Methyl Acrylate: Unpacking the Backbone of Modern Chemistry
The Roots and Rise of Methyl Acrylate in Industry
Methyl acrylate didn’t step into the world quietly. Back in the early twentieth century, chemists began exploring acrylate esters to answer mounting needs for flexible, durable materials. It proved its value early on, catching the attention of manufacturers obsessed with improving everything from glass alternatives to adhesives. Its popularity only snowballed as the plastics industry spread across the globe post-World War II, driven by the desire to build lighter, tougher products. My own work in a polymer lab hammered home the real weight of this compound—just one variable in a recipe, but with the power to shift how a material bends, sticks, or shrugs off water.
Getting to Know Methyl Acrylate
Think of methyl acrylate like a sharp, pungent tool in the chemist’s toolbox. As a clear, colorless liquid, it jumps into action in many industries hungry for speed and reliability. Its low boiling point makes it easy to distill, and its flammable nature is a double-edged sword—convenient for some processes, but only if you’re careful not to set the place ablaze. Handling it for the first time, the smell almost knocks you off your feet, and you quickly learn an ungloved finger is a mistake you only make once.
Physical and Chemical Reality of Methyl Acrylate
This material has a boiling point around 80°C and melts below freezing, landing it in a narrow liquid range that matters for process engineers. It dissolves easily in most organic solvents and can be mixed with water in limited amounts. In chemical terms, its acrylate backbone holds a reactive double bond that loves to connect with itself and other molecules—a trait that underlies its key applications in resins and coatings. Looking at reactivity, methyl acrylate races ahead in polymerization, explaining why it’s often stored with inhibitors that stop runaway reactions. What’s fascinating from a scientific perspective is how that same double bond lets you modify the molecule in a thousand ways, making it a launchpad for further tweaks and specialty chemicals.
Technical Specs and Label Talk
Anyone reading a barrel of methyl acrylate’s label learns quickly why chemistry requires respect and routine. You deal with UN numbers, hazard pictograms, and plenty of reminders to avoid open flames and skin contact. The paperwork isn’t just legal red tape—it’s about protecting workers and the wider community from exposure, spillage, or worse. In every facility I’ve walked through, protocols around ventilation and containment seem excessive until you hear stories of what happens if they’re ignored. Specification sheets focus on purity, acidity, and stabilizer content, all crucial for maintaining quality and preventing side reactions during storage and transport.
How Methyl Acrylate Comes to Life
Modern producers favor a streamlined process that reacts acrylic acid with methanol, using strong acids as catalysts. The chemistry looks simple on the surface—mix, heat, separate—the real challenge comes with purifying the product and handling vapor that could spark or leak. The technical teams involved in scaling up from test tube to tank truck deserve more credit than they get. Even slight shifts in raw material quality or process conditions can leave behind unwanted by-products, making tight quality control non-negotiable.
Chemical Reactions: Methyl Acrylate as a Starting Point
Methyl acrylate plays the role of an eager participant in reactions that graft new functionalities or build long chemical chains. Radical polymerization dominates its industrial use, generating acrylate polymers that end up in paints, films, and flexible packaging. In the lab, you watch it open doors to specialty esters, modified resins, and new copolymers by tweaking what gets added to the mix or by changing the reaction pressure and temperature. I’ve seen how swapping this single monomer for a slightly different one can turn a brittle plastic into a resilient film or a sticky glue—proof of the transformative punch locked into a simple molecule.
Other Names, Same Substance
Methyl acrylate hides behind several aliases. Some call it MA or acrylic acid, methyl ester. Old texts sometimes use less familiar terms, but in practical settings, the short names stick for clarity. This matters because in global trade, misunderstandings over naming conventions have tangled more than a few shipments—or led to processing errors that cost companies dearly.
Keeping Safety Front and Center
No one handles methyl acrylate without proper gear. It burns exposed skin, and inhaling its vapors causes headaches, coughing, and worse. Ventilated hoods—and in some settings, full-face respirators—aren’t optional. Fire departments don’t take kindly to leaky tanks, either, because methyl acrylate spills can ignite with ease and generate toxic smoke. Strict handling also follows from environmental impacts—uncontrolled leaks don’t just harm workers but pollute air and water around plants. That’s why incidents motivate regulators and company leaders to invest in training and technology for immediate containment and remediation.
Where Methyl Acrylate Ends Up
Few chemicals boast a resume as broad as methyl acrylate. It gives plastics their stretch in packaging films and pressure-sensitive adhesives. Textile finishing agents draw on it to keep garments soft or wrinkle-free. Water treatment operators use it to grab hold of unwanted ions and dirt. In paints and coatings, it improves durability and weather resistance. Biomaterial research teams experiment with it in controlled drug delivery and medical devices, since its backbone offers a flexible route to novel polymers with tailored properties. I remember collaborating with a coatings manufacturer upset with cracking and brittleness in a new line—switching a portion of their monomer mix to methyl acrylate fixed the problem almost overnight.
Pushing the Limits: Research and Development
Researchers keep digging for new uses and better processes. Advances in green chemistry now target safer, cleaner production routes, ditching heavy-metal catalysts and minimizing waste. Polymer science moves forward too, as teams develop “smart” materials that respond to stimuli like heat, light, or pH, all thanks to tuning the properties of methyl acrylate-derived chains. Several research groups focus on improving how these polymers break down or recycle, a sore point as society wrestles with plastic waste clogging up landfills and waterways.
Toxicity and Public Health Questions
There’s no sugar-coating the hazards. Animal studies point to acute toxicity, especially with high-level exposure. Chronic effects remain a topic of ongoing research, with legitimate questions about cancer risks and long-term health impacts for exposed workers. Robust monitoring and personal protection stay critical in every workplace, especially as epidemiology data sometimes lags behind real-world exposure. Neighborhoods near production sites also demand transparency, pushing authorities to set and enforce strict air and water quality limits.
Methyl Acrylate’s Way Forward
As the world calls for greener, safer chemistry, methyl acrylate finds itself both a challenge and a solution. Demand won’t disappear, since industries from electronics to construction lean on its versatile backbone, but opportunities lie in cleaner synthesis, improved recycling paths, and smarter end-use products. Academic teams explore bio-based acrylates as substitutes, and start-ups trial new methods to lower hazards in transport and storage. From experience, the future of this chemistry depends as much on honest risk assessment and open conversation as on technical fixes. Progress grows from respecting the lessons of the past without closing the door to smarter ways of harnessing the power in a drop of methyl acrylate.
What is methyl acrylate used for?
The Glue Behind Countless Products
Walk down any hardware or grocery store aisle and you pass by dozens of items that rely on methyl acrylate, often without even spotting its presence. I spent years working in a facility that produced coatings, and every day I saw tankers pull in loaded with methyl acrylate. This chemical sits at the center of a huge family of tough, flexible, and long-lasting plastics and coatings.
Making Things Stick, Seal, and Shine
People know super glue, but they don’t always realize its key power comes from chemicals like methyl acrylate. Manufacturers blend this liquid into adhesives for packaging, woodworking, and automotive parts. It helps create glues that bond fast and hold tight. Building contractors often trust adhesives based on methyl acrylate for weatherproof jobs — sealing windows, roofing, or floors where moisture threatens less durable materials.
Paints and coatings draw on methyl acrylate for resilience. If you have ever cleaned graffiti from a wall or scrubbed mud off house siding, you’ve seen how modern coatings shrug off abuse. Protective films formed by acrylics reach their stretch and strength thanks to methyl acrylate in the base formula.
Plastics that Touch Everyday Life
Shopping bags, food packaging, and clear display cases depend on plastics like polyacrylate and polymethyl methacrylate that trace their genealogy to methyl acrylate. The plastics keep food fresh, seal electronics against moisture, and provide the see-through shields in banks and buses.
This chemical shows up in those soft squeezable bottles used in lotions and hand sanitizers. Flexible enough to bounce back when squeezed, these plastics resist cracking and keep out germs. A few years back, I watched a packager battle issues with brittle bottles until switching to an acrylic copolymer that owed much of its flexibility to methyl acrylate. It made a world of difference in shelf life and durability.
Textiles and Water Treatments
Textile workers count on methyl acrylate for making brighter, more flexible fabrics. The chemical goes into finishes that help garments hold color, shed water, and feel smoother. Clothing manufacturers, especially those producing athletic wear, target these acrylic blends for lightweight performance fibers.
In water treatment, methyl acrylate lays the foundation for polymers that filter dirt from drinking water or catch tiny solids in sewage plants. As the need for cleaner water rises, chemical engineers lean harder on sturdy, reliable polymers built around this key ingredient.
Balancing Use and Safety
All this utility comes with a real need for care. The vapors from methyl acrylate can irritate lungs, eyes, and skin. Safety teams in the plants where I worked double-checked monitoring systems and insisted on ventilation and protective gear. Regulators in the U.S. (OSHA, EPA) and Europe set exposure limits and push for containment, which helps keep workers and consumers safer.
Alternatives for some uses exist, but most don’t yet match the balance methyl acrylate brings: toughness, flexibility, affordability. Green chemistry researchers keep searching for safer and more sustainable polymers. Some new acrylate variations use bio-based ingredients or break down faster in the environment, aiming at the same performance targets.
Why Consumers Should Care
Methyl acrylate helps package food safely, keeps buildings sealed, and supports industries from automotive to cosmetics. The scale of demand means responsible management matters at every point, from plant floor to end-user. Progress in worker safety and greener chemistry benefits all of us. Shoppers who ask about packaging or check for low-VOC (volatile organic compound) paints encourage companies to raise their standards. As someone who has worked in chemical manufacturing and now pays attention to what goes into the goods I buy, I can tell you — every question and every upgrade makes a difference.
What are the safety precautions when handling methyl acrylate?
Understanding the Risks Up Close
Methyl acrylate shows up across many industries, including plastics, coatings, adhesives, and textiles. It might seem harmless in a drum or bottle, but experience in chemical environments makes one thing clear: you can’t cut corners with this stuff. Its strong, almost sweet odor signals a hidden danger—this liquid can do real harm if ignored. Skin contact can trigger quick, nasty irritation and absorption. Eyes take an even harder hit, often with lasting consequences. Inhalation can set off coughing, headaches, dizziness, even more serious health problems if you don’t get out of the way. Observing a careless coworker once with a failed glove demonstrated just how fast symptoms creep up. Safety starts before one drop leaves the container.
PPE and Controls: Basics No One Should Skip
Basic rules save lives. Chemical-resistant gloves, splash goggles, and face shields protect skin and eyes. Old habits die hard, but leaving regular latex or vinyl gloves behind and choosing the right material—nitrile or neoprene—keeps hands safer for longer. Lab coats, aprons, and reliable closed footwear create an extra layer between worker and chemical. Respiratory risks lurk especially in tight spaces. Using properly fitted respirators with organic vapor cartridges stops dangerous fumes before they hit the lungs.
Ventilation: The Unsung Hero
Never underestimate air movement. Working in a confined or poorly ventilated lab once taught everyone involved a harsh lesson. Fume hoods and well-designed local exhaust systems draw methyl acrylate vapors away, cutting exposure. No hood? Fans and open windows help, but nothing beats mechanical ventilation built into the workspace. Companies like Dow and BASF (who manufacture bulk methyl acrylate) typically invest heavily in these controls—small shops should learn from them. Routine checks ensure their systems actually work instead of just ticking a box.
Handling and Storage: More Than Common Sense
Transferring methyl acrylate uses grounded, spark-proof equipment. The flash point sits dangerously low—static, faulty electronics, or a tiny flame spell disaster. Drip trays, absorbent mats, and spill kits should stay within reach. Sealed, labeled containers make accidental exposure less likely. Personal experience says never trust an unlabeled drum, especially with chemicals notorious for similar smells or appearances.
Spills and Emergencies: Practiced, Not Panicked
Even the most careful setups face spills or leaks. New hires often freeze up unless they’ve seen a drill done properly. Training builds muscle memory that carries people through—the rush for the eyewash station, rinsing for fifteen minutes, heading straight for medical help. Neutral absorbents and non-sparking tools come out of supply rooms—not paper towels or bare hands. Companies that run regular emergency response drills always recover quicker.
Bigger Picture: Training and Culture
Of the lessons chemical safety has taught over years, culture makes the biggest difference. Documented procedures belong in every company, kept practical and lived—not hidden in binders. Regular walk-throughs, short check-ins, and honest reporting must edge out fear or complacency. Regulations from OSHA and similar agencies exist for a reason, but personal buy-in keeps colleagues safe. Frequent review and reminders, not just annual retraining, reinforce good habits. Everyone learns from their own small mistakes, but group commitment means fewer mistakes, plain and simple.
What is the chemical formula of methyl acrylate?
What Is Methyl Acrylate?
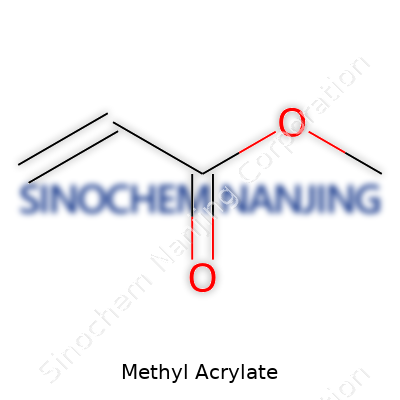
Methyl acrylate pops up in so many products that most folks never realize how often they’re around it. It’s an organic compound used heavily in making plastics, paints, textiles, and adhesives. Its chemical formula—C4H6O2—captures a world of industry tucked into one short string of letters and numbers. This small molecule packs a punch, with a structure that lets it react easily with others, building longer chains. That property turns it into a vital building block in many manufacturing plants.
Why Knowing Its Formula Matters
It’s worth more than just trivia for chemistry students. The makeup of methyl acrylate tells chemists a lot about what it can do, and, just as important, about its hazards. Methyl acrylate contains a double bond between two carbon atoms and an ester group. That double bond means it’s quick to react, which is a blessing for polymer production but also raises concerns about accidental reactions. The formula guides safety data sheets, shipping rules, and labels found in factories worldwide—one mistake in that formula could put real people in danger.
I’ve helped train new lab workers, and it always helps to connect the written formula to what someone handles in the lab. Seeing that oxygen atom, for instance, hints at possible reactions, including polymerization and combustion. People who know their chemicals by formula learn to spot trouble long before a problem develops.
Real-World Examples and Hazards
C4H6O2 looks harmless on paper, but methyl acrylate can irritate skin, eyes, and lungs at very small exposures. Around 50 parts per million in the air can start bothering people, which is why plant engineers set up strong ventilation and monitoring systems. Polymer workers remember to check their gloves and goggles, all because they know those atoms rearrange themselves into something sticky and sometimes, hazardous.
Take one example from the early 2000s: an operator in a textile plant once forgot to cap a container of methyl acrylate. Not only did the fumes cause a temporary evacuation, the cleanup forced a full halt in production. No one got hurt, but the schedule slipped and the costs stacked up. This wasn’t just because of a spill—it was a direct result of underestimating the power hiding behind a simple formula.
Better Practices and Safer Handling
Companies who value safety keep their teams well-versed in chemical formulas and what they represent. I’ve learned that strict labeling and regular training make a difference, but that’s just a start. Installing up-to-date ventilation, using sealed containers, and selecting personal protective equipment suited for acrylates bring down the risk. No one can avoid all accidents, but understanding what C4H6O2 means pushes people to treat even a simple flask with due respect.
Looking beyond the lab, researchers hunt for safer alternatives or systems that neutralize methyl acrylate faster in case of a leak. Sharing findings and best practices with other organizations keeps people safer across the whole supply chain. That’s the value of knowing a chemical’s formula inside out—it isn’t just about numbers and letters, it’s about keeping people and communities safe while pushing industries forward.
How should methyl acrylate be stored?
Why Proper Storage Isn’t Optional
Methyl acrylate can pose a real threat in a careless storage setup. This isn’t just about ticking boxes on a safety audit, but making sure that everybody in the facility gets home in one piece. I once worked at a site where a single loose drum nearly caused a disaster. No one felt proud explaining that to the emergency crew. Storing chemicals correctly protects both people and expensive equipment.
Key Hazards of Methyl Acrylate
This compound won’t let you off easy if you make mistakes. Methyl acrylate gives off fumes that can irritate skin, eyes, and lungs. It ignites at relatively low temperatures and reacts fast with oxidizers or acids. Experienced handlers know to watch for polymerization—think of containers heating up on their own and swelling until something gives. That risk alone means taking the time for proper storage makes sense every single day.
Containers That Make a Difference
Steel drums with tight-fitting, corrosion-resistant seals have always been the standard in every job I’ve had. Glass or plastic containers crack, warp, or sometimes even dissolve with chemicals like this. The label must always stay visible and readable—scribbles and peeled corners can cause costly accidents. You don’t want to leave questions about which drum holds what, especially during a busy shift.
Handling Temperature and Ventilation
Heat turns a safe chemical store into trouble fast. Methyl acrylate belongs in a cool room set away from direct sunlight, hot motors, or steam pipes. I’ve seen more than one facility pay for new ventilation after noticing that the old system couldn't clear out fumes. Good air flow means workers can breathe easy, and sensors catch leaks before they grow. Automated ventilation systems connected to alarms have become standard for good reason—nobody has time to chase down a faint odor in a full warehouse.
No-Nonsense Storage Rules
Keep methyl acrylate away from acids, alkalis, peroxides, and oxidizers. Even shelf neighbors matter, as accidental mix-ups have led to warehouse fires more than once. Whiteboards or digital tracking help catch errors in placement. Not every site uses technology, but even old-school spreadsheets beat “trusting your memory.” Fire-resistant storage cabinets separate volatile chemicals from the rest and buy precious time if something goes wrong.
Sensible Safety Companions
Storing this compound near spill kits, absorbents, and eye-wash stations is not an overreaction—it’s common sense. I keep an emergency checklist taped inside the cabinet; everyone deserves a reminder before things get busy. Every worker should know the evacuation routes and have access to protective gloves, goggles, and face protection. Refresher training after an incident isn’t overkill. It’s how teams avoid repeating close calls.
Learning from Incidents
The industry holds plenty of cautionary tales. Years ago, poor labeling and rushed placement led to a container rupture and a hot afternoon spent in hazmat suits. Every time a team completes a storage audit, you know they learned something from stories like that. You can’t cut corners without risking health, reputation, or even someone’s life. Safer storage means better business and fewer regrets afterward.
What are the health hazards of methyl acrylate exposure?
What Makes Methyl Acrylate So Concerning?
Factories use methyl acrylate every day in paints, adhesives, textiles, and coatings. The chemical brings flexibility to plastics and helps glue stick to surfaces, but at a cost most folks never see. Methyl acrylate hits the body hard. Even a little in the air can sting your eyes, burn your nose, and set off a cough. Larger amounts do more damage. Exposure often slips under the radar until a headache or dizziness creeps in. Medical journals have reported chest pain, trouble breathing, and even nervous system problems after workers breathed it in on the job.
Pain at First Contact
Skin contact gives the first warning. I’ve worked in shops where workers splash a little on their hands. The burning comes quick. Redness and swelling follow. Folks who touched the liquid day after day grew thick, cracked skin. One coworker complained of blisters that broke open after a shift. Eyewashes and gloves took some sting out, but not everyone used them each time. Many shrugged it off, saying, “It’s just part of the job.” Over time, these small exposures built up into raw, peeling hands that couldn’t grip a wrench.
The Unseen Damage From Breathing Fumes
Methyl acrylate fumes fill closed rooms. The sweet, sharp smell lingers in the nose and throat, making it hard to breathe deep. Researchers from the Centers for Disease Control and Prevention (CDC) found that exposure leads to coughing, wheezing, and asthma-like symptoms. Repeated inhalation has been linked to long-term lung function loss. In my experience, after eight hours in a room with poor airflow, even the tough old-timers developed sore throats and raspy voices. Some felt dizzy or nauseous yet pushed through, worried about missing hours on their paycheck.
Risk Beyond the Factory Floor
Risk does not stop with workers on the line. Methyl acrylate can seep into air, soil, and water if spills or leaks happen. I lived near a plastics plant where an accident sent a chemical plume drifting toward homes. Residents developed headaches, eye irritation, and sore throats in just a few hours. Local clinics saw cases spike. Long-term effects from such incidents still need more study, but short-term misery becomes clear fast.
Solutions That Can’t Wait
Simple steps protect health. Ventilation stands out. Open windows, run fans, and set up exhaust systems so fumes do not hang in the air. Personal protective equipment is not negotiable: strong gloves, goggles, and proper respirators cut down on skin and lung risk. Clean up spills fast. Employers should offer regular safety training that shows real-world risks and not just check boxes. Medical monitoring helps catch problems early. If companies share honest data with workers, trust builds—no one should feel blindsided by dangerous exposure.
Companies might chase profit, but skipping safety ends up costing more in medical bills, sick days, and broken trust. Telling the truth about chemical dangers gives every worker a fair chance at good health, not just those lucky enough to avoid a spill or a leak. Real protection starts with seeing risk as more than a line of fine print on a label.
| Names | |
| Preferred IUPAC name | methyl prop-2-enoate |
| Other names |
Acrylic acid methyl ester 2-Propenoic acid, methyl ester Methyl propenoate MA Methylacrylic acid |
| Pronunciation | /ˈmɛθ.ɪl ˈæk.rɪ.leɪt/ |
| Identifiers | |
| CAS Number | 96-33-3 |
| 3D model (JSmol) | `C=CC(=O)OC` |
| Beilstein Reference | 635068 |
| ChEBI | CHEBI:4636 |
| ChEMBL | CHEMBL149786 |
| ChemSpider | 8192 |
| DrugBank | DB04169 |
| ECHA InfoCard | 22fbd515-3c7d-4186-84c6-1d237f21ba58 |
| EC Number | 201-185-2 |
| Gmelin Reference | Gmelin Reference: **5547** |
| KEGG | C01780 |
| MeSH | D000426 |
| PubChem CID | 10797 |
| RTECS number | GF1925000 |
| UNII | 4JO701U16R |
| UN number | UN2031 |
| Properties | |
| Chemical formula | C4H6O2 |
| Molar mass | 86.09 g/mol |
| Appearance | Colorless liquid with a characteristic odor. |
| Odor | Acrid, pungent |
| Density | 0.956 g/cm³ |
| Solubility in water | 7.5 g/100 mL (20 °C) |
| log P | 0.739 |
| Vapor pressure | 86 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 13 |
| Basicity (pKb) | pKb ≈ 12.5 |
| Magnetic susceptibility (χ) | -7.67×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.401 |
| Viscosity | 0.601 mPa·s at 25°C |
| Dipole moment | 1.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 276.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -425.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1782 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H317, H332, H335, H341, H351, H412 |
| Precautionary statements | P210, P261, P273, P280, P301+P310, P305+P351+P338, P304+P340, P403+P233, P403+P235 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 10 °C (50 °F) |
| Autoignition temperature | ```string 393 °C ``` |
| Explosive limits | 2.8% - 16.3% |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 870 mg/kg |
| NIOSH | NIOSH: MAH |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Methyl Acrylate: 10 ppm (parts per million) |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Acrylic acid Methyl methacrylate Ethyl acrylate Butyl acrylate Ethyl methacrylate |

