Methyl Formate: Tracing the Journey, Understanding the Impact
A Look Back: The Early Years and Evolution
People rarely think about the history of chemicals, but methyl formate has a story rooted deep in the early days of industrial chemistry. Back in the 19th century, researchers working to understand formic acid—named for its presence in ants—discovered that combining it with methanol produced methyl formate. Initially, curiosity drove investigations, but practical needs soon took center stage. Engineers began using methyl formate for its unique solvent properties. Over time, its uses multiplied, walking hand-in-hand with both industrial growth and regulatory change. Industries developed greener, safer processes to keep up with environmental and health concerns. The chemical’s journey tells a lot about the changing priorities in science and manufacturing: early excitement about new reactions slowly gave way to intentional design and efficiency improvements in laboratories and factories. Looking at this evolution, we get a sense for how historical trends in chemistry have responded to society’s shifting needs.
Understanding What Methyl Formate Offers
Methyl formate isn’t just another building block in the giant warehouse of organic chemicals. Its sweet, fruity smell might remind some of apples, but its performance in the lab deserves more attention than its fragrance. Synthetically, this compound works as a powerful solvent, delivering strong solvency for cellulose, resins, and oils, but evaporates quickly enough to make it a star in quick-drying coatings and adhesives. This sort of versatility gives industries an extra tool, whether it’s in electronics or flavor production. For example, companies producing synthetic flavors rely on methyl formate to create nuanced notes that wouldn’t be possible using just raw plant extracts. The practical know-how needed to balance odor, volatility, and safety is something only time, trial, and error have refined.
Physical and Chemical Identity: Why It Matters
Walk into a lab and open a bottle of methyl formate, and the odor quickly highlights its volatility: this compound boils just above room temperature at 31.5°C, and it freezes far below zero. Unlike heavier organic solvents, it wafts away before you notice, which means engineers have to use special storage techniques and work under strict ventilation. Its density and miscibility with other solvents make blending easy in industrial processes. Chemically, methyl formate’s simple ester structure—one carbonyl group connected to a methyl—lets it participate in reactions that change its core and create new products. Understanding these traits isn’t a matter of curiosity; it directly affects how safe and successful a chemical plant can be. The risks linked with high vapor pressure and flammability demand both respect and technical care.
Getting Specific: Technical Standards and Label Details
Chemical producers print detailed labels for methyl formate to keep everyone on the same page. Purity, water content, acidity—all these details can decide whether a reaction takes off or fizzles. For example, trace acid or moisture can ruin sensitive organic syntheses or catalyze side reactions that compromise product quality. Technicians analyze each drum using gas chromatography or titration before approving it for use in pharmaceutical or electronics fabrication. Regulations require clear hazard markings, but anyone who’s worked with the material knows that practical safety takes more than a GHS diamond or bolded warnings. Reliable labeling couples with vigilant laboratory procedure, and both have been shaped by a long line of near-misses and lessons learned.
Preparation: A Marriage of Chemistry and Engineering
Anyone searching for methyl formate on an industrial scale needs to juggle raw material costs, purity targets, and environmental compliance. Most modern processes feed methanol and carbon monoxide over an alkali metal catalyst, typically sodium methoxide, to generate large quantities efficiently. Companies carefully tune temperature and pressure to harvest high yields without drifting into hazardous territory. Historical methods involving formic acid or less selective pathways faded due to poor efficiency and growing environmental awareness. These advances weren’t handed down overnight—they emerged from years of process optimization, worker advocacy, and the realization that cleaner reactions keep both profits and safety records strong.
It’s Not Just Synthesis: Reactivity and Real-World Modifications
Chemists value methyl formate beyond simple synthesis. This ester readily breaks down to yield formic acid and methanol again, and under hydrogenation, it offers a route to methanol alone. As an intermediate, it helps produce formamides, formamidines, and even dimethylformamide, which all feature in high-performance polymers, pesticides, and solvents. Tinkering with reaction conditions lets skilled chemists guide the molecule’s fate with surprising precision. These transformations find use not just in theory but across major segments of the modern economy, from agricultural supply chains to electronics manufacturing.
Other Names, Same Molecule: Navigating Synonyms and Brands
In chemical literature, methyl formate sometimes appears as “methyl methanoate” or “formic acid methyl ester.” Browsing through research articles or safety data sheets, one sees a long lineage of trade names and translations, shaped by evolving IUPAC conventions and branding choices. Each synonym keeps the molecule’s essential features consistent. For newcomers, this can create confusion, but professionals keep glossaries and up-to-date documentation close at hand. This dance of names reflects a global industry’s need for both precision and marketing distinction, illustrating another way chemistry has entered the world stage.
Raising the Bar: Safety Knowledge Grows by Experience
Methyl formate’s volatility brings more than technical interest—it creates a constant thread of concern for safety. Its low flash point, readiness to vaporize, and narcotic effects at high concentration have prompted factories to upgrade ventilation systems, rethink personal protective equipment, and expand fire suppression networks. Decades ago, incidents forced regulators and industry groups to set exposure limits and reporting requirements, shaping protocols still used in facilities around the world. Responsible operators invest in real-world training rather than relying on paperwork alone. Talking to plant workers reveals a clear message: practical safety emerges from on-the-ground experience and a willingness to learn from past mistakes.
Application Areas: Finding Value in Surprising Places
Today, methyl formate has found its way into a remarkable variety of products. Foam manufacturers appreciate its low toxicity compared to legacy blowing agents like CFCs, as it helps create polyurethane foams for furniture and insulation. Electronics companies turn to methyl formate in cleaning or etching applications, taking advantage of its rapid evaporation and solvent power. Flavor and fragrance creators use it to build synthetic aromas, while smaller specialty firms find niche uses in extracting plant oils or cleaning fine instruments. Real gains come when industries find new applications for a familiar substance—sometimes inspired by regulatory change, sometimes by clever chemistry.
Pushing Forward: Research, Development, and Unanswered Questions
Despite its long history, methyl formate keeps appearing on research agendas. Scientists investigate new catalysts to make its preparation greener and more cost-effective, and study its breakdown pathways to reduce emissions from factories. Ongoing research focuses on boosting yields for pharmaceutically relevant intermediates, often targeting energy efficiency and reduced byproduct formation. Others search for bio-based routes, aiming to bypass fossil feedstocks entirely. Even after a century of use, methanol-carbon monoxide processes still see tweaks and updates, usually tied to tighter environmental rules or the rising cost of raw materials.
Understanding Risks: Toxicity in Context
Anyone coming near methyl formate often asks about health effects. Low concentrations in air typically cause headaches and dizziness, while higher exposures raise alarms for rapid anesthetic effects and even risk of death—especially in poorly ventilated spaces. Researchers investigate long-term effects, but short-term toxicity draws the clearest focus. Animal studies guide exposure limits, which workers and companies enforce with ventilation and monitoring. Past accidents—fires, spills, and overexposures—have left a clear impression: no amount of routine can substitute for vigilance and training. These lessons anchor both regulatory frameworks and workplace cultures.
Looking Down the Road: Where Methyl Formate Fits Tomorrow
The story of methyl formate isn’t finished. Sustainability conversations push producers toward renewable routes using captured carbon sources or bio-methanol, blending industrial innovation with climate action. Some companies experiment with new reactions, placing methyl formate at the heart of carbon recycling schemes, or using it in solar-fuel processes for energy storage. The growing demand for safer, greener solvents seems ready to secure methyl formate’s relevance—though always with an eye on keeping workers and the environment out of harm’s way. Chemical development rarely follows a straight path, but methyl formate’s resilience in the face of change offers a useful model for the rest of the chemical world. Every year, new uses, new precautions, and new policies emerge, ensuring that the story grows richer with every experiment, every policy debate, and every successful production run.
What is methyl formate used for?
What’s the Real Use of Methyl Formate?
Methyl formate doesn’t make headlines like gasoline or plastics, but it’s the sort of chemical that quietly shapes modern life. Most people never think about where it fits in, so let’s put a spotlight on it for a moment.
Methyl formate, with its faintly sweet smell, rolls off production lines mostly to help larger industries make things run smoothly and cheaply. Companies use it as an ingredient for producing formic acid. That’s not something the average person keeps in the pantry, but farmers and factories depend on it. Formic acid shows up in animal feed additives, rubber production, even leather tanning. The fun thing is, methyl formate makes these processes cheaper and a bit cleaner on the back end.
Methyl Formate and Everyday Products
Growing up, I watched workers strip paint from old doors with strong-smelling chemicals. Some of those paint removers relied on methyl formate. Its ability to dissolve stubborn paints and finishes, mixed with less hazardous chemicals than older paint strippers, means better safety for workers. Methyl formate replaced a few more toxic ingredients, so it’s less likely to linger in people’s bodies or poison groundwater.
Foam insulation sandwiching new homes together owes a lot to this simple molecule, too. Methyl formate acts as a blowing agent—basically, it helps puff up the foam by forming bubbles inside. So if your winter heating bills stay low, there’s a solid chance methyl formate had a hand in stopping your house from leaking warm air. It replaced some of the nastier older blowing agents that chipped away at the ozone layer for years. That’s a win for both homeowners and the planet.
Safer Choices and Climate Connections
In recent years, more companies have swapped their traditional solvents out for methyl formate. Strong science backs the switch. Researchers have pointed out how methyl formate breaks down quickly in the environment, avoiding the toxic buildup linked with older industrial choices. It doesn’t hang around in air or soil, and it skips out on the greenhouse gas issues baked into CFCs and HCFCs. Regulatory agencies like the EPA flag it as a “greener” substance thanks to its low toxicity and fast breakdown.
That doesn’t mean it’s harmless. In workshops without good airflow, breathing high doses can cause dizziness or irritation. Decent safety training and protective gear make all the difference. Workers need to understand the risks—no one wants a “green” solvent turning into another hospital visit story.
The Road Ahead
People want their products cleaner, safer, and cheaper. Methyl formate fits into that push, but it’s on manufacturers to keep safety high and emissions low. Industry leaders can keep testing alternatives and tracking health impacts. Governments can keep their hazard ratings up to date as new evidence comes in. Homeowners, workers, and everyone in between benefit from this kind of steady attention—small steps like these made methyl formate a smarter pick for modern manufacturing.
Is methyl formate hazardous to health?
Looking Closer at the Chemical
Methyl formate pops up in all sorts of industries. Factories use it to make things like solvents, blowing agents for foams, and some adhesives. It has a strong, sweet odor, often catching people off guard in the workplace. Plenty of folks may handle it daily without knowing the long-term effects or what that whiff really means for their health. Having spent time in a manufacturing plant, I’ve watched workers handling barrels and hoses with gloves and masks—not because they’re overly cautious, but because they’ve seen what happens when someone takes a shortcut.
Exposure and What It Does to the Body
People usually come into contact with methyl formate by inhaling its vapors or letting the liquid touch their skin. The substance evaporates quickly when left open, filling the workspace with fumes. Breathing it in can irritate the nose, throat, and lungs pretty quickly. Folks in the field have reported headaches, dizziness, and even nausea after a long shift. The Centers for Disease Control and Prevention lists it as a respiratory hazard. Skin contact can cause redness, dryness, and even blistering in higher concentrations. Eyes take the hit worst. Even a small splash or strong vapor can make them burn and water. Not everyone takes the same hit—sensitivity varies.
Long-Term Health Problems
Research hasn’t revealed everything about methyl formate’s long-term effects, but there are reasons for concern. Chronic exposure over weeks or even months could cause nerve problems and impact memory. Some animal studies have shown organ effects, particularly involving the liver and kidneys, after repeated exposure. The National Institute for Occupational Safety and Health has set workplace exposure guidelines, reflecting concern over accidents and cumulative health issues.
Workplace Safety and Accountability
Staying safe at work means more than just a mask tossed over your face. Companies have a duty to train workers on chemical risks. Proper ventilation, robust exhaust systems, and reliable personal protective equipment matter as much as following a recipe in the kitchen. Workers who understand what methyl formate does and how to handle spills or leaks can spot early warning signs and avoid costly mistakes. Years back, my crew went through a chemical safety drill—the sort nobody thinks they'll need until something sprays or drips. That drill paid off more than once. Training creates a culture of caution, and the investment shows in fewer accidents and illnesses.
Policy and Practical Adjustments
Stronger rules can make a difference. Governments can enforce stricter air quality monitoring and push for safer chemical substitutes. Incentives to switch to less hazardous solvents also work—money spent upfront saves health costs later. On a smaller scale, routine air checks and open communication between management and staff keep everyone aware and prepared. In communities near large plants, public reports and neighborhood meetings bring light to chemical use and give locals a voice. Honest, accessible information reduces fear and builds trust.
Balancing Progress and People’s Health
Industries need chemicals like methyl formate, but health shouldn’t take a back seat. Paying attention to new research, respecting science-backed guidelines, and prioritizing safety keep both productivity and people in good shape. Everyday choices in the plant—wearing protection, checking for leaks, and speaking up about symptoms—protect workers today and for years down the road.
What are the storage requirements for methyl formate?
Looking After a Volatile Chemical
Methyl formate stands out for its role in industrial settings, found everywhere from manufacturing to labs. Its sharp odor and fast evaporation rate demand attention, not just for peace of mind but for the health and safety of workers and anybody nearby. Growing up near a chemical plant, I saw first-hand how one careless mistake brought out the fire department and shut down half the block—never a fun day for anyone. So, treating methyl formate with care isn’t just a rule. It’s the way you keep people and the neighborhood safe.
Containers Matter More Than You Think
Let’s talk storage containers. Forget about reusing any old drum or plastic jug. Methyl formate reacts with a lot of metals and breaks down certain plastics. Weak seals or improper linings can leak vapors, and you don’t want those hanging in the air. The safest bet is picking solid, completely sealed metal drums—stainless steel or those with a good internal lining. Drums and tanks must handle pressure, since this stuff vaporizes at lower temperatures. I remember an incident at a cousin’s hardware shop where one subpar barrel inflated slightly and caught everyone’s attention. Luckily, a quick fix worked that time. Relying on proper containers and strict inspection routines keeps small problems from turning into emergencies.
Temperature and Ventilation
Methyl formate boils well below water’s boiling point. Even on a warm spring day, it’s pushing to turn into gas. Warm rooms, sunlight, or lack of shade aren’t just nuisances—they’re a recipe for vapor buildup. Airflow in the storage area makes a huge difference. Mechanical exhaust systems pull vapors away from people and wiring. Open flames and sparks don’t mix with the fumes, so using spark-proof fans and switches is part of the groundwork. Insulation material helps too; I’ve visited sites that kept methyl formate in climate-controlled cabinets with zero incidents to show for it.
Keep It Dry and Free From Incompatibles
Moisture in the storage area means trouble. Water triggers hydrolysis, which leads to more acidic products, increased pressure, and sometimes enough heat to bring about a fire. Damp basement corners or open-slat warehouse floors just don’t cut it. This isn’t just about being careful; it’s about understanding how chemistry works inside every drum.
Nearby storage also counts. Acids, bases, and strong oxidizers should stay as far away as possible. Cross-contamination or a spill mixing with another chemical can turn a routine day into one no one forgets. Labels aren’t enough. Good signage and clear separation stop problems before they start. Safety data sheets should hang in plain sight, not hidden in a file cabinet.
Early Detection = Early Action
Even with airtight barrels and well-kept rooms, methyl formate’s distinctive smell can sneak up if something starts to go wrong. Monitoring with vapor detectors, not just relying on a worker’s nose, catches leaks before the air turns dangerous. Sprinklers and foam extinguishers belong close at hand—water won’t cut it with chemical fires.
Training and Routine Make All the Difference
People sometimes overlook hands-on training, thinking printed instructions are enough. Comprehensive training means practice drills, checks before and after every shift, and regular review of emergency steps. After seeing a few near-misses in my early days at a chemical distributor, I’ve watched experienced teams spot a faulty seal or suspicious bulge in a drum at a glance, preventing trouble before it starts.
Methyl formate isn’t rare, but safe handling and smart storage remain rare enough that sharing practical know-how matters. Keeping up with the rules and operating above the bare minimum goes beyond compliance—it’s just the right thing to do for everyone involved.
What is the chemical formula of methyl formate?
Getting to Know Methyl Formate: Formula and Structure
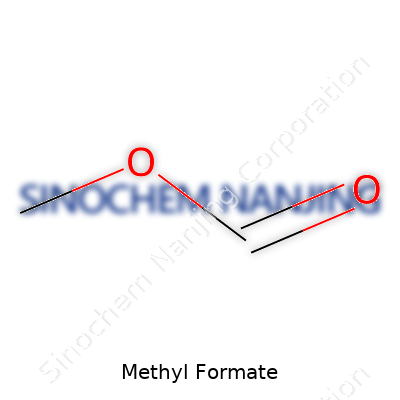
Methyl formate comes with a tidy chemical formula: C2H4O2. In everyday language, this means two carbon atoms, four hydrogen atoms, and two oxygen atoms link up to create a small, volatile ester. Scientists and industry folks mark this compound for its sharp, sweet aroma. It gets a lot more attention than the sum of its parts might suggest.
Spotting Methyl Formate in the Real World
Methyl formate isn’t some exotic compound tucked away in a lab. It surfaces where coffee beans roast, shoots up during fermentation, and finds regular use in factories. It serves as a key step in making formamide and has its fingerprints all over the production of solvents and blowing agents. I once worked with colleagues who dreaded methyl formate leaks — one whiff and the whole room smelled impossibly sweet, almost like spoiled apples and alcohol all at once. It’s easy to underestimate the punch these small molecules can pack.
Health and Safety: Risks Can’t Be Ignored
Safety matters every time methyl formate comes out. At workplace levels above a certain threshold, exposure can irritate eyes, skin, and the lining of the nose. Larger spills displace air and can make workers light-headed or dizzy fast. In my experience, factories with strict ventilation and clear emergency procedures keep trouble at bay. The substance catches fire easily, so reliable storage with tight-sealing containers and away from sparks ranks pretty high on the priority list. As a community, we’ve learned through hard stories that strong safety training paired with airtight containers makes a difference in preventing injuries and property damage.
Environmental Footprint: Not Always Transparent
Methyl formate breaks down in water and air, and it doesn’t stick around the way some persistent organic pollutants do. Still, there’s more to the story. Leaks during manufacturing send fumes into the air, and nobody wants a buildup in crowded industrial parks. The sweet odor often acts as a red flag, alerting workers long before the numbers on a sensor might change. Responsible companies run regular checks and redesign pipes and valves to keep accidental releases low. Production lines that rely on green chemistry cut out a lot of headaches down the line for both workers and communities nearby.
Innovation and Safer Practices
Methyl formate has carved out a role in cleaner manufacturing. Its presence in making less-harmful foam blows a hole in the need for ozone-depleting gases. One neighbor of mine, who runs a small furniture business, insisted on eco-friendly foam because his customers started asking questions about chemical exposure in their homes. Methyl formate-based solutions gave companies like his a way to meet both regulatory requirements and consumer trust. Savvy manufacturers invest in closed-loop systems, recycling waste gases and reducing the total volume let out into the air. Folks have seen profits and worker morale swing up as a result.
Smarter Rules and Community Engagement
Better standards don’t happen overnight, but every advance counts. Regulators and advocacy groups keep pressure on, pushing for updates as new data on health and environmental impacts come forward. Real progress happens when industry workers, researchers, and local communities share firsthand stories and push for more transparent reporting. Over time, these small wins add up, driving policies that keep everyone a little safer around chemicals like methyl formate.
How is methyl formate produced industrially?
A Closer Look at Production
Factories crank out methyl formate through methods that don’t just pop up out of a textbook. The main approach chugs along by taking methanol and carbon monoxide, then firing them through a reactor in the presence of a solid base like sodium methoxide. Toss in some pressure, tweak the temperature, and you get methyl formate. Over the years, factories have adopted this direct carbonylation route for a reason—it brings big volumes, fewer steps, and keeps side reactions out of the way.
It’s easy to talk chemistry all day, but anyone who’s stepped foot in a plant knows the nuts and bolts matter. Carbon monoxide shows up dangerously toxic, so any mishap turns into a story no one wants to tell. Systems use tight containment and plenty of sensors. The importance of solid safety training stands out here, because nobody gets a second chance once CO starts leaking.
Why Bother With Efficiency?
Money doesn’t grow on trees. Efficiency cuts down the raw inputs and saves on energy bills. In hard times, that matters more than ever. Methanol’s cheap, widely available, and easy to move, so it fits perfectly into big plants looking to make metric tons. Plants using recycled CO from other chemical streams help too. Tossing less out the stack and cutting costs hits both profit and responsibility marks in the industry.
Some processes run at lower temperatures to avoid blasting through energy. A few operators use heat exchangers and tweak catalyst choices, squeezing the most out of their reactors. In my early years in chemical plants, I noticed old systems would overheat, wasting fuel. Updating can sound pricey, but with rising energy costs, most companies shake loose some capital for upgrades.
Pursuing Purity
Most folks outside the plant don't think much about purity, but it shapes what you can do with your final product. Raw methyl formate contains water, methanol, or by-products—nasty for downstream reactions or fine chemical use. That’s why distillation towers run around the clock, separating out impurities and squeezing every drop of value out of what’s produced. People spend a lot of time tracking tiny trace chemicals, because getting quality wrong means failed batches further down the line.
Environmental and Safety Hurdles
CO brings big risks. Leaks threaten workers, but venting carbon monoxide also throws greenhouse gases into the air. Smart plants recycle unused CO, trapping and piping it back in. The trick isn’t just bigger scrubbers or fancier fans; it’s repeat inspections and actual people walking lines checking for trouble. Wastewater from the process picks up organics, so on-site treatment plants pop up as mandatory stops before anything heads back out. Management often forgets these systems when chasing production targets, but ignoring them leads to real fines and angry neighbors.
Room for Change
Sustainability trends don’t skip chemicals. Green chemistry gets a lot of talk, yet practical steps win out. Some groups look for renewable sources—pulling methanol from captured CO2 instead of fossil fuels. I’ve seen pilot plants work with bio-methanol and alternative sources, but scaling beats enthusiasm every time. Any new method must match the reliability and cost of the old standby, or nobody in the front office signs off.
Methyl formate production looks simple on paper, but real life means constant tweaks and tough judgment calls. With more eyes on climate and safety, chemical plants push toward cleaner, safer, and cleverer ways to get the job done.
| Names | |
| Preferred IUPAC name | methyl methanoate |
| Other names |
Methyl methanoate Formic acid methyl ester Methylester kyseliny mravenci Rocanol HCOOCH3 |
| Pronunciation | /ˈmiːθɪl ˈfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 107-31-3 |
| 3D model (JSmol) | `COC=O` |
| Beilstein Reference | Beilstein Reference: 1739977 |
| ChEBI | CHEBI:48250 |
| ChEMBL | CHEMBL44897 |
| ChemSpider | 7276 |
| DrugBank | DB14116 |
| ECHA InfoCard | 100.000.594 |
| EC Number | 203-481-7 |
| Gmelin Reference | Gmelin Reference: 82877 |
| KEGG | C01741 |
| MeSH | D008736 |
| PubChem CID | 6587 |
| RTECS number | LQ9625000 |
| UNII | 26OD7PFO3A |
| UN number | UN1243 |
| CompTox Dashboard (EPA) | DJ6B9H61G2 |
| Properties | |
| Chemical formula | C2H4O2 |
| Molar mass | 60.05 g/mol |
| Appearance | Colorless liquid with a pleasant odor |
| Odor | fruity odor |
| Density | 0.97 g/cm³ |
| Solubility in water | soluble |
| log P | -0.26 |
| Vapor pressure | 514 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 7.8 |
| Basicity (pKb) | 13.23 |
| Magnetic susceptibility (χ) | -35.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.344 |
| Viscosity | 0.46 mPa·s (at 25 °C) |
| Dipole moment | 1.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 138 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -413.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -561.5 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P311, P337+P313, P370+P378, P403+P235, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-4-1 |
| Flash point | -19 °C |
| Autoignition temperature | 451°C |
| Explosive limits | 5.5% - 26.7% |
| Lethal dose or concentration | LD50 Oral rat 1,800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,600 mg/kg (rat, oral) |
| NIOSH | MFJ6000000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Methyl Formate: "100 ppm (parts per million) or 246 mg/m³ (OSHA) |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Formic acid Formate esters Methyl alcohol (methanol) Formamide Dimethyl carbonate |

