Methyl Acetoacetate: Chemistry’s Swiss Army Knife Faces New Frontiers
Digging into Its Roots
Methyl acetoacetate has a story that echoes the changing tides of organic chemistry. In the late nineteenth century, as chemists learned to master the synthesis of versatile building blocks, this simple beta-keto ester quickly became a favorite. Its structure—two carbonyl groups separated by a methylene bridge—gave it a reactivity that dreamers in a flask could count on. German and French chemists spearheaded early investigations, not just out of academic curiosity but because factories already relied on simple esters for flavorings, plastics, and dyes. In labs I’ve worked in, old leather-bound journals sit on dusty shelves, many with yellowed pages chronicling the earliest preparations of methyl acetoacetate. That tradition still runs strong, with modern researchers circling back to old methods as the need for cleaner, greener synthesis grows.
What Makes Methyl Acetoacetate Tick
For anyone who has cracked open a bottle of methyl acetoacetate, the experience registers quickly—colorless, low-viscosity liquid with a fruity odor hinting at its role in flavors and fragrances. Simple esters often get attention for their pleasant smells, but the secret to methyl acetoacetate’s widespread use lies beneath the surface. The molecule carries both an ester and a ketone, granting it impressive flexibility in chemical reactions. Specific gravity clocks in around 1.07 g/cm³, which is enough to remind handlers that this isn’t just another food-grade additive. Its boiling range stays within the 169–171°C neighborhood, and it shows strong solvency in many organic liquids, making it a go-to for blending, dissolving, or carrying other molecules. Its structure and function cover so much ground that it carves out a niche in synthesis as both a starting point and an intermediate.
Technical Notes, Real-World Labels
Labeling isn’t always as clear-cut as industry guides suggest. In a chemical storeroom, methyl acetoacetate carries a slightly ominous collection of warning stickers. Vapors can irritate eyes and lungs, so common sense points to ventilated racks and safe-handling reminders. Most standard containers mention purity upwards of 99 percent, keeping impurities in check for those who measure yields in fractions of a percent. Its CAS number pops up on digital inventories everywhere, as regulators and labs alike treat such numbers as shorthand for safety audits, disposal documentation, and supply-chain traceability. Labels set professional boundaries but don’t capture the day-in, day-out vigilance hardwired into every technician who measures and mixes the stuff.
Chemistry Behind the Curtain
Making methyl acetoacetate sits right in the sweet spot between straightforward and clever. Most processes start with diketene and methanol. This approach took off for good reason—it offers predictability with relatively mild conditions, making it popular in settings ranging from industrial reactors to academic setups. Some outfits lean on the Claisen condensation, where ethyl acetate plays a starring role alongside sodium. Others swing back to older methods, especially in regions where access to modern reagents hits snags due to logistics or regulation. Every route brings questions about cost, yield, waste, and compliance, and chemists often return to bench-scale experiments for answers when large-scale production needs tweaking to satisfy both scientists and policymakers.
What Happens When You Put Methyl Acetoacetate to Work
Put methyl acetoacetate under the right conditions, and it steals the show. Its methylene bridge is unusually acidic for an ester, letting it form enolates and undergo a long menu of addition, substitution, and condensation reactions. Chemists reach for it to build up molecular complexity in pharmaceuticals, agrochemicals, and specialty polymers. The molecule stands up under the spotlight in the acetoacetic ester synthesis, a classic method for constructing new carbon-carbon bonds. In the hands of curious researchers, it’s been twisted into cyclic ketones, heterocycles, or even amino acid derivatives. Each modification leverages the molecule’s structure, providing a scaffold that can swing from flavors to medicines within a few careful steps.
Synonyms and Product Aliases
Walk through a chemistry catalog, and methyl acetoacetate will show up under a handful of names: MAA, 2-oxo-3-butanoic acid methyl ester, methyl 3-oxobutanoate, or even methyl β-ketobutyrate. The shifting vocabulary reflects regional habits, legacy naming conventions, and years of different research traditions talking past each other. Still, no matter the name, the signature properties and core reactivity remain.
Workplace Hazards, Safety Gaps, and Staying Sharp
Daily handling of methyl acetoacetate means keeping safety protocols within arm’s reach. The ester isn’t the most dangerous chemical on the shelf, but its flammability and potential to irritate mucous membranes make every spill a learning moment. I’ve seen training sessions where new hires practice with colored water, treating every drop as hazardous for muscle memory’s sake. Safety data sheets matter, but institutional culture does more to prevent accidents—peer checks, open communication about near misses, and routine updates as standards shift. Regional regulations ask for adequate PPE, but the real safeguard turns out to be colleagues willing to step in and remind each other about gloves, goggles, and fume hood doors. Even as self-contained respirators sit in emergency cabinets, most labs experience their closest calls through complacency rather than catastrophic error.
Industry Reach: Versatility Outpaces Stereotypes
Many know methyl acetoacetate through its supporting role in the fragrance and flavor industry. Combinatorial chemistry has shown how a few changes to its structure result in a spectrum of tastes and aromas—from tart apple to pear. Yet the compound shoulders serious responsibility outside food labs. In pharmaceutical plants, it supplies key steps in the creation of painkillers, sedatives, and antibiotics. Agricultural science teams, pressed by the demand for new crop protection agents, use it to create active ingredients that fight weeds, insects, and fungal blights. Studies run out of university and government consortia consistently highlight this molecule as a precursor for novel dyes, specialty coatings, UV absorbers, and even complex polymers. This wide application base doesn’t let up and shows no sign of waning, thanks in part to the geographic spread of the chemical industry beyond North America and Europe.
Research, Toxicity, and What’s Around the Bend
Decades of research haven’t stopped the questions about methyl acetoacetate’s safety. Toxicological profiles show moderate acute toxicity, transient irritation, and low risk of chronic harm, though researchers emphasize routes of exposure and cumulative effects can turn gentle chemistries risky under lax conditions. Studies funded by consortia track metabolic breakdown products, tying regulatory limits to measured systemic impacts in animal models. The European Chemicals Agency and American Environmental Protection Agency periodically revisit recommendations, nudged by new inhalation studies or environmental fate reports. Most urban wastewater treatment systems struggle to perfectly remove organic esters, so some data tracks persistence and bioaccumulation in aquatic environments, raising concerns among conservation groups. Occupational health studies—powered by unions or advocacy organizations—press for stronger controls, especially in countries without high compliance standards. All of this marks a shift in attitude; what used to pass with a wink now gets serious attention from plant managers and safety officers.
Charting the Future: Green Chemistry and Emerging Uses
Every few years, a surge of innovation brings new focus to molecules like methyl acetoacetate. Green chemistry drives a lot of that interest—industry leaders set ambitious goals to cut waste, lower emissions, and replace hazardous feedstocks. Researchers look for catalytic methods that skip corrosive bases or work at ambient temperatures, taking cues from nature’s enzymes. Patent filings spike as startups and universities find new reactions, sometimes borrowing biotechnological approaches to skirt older, dirtier methods. Demand for biodegradable plastics and advanced films spotlights methyl acetoacetate as a functional monomer. A younger generation of chemical engineers anticipates pressure from both regulators and consumers, prompting broader scrutiny over every raw material’s life span from cradle to grave. Investment in alternative synthesis methods, environmental analytics, and human health impact studies ensures that even a familiar molecule stands at the edge of transformation. What once counted as a workhorse of twentieth-century chemistry just might become a talking point for the next big shift in industry sustainability.
What is the chemical formula of Methyl Acetoacetate?
A Close Look at a Key Chemical
Methyl acetoacetate shows up in a lot of places—labs, industry sites, and even some food labs. Its chemical formula, C5H8O3, might look like just a string of numbers and letters, but there’s a story in those bonds. If you’ve ever held a small bottle of this stuff, you know right away this isn’t some rare lab oddity; it’s central to making flavors, pharmaceuticals, and certain plastics work the way they should.
C5H8O3 unlocks a lot of possibilities for chemists and manufacturers. Even as a high school student, the idea that single molecules shaped all sorts of products fascinated me. Here, two carbonyl groups—the stuff that helps reactions move—hang on the backbone, letting this methyl ester act like a Swiss Army knife. Each atom’s position isn’t just a detail; the way they arrange themselves gives methyl acetoacetate its usefulness in making both adhesives and medicines.
Why Methyl Acetoacetate Matters
Plenty of factory lines owe their efficiency to this compound. Factories crank out flavors that mimic natural fruits, aided by C5H8O3. In the pharma world, it builds blocks for barbiturates and vitamins. Chemists like its easy reactivity. Even in my undergrad labs, one basic reaction stood out: using methyl acetoacetate as a nucleophile in alkylation. The lab smelled, reactions bubbled, and each molecule played its part without fanfare.
Think about flavors. Some of the fruity notes in drinks trace back to methyl acetoacetate as a precursor. Those candies and soft drinks don’t owe their taste just to natural ingredients—this molecule slips quietly into recipes. In medicine, it barely gets mention when pills change hands at a pharmacy counter, but its presence in the manufacturing chain shapes outcomes for patients every single day.
Safety and Environmental Concerns
Chemicals like this come with baggage. Even strong-smelling esters bring their own risks. Breathing vapors in a closed space can make someone dizzy. Methyl acetoacetate needs careful handling, since spills mean more than a mess—the health of workers and the wider environment faces threats if waste isn’t managed. I remember seeing barrels of chemicals lined up behind a production facility. One small leak left a sharp, sweet odor in the air—an easy temptation to overlook, but a real problem if safety teams don’t keep watch.
Regulations step in here. Companies get challenged to create better containment, improved training, and quick clean-up strategies. The industry has seen fines and regulatory pressure push toward closed handling systems and good ventilation. Personally, working in a lab with basic protective gear gave me respect for how quickly a lapse could turn routine work into a crisis.
Where Chemistry Can Go Better
Green chemistry methods set new expectations for industries that use methyl acetoacetate. Switching solvents, cutting waste, and monitoring emissions show up more in both research papers and practical guidelines. Engineers and chemists focus not just on yield, but also on how to keep reactions efficient and safe for both humans and the ecosystem. This kind of mindfulness can cut costs and health complaints, and it shows a better way of working with chemicals that touch so many parts of our lives.
Looking at methyl acetoacetate, it’s clear how even a small molecule can shape industries, jobs, and health. What sits inside that neat C5H8O3 package connects science classrooms to factory floors, and links safety protocols with the everyday things we taste and use.
What are the typical uses of Methyl Acetoacetate?
Practical Value in Chemistry Labs
Every chemist gets acquainted with Methyl Acetoacetate early in their career, usually because it’s such a versatile building block. This clear liquid has a faint, fruity smell, and it shows up again and again in synthesis projects for one important reason: it plays well with other chemicals. Imagine you’re in an organic chemistry lab, staring at your list of possible starting materials. If there’s a need to make heterocyclic compounds, vitamins, or dyes, this reagent stands out. It works as a bridge between basic starting compounds and much more complicated structures. Those who’ve spent hours pipetting can tell how the intermediate steps made possible by Methyl Acetoacetate help shape crucial molecules.
Pharmaceuticals: Quiet Helper Behind the Scenes
Most people wouldn’t guess that part of their headache relief, antibiotics, or asthma medication might owe its existence to a colorless, unassuming liquid. In pharmaceutical manufacturing, it’s a staple for producing active drug molecules like antipyretics or barbiturates. For instance, the core structure of many over-the-counter drugs comes from reactions where Methyl Acetoacetate steps in as a reactive partner. One thing that stuck with me from a stint working with pharmaceutical chemists—the smallest adjustments in those reactions can drastically improve purity and yield, which saves resources and makes medicine more accessible.
Flavors, Fragrances, and Beyond
Food and fragrance industries often rely on science more than people realize. Methyl Acetoacetate enters as a precursor for artificial flavor compounds like raspberry, pineapple, and even vanilla. It doesn’t land in the final taste itself, but without it, kitchens and factories would run into roadblocks creating uniform, safe flavorings. Anyone who has toured a fragrance-making facility will remember the odd mix of scents hovering in the air—a backdrop where chemists pull off precise transformations using this chemical to produce the rich palette of modern perfumes.
Dyes, Pigments, and Everyday Color
Turning blank, pale fabrics into bright reds or deep blues calls for chemistry that binds color molecules firmly to fibers. Methyl Acetoacetate proves itself here as a partner in creating azo dyes. These are the bright, durable pigments that color clothing, plastics, and even inks. Artists may never realize the work that takes place at the chemical plant, but those manufacturing teams constantly adjust their recipes, knowing any shortcut in raw materials can impact the vibrancy and longevity of their products.
Sustainable Production and Safety: The Next Steps
Years spent working around lab benches and factory settings make one thing clear—handling chemicals like Methyl Acetoacetate safely always matters. There’s no shortcut around protective gear or well-ventilated stations, especially since exposure can cause skin and eye irritation. As demand for pharmaceuticals, perfumes, and dyes grows, companies look for cleaner ways to produce and recycle these chemicals. Some organizations have started using greener solvents or developing closed-loop systems that cut waste. These efforts protect workers and the environment. The lessons learned here show that progress doesn’t just mean bigger output but safer, more responsible chemistry for everyone.
What is the storage condition for Methyl Acetoacetate?
The Real Risks Behind a Simple Bottle
I’ve worked in labs where a single careless decision turned a tool into a hazard. Methyl acetoacetate sounds technical, but it’s found everywhere you see synthetic flavors, solvent work, or specialty medicine production. Despite its popularity, this clear liquid carries some punch. It’s flammable, doesn’t mix well with moisture, and tends to break down under heat. Ignoring those facts turns an ordinary shelf into the start of a bad day.
Why Extra Attention Protects More Than Just Property
Flammable material catches attention in any chemistry storeroom. Still, after years around solvents, I notice folks get numb to the danger when bottles line up beside harmless-looking reagents. Methyl acetoacetate vapor can ignite with just a stray spark or static discharge. Just as importantly, improper storage can break it down into substances that are much nastier and harder to clean. Proper storage isn’t about just checking off a box; it’s about looking out for people you’ll never meet—your colleagues, the janitor making rounds, or the next person replacing an empty bottle.
Temperature, Humidity, and Light All Play a Role
Some people toss almost anything into a room with air conditioning and call it good. But experience says not all cool rooms work the same. Methyl acetoacetate prefers storage below 30°C. Above that, vapor builds up, and when the temperature spikes, pressure in the container rises fast enough to force open even a “tight” cap. Ever cleaned up a sticky chemical that seeped down a shelving unit? It’s memorable for all the wrong reasons.
Humidity matters just as much. This compound reacts with water in the air, slowly tearing itself apart and making acids you don’t want hanging around. Dry, well-sealed storage keeps it stable longer. Pair those basics with storage away from direct sunlight, and the bottle lasts through its whole intended shelf life. Light speeds up breakdown, ruining chemical purity and creating extra work.
Simple Solutions Make a Big Difference
A metal flammables cabinet is always a better choice than an open wooden shelf. Organize the shelf so that acids, bases, and high-energy oxidizers don't stand nearby. Good airflow keeps vapors from pooling and helps staff spot small leaks before they turn into emergencies. Labels on all sides—one look and everyone knows what’s inside.
From personal experience, one of the most overlooked details is rotation. Don’t keep aging stock just because space exists. Dispose of old, partially used bottles following local hazardous waste rules. Inventory checks every couple of months flag problems before they get serious.
Training Matters Every Day
No fancy engineering lasts if people don’t understand what they’re handling. Training—the kind that skips the technical jargon and shows real accidents from real workplaces—leaves a bigger impression than hours reading safety data sheets. Regular safety meetings, open access to MSDS, and clear instructions for spill response go a long way toward protecting everybody.
Labs and warehouses shape their safety cultures from the ground up. It’s not overkill to double-check that doors close tightly, that the right absorbent materials stand nearby, or that emergency numbers are posted in plain view. Doing these things doesn’t slow down the job; it protects the people doing it, every day.
Better Habits Create Safer Workplaces
Storing methyl acetoacetate properly isn’t about fear—it’s about respect for chemistry and each other. These routines don’t take much extra time, but they make every project run safer and smoother in the end.
Is Methyl Acetoacetate hazardous to health or the environment?
Understanding What Methyl Acetoacetate Is
Methyl acetoacetate pops up in labs and factories, mostly where chemicals get cooked and tweaked for paints, flavors, or medicines. This clear liquid belongs to a group called beta-keto esters, and it carries a sharp, fruity smell. If you’ve ever worked with synthetic flavors or certain pharmaceuticals, you probably ran into its presence somewhere on a label or a shipment.
Exposure Risks and Health Effects
Anytime we talk about chemical exposure, personal experience reminds me that proper ventilation and gloves aren’t just lab recommendations. Methyl acetoacetate could irritate your eyes, nose, or throat if vapors float in high enough concentrations. Skin contact can cause redness or dryness. Accidental splashes burn like other esters—quick, aggressive rinse is the only fix I trust.
Health agencies, including the CDC and European chemical watchdogs, rate methyl acetoacetate as harmful if inhaled or swallowed in quantity. Large doses show toxicity in animal studies, affecting the central nervous system and producing symptoms like drowsiness or headaches. There’s no solid evidence of it causing cancer in humans, but as someone who’s handled solvent spills, I treat any unfamiliar chemical with caution until long-term effects become clearer. Chronic exposure, especially without protective gear, shouldn’t be taken lightly.
Environmental Concerns
Methyl acetoacetate seeps quickly into soil or water if spilled, and its chemical structure doesn’t hang around the environment for long. Sunlight and bacteria usually break it down within a few days. But in high concentrations, this compound can stunt aquatic life—fish and water bugs lose out when oxygen drops or the water gets muddied by chemical runoff.
I’ve seen community ponds near chemical plants struggle with algal blooms and wildlife die-offs after heavy rains. Even if methyl acetoacetate isn’t the headline culprit, accidental spills from careless disposal put unnecessary pressure on local ecosystems.
Regulation and Industry Practices
Rules exist for a reason. Factories in Europe and North America face restrictions on how much methyl acetoacetate escapes into air or waterways. The EPA tracks industrial emissions, and companies must report significant leaks. Workers receive training on handling and storage, as required by OSHA or equivalent bodies abroad.
From what I’ve witnessed on shop floors and in warehouse yards, accidents often stem from poor labeling, rushed transfers, or aging containers. Routine inspection and plain-language safety sheets make the difference. Where leaks happen, fast cleanup and proper disposal prevent a small mistake from becoming an environmental headline.
Alternatives and Safer Practices
Green chemistry isn’t just a buzzword—it shapes how teams pick solvents and additives. Some newer projects swap out traditional esters for options with lower toxicity or easier biodegradability, especially where food or consumer use enters the picture.
Labs now emphasize closed systems, scrubbers, and sensors that catch leaks early. In my own work, guidance from environmental engineers and seasoned safety officers has reduced unnecessary exposures. Home chemists and educators benefit from smaller batch sizes and learning how to neutralize spills with basic supplies.
Choosing Safety
Chemicals like methyl acetoacetate serve practical roles, but ignoring their risks does nobody any favors. Clear labeling, regular inspections, protective gear, and real talks about disposal—not just written rules—make things safer for workers and the local environment. Much like cleaning up after yourself in the kitchen, putting in a little effort up front saves headaches down the line.
What is the shelf life of Methyl Acetoacetate?
Storing Chemicals is Not a One-Size-Fits-All Game
Methyl acetoacetate might not get featured in flashy magazine spreads, but it’s a mainstay in many labs, flavor and fragrance companies, and chemical plants. Most folks using chemicals care about purity and stability because anything that messes with quality can end up souring a big batch of product, triggering safety headaches, or wasting money. That’s why shelf life matters.
How Long Does Methyl Acetoacetate Last?
Methyl acetoacetate usually lasts about twelve months if you store it right—meaning sealed tight, away from sunlight, in cool spots, with low moisture. Many chemical suppliers stamp a one-year expiry date from production for this reason. Heat, air, and light speed up the breakdown. Over time, you might spot more impurities or get weird odors coming from the bottle. If a manufacturer claims their methyl acetoacetate can last two years or longer, make sure to ask about their stabilizers or extra steps in production.
Why Shelf Life Shortens
Exposure to air leads to slow oxidation, especially if the bottle is opened too much. Heat cranks up reaction rates, so a warm storeroom means trouble. Moisture or leaks can set the stage for hydrolysis, splitting molecules and leaving the contents less useful. If the cap’s not tight, you’ll end up with evaporation—the chemical quietly shrinking away and losing strength or changing composition. Basically, bad storage shortens shelf life and can make the product unsafe.
Real-World Consequences in the Lab and Plant
In research or on the factory floor, expired methyl acetoacetate can lead to faulty reactions, low yields, or waste. I’ve seen more than one project stall when someone ignored a cloudy bottle or pushed past the manufacturer’s date, thinking a sniff test would do. Labs that run tight quality control check methyl acetoacetate with HPLC or GC to pick up subtle shifts over time. Sadly, not everyone checks every bottle, especially small companies running on thin margins. Cutting corners here means risking product recalls, failed syntheses, or rough audits.
Steps for Keeping Methyl Acetoacetate Stable
Chemicals need attention to detail if you want to get the most from your money and avoid headaches. So, use amber glass bottles to shield from light. Keep storage temps below 25°C; a chemical fridge works even better. Lay in a supply of silica packs to trap stray moisture. Write the opening date on every container, track how often it’s used, and never trust a leaking seal. For bulk storage, nitrogen or argon blankets offer an extra barrier against oxygen and help keep the product closer to its original quality.
Solutions for Safer, Smarter Handling
Train staff to check for changes in smell, color, or clarity, and don’t ignore what your senses flag. Rotate stock so that older bottles get used first. If you’re running a large operation, digital inventory systems cut down on forgotten containers. For smaller teams, old-fashioned logbooks and sharp eyes do the trick. Waste disposal rules matter—a bottle past its prime can’t just go in the trash.
Trust but Verify
Good practice means no shortcuts. Always ask for a Certificate of Analysis and check the batch date yourself. If there’s ever doubt about purity or stability, confirm with testing. Shelf life isn’t just a numbers game—it connects directly to lab safety, product quality, and hard-won profit.
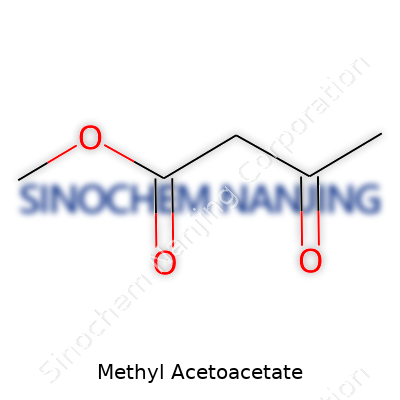
| Names | |
| Preferred IUPAC name | Methyl 3-oxobutanoate |
| Other names |
Acetoacetic acid methyl ester Methyl 3-oxobutanoate Methyl 3-oxobutyrate 3-Oxobutyric acid methyl ester |
| Pronunciation | /ˌmɛθɪl əˌsiːtəʊˈæsɪteɪt/ |
| Identifiers | |
| CAS Number | 105-45-3 |
| Beilstein Reference | 1462127 |
| ChEBI | CHEBI:17862 |
| ChEMBL | CHEMBL14236 |
| ChemSpider | 66507 |
| DrugBank | DB03832 |
| ECHA InfoCard | 100.047.907 |
| EC Number | 203-572-1 |
| Gmelin Reference | 84153 |
| KEGG | C06170 |
| MeSH | D008749 |
| PubChem CID | 7979 |
| RTECS number | EL5250000 |
| UNII | 5E42W7X4B3 |
| UN number | UN2353 |
| Properties | |
| Chemical formula | C5H8O3 |
| Molar mass | 130.14 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Fruity |
| Density | 1.050 g/mL at 25 °C |
| Solubility in water | Soluble in water |
| log P | -0.14 |
| Vapor pressure | 0.7 hPa (20 °C) |
| Acidity (pKa) | 7.7 |
| Basicity (pKb) | 7.96 |
| Magnetic susceptibility (χ) | -7.64 × 10⁻⁶ |
| Refractive index (nD) | nD 1.410 |
| Viscosity | 1.149 cP (20°C) |
| Dipole moment | 2.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -487.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2022 kJ/mol |
| Pharmacology | |
| ATC code | M01AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | NFPA 704: 2-2-2 |
| Flash point | Flash point: 83 °C |
| Autoignition temperature | 229 °C (444 °F; 502 K) |
| Explosive limits | Upper: 13%, Lower: 2.3% |
| Lethal dose or concentration | LD50 oral rat 3200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 1,600 mg/kg |
| NIOSH | WA2275000 |
| REL (Recommended) | 5 mg/m³ |
| Related compounds | |
| Related compounds |
Acetoacetic acid Ethyl acetoacetate Diethyl malonate Methyl acetate Methyl malonate |

