Incompatible Materials
Certain substances should not share a shelf with methyl propionate. Bases, oxidizers, and acids fall into this category. Too-close storage increases the chance of spills mixing—something that’s triggered more emergencies than I’d like to remember. Pay attention to the shelf plan. Use secondary containment trays to catch leaks. A bit of foresight saves cleanup time, costly shutdowns, or worse.
The Human Factor: Training and Accountability
Even the best storage setup won’t hold up if people use shortcuts. Training matters. In every workplace I’ve been, the labs that run safety drills see fewer problems. People follow clear protocols, so mistakes get noticed quickly and corrected early. Anyone opening, transferring, or even inspecting bottles of methyl propionate should know the risks, understand local emergency procedures, and wear suitable gear like gloves and goggles.
Insisting on checklists and regular inspections keeps everyone honest. I’ve seen places lose sight of chemicals that quietly evaporated out over the years. Inventory tracking using barcodes or digital logs halts that problem, making sure outdated or leaking stock never sits around unnoticed.
Practical Responsibility in Everyday Action
Whether working in a large facility or a small workshop, storing methyl propionate safely doesn’t boil down to fancy technology or endless rules. The basics—ventilation, protection from heat, compatibility, secure storage, and staff training—shape a real defense. Accepting shortcuts in storage can cost money, reputation, and lives. Practicing responsible storage routines every day pays off in quiet, uneventful shifts—a small price for peace of mind.
What are the physical properties of Methyl Propionate?
A Look at What Makes Methyl Propionate Distinct
Methyl propionate brings a sweet, pleasant scent that most folks have come across in fruit flavorings, perfumes, or even nail polish removers. The clear, colorless liquid flows smoothly and evaporates pretty quickly when left in the open. That light, fruity smell is no accident; the chemical comes from a group called esters, known for these aromas. But beneath its gentle exterior, this substance has traits that shape how it’s handled in labs or in large-scale factories.
Temperature Matters
You don’t need heavy equipment to notice how methyl propionate reacts to heat. It boils at around 80°C (about 176°F). This point is lower than water, so it escapes as vapor faster than you might expect. Folks working with it in summer or in hot spaces run a higher risk of breathing in the fumes, which can irritate the eyes or throat. Its melting point sits near –96°C (–141°F), almost as cold as you can get in a regular freezer, so you never see it freeze under normal storage.
Solubility and Evaporation
Drop some methyl propionate into water and it dissolves, but not entirely. If you pour it onto your workbench, most of it just disappears, evaporating off in no time. Behind this behavior stands its vapor pressure, which is higher than that of water even at room temperature. This makes the liquid not only easy to smell but also difficult to keep in a bottle left open. The chemical spreads into the air and could add to air quality issues if not managed well.
Handling and Storage Concerns
Methyl propionate isn’t thick or sticky; it pours almost as readily as alcohol. One important physical property stands out when thinking about safety: flammability. It catches fire at around 10°C (about 50°F), which is well below the temperature of a warm room. Whether in a lab, a factory, or transport, keeping the substance away from sparks and heat sources isn’t optional. If it ignites, it can produce dangerous fumes, so ventilation, flame-resistant containers, and clear labeling play key roles in safe use.
Uses and the Need for Respect
Its physical traits turn methyl propionate into a handy solvent for coatings, inks, and flavors. That pleasant aroma brings it into perfumes, while the fast evaporation fits for cleaning products. At home or work, people often don’t realize what a flammable and quick-to-evaporate liquid they’re around. Not everyone stops to read a safety data sheet, but those details really do matter—any slip in temperature control or careless storage could lead to a fire or unwanted health issues.
Ways to Handle Risks
Smart control keeps methyl propionate working for us, not against us. Factories and research labs use special containers and proper exhaust systems to keep vapors low. Simple choices, like storing it away from sunlight and out of hot rooms, go a long way. Lab workers reach for goggles and gloves, especially on warm days, and always cover containers if they walk away. Ventilation, fire extinguishers, and training for everyone in the building help keep incidents rare. Clear information and practical steps turn a risky substance into a useful tool, without sacrificing health or safety.
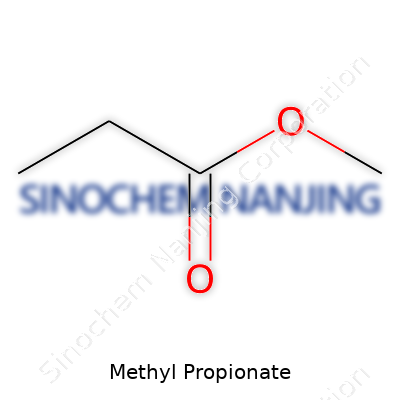
| Names | |
| Preferred IUPAC name | methyl propanoate |
| Other names |
Methyl propanoate Propionic acid methyl ester |
| Pronunciation | /ˈmɛθɪl proʊˈpiə.neɪt/ |
| Identifiers | |
| CAS Number | 554-12-1 |
| 3D model (JSmol) | `CCCC(=O)OC` |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:7770 |
| ChEMBL | CHEMBL1506844 |
| ChemSpider | 6928 |
| DrugBank | DB04181 |
| ECHA InfoCard | 100.082.350 |
| EC Number | 203-547-4 |
| Gmelin Reference | Gmelin Reference: **8086** |
| KEGG | C10456 |
| MeSH | D008762 |
| PubChem CID | 7754 |
| RTECS number | AV3325000 |
| UNII | RTI6W8N0TU |
| UN number | UN1248 |
| Properties | |
| Chemical formula | C4H8O2 |
| Molar mass | 74.08 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | fruity |
| Density | 0.915 g/mL at 25 °C |
| Solubility in water | 16 g/100 mL (20 °C) |
| log P | 0.89 |
| Vapor pressure | 31.6 mmHg (20°C) |
| Acidity (pKa) | 24.3 |
| Basicity (pKb) | pKb ≈ 15.5 |
| Magnetic susceptibility (χ) | -55.0e-6 cm³/mol |
| Refractive index (nD) | 1.369 |
| Viscosity | 0.502 cP (25°C) |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 180.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -403.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2021 kJ/mol |
| Pharmacology | |
| ATC code | M01AB15 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | Health: 1, Flammability: 3, Instability: 1, Special: - |
| Flash point | -1 °C (30.2 °F) (Closed cup) |
| Autoignition temperature | 464 °C |
| Explosive limits | 2.1% - 10.1% |
| Lethal dose or concentration | LD50 oral rat 6,980 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 6,920 mg/kg |
| NIOSH | MPY |
| PEL (Permissible) | PEL: 150 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 1500 ppm |
| Related compounds | |
| Related compounds |
Propionic acid Propionyl chloride Ethyl propionate Methyl acetate Methyl butyrate |

