Looking Beyond the Beaker: Methyl Lactate in the Modern Chemical Landscape
Tracing the Path: Historical Development
Methyl lactate barely draws a crowd outside industrial circles, but look a little deeper and its story walks in step with chemistry’s evolution. Early in the nineteenth century, chemists scrambled to convert organic feedstocks into useful compounds, and methyl lactate came out of those bubbling, steamy glass flasks. It links to the rise of green chemistry too. Lab folks discovered that lactic acid, sourced from fermentation, could react with methanol—unlocking a whole family of esters with practical application. Farmers, tinkerers, and academics were all looking for safer, renewable chemical routes. Over time, the bustling demand for solvents that sidestep the nastier routes pushed methyl lactate out of anonymity and into many labs and factories. The attention speaks volumes about technology’s shift from brute force synthesis to smarter, cleaner approaches.
Getting a Handle on the Product
Methyl lactate belongs to the methyl esters and stands out for its gentle fruity smell. Folks in various industries reach for it thanks to its solubility in water and organic solvents; it combines an ease of handling with low toxicity, which can’t be said for every chemical in the lab. While some solvents stick around by tradition alone, methyl lactate makes its case by behaving predictably, breaking down easily, and fitting into clean label product formulations. It serves as both a solvent and an intermediate, bridging the gap between pure organic chemistry and practical manufacturing.
Physical & Chemical Properties: Straight from the Bench
On the bench, methyl lactate looks like a clear liquid and has a slightly sweet aroma. This fluid mixes well in water, ethanol, and many organic media, so it slots into formulations where engineers need flexibility. Lower boiling point compared to its parent acid or methanol means recoveries from distillation can be managed without excessive energy input. Combustibility draws a line in the sand for risk, but with sound ventilation and controlled sources, the material avoids the drama of highly volatile solvents. Its chemical backbone—the ester linkage from lactic acid and methanol—means it takes part in plenty of organic synthesis, either as a building block or a mild, approachable solvent.
Technical Specifications & Labeling: Stepping Up to Standards
Take a walk through any chemical storeroom and standardized labeling becomes non-negotiable. Methyl lactate typically arrives labeled with purity grades, safety statements, and handling instructions tailored for the end-user’s environment. Whether the batch aims at the food industry, pharmaceuticals, or coatings, purity drives the narrative. Impurities, water content, and acidity keep quality managers up at night. Regulatory frameworks mean producers commit to quality assurance and traceability—nothing short of what modern supply chains expect.
Preparation Methods: Getting Down to Process
Methyl lactate’s usual journey starts with fermentation. Industrial players use renewable resources—think corn or sugarcane—as sugar feeds friendly bacteria. Batches churn lactic acid out, which gets collected and dried. At this point, lactic acid meets methanol under acid catalysis, creating methyl lactate in a reaction called esterification. Sometimes continuous reactors churn day and night for efficient and scalable outcomes. Vacuum distillation purifies the product, and smart process control systems keep an eye on yield and quality. The process fits with sustainable ethos, and energy improvements keep environmental footprints shrinking every year.
Chemical Reactions & Modifications: Flexibility in Action
In the toolkit of chemists, methyl lactate stands as more than a passive player. The ester group remains reactive, letting creative folks swap out the methoxy or manipulate the lactic acid skeleton. Hydrolysis brings methyl lactate back to its parents: lactic acid and methanol—clean, reversible chemistry that’s handy in process design. When folks want to extend the molecule or tweak properties for specialty products, the lactic backbone allows for modification—polymerization, reduction, and even more exotic tricks. This flexibility signals why methyl lactate keeps showing up in unexpected places, from electronics to biodegradable materials.
Synonyms & Product Names: The Language of Industry
Sometimes it reads as lactic acid methyl ester. Other times, lab books refer simply to methyl 2-hydroxypropanoate. In regulatory documents, the CAS number acts as the true fingerprint, but back on the plant floor, old timers just call it what the supplier prints on the drum. This duality—one foot in technical jargon, the other in common industry lingo—mirrors how widely the chemical seeps into modern practice. Having these alternate names can complicate logistics, so not surprising that digital systems now cross-link every permutation.
Safety & Operational Standards: Doing the Job Right
No free passes on safety—methyl lactate reminds us that every solvent brings a mix of hazard and utility. Its relatively low toxicity tempts complacency, but prudent operators never treat solvents as benign. Proper gloves, eye protection, hard hats, and lab ventilation stand as crucial as ever. Regulatory rules—OSHA in the States, REACH in Europe—demand clear documentation and hazard communication. Spill response, waste handling protocols, and engineered controls round out safe operations. Every mishap avoided comes down to a culture of training and vigilance, not just paperwork.
Where Does It Go? Application Areas Big and Small
Ask a paint formulator or a cosmetics chemist, methyl lactate has cred. It steps in as a green solvent, offering performance without punching up the environmental liabilities. Paint removers, coatings, inks, and cleaning products all draw on its dissolving power. Pharmaceutical and food industries use pharmaceutical grade methyl lactate as an ingredient or processing aid. Lately, companies looking to go green began eyeing it for biodegradable plastics, lactic acid derivatives, and even as a potential feedstock for biobased surfactants. Its profile—renewable, manageable, effective—lets companies clean up both their product lines and their act.
Digging into R&D: Ongoing Curiosity
Research teams keep poking at methyl lactate’s untapped potential. Academic consortia and industrial labs both hunt for catalysts that make esterification faster and less resource-hungry, trying to squeeze more product from renewable streams. Bioprocess engineers run pilot plants to scale up fermentation and post-processing in smarter ways. As global supply chains face crunches, methyl lactate’s renewability and flexible process chemistry look increasingly attractive. Smart folks examine its breakdown products for toxicity and look for synthetic tweaks that improve biodegradability and performance in targeted applications. The field keeps inching closer to a kind of green-plastics-and-chemicals loop that actually closes.
Toxicity Under the Microscope: What’s Safe, What’s Not
Ingredient safety never fades into the background. Toxicological studies show methyl lactate does not accumulate in animal tissue and breaks down to lactic acid and methanol, both present in natural metabolic pathways. High doses can stress organs in animal studies, primarily due to methanol, so regulatory guidelines limit exposure, especially in workplace situations. Air monitoring, closed systems, and personal protection help keep exposures way below danger levels. Comparing its toxic load to traditional petrochemical solvents, methyl lactate rates well for both episodic and chronic toxicity, helping shift industry toward safer practices—though vigilance in research and regulatory review never goes out of style.
What Lies Ahead: Future Prospects
Every decade brings new pressures—tighter environmental regulations, consumer demand for transparency, and an industry hungry for renewable options. Methyl lactate, with its sturdy ties to bio-based feedstocks and mild operating properties, stands poised for a bigger future. Process engineers redesign supply chains to build in resilience, using fermentation as a stable platform for specialty chemicals. Materials scientists tinker with methyl lactate derivatives for advanced polymers and coatings, all aiming to close the loop on waste and cradle-to-cradle product life. Trends suggest even broader roles for solvent blends, green chemistry, and specialty syntheses fed by methyl lactate. If cleaner, safer, smarter industry matters, its trajectory offers lessons for the whole sector.
What is Methyl Lactate used for?
What Exactly Is Methyl Lactate?
Methyl lactate comes from lactic acid, which everyone has heard about thanks to fitness enthusiasts and yogurt commercials. This chemical forms when lactic acid teams up with methanol. The result is a clear liquid that smells a bit like fruit. It shows up in more places than most people realize, mainly because it’s considered less toxic than a lot of historical solvents.
The Role of Methyl Lactate in Industry
Chemists like working with methyl lactate, especially in cleaning formulations. Walk into a factory or a large print shop and you’ll spot this compound in inks and cleaners. Factories dealing with electronics often rely on it to remove tough residues from delicate circuit boards. It works hard without breaking down sensitive components. Painting professionals sometimes reach for it to clean brushes or thin coatings, since it pulls oil and pigment away without the harshness of older chemical solvents.
Big companies searching for “greener” options keep an eye on methyl lactate. Most traditional solvents carry strong fumes and environmental risks. Methyl lactate, since it comes from fermenting plant sugars, fits better with efforts to cut down on fossil fuel dependence. Several brands in cosmetics and personal care put it to work as well, because it dissolves oils without leaving nasty byproducts behind. I’ve seen the stuff pop up in makeup removers and exfoliating creams—quietly doing its job and letting natural fragrances shine through.
Medical and Food Touchpoints
Pharmaceutical labs see methyl lactate as a helper for creating medicines. It helps in making active ingredients more available in topical creams or patches. Some researchers mention it as a safe option in drug delivery, though they don't splash the news across grocery shelves. The food industry doesn’t ignore this chemical either. In food flavoring processes, its mild solvent powers gently carry delicate flavors while avoiding residues that raise red flags with regulators.
Risks and Environmental Concerns
Nothing gets a free ride in chemistry, not even methyl lactate. It scores higher for safety than old-school cleaning agents, yet direct contact—especially on the skin—can irritate some people. Breathing in concentrated vapors isn’t healthy. Having handled it in a lab, I always trusted my gloves and vent hoods. Environmentally, methyl lactate breaks down faster than many petrochemical solvents. It doesn’t linger long in water or soil, which keeps wildlife and drinking water safer. Still, overuse or careless disposal can undo those benefits. Industries bear the burden to handle it with respect and not treat “biodegradable” as a license for dumping.
Moving Toward Safer Chemistry
Consumers increasingly care about what’s in their cleaning sprays and body lotions. Companies that substitute harsh petrochemicals with methyl lactate stand to gain not just regulatory approval, but also goodwill from customers looking for cleaner labels. A few major paint brands now tout “bio-based solvents” in marketing materials, listing methyl lactate front and center. If manufacturers keep listening to safety data and push to recover and recycle even the less-harmful solvents, the trajectory for this chemical looks increasingly positive.
Is Methyl Lactate safe to use in cosmetics?
Methyl Lactate: Where It Shows Up and Why
Most folks won’t recognize methyl lactate on an ingredient label, but this compound turns up in all sorts of things—lotions, creams, and even makeup removers. It acts as a solvent and gives off a mild minty scent. Brands use it to thin heavy oils or to add a cooling touch to a formula. After working with cosmetic chemists and talking with folks on the development side, it’s clear that convenience and effect drive its popularity, especially for rinse-off products.
Safety: What Science and Regulators Say
Every so often, an ingredient makes beauty headlines because someone heard about a new study or a vague concern. The real measure for safety comes from scientific research and what regulators lay out. The Cosmetic Ingredient Review (CIR), which goes over ingredients used in the United States, lists methyl lactate as low-risk in rinse-off formulas at the usual concentrations. The European Union allows it under similar guidelines. These groups study irritation, allergic reactions, and hormone disruption for a reason—they aren’t letting just anything through.
A 2012 study from the International Journal of Toxicology covered both immediate effects and concerns from long-term use. Researchers checked for allergic reactions and signs of irritation on healthy skin and found methyl lactate rarely causes trouble in small doses, especially in diluted or rinse-off products. On broken or compromised skin, higher concentrations will sting. My experience working hands-on with test batches matches those findings—getting pure methyl lactate on a paper cut is no fun.
Concerns in the Real World
Anyone hunting for “natural” or “gentle” ingredients often pauses at anything that sounds too chemical. I hear the same questions from clients again and again: Does it build up in your body? Will it make skin worse? There isn’t evidence that methyl lactate builds up in tissues or causes issues with regular use in skin care. The body breaks it down into lactic acid and methanol, both processed by normal metabolic pathways.
Where problems crop up tends to be in products left on skin for long periods, or in rare cases of allergy. There’s always a small slice of people whose skin reacts to anything new. Contact dermatitis can show up as redness, itchiness, or dryness, but these responses stay rare for methyl lactate at the levels approved for cosmetics.
Solutions and Steps Forward
Skin safety isn’t about chasing ingredients off the shelves; it’s about real understanding and clear labeling. Brands should stick with the concentrations proven safe by solid research. Better communication also helps: transparent ingredient lists and a quick heads-up about what to expect will help folks with sensitive skin. I like to see companies offer patch-test recommendations, so customers know both what’s inside and how to try it out on a small patch of skin first.
Cosmetic safety moves forward when brands pay attention, regulators keep up with research, and shoppers have a say in what they want. With methyl lactate, small amounts in creams, balms, and cleansers meet the science-backed safety bar most of the time. Still, anyone who knows their skin flares up with new additions can always check in with a dermatologist before rolling out a new routine.
What are the storage requirements for Methyl Lactate?
Why Storage Conditions Matter So Much
Methyl lactate finds its way into labs and factories around the world for a reason. As a solvent and intermediate, plenty of people rely on its consistent quality. I’ve seen problems pop up when storage goes wrong—not just headaches from lost product, but real risks to health and safety. Folks sometimes think, “It’s just another chemical.” But methyl lactate proves unforgiving when overlooked. Its low flash point and sensitivity to air can spell disaster if left in a drafty, hot, or carelessly sealed spot.
The Core Storage Requirements
Let’s start with the basics. People working with methyl lactate want container seals that keep the air out. Exposure to humidity or oxygen speeds up hydrolysis, breaking it down into lactic acid and methanol. Not only does this mess with the material’s purity, it also builds up pressure and causes containers to bulge or rupture. Drums or bottles must shut tight and stay that way until the last drop gets used.
Anyone who stores this liquid knows high heat can be a real problem. Its flash point hovers near 40ºC (104ºF); that’s lower than you might expect. Park a barrel near a sunny loading dock, or inside a room that gets hot in summer, and it turns risky real quick. Fires start from warm rooms and stray sparks more often than folks realize. Methyl lactate likes temperatures under 25ºC (77ºF); cool, dry storage matters more here than with many other solvents.
Ventilation and Fire Protection
Tight storage doesn’t mean you skip fresh air. Ventilated storage areas help keep vapors from drifting and building up. Fume hoods, ventilated cabinets, and open space for any larger stock make sense—both for people working nearby and for insurance against unexpected leaks. Good air flow cuts the odds of inhalation or flash fires.
Fire-rated storage cabinets stand out as the gold standard. Anyone who’s worked in a busy shop knows accidents happen: A forklift rams a drum, a container drops during rush hour, or a bottle tips over. Putting flammable chemicals like methyl lactate in proper fire-proofed shelving limits trouble. Sprinkler systems add a final layer, catching sparks or fires before they have a chance to spread. Local regulations sometimes force the issue, but safety forward thinking goes beyond checking legal boxes.
Labeling and Regular Inspection
Every workplace dealing with chemicals should keep labeling crisp and routine inspections regular. People get busy, and old stock gets forgotten. Over time, seals degrade, labels fade, and what’s inside may not match what’s written outside. Old methyl lactate sometimes changes color or smells different if it’s started breaking down. Spotting those signs comes with experience, but having a schedule to check every drum and bottle makes things routine. I’ve worked with folks who set phone reminders or run weekly walk-throughs. It pays to stay watchful so product loss and safety worries don’t catch anyone off guard.
Smart Solutions Going Forward
Switching to smaller, better-sealed bottles can help smaller labs limit loss from degradation. Large factories benefit from investing in cool, segregated storage spaces that stay locked and inspected. A few places now use monitoring tech to track temperatures right at the shelf, with alarms pinging if things get too warm; these sorts of steps save time, money, and possibly lives. Clear workplace training, such as regular reminders and hands-on drills, rounds out a smart plan for anyone looking to handle methyl lactate with confidence and peace of mind.
What is the chemical formula of Methyl Lactate?
Understanding Methyl Lactate’s Structure
The chemical formula for methyl lactate is C4H8O3. Chemists identify it as the methyl ester of lactic acid. Take one lactic acid molecule, replace the hydrogen atom in the carboxy group with a methyl group, and you get methyl lactate. It carries both an ester group and a hydroxyl group, which gives it a unique set of properties, especially in industrial and lab settings.
Real-World Uses Matter
Industrial processes appreciate straightforward, stable chemicals. In my years working in labs and occasionally consulting for small manufacturing outfits, methyl lactate sat among the reliable green solvents. Its low toxicity and biodegradability help manufacturers reduce environmental impact. Methyl lactate allows companies to meet sustainability targets laid out by both regulatory boards and retailers demanding ‘greener’ products on their shelves.
Take paint stripping as an example. Traditional solvents, toluene or methylene chloride, create headaches for safety managers. Methyl lactate offers similar results without the harsh fumes or challenging waste disposal. It breaks down safely, both in the environment and in the human body, making it safer for workers and less burdensome for wastewater treatment.
Food and Flavor
While I never set out to taste chemicals, methyl lactate finds its way into food as a flavoring agent. That is because its breakdown products — methanol and lactic acid — both occur naturally in a host of vegetables and fermented products. Regulatory agencies in North America and Europe have approved its use for specific applications. It’s powerful in tiny amounts, which reminds me of how some ingredients can define a dish with just a dash.
Lab and Research Applications
Biochemistry labs use methyl lactate for preparing certain samples. It offers mild solvent properties for processes like tissue clearing for microscopy. In synthetic chemistry, its polarity helps dissolve both hydrophilic and hydrophobic compounds, making it useful in research protocols that need gentle but effective solvents.
Methyl lactate can act as a green solvent alternative in thin-layer chromatography and HPLC applications. Many researchers value safer lab environments, encouraged by shifting attitudes around solvent exposure and waste handling. Institutions face mounting pressure not just to keep researchers safe but to dispose of chemicals in responsible ways. After teaching college lab courses, I noticed students preferred methyl lactate over harsher options, and disposal protocols ran smoother since the solvent wasn’t classified as hazardous.
Challenges and Next Steps
Wider adoption of methyl lactate gets slowed down by pricing and availability. Green chemistry often costs more than legacy chemicals. Companies wanting to switch will do so if public pressure builds and if supply chains can consistently meet demand. Upstream, more investment goes into bio-based production methods to scale up output and lower costs. Once manufacturers have a steady, affordable supply, the shift away from more hazardous chemicals could speed up, with real benefits for workers and water sources downstream.
So, while methyl lactate’s formula — C4H8O3 — may look simple, understanding its role in safer manufacturing and lab practices opens the door to more sustainable chemistry in everyday life.
Is Methyl Lactate biodegradable?
Everyday Chemicals and Real Impact
Most people will never spot methyl lactate on a grocery store label, but this chemical crosses into dozens of everyday products. It comes from lactic acid, which also happens to occur naturally as your body breaks down sugar for energy. Manufacturers use methyl lactate as a solvent, so it winds up in paints, coatings, and cleaners. Its rise owes a lot to growing pressure to swap out harsher petrochemical ingredients for gentler alternatives. Still, the conversation about its actual environmental benefits deserves a closer look—especially the claim that it biodegrades safely and quickly after use.
What Happens After Use
The journey of methyl lactate doesn’t end down the drain. Environmental impact matters long after the bottle empties. Biodegradability means soil microbes and waterborne bacteria can break the chemical into natural byproducts without trouble, lessening buildup in nature or risk to wildlife. Research provides some comfort. Studies point out that under aerobic conditions, methyl lactate tends to degrade swiftly—often weeks rather than months. The European Chemicals Agency notes ready biodegradability in test conditions, and reports show bacteria found in rivers and soil munch their way through it fairly efficiently. That difference matters. Solvents that persist or bioaccumulate can turn up in groundwater and organisms, climbing food chains and setting off health alarms, as seen with traditional petrochemical solvents.
Real-World Nuances
Lab claims can overpromise if we take them at face value. Not all breakdown processes outside test tubes run as smoothly. Soil and water temperature, oxygen levels, and even chemical mixing play roles. Cold or low-oxygen conditions can slow down the process. Wastewater treatment plants—main landing spots for household chemicals—vary in their ability to break things down, depending on their setup. Lessons from other “green” solvents remind us that marketing a chemical as biodegradable sometimes skips over the practical hurdles. Still, methyl lactate holds up better than most due to its simple structure and origin in renewable sources.
Why It Matters for Consumers and Industries
Many folks don’t realize their cleaning choices link back to rivers and farmland. Picking products based on evidence—not buzzwords—means looking beyond the front label. For small businesses like eco-friendly cleaning startups, the choice of methyl lactate helps improve environmental performance reports. I’ve worked on consumer campaigns where the challenge was not only finding alternatives that clean well but also making sure waste doesn’t pile up in nature. Details from reputable sources and peer-reviewed studies made a difference in steering product development and shaping consumer trust.
What Can Drive Things Forward
Moving the needle in chemical stewardship always takes broader teamwork. Regulators, scientists, and industry groups can push for more transparency in data collection and reporting, not just assuming lab data translates to real-world safety. Investing in advanced wastewater treatment technology matters, too. On an individual level, folks can stay engaged by asking questions about product ingredients and environmental certifications. Companies making claims should show supporting data, audited by third parties.
Communities share a stake in how chemistry unfolds in daily life. The story of methyl lactate’s biodegradability shows that science, transparency, and practical application must work together for any chemical to earn its “green” badge.
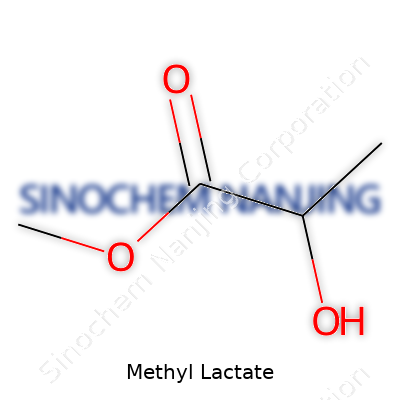
| Names | |
| Preferred IUPAC name | Methyl 2-hydroxypropanoate |
| Other names |
Lactic acid methyl ester Methyl 2-hydroxypropanoate Methyl α-hydroxypropionate Methyl lactate Methyllactate |
| Pronunciation | /ˈmɛθ.ɪl ˈlæk.teɪt/ |
| Identifiers | |
| CAS Number | 547-64-8 |
| 3D model (JSmol) | `JSmol` 3D model string for Methyl Lactate (InChI string format): ``` InChI=1S/C4H8O3/c1-3(5)2-7-4(6)8/h3,5H,2H2,1H3,(H,6,8) ``` |
| Beilstein Reference | 635950 |
| ChEBI | CHEBI:17802 |
| ChEMBL | CHEMBL142437 |
| ChemSpider | 52608 |
| DrugBank | DB11255 |
| ECHA InfoCard | EU-InfoCard-100.029.207 |
| EC Number | 201-184-2 |
| Gmelin Reference | 6764 |
| KEGG | C14416 |
| MeSH | D008771 |
| PubChem CID | 6131 |
| RTECS number | OD9625000 |
| UNII | 9B27489Z69 |
| UN number | UN3278 |
| Properties | |
| Chemical formula | C4H8O3 |
| Molar mass | 104.12 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Faint, pleasant odor |
| Density | 1.06 g/cm3 |
| Solubility in water | miscible |
| log P | -0.18 |
| Vapor pressure | 0.26 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 15.1 (hydroxyl), 25 (alpha proton) |
| Basicity (pKb) | 15.51 |
| Magnetic susceptibility (χ) | -7.62×10⁻⁶ |
| Refractive index (nD) | nD 1.414 |
| Viscosity | 2.63 mPa·s (25 °C) |
| Dipole moment | 2.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 111.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -682.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1346.7 kJ/mol |
| Pharmacology | |
| ATC code | A03AX12 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| Flash point | Approximately 83 °C (181 °F) |
| Autoignition temperature | 215 °C |
| Explosive limits | Explosive limits: 2.6% - 20.4% |
| Lethal dose or concentration | LD50 (oral, rat): 2,600 mg/kg |
| LD50 (median dose) | 5,000 mg/kg (rat, oral) |
| NIOSH | WI6725000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Methyl Lactate: Not established |
| REL (Recommended) | 0.5% |
| IDLH (Immediate danger) | No IDLH Established |
| Related compounds | |
| Related compounds |
Lactic acid Ethyl lactate Methyl acetate Methyl pyruvate Propylene glycol Methyl formate |

