Methyl Bromoacetate: More Than a Building Block in Modern Chemistry
Historical Roots and Shifting Landscape
Methyl bromoacetate traces back to some serious foundational roots in organic synthesis, with its story running parallel to the age of rapid chemical discovery during the late 1800s and the burst of synthetic dyes and pharmaceuticals. Many early chemists took to halogenated esters like methyl bromoacetate because these compounds unlocked new pathways for targeted modifications on a molecular level. Reactions involving methyl bromoacetate weren’t just props in lab demonstrations; they pushed forward the toolkit for medicinal chemists and advanced the production of important compounds at a time when the world was opening up to industrial-scale medicines. That history matters because each generation builds upon the last, relying on small, reactive molecules like this to step into new frontiers in chemistry, especially now with pharmaceuticals looking to do more with less starting material.
What Sets It Apart: Product Overview and Key Features
This molecule isn’t exactly a household name, but its reputation among chemists is pretty solid. Methyl bromoacetate shows up as a clear, colorless to slightly yellow liquid, giving just a hint of a sharp, irritating odor. It’s got a lot of punch in a small package because its carbon-bromine bond is reactive, making it much more than a routine ester. While you might pass by a jug of methyl bromoacetate in a chemical warehouse and not give it a second glance, chemists see a versatile actor for all kinds of substitution and addition reactions. The double threat of its mild solubility in water and ready blending in organic solvents makes it a prime choice for controlled lab reactions where you want to balance speed with precision.
What’s Under the Hood: Properties and Points to Watch
Methyl bromoacetate clocks in with a molecular weight right in the range that suits most organic synthesis work, boiling at around 103-105°C under standard atmospheric pressure, which means its volatility is something to keep an eye on. It's heavier than water and denser than many solvents, with a refractive index that points to its halogen content. From a technical user’s standpoint, the big draw is its reactivity profile — that bromine atom doesn’t wait around for long, making this molecule a reliable alkylating agent. That’s crucial for scientists hoping to tag a new functional group onto a substrate. Anyone who’s ever run a basic alkylation reaction with this compound knows the reaction usually finishes fast, demanding decent ventilation and airtight glassware to avoid vapor loss and lab safety headaches.
Making It: How It’s Prepared
Manufacturing methyl bromoacetate isn’t a dark art reserved for specialists, but there’s enough care involved to keep things interesting. Synthesis generally starts with the reaction of bromoacetic acid with methanol, using a suitable acid catalyst. This method gives chemists a reasonably straightforward way to get a decent yield, assuming you keep things dry and your methanol free of water. Anyone who’s been around a prep lab will tell you the reputation this method has for reliability, but it also pushes the need for safe handling because of the fume risk. Old-school chemists sometimes mention alternate routes, but there’s a reason most stick with methylation and bromination — it works, the gear is standard, and there’s less room for nasty surprises unless the starting materials are off-grade.
Names in the Crowd: Synonyms and Aliases
Nobody expects an ester to be a celebrity, but methyl bromoacetate has picked up a few names over the years that show up in papers and catalogs. You’ll see it listed as Bromoacetic acid methyl ester or Methyl 2-bromoacetate, and on rare occasions just BME among those who use it often. These alternate names matter for research indexing and procurement — mix-ups or confusion with similar-sounding esters can lead to costly setbacks, especially for newcomers or labs switching suppliers.
Reactivity and Chemical Versatility
Almost every researcher handling methyl bromoacetate does so for its ability to add bromoacetate fragments onto a host molecule. The compound stands out for nucleophilic substitution, making it a go-to reagent for alkylating thiols, amines, and phosphines. You see it featured not only in simple lab exercises but in the published procedures behind advanced pharmaceuticals and new agrochemicals. Changing that bromine for other functional groups launches new compounds with interesting properties. For synthetic chemists, the compound’s value shines when building up complex molecules — reactions using Grignard reagents, for example, can tack on tailored side chains with impressive precision. That kind of control positions methyl bromoacetate as a mainstay for those who need results they can reproduce, not just hope for.
Working With It: Safety Realities and Operational Protocols
There’s no denying the hazards involved with methyl bromoacetate. It’s a skin, eye, and respiratory irritant, capable of causing burns or worse in concentrated form. Anyone who’s fumbled a reaction or caught a noseful of vapor learns fast that caution isn’t optional. Labs need to follow strict fume hood usage, glove and eye protection, and diligent spill management. Given its volatility and risk profile, storage demands tightly sealed glass, cool temperatures, and solid labeling to prevent mistaken use. Anyone who works with this compound should keep antidotes for bromide poisoning on hand and ensure clear SOPs to avoid exposure. Experienced chemists remember small accidents can quickly escalate; that reality shapes how we structure training protocols and how often we review safety drills.
Practical Applications: Where It Delivers Real Value
Methyl bromoacetate doesn’t usually get the headlines, but it helps drive progress in pharma, fine chemical synthesis, and even new agricultural solutions. Drug developers rely on this compound for building-block strategies to make complex active ingredients. In agrochemical development, it brings options for synthesizing more selective pesticides, often cutting down on environmental impact thanks to the specificity introduced during the synthesis step. Bioconjugation strategies — attaching fluorescent tags or therapeutic moieties to proteins for research — often start with small, reactive esters like methyl bromoacetate. It finds its way into specialty plastics and as a precursor in below-the-radar industrial processes, quietly enabling advances that help shape the future of modern materials and medicines.
Research and Development: What’s Driving Progress?
Every innovation with methyl bromoacetate has come from looking past the bottle and asking bigger questions about sustainability, process safety, and molecular design. Researchers are taking hard looks at greener routes for both synthesis and disposal, aware of the health and ecological impacts. Screening for better catalysts, developing bio-based feedstocks, and dialing in process parameters to reduce waste all show up in the latest literature. Efforts to tame its volatility and unpredictability have already yielded improvements in containment, dosing, and automation, which in turn opens doors for broader adoption in settings where only veteran chemists dared work before. Researchers keep seeking out new applications beyond legacy pharmaceuticals, like new polymer modifications and targeted biochemistry projects. That’s the kind of work that will keep this compound enmeshed in cutting-edge research.
Toxicity: Facts, Risks, and Real-World Impact
Nobody in the know treats methyl bromoacetate lightly — animal studies point to significant acute toxicity, with exposure routes including inhalation, ingestion, and skin absorption. Toxic effect thresholds get published in MSDS sheets and the warning signs of bromine toxicity stick in the minds of regular users: delayed pulmonary edema, nervous system depression, and corrosive tissue injury. The compound’s volatility bumps up the risk, making vigilance in storage, labeling, and daily practice essential. Some regulatory agencies place stringent controls on shipment and usage. Toxicology will only get more thorough, as future work finds better biomarkers and mitigation strategies. Ignoring the risk profile would slow research and jeopardize entire lines of development; facing it head-on has forced industry and academia to collaborate on safer alternatives and smarter, smaller-scale synthesis.
Looking Ahead: Where Methyl Bromoacetate Might Go from Here
Looking at the next chapter for methyl bromoacetate, the picture is all about evolving roles and smarter chemistry. The drive for greener, safer production lines is pushing suppliers toward cleaner synthesis, tighter waste controls, and closed-loop reaction designs. Chemists continue to work out new catalytic systems to either bypass or recover halides, spurred by regulatory and environmental pressure. New uses will tie this molecule to fields like protein bioconjugation and molecular diagnostics, where precision matters more than ever. That’s how methyl bromoacetate moves from a workhorse reagent of old-school chemistry to a linchpin in tomorrow’s scientific breakthroughs — not by resting on reputation, but by adapting to the ever-shifting boundaries of chemical research and human need.
What is Methyl Bromoacetate used for?
A Closer Look at Methyl Bromoacetate
Methyl bromoacetate sits on the shelves of many labs for a reason. In my chemistry days, this compound popped up in nearly every organic chemistry textbook but rarely in common conversation. Still, its usefulness can't be shrugged off. It has a strong reputation among researchers, especially for crafting new molecules quickly and efficiently. This colorless liquid carries the tag of an alkylating agent. That means it helps build bigger, more complex molecules by linking up small, active chemical pieces. People in the pharmaceutical and chemical research sector count on it for that very skill.
Why Do Labs Use Methyl Bromoacetate?
A lot of drug design starts with tiny tweaks. Scientists often need to attach a specific group to a molecule in a reliable way. Methyl bromoacetate handles this job. By donating its “bromoacetyl” group, it offers a shortcut in building intricate drug candidates, new pesticide leads, and other specialty molecules. Even academic research often leans on it for exploring new chemical territory.
The compound’s actions stretch further than medicine. Agrochemical businesses employ it to try out new active ingredients—think of herbicides or insect controls. Fine chemicals, like flavors, fragrances, and certain plastics, owe their development to those same reliable building skills. In my own undergrad lab, I saw it spark chain reactions that made designing experiments feel more like playing chess than following recipes.
The Trouble Beneath the Surface
Yet, gifts like these do not come without setbacks. Methyl bromoacetate earns respect and caution. It packs toxicity that can do plenty of damage if handled poorly. Skin and lung irritation, damage to the nervous system, even long-term cancer risks—all mark the warning labels. Most labs keep it locked up with careful training for the students and researchers who need to touch it. This isn’t just a bureaucratic measure. The dangers link to its reactive nature. What helps it join molecules so well, also turns it threatening to living tissues. The Environmental Protection Agency places strong restrictions on workplace exposure and disposal. Breathing in vapors, letting it touch the skin, or mishandling waste can all end badly. The need for gloves, fume hoods, and face shields is not simply best practice; it is non-negotiable.
Finding Safer and Smarter Approaches
Scientists keep searching for safer options, especially as demand grows for cleaner technology in laboratories. Researchers look for chemical substitutes that offer similar results without health hazards. Thanks to advances in “green chemistry,” more labs test alternatives that avoid bromine-based chemicals or cut toxic byproducts. Commercial providers now list “greener” alkylating agents aiming to replace methyl bromoacetate in routine labs. I remember mentors pushing me to consider not just what a chemical could do, but whether the cost to safety or health outweighed the convenience.
Methyl bromoacetate will likely stick around in advanced chemical synthesis, but its risks push us toward smarter habits. Wearing the right protection, using proper engineering controls, and never working alone spell out good lab culture. Open conversation on new research helps. Sharing successful replacements or improved methods keeps science moving forward—toward breakthroughs that don’t ask people to trade safety for results.
What is the chemical formula and structure of Methyl Bromoacetate?
Getting Down to the Basics
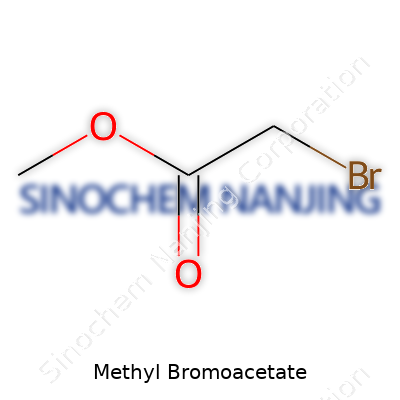
Methyl bromoacetate is a name that pops up in labs working with synthesis, pharmaceuticals, and organic chemistry teaching. Its chemical formula is C3H5BrO2. With this formula, there’s no confusion about the atoms involved — three carbons, five hydrogens, one bromine, and two oxygens. That single bromine atom changes everything, making this compound both a useful tool and a chemical to treat with care.
Structure: More Than a Simple Drawing
I can picture methyl bromoacetate as a short chain of three carbon atoms. Right at the start of that chain, one carbon carries a bromine atom and a hydrogen (a bromo group). The next carbon sits between that and an ester group at the far end. The methyl part comes from a -OCH3 attached through an ester linkage to the last carbon. Sometimes, in textbooks or during lectures, I sketched its condensed structure: BrCH2COOCH3. The placement of the bromine and the ester both open doors for reactions.
Why the Structure Matters in Real Life
The bromine atom makes methyl bromoacetate reactive. In the lab, this reactivity isn’t just a detail — it’s the reason people use it to build other molecules. It acts as an alkylating agent, something that attaches a piece of itself onto other molecules. In the classroom, professors use methyl bromoacetate to teach SN2 reactions, one of the most reliable ways to introduce new groups into a molecule. It’s hard to overstate how much practical organic chemistry relies on choices like this, where every built-in functional group counts.
Safety and Health: Respect Required
That extra snap in reactivity from the bromine also means risk. I’ve spent time in teaching labs where the difference between a careful student and a careless one showed up fast around chemicals like this. Methyl bromoacetate is toxic, with risks if you inhale its vapors or spill it on your skin. It can also be an irritant to the eyes. Beyond the general warnings, there’s an ethical angle here: experience in the lab offers a reminder that everyone needs to treat such reagents with respect. Good training, gloves, and fume hoods aren’t optional luxuries; they keep people healthy and projects on course.
Supporting Facts and Concerns
I recall reading safety data that highlights methyl bromoacetate’s dangers. According to the U.S. National Library of Medicine, the compound has been classified as a hazardous substance. It can cause coughing, shortness of breath, and headaches if inhaled. Contact with skin doesn’t just sting; it can be absorbed and cause systemic effects. For this reason, regulatory agencies in several countries include it on controlled chemical lists. Labs using this chemical must track quantities, secure their storage areas, and plan for emergencies.
Thinking About Solutions
Rather than avoid methyl bromoacetate, many have improved how they work with it. Substitutions aren’t always possible due to its unique properties, but ventilation, protective gear, and proper labeling make all the difference. Some institutions run drills for chemical spills, train staff and students on handling emergencies, and require up-to-date records of who uses the material. In my experience, a culture of accountability beats any single safety rule. Colleagues looking out for one another, calling out unsafe habits, and updating procedures when better information comes along, help everyone sustain a safe environment.
What are the safety precautions when handling Methyl Bromoacetate?
Why Safety Matters with Methyl Bromoacetate
Methyl bromoacetate is nothing to take lightly. This clear, colorless liquid belongs in a chemical class that brings real risk into any workspace. I remember my first experience with it in an organic synthesis project during graduate research. My lab supervisor warned me, “Respect the fumes, or you’ll regret it.” He was right. Within minutes of opening the bottle without proper ventilation, a colleague complained of a burning sensation and quickly left the room—luckily, with no lasting effects. This incident drove home a lesson about real-life consequences when ignoring established precautions.
Hazards and Exposure
People don’t always realize how harmful methyl bromoacetate can get. Just a small spill can send vapors rolling across the bench, regardless of how careful you think you’re being. While it’s easy to underestimate, methyl bromoacetate poses a dual threat—both as a skin contact hazard and as a vapor that irritates eyes, nose, and lungs. The molecule acts as an alkylating agent, which means it can damage proteins and DNA, not just for someone in the lab, but for anyone passing nearby.
Protective Equipment
No gloves, no deal. I’ve come to treat nitrile or neoprene gloves like entry tickets to the lab. Latex fails to provide protection here. Whenever handling methyl bromoacetate, I throw on proper splash goggles and a lab coat. Even a brief slip fills the air with a biting odor, so I always favor a chemical fume hood over a crowded workbench. Respirator use sounds dramatic, but in a spill or clean-up scenario, that piece of gear can make a real difference according to OSHA’s chemical safety guidelines.
Storage and Spill Management
Secure storage is often the most overlooked part. Methyl bromoacetate belongs in a tightly sealed glass bottle, kept in a safety cabinet dedicated for toxic reagents. I still recall a case when someone placed the bottle on a corridor bench—security noticed, and the lab got a stern warning about how easy it is for vapor to spread. Proper signage and segregation from acids or combustibles cut down on the potential for uncontrolled reactions.
Spills demand immediate action. In my lab, each team member runs periodic drills with spill kits—absorbent pads, neutralizers, and heavy-duty gloves. Every kit includes a written plan that doesn’t leave anyone guessing who’s responsible. Any splash on skin calls for a trip to the safety shower, no hesitation. The material safety data sheet recommends at least 15 minutes of water rinse. Having emergency eyewash stations within arm’s reach makes a significant impact during these incidents.
Training and Best Practices
Lab training does not end with a safety walkthrough. I encourage hands-on sessions for new workers, where they practice opening, measuring, and disposing of volatile materials. That builds habits, not just compliance. The goal is to get everyone thinking ahead: double-check container seals before moving them, keep only the quantity needed for one experiment, and store waste in labeled, vented bottles for prompt disposal through professional hazmat services.
Working Together to Stay Safe
Very few chemicals demand respect like methyl bromoacetate does. Personal discipline and team culture go hand in hand. It only takes one shortcut to learn a lesson the hard way. Following up-to-date references from organizations such as NIOSH and OSHA, and treating every step as essential, keeps everyone safe and research on track.
How should Methyl Bromoacetate be stored?
Understanding the Risks and Learning from Experience
I’ve seen plenty of labs treat chemicals like afterthoughts—shoved into any spare shelf space, tucked in unlabeled bottles, or stashed near the nearest vent. It’s easy to cut corners, especially with a schedule full of reactions and deadlines, but with methyl bromoacetate, carelessness can mean real trouble. This liquid won’t give much warning: its fumes pack a punch, and contact with your skin or eyes brings nasty burns. I once watched a co-worker forget his gloves while decanting it, only to end up with a red, burning splotch that haunted him for weeks. No need for a chemistry degree to figure out why treating this stuff with respect matters.
Why the Details Matter
Methyl bromoacetate soaks up moisture and attacks plastics and metals you’d expect to be immune. That means storing it in a glass container with a tight, sealed top is more than good practice; it’s smart survival. Anyone who’s ever watched an old plastic cap crumble off a reagent bottle knows the mess it leaves behind—mix that with highly toxic vapors, and the risk multiplies. Glass stands up well and keeps its shape, which matches what the literature and Material Safety Data Sheets have said for years.
Keep it cool, dark, and well away from any acids, bases, or water. Some folks ignore that advice and line solvent shelves with whatever’s handy. One mistake, a quick spill or a container left open, and those vapors drift out—throat burning, headache-building, serious stuff. A ventilated chemical cabinet with proper signage makes all the difference. It’s not just about following rules; it’s about making sure the space you work in doesn’t turn on you.
Accountability and Easy Access
Whenever a student asks me how to set up their first storage system, I point straight to clear labeling and inventory checks. Making sure every bottle is named and dated takes a few extra minutes, but if something gets knocked over or leaks, you’ll know exactly what you’re dealing with. In one university lab I visited, a half-used bottle of methyl bromoacetate got shoved behind old sodium hydroxide and left for months, only to be discovered in a sticky puddle. That sort of slip-up costs time and exposes everyone nearby to risk. Regular checks might seem fussy, but they keep everyone honest—and safe.
Room for Improvement
On the practical side, investing in proper fume hoods and spill kits cuts risk even further. If budget is tight, I always tell people to at least set up a small, vented enclosure; leaving methyl bromoacetate out in the open gives its fumes free rein. Wearing goggles and gloves is non-negotiable—lab coats, too, since one splash does serious damage. Easy access to emergency showers and eyewash stations saves more than pride; fast response often means the difference between a scare and a scar.
At the end of the day, getting the basics right—secure glass containers, cold and dry storage, good ventilation, and accountability with labeling—protects not just the person handling methyl bromoacetate, but everyone who shares the lab. It’s not just a safety box to check; it’s a habit that sticks with you long after the experiment ends.
What are the potential hazards or toxicity of Methyl Bromoacetate?
A Close Look at Toxicity Risks
Methyl bromoacetate looks like a simple clear liquid, but it packs some serious punch in both lab and industrial settings. This chemical helps make medicines, dyes, and different organic compounds, yet its hazards often fly under the radar. Just one whiff or splash can land someone in real trouble. It threatens health both quickly and over time, so the danger should not be shrugged off.
Let’s dig into what happens when this stuff enters the scene. Breathing it in feels like inhaling a sharp, biting vapor. Throat, nose, and lungs burn. Headaches settle in fast, even with just a little airborne exposure. Eyes sting, and skin becomes red or blistered if any droplets land. Most people don’t realize how quickly symptoms can creep up. I once watched a colleague pull off his gloves after cleaning a glassware spill—within minutes, his fingers had reddened and his breathing grew shallow. Quick action made a difference that day, but we learned to stay alert.
This chemical doesn’t just bother the nose and eyes; the real trouble comes after contact. The body absorbs methyl bromoacetate easily, and it won’t matter if it hits skin, eyes, or lungs. Systemic symptoms may show up later—nausea, dizziness, muscle weakness, and in bad cases, convulsions or collapse. Research shows that even low concentrations can damage the nervous system. Animal studies back this up: after exposure, rats showed tremors and changes in coordination, a clear sign of neurotoxicity.
Chemical Behavior and Accidental Release
Methyl bromoacetate doesn’t just linger; it drifts easily into the air, especially if a bottle breaks or a reaction runs hot. In a closed room, vapors can build up in minutes. This volatile property increases the risk of accidental inhalation. Older labs without proper fume hoods turn into hazard zones as a result. I’ve seen this firsthand: a tight window and fume hood off led to a week-long cleanup in a student lab, after a tiny spill caused throat irritation all the way down the hall.
This chemical reacts with water, too. The hydrolysis forms corrosive and irritating byproducts, including hydrobromic acid. Floors, tools, and waste containers corrode fast. A little moisture, a small leak, and you’re left with damaged equipment—or much worse, tissue burns.
Working Toward Safer Handling
Reducing the risk means building habits as much as adding barriers. Well-trained staff spot leaks or vapor before things get out of hand. Most experts recommend wearing splash goggles, gloves made from nitrile or neoprene, and using chemical fume hoods every time. Emergency showers and eye wash stations stay more than decoration; quick rinsing after accidental contact really matters, as it did for my colleague.
Regulators have set exposure limits for a reason. The threshold limit value sits low for a chemical like this—just a few parts per million already raise a red flag. Routine air monitoring and maintenance of lab ventilation cut down surprises. Some research groups now substitute less toxic alternatives in student labs, trading small losses in yield for much less risk. Skipping shortcuts and communicating about hazards keeps everyone safer and ensures that one careless mistake doesn’t spiral into something far worse.
| Names | |
| Preferred IUPAC name | Methyl 2-bromoacetate |
| Other names |
Bromoacetic acid methyl ester Methyl 2-bromoacetate 2-Bromoacetic acid methyl ester Methyl monobromoacetate Methyl α-bromoacetate |
| Pronunciation | /ˈmɛθ.ɪl ˌbroʊ.moʊ.əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | `96-32-2` |
| Beilstein Reference | 1209229 |
| ChEBI | CHEBI:41261 |
| ChEMBL | CHEMBL133778 |
| ChemSpider | 16521 |
| DrugBank | DB04261 |
| ECHA InfoCard | echa.infoCard: "03b1a8e4-5abe-4c5c-8d70-814503da9e93 |
| EC Number | 209-150-3 |
| Gmelin Reference | Gm. 8363 |
| KEGG | C19268 |
| MeSH | D008763 |
| PubChem CID | 7519 |
| RTECS number | AG9625000 |
| UNII | 1Z41ODP7QF |
| UN number | UN2660 |
| CompTox Dashboard (EPA) | DTXSID8021393 |
| Properties | |
| Chemical formula | C3H5BrO2 |
| Molar mass | 167.99 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | pungent |
| Density | 1.594 g/mL at 25 °C (lit.) |
| Solubility in water | Soluble |
| log P | -0.2 |
| Vapor pressure | 12 mmHg (20°C) |
| Acidity (pKa) | 2.79 |
| Basicity (pKb) | 2.53 |
| Magnetic susceptibility (χ) | -9.64×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.456 |
| Viscosity | 0.97 cP (20°C) |
| Dipole moment | 2.20 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -389.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -722.6 kJ/mol |
| Pharmacology | |
| ATC code | Methyl Bromoacetate does not have an ATC code. |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. H317: May cause an allergic skin reaction. H331: Toxic if inhaled. H411: Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 49 °C (120 °F; 322 K) |
| Autoignition temperature | 410°C |
| Explosive limits | Lower: 2.5% Upper: 19% |
| Lethal dose or concentration | LD50 oral rat 95 mg/kg |
| LD50 (median dose) | LD50 (median dose): 175 mg/kg (oral, rat) |
| NIOSH | PS9450000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Bromoacetic acid Methyl chloroacetate Ethyl bromoacetate Methyl iodoacetate Methyl acetate |

