Methyl Cyclohexane: Behind the Chemistry and Beyond
Historical Roots That Shape Today
Methyl cyclohexane sits in the shadow of bigger chemical names, but its story stretches back to the days when organic chemistry split from curious tinkering into the backbone of modern industry. Early researchers chasing better fuels and solvents found themselves circling back to cycloalkanes like this one. The drive to decode petroleum distillation, paired with the want for refined automotive fuels as engines evolved, pushed scientists to trace every fraction of crude oil. In this process, methyl cyclohexane cropped up not as a theoretical oddity, but as a practical building block.
What It Really Is—And What It’s Good For
If you ask any chemist who sweated through orgo lab, methyl cyclohexane isn’t just another name to memorize. Its six-membered ring, decorated with that single methyl group, brings up lessons in molecular strain, ring-flipping, and how even small tweaks change a liquid’s behavior. Pour it into a beaker and you get a clear, colorless liquid with a gasoline-like odor. Boiling at around 101°C, with a density less than water, it floats and evaporates way faster than most folks expect. You don’t find methyl cyclohexane at the grocery store or pharmacy, but anyone who’s used paint removers, adhesive thinners, or certain degreasers likely came across it in action. Chemical plants rely on its stability and solvent powers for both research and big-volume industrial tasks.
Properties That Steer Its Role
Methyl cyclohexane’s charm sits in its low reactivity under normal conditions, mixed with an ability to dissolve oils, waxes, and non-polar compounds that stump water. React it with the right catalyst or put it under the right pressures, and now it’s a springboard for a cascade of reactions that matter for synthetic rubbers, resins, or fuel chemistry. In my own lab experience, we’d record its flash point—about 25°C—and remind newcomers that its rapid evaporation made fire safety more than a footnote in the binder. It burns clean but vapor travels farther than folks expect, sometimes drifting to ignition sources that no one gave a second thought. This simple hydrocarbon warrants respect, not indifference.
Getting a Handle on the Numbers and Labels
Every barrel or container tells the story: methyl cyclohexane’s CAS number (108-87-2) follows it around the world, as do its synonyms—“1-methylcyclohexane,” “hexahydrotoluene.” Labels might focus on flammability, keeping it clear that a spark means trouble even at low concentrations. Storage isn’t about stashing it on any old shelf; temperature controls and grounding wires matter to prevent static discharge in busy work areas. I’ve seen more than one rookie swept up by the temptation to downplay the MSDS, until a waft of vapor in a closed room brings on dizziness. These health markers aren’t bureaucratic red tape; the headaches, nausea, and even longer-term risks can catch up fast with poor habits.
How It’s Made and Changed
Dig into refinery chemistry and methyl cyclohexane falls out of the jumble of hydrocarbon streams via catalytic hydrogenation of toluene. It’s a textbook example of turning aromatic rings into saturated rings, trading reactivity for the more docile bonds of a cycloalkane. In research labs, we’d walk through this process, noting yield bumps depending on catalyst purity or hydrogen pressure. Once isolated, methyl cyclohexane doesn’t just sit idle. Through halogenation, sulfonation, or oxidation, chemists tug more “handles” onto the molecule, shaping intermediates for plastics, agrochemicals, or specialty solvents. The history of its modifications reads like a material science journal in miniature—each tweak opens another path for product design or reactivity.
Applications That Go Beyond the Obvious
Those outside the chemical world might never know methyl cyclohexane’s fingerprints are all over industries. Petroleum refining ranks high—blending agents for higher-octane fuels depend on it. Labs use it to separate precious analytes in chromatography or to strip out moisture without harsh chemistry. It steps in as a non-polar solvent where hexane or heptane used to rule but environmental and health rules clipped their use. Newer research circles revisit methyl cyclohexane in hydrogen storage systems; its reversible hydrogenation and dehydrogenation cycles look attractive for moving energy, drawing on the decades of thermodynamic data that researchers built up alongside worries over carbon footprints and storage safety.
Safety and Operational Habits
Any work with methyl cyclohexane puts stewardship in the spotlight. Fire marshals and safety engineers make sure that chemical rooms get proper ventilation, that spill kits and absorbents aren’t just box-ticking but real responses to real leaks. Flammable vapors hang low and travel silently, so open flames and static-prone equipment find themselves banned from these settings. Carelessness leads to rapid headaches, dizziness, or worse. Gloves—nitrile, not latex—stay near every bottle, along with goggles. Respiratory risks ramp up with prolonged exposure, so closed systems and fume hoods aren’t suggestions. Institutional memory, shaped by close calls and accident reports, shapes how every bottle gets used or stored.
Where Research Is Pointing
Across the labs fixing their gaze on clean fuels and alternative energy carriers, methyl cyclohexane has found itself at the center of debates on hydrogen logistics. The reversible nature of its hydrogenation wins the interest of engineers looking for practical storage vectors. Research arms of chemical companies keep dissecting its life cycle—from extraction to combustion—to weigh the real environmental burdens. Safety studies have drilled in on occupational exposures, too. Reports from Japan and Europe, where hydrogen infrastructure hopes run strong, show sustained funding and publication rates in this area. The challenge links back to handling, throughput, and ensuring containment matches scale—building confidence in safety while keeping eyes on real-world feasibility.
Looking at Toxicity and What It Means
Methyl cyclohexane doesn’t evoke the horror stories tied to older solvents like benzene, but that doesn’t give blanket approval. Chronic exposure, especially through inhalation, knocks cognition, irritates airways, and raises questions about liver and kidney health if handled without care. Environmental watchdogs worry about what seeps into groundwater from spills or accidents, since the compound’s mobility and persistence drift into larger water tables. Toxicity research lags behind for long-term low-dose exposures compared to headline-making substances; that signals a need for better tracking and thorough studies. Occupational health circles recognize this gap and push for more epidemiological data from workforces who logged hours in paint shops, refiners, or chemical plants.
The Road Ahead for Methyl Cyclohexane
Looking past current practice, methyl cyclohexane stands as a case study in balancing technical need and responsible application. Interest in sustainable chemistry shapes the way companies invest in refining processes with lower emissions. Stricter regulations on volatile organic compounds encourage research into safer, greener alternatives, but for critical industrial contexts, familiarity with methyl cyclohexane’s quirks and limits remains key. Instead of sweeping it off the table, the conversation shifts toward safer handling, smarter engineering, and ongoing testing to catch risks before they cascade. For all its perceived simplicity, this molecule reflects broader truths about the chemical industry—progress only counts if knowledge and vigilance move alongside it.
What is methyl cyclohexane used for?
Everyday Utility in Industry
Factories and laboratories have a regular companion, and that’s methyl cyclohexane. Walk through a processing plant, and this clear, flammable liquid probably sits on a storage shelf. Paint manufacturers, adhesive makers, and those dealing in resins rely on methyl cyclohexane as a solvent. It dissolves tough compounds that water or alcohol can’t touch, so it keeps paints smooth and resins blended.
Pharmaceutical firms, too, have good reason to keep this chemical close. Key reactions—the kind that spit out pain relievers or anti-inflammatories—move along smoothly in its presence. You’ll also find methyl cyclohexane in cleaning solutions, helping degrease machinery and electronics. Anyone dealing with precision components knows how crucial that kind of thorough wash can be.
Deep Ties to Fuel
Refineries draw on methyl cyclohexane during the long journey from crude oil to gasoline. It hops from intermediate to intermediate before ending up in fuel that powers cars. The auto industry benefits as well: it’s mixed into some aviation fuels, chosen for its stable, high-octane traits. A decade ago, advances in fuel chemistry brought methyl cyclohexane into the spotlight again. People began to discuss it as a possible hydrogen carrier, since it can store hydrogen safely in a kind of “liquid battery.” Some Japanese energy projects run field tests using methyl cyclohexane to ferry hydrogen from one spot on the planet to another.
Everyday Safety and Environmental Questions
Coming across methyl cyclohexane isn’t something most people will do at home, and that’s a relief. Those who handle drums of it at work know about the headaches and nausea that hit after careless contact. The liquid evaporates with a sharp smell, and anyone fumbling with it needs gloves, goggles, and good ventilation.
Runoff after a spill doesn’t just disappear. City water treatment plants aren’t properly designed to break down something like this. Instead, it can slip through to rivers, heading downstream past fish and wildlife. Back in the 1980s, several spills led the U.S. Environmental Protection Agency to put stricter reporting rules in place. Modern equipment reduces some of the risk, but mistakes or old tanks can still lead to preventable accidents.
Building Safer and Smarter Standards
Industry groups and regulators stay on their toes, testing new ways to move and store methyl cyclohexane. Fire safety guidelines have improved, with better ventilation and sensors checking for leaks. Chemistry researchers keep seeking alternatives or new catalysts so that reactions can take place in milder solvents.
Sharing what goes wrong, as well as what works, matters. Engineers, chemists, and health officials need access to clear incident reporting and up-to-date scientific reviews. Safety training can’t happen just once, and glove standards shouldn’t go unchanged for a decade.
Long-term, we need more chemical transparency and safer substitutes where possible. Substitution programs, better recycling methods, and investments in greener solvents will keep people healthy and manufacturing robust. Where we still use methyl cyclohexane, careful management limits the downside—reminding us that progress means not just making, but making wisely.
What is the chemical formula of methyl cyclohexane?
Looking into the Structure
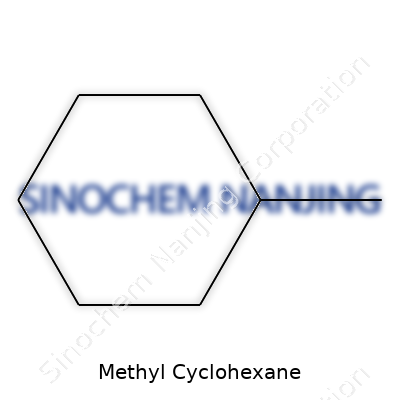
Methyl cyclohexane grabs attention partly because of its straightforward composition, C7H14. Seven carbons, fourteen hydrogens—engineered by nature and refined by chemists in the lab. The structure itself tells a story about how a single methyl group attaches to a cyclohexane ring and changes the entire behavior of the molecule. In practical terms, you take a cyclohexane ring, slap on a methyl group, and you end up with something that handles differently in both lab reactions and real-world uses. Seeing that shift makes the study of organic chemistry come alive, because minor adjustments can lead to big differences downstream.
The Reality of Working with Methyl Cyclohexane
During college, most people probably hear about methyl cyclohexane for the first time in organic chem class. My first real encounter, though, happened in the back corner of an undergraduate research lab where solvent choice meant the difference between a clean separation and a failed experiment. The reason methyl cyclohexane shows up on so many reagent shelves is not some fluke. It’s because of its stability, nonpolarity, and its knack for dissolving other nonpolar compounds without breaking a sweat. Unlike aromatic solvents, it doesn’t carry the same risks of benzene content, which lets labs work a little safer.
What Sets C7H14 Apart?
Methyl cyclohexane sits in an interesting niche. As a hydrocarbon, it steps up as a safer, less volatile alternative to materials like toluene or hexane. Engineering programs and manufacturing processes lean on it for applications like paint thinners, adhesives, or as a reference standard in analytical labs. There’s real value in replacing harsh solvents with something that handles with less toxicity and flammability. Studies from the International Agency for Research on Cancer support efforts to use solvents with lower health risks, and methyl cyclohexane ranks lower on the danger scale compared with older options containing benzene.
Concerns and Environmental Impact
No solvent is perfect, of course. Methyl cyclohexane brings some questions about VOC emissions into the picture. The chemical won’t pollute groundwater as aggressively as halogenated solvents or some persistent hydrocarbons, but plenty of it in the air can add to smog and respiratory irritation. Researchers have examined how rapidly it breaks down in the environment and found it doesn’t stick around for years the way others do. Even so, responsibility falls on industries to keep emissions down and invest in recovery systems that sniff out vapors before they head out the stacks.
Charting a Responsible Path Forward
Training teams in chemistry and safety circles often focus on handling solvents with respect and care, especially when those solvents show up in gallons instead of test tubes. Switching to greener processes calls for more than picking the right formula. Sourcing, storage, and recycling matter as much as formulae and flash points. Some research labs have built mini-reclamation units to recapture used methyl cyclohexane, cutting costs while easing the environmental toll. In commercial settings, efforts to track usage and substitute with bio-derived options continue to gain traction, answering concerns over resource depletion.
Everyday Chemistry with a Personal Connection
From the desks of students to the benches in industrial plants, methyl cyclohexane provides a lesson in chemistry with real stakes. Drawing that line between simple formula—C7H14—and the cascade of choices it sets off in safety, supply, and sustainability, shows why a molecule is rarely just a molecule. It’s a decision point with health, science, and the next project all waiting on the outcome.
Is methyl cyclohexane hazardous or toxic?
What Methyl Cyclohexane Actually Does
Methyl cyclohexane pops up in all sorts of places—paints, adhesives, fuels, and even in the labs that make the building blocks for plastics and rubber. A colorless liquid with a slightly sweet odor, it vanishes into the air pretty quickly. Some have probably smelled it in a hardware store or while working on a home project. On its own, it seems harmless enough, but there’s a bigger story here.
The Hazards: What Science Says
Diving into the safety data, it’s clear this is a flammable substance. One spark, and methyl cyclohexane may start a fire. Labs and factories know this, so strict protocols stay in place to limit open flames, static, and sparks anywhere near it. People take flammability like this seriously, because workplace accidents with volatile chemicals create real danger. A big spill creates a fire risk not just for property, but for every worker nearby.
Breathing in high concentrations of the vapor creates another set of problems. Health agencies such as the CDC and OSHA say exposure to methyl cyclohexane vapor can irritate the nose, throat, and lungs. It causes headache, nausea, dizziness, and in high enough doses, workers can even pass out. Long-term effects aren’t as well-documented, but frequent exposure to high levels of hydrocarbons sometimes harms the nervous system. Simple protective equipment like gloves and masks, and using proper ventilation, does cut down on risk.
Toxicity: Is It Dangerous in the Body?
Swallowing or inhaling a lot of this chemical is definitely dangerous. Animal studies found that large doses harmed the liver and kidneys, though these doses were far higher than what most people ever encounter. That being said, methyl cyclohexane doesn’t build up in the body over years. The body can break it down and push it out, which sets it apart from some truly nasty industrial toxins. But that doesn’t make it safe to handle without care—just less likely to cause trouble at normal exposure levels.
Environmental impact deserves a mention. This compound hangs out in the atmosphere for a while, and spills on land or in water can threaten fish and plants. In high enough amounts, hydrocarbons kill aquatic life. Any site that handles enough to risk a spill should have a real emergency plan. Rushing through cleanups or pretending small spills don’t matter just ends up hurting everyone.
Practical Safety: What Works in Real Life
Most people never run into methyl cyclohexane on purpose. For folks working in research, transportation, or manufacturing, common sense goes a long way—use gloves and goggles, keep good airflow, and do not smoke or eat near this stuff. Regular safety drills make a difference, because panic never helps during emergencies. I’ve known plant workers who swear by clear labels and color-coded spills kits—no one mistakes methyl cyclohexane for something safe, and cleanups happen fast.
Personal experience reminds me: danger from chemicals rarely comes from a single mishap. It comes from ignoring labels, letting safety gear gather dust, and acting like basic training isn’t needed. Everyone in supply chains, from truck drivers to warehouse teams, deserves real education about chemicals like this one. Honest conversations reduce accidents a whole lot more than just relying on posted warnings.
Learning from the Facts
Methyl cyclohexane brings risk because of its flammability and health effects after high exposures—not because it builds up like mercury or polychlorinated biphenyls (PCBs). Just calling something ‘hazardous’ doesn’t tell the whole story. By staying alert, following rules that make sense, and keeping up with new data from trusted organizations, workplaces and communities can count on a safer environment. That’s an outcome everyone wants, no matter which chemicals cross the loading dock.
How should methyl cyclohexane be stored and handled?
Getting Real About Risks
Anyone who has spent time around a chemical lab or industrial site knows that some liquids demand more respect than others. Methyl cyclohexane fits that category. It’s clear, easy to mistake for other solvents, and it evaporates into a vapor that lights up with just a spark. That’s not a theoretical hazard—it’s something I have seen go wrong with similar solvents, even from a forgotten rag or a spark from a nearby tool. The flashpoint for methyl cyclohexane is low; a warm room can make vapor levels rise fast, turning an ordinary day into a serious emergency.
Simple Steps Make a Real Difference
Working around flammable chemicals pushes people to pay attention—not because rules say so, but because the risks are real. Use grounded, sealed metal containers with tight lids, and keep them far from any ignition sources. Methyl cyclohexane is no friend to open flames or faulty wiring. I’ve watched even seasoned colleagues double-check to make sure containers stay closed and labeled, especially after a close call a few years back. Those simple rituals prevent stories from becoming news headlines.
Ventilation Isn’t an Option
Vapor builds up quietly. One week, old ventilation in the backroom let vapors cling to the air and workers only noticed when headaches started spreading. Proper storage means using spaces with reliable exhaust fans drawing fumes away. The best setups pull air outside, not just toward a corner. Any whiff of solvent in the air signals a problem needing attention now—not after lunch break. Storing drums or larger volumes outside, under shade, keeps things cooler, though leaks or spills call for speedy cleanup using absorbent material—the kind made to handle solvents.
Watch What You Store Together
Some might stash different chemicals together to save space, but that just multiplies risks. Methyl cyclohexane won’t play nicely with strong oxidizers, acids, or alkalis. I still remember a shelf collapse years ago where incompatible chemicals mixed—nobody wants to see that reaction firsthand. Separate storage works for a reason. Keep things labeled clearly and separated based on compatibility, not convenience.
Personal Gear Makes a Difference
No one should skip gloves or goggles for “just a quick job.” Direct skin contact with methyl cyclohexane causes irritation, and getting a splash in the eyes means a straight trip to the eye wash. Chemical-resistant gloves, splash-proof goggles, and work aprons cost less than a doctor’s visit or lost work. Most folks learn that lesson once and don’t gamble again. I’ve seen that moment of regret from people who thought they were saving time.
Training Matters More Than Signs
Paperwork and safety signs alone won’t cut it. Regular training keeps everyone sharp. Practical drills—how to react to a spill, how to use the eyewash, when to call for help—these build skills that stick. Repeating the basics every few months beats having to explain what went wrong after an accident. Leadership, clear rules, and a safety-minded crew keep emergencies in the “never happened” column.
Better Tools for Routine Tasks
Technologies like flame arrestors, grounded pumps, and chemical spill kits don’t just meet regulations—they make daily tasks less stressful. Automated sensors or alarms that trigger above safe vapor levels shut down situations before they escalate. These upgrades pay for themselves the first time they catch a mistake before someone gets hurt.
Patching Leaks Early, Not Later
No one wants to discover a slow leak after the floor’s sticky or the smell hangs in the air. Daily visual checks and well-maintained equipment catch drips before they grow. Escaping vapors cost money, threaten health, and, left unchecked, invite disaster.
What are the safety precautions for working with methyl cyclohexane?
Understanding the Risks
Methyl cyclohexane gets used a lot in labs and plants, often as a solvent or in chemical synthesis. Over the years, I’ve seen folks treat it like it’s harmless just because it looks clear and flows like water. The facts don’t support that laid-back thinking. Breathing in those vapors can irritate your lungs, cause headaches, or get you feeling dizzy—sometimes even knocking you off your feet. It’s highly flammable, so a stray spark or a warm surface can start trouble in an instant.
Respecting the Hazards: Practical Steps
Forget open windows and a desk fan. Working with methyl cyclohexane demands real ventilation. Fume hoods pull vapors away before you even know they’re building up. I’ve spilled my share of chemicals, and trust me, you notice if you miss a fume hood only once—the coughing and burning in your throat does the teaching.
Eye protection goes beyond the basic safety glasses. Splashing a solvent in your eye will have you racing to the eyewash station and hoping for the best. Tight-fitting goggles block surprises that regular glasses let slip through. I always put them on, even if I’m just pouring from one bottle to another.
Nitrile gloves stand up best to methyl cyclohexane. Latex breaks down, and nobody wants that stuff soaking straight through to your skin. Gloves need changing every time you suspect a splash or after a long session, long before you actually see evidence of a leak.
Fire Safety Can’t Wait
A lot of warehouse fires happen because people store solvents like this near ignition sources. Every drum and bottle needs a cool, spark-free home. Bonding and grounding during transfers cut the risk of static sparks—run a copper wire between containers, and there goes one possible fire. It sounds simple, but too many places ignore it until the fire chief is filling out a report.
Keeping a Class B fire extinguisher within arm’s reach isn’t optional. These extinguishers tackle flammable liquid fires, and minutes matter when a spill ignites. I’ve seen more than one accident where workers remembered training and cut damage in half just by having that extinguisher ready.
Emergency Planning Isn’t Just for Show
Every site using methyl cyclohexane should run emergency drills until the routine sticks. Fire alarms, spill kits, evacuation routes—all those things look boring on checklists, but they help when the room fills with smoke or your friend grabs their throat after a spill. I’ve been in enough drills to know which sites take training seriously. The difference in real emergencies always shows.
Labeling every bottle matters as much as the contents themselves. I once grabbed what I thought was acetone and realized by the smell that something was off. Luckily, the label corrected my mistake, sparing me a ruined experiment and a possible injury.
Better Choices Mean Fewer Regrets
Training new staff to respect chemicals—not fear them—builds a safer workplace. Sharing near-misses and stories keeps everyone alert. Mistakes with methyl cyclohexane don’t give many second chances, so up-to-date knowledge and teamwork really save lives. Reviewing safety data with the crew and checking equipment from time to time keep old habits from slipping in.
It always comes down to common sense and respect for what you’re handling. Methyl cyclohexane won’t forgive shortcuts, and neither will health or property losses after a serious accident.
| Names | |
| Preferred IUPAC name | methylcyclohexane |
| Other names |
Hexahydrotoluene Cyclohexylmethane |
| Pronunciation | /ˌmɛθɪlˌsaɪkloʊˈhɛkseɪn/ |
| Identifiers | |
| CAS Number | 108-87-2 |
| 3D model (JSmol) | `CCCC1CCCCC1` |
| Beilstein Reference | 1721441 |
| ChEBI | CHEBI:15604 |
| ChEMBL | CHEMBL15830 |
| ChemSpider | 8077 |
| DrugBank | DB01982 |
| ECHA InfoCard | 100.116.255 |
| EC Number | 203-624-3 |
| Gmelin Reference | 100793 |
| KEGG | C06588 |
| MeSH | D008778 |
| PubChem CID | 8077 |
| RTECS number | GV7875000 |
| UNII | 6RL3M1ZN02 |
| UN number | UN2296 |
| Properties | |
| Chemical formula | C7H14 |
| Molar mass | 98.19 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet, gasoline-like |
| Density | 0.77 g/cm³ |
| Solubility in water | insoluble |
| log P | 3.61 |
| Vapor pressure | 52.7 mmHg (20°C) |
| Acidity (pKa) | ~50 |
| Basicity (pKb) | 16.7 |
| Magnetic susceptibility (χ) | -74.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4231 |
| Viscosity | 0.68 mPa·s (25 °C) |
| Dipole moment | 0.36 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -156.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4774.3 kJ/mol |
| Pharmacology | |
| ATC code | Methyl Cyclohexane does not have an ATC code. |
| Hazards | |
| GHS labelling | GHS02, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P240, P241, P261, P273, P301+P310, P331, P303+P361+P353, P304+P340, P403+P235 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 25 °C (77 °F) |
| Autoignition temperature | 223 °C (433 °F; 496 K) |
| Explosive limits | Explosive limits of Methyl Cyclohexane: 1.2% - 7.0% |
| Lethal dose or concentration | LD50 oral rat 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 3200 mg/kg |
| NIOSH | LH1400000 |
| PEL (Permissible) | 500 ppm |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 1500 ppm |
| Related compounds | |
| Related compounds |
Cyclohexane Toluene Methylcyclopentane Ethylcyclohexane Cyclohexene |

