Methoxyethanol: The Chemical’s Role, Risks, and Road Ahead
Historical Development
Methoxyethanol, known to some in the lab as 2-methoxyethanol or methyl cellosolve, popped up during the early 20th century, riding the wave of synthetic chemistry’s industrial expansion. It drew early attention as a polar solvent that handled both water-loving and oil-loving substances, giving chemists a new edge in solving stubborn problems. Factories gravitated toward methoxyethanol because it simplified processes in coatings, plastics, and the synthesis of certain resins. By the 1940s and ‘50s, paint shops and electronics plants leaned heavily on solvent blends based around this compound for its unique properties. That demand led to widespread adoption, and with that, concerns began brewing about what else it might bring into human environments.
Product Overview
People know methoxyethanol mainly for what it does in manufacturing and research settings. It takes on roles as a solvent for resins, inks, dyes, and certain organic reactions. Its presence in household or consumer-facing products is rare, mainly because health issues started cropping up after decades of industrial use. Companies concentrated on this compound for its ability to dissolve pretty much anything with a polar or semi-polar structure, making it almost indispensable at one time for cleaning and surface preparation in microelectronics production.
Physical and Chemical Properties
This clear, colorless liquid has a faintly sweet scent and a low viscosity, making it easy to pour but just as quick to soak through gloves or certain plastics if safety takes a back seat. Methoxyethanol can mix with water, alcohols, ethers, and many organic solvents. Its boiling point hovers in the low 120s Celsius, leading to moderate volatility—enough to create workplace vapor. Chemically, it stands out due to the ether and alcohol groups sitting on a two-carbon backbone. Those features invite reactions with acids, bases, and oxidizers, but also let this molecule dissolve tricky resins in ways water or straight alcohols can’t touch.
Technical Specifications and Labeling
Real lab work doesn’t coast on idealized numbers. Practical specs focus on purity—often above 99% to avoid reaction mess or equipment gunk. Labels on commercial packaging tend to spell out the usual suspects: 2-methoxyethanol, methyl cellosolve, and maybe its CAS number to stop confusion between similar glycol ethers. Companies mark important warnings about toxicity, flammability, and storage, which grew more prominent as research uncovered reproductive risks, kidney and liver stress, and other health hazards.
Preparation Method
Industrial players produce methoxyethanol by reacting ethylene oxide with methanol under pressure and a catalyst. This approach keeps yields high and costs reasonable, though the reaction’s exothermic nature demands careful temperature control to prevent runaway events. The process spins off few significant byproducts if run tightly, but leaks, spills, or improper handling during or after synthesis create headaches for both worker safety and environmental impact.
Chemical Reactions and Modifications
Methoxyethanol’s chemistry extends past simple solvency. It has proven valuable as a starting material for synthesizing specialty surfactants and plasticizers. It can undergo esterification, etherification, or even form acetals under the right conditions. The alcohol group persists as a practical handle for further manipulation in organic synthesis, while the ether linkage opens doors for reactivity not always predictable without some hands-on experimentation.
Synonyms and Product Names
2-methoxyethanol goes by more than one name. In industry, methyl cellosolve appears frequently. Others tag it as ethylene glycol monomethyl ether, EGME, or monomethyl glycol ether. These aliases matter if you’re chasing references in old research, finding old stock in a dusty storeroom, or comparing regulatory restrictions across regions with different naming standards.
Safety and Operational Standards
Early decades kept more focus on chemistries and profit than on human consequences. Time and research forced companies and regulators to tighten their grip. Several occupational safety bodies set airborne limits, often a handful of parts per million. Strict workplace procedures got written down, stressing gloves, goggles, and proper ventilation. Methoxyethanol draws special scrutiny for its effect on reproductive health, so some countries ban or sharply restrict its use in consumer products and even phase it out entirely in favor of less hazardous glycol ethers for new operations. Spill response and disposal guidelines stress containment, specialized waste treatment, and immediate evacuation for large releases.
Application Area
People sometimes overlook the range of fields that mapped onto methoxyethanol over the decades. Paint and coating developers relied on it to thin formulations without losing performance. Microchip and photographic industries exploited its mild solvency and low residue to clean delicate surfaces. Some pharmaceutical labs ran synthesis steps in its presence, chasing specific yields or molecular arrangements unattainable by other means. Those who soldered circuit boards or mixed specialty printing inks often smelled its sweet edge in the workplace air, sometimes before they understood the compound’s deeper risks. Environmental testing now keeps close watch on emission points inside plants handling glycol ethers, learning from earlier generations who dealt with exposure consequences.
Research and Development
Over recent years, research labs have tried to edge methoxyethanol out of processes where possible, spurred by evidence linking chronic exposure to organ damage and birth defects. Green chemistry research now digs into safer replacements, often targeting bio-based solvents or customized molecules tuned for lower human toxicity. Analytical chemists refine detection and removal methods to handle legacy stockpiles. In environmental research, the compound serves as a marker for certain sources of pollution or as a test case for vapor intrusion studies. Development now leans toward materials with similar solvency but without the human health baggage.
Toxicity Research
Scientific studies and workplace health records keep pointing to problems that come from skin contact or inhaling methoxyethanol. Reproductive toxicity concerns started in animal studies, then echoed in human health logs from industrial sites. Chronic exposure links to anemia, kidney and liver stress, and birth defects became too loud to ignore. Animal models provided clear warnings about its ability to cross biological membranes and interfere with fetal development. Public health agencies list methoxyethanol alongside those chemicals flagged for phase-out, especially in jobs where proper personal protective gear lags behind standards. The research became pivotal for policy changes that favor substitution or outright bans, not just risk reduction.
Future Prospects
The story of methoxyethanol looks different now than it did decades ago. Its unique chemistry once made it a staple; now, its health risks push it to the sidelines. Many major chemical producers are phasing out its use except where no substitutes exist, and downstream industries move to engineered solvents, ionic liquids, or upgraded processing hardware as replacements. Regulations keep tightening, especially in jurisdictions that prioritize green chemistry. Some developing regions still rely on legacy solvents like this due to cost and infrastructure limits, so global phase-out remains a long-term goal. Ongoing research into human exposure and environmental persistence continues to shape how we think about transitioning away from compounds that once looked like progress but eventually demanded a hard tradeoff between convenience and care for health and safety.
What is Methoxyethanol used for?
The Role of Methoxyethanol in Industry
Methoxyethanol shows up a lot in chemical plants and paint factories. My first contact with the stuff came during a short stint at a small printing company, where the shop manager explained why methoxyethanol kept paint from drying on rollers and helped ink move smoothly over giant sheets of paper. Solvents like this clear out jams and give paints or inks the consistency needed to work on large jobs without clogging up the machinery.
Factories making electronics or plastics also keep plenty of methoxyethanol on hand. The chemical helps carry dyes into parts during manufacturing, giving plastics solid and even color. Shoe factories too—yes, even those—use it to soften leather. Methoxyethanol soaks into the material and makes it easier to shape into sneakers or boots.
Health and Safety: Why Attention Matters
So, why should I care so much about methoxyethanol? It all comes back to health. I remember one co-worker at the print shop mentioning headaches after a long shift near the press. That’s a classic sign of solvent exposure. According to the CDC, breathing in methoxyethanol can mess with the blood and cause long-term nerve and kidney damage. Some studies link exposure to fertility problems. In California, I saw how state safety regulators crack down on companies that don’t protect their workers from vapors—inspections, fines, and new rules keep popping up as researchers learn more.
In the past, regulations let factories use these types of solvents with barely any oversight. Now, companies have to install exhaust systems, hand out gloves and goggles, and swap in less risky chemicals if they exist. Reading through OSHA reports, it’s easy to spot a trend—workplaces doing a better job of testing air and protecting workers.
Why the Industry Still Needs Methoxyethanol
Plenty of painters still rely on methoxyethanol for its strong solvency and how well it blends with water. Other chemicals sometimes fall short when it comes to keeping paints and coatings from separating or getting globby. In electronics factories, workers rely on how easily methoxyethanol can pull greasy stains off circuit boards or metal parts.
Chemical labs, especially those tied to universities and hospitals, use it for extracting and cleaning biological samples. Some research projects would stall without a chemical that works exactly like methoxyethanol, since it dissolves both water-based and oily substances. I’ve seen how one bottle does the work of several weaker cleaners.
Solutions and Safer Alternatives
To keep workers safe, plant managers now look for substitutes where possible. Ethanol or propylene glycol offer slightly milder profiles, though sometimes they come at a higher price or lower performance. Switching can mean longer drying times or unexpected issues in production, so shifts to safer chemicals come slowly.
Education helps—many shops now run classes on handling solvents, reading labels, and knowing what health symptoms to watch for. Open windows, good exhaust fans, and frequent breaks reduce irritation and headaches. I always keep a fresh pair of gloves and a sealed bin for rags, since even a few drops on the skin can start to cause problems.
Methoxyethanol sits in a tricky spot: valued for its unique properties but clearly risky. Weighing the risks and taking every measure to protect workers isn’t just a matter of checking off boxes—it’s about real people and their long-term health.
Is Methoxyethanol hazardous to health?
Everyday Exposure and the Invisible Dangers
Working in factories or labs puts you right next to chemicals most people don’t hear about in the news. Methoxyethanol often shows up on ingredient labels in paints, inks, and cleaning fluids. I once spent a summer in a printing workshop, and that sweet, slightly piercing smell of certain solvents stuck with me long after my shift ended. Many don’t realize the risks tied to this compound, since it doesn’t raise alarms like more famous chemicals do.
What Science Says About Methoxyethanol
Research going back decades draws a pretty clear picture. The U.S. Environmental Protection Agency classifies methoxyethanol among the chemicals with the potential to cause serious health problems. Animal studies show birth defects and fertility issues after repeated exposure. Human cases raise similar concerns. Folks regularly working with methoxyethanol in factories report headaches, dizziness, and symptoms affecting the nervous system. Some research links long-term contact to blood disorders and weakened immune response.
A report from Sweden’s National Institute for Working Life highlights that breathing in methoxyethanol fumes increases the risk of anemia and can damage bone marrow. In the real world, that means more sick days, trouble fighting off infections, and in rare cases, lasting disability. In families where one partner works long hours with chemicals, the risk can extend beyond the workplace. Fumes carried home in clothes or on skin don’t stop at the factory gate.
Why Workplace Safety Doesn’t Always Cut It
Safety rules exist. Labels warn workers to wear gloves, goggles, and masks. Yet rules rarely match reality on busy shop floors, where speed outruns caution. I remember colleagues skipping gloves on hot days or ignoring fume hoods after a long shift. Companies sometimes delay upgrades because of costs or lack of clear regulations. Small businesses, especially, struggle to stay up-to-date. OSHA sets legal limits, but those numbers sometimes trail behind new scientific findings. A recent study by the National Institute for Occupational Safety and Health pointed out that even so-called “safe” levels can still threaten health over years of exposure.
Protecting People—Not Just Checking Boxes
No checklist replaces real-world thinking. Workers and families deserve more than regulatory minimums. Providing better ventilation, automating processes, and swapping out methoxyethanol for safer alternatives where possible all make a difference. A paint manufacturer in Ohio, for example, cut health complaints in half by switching to low-toxicity solvents and adding air purifiers to its production floors.
It takes strong workplace culture. Open conversations about risks, regular training, and easy reporting channels help people stay alert to early symptoms. Unions and industry groups have power to press for stricter rules and regular health screenings. Doctors can learn to ask about chemical exposure when workers show up with ambiguous symptoms. It’s not only about chemical literacy; it’s about building a sense of responsibility from the top down.
Toward Smarter Choices
Methoxyethanol proves that hidden dangers still lurk in many common products and workplaces. Science continually uncovers new risks, so listening to those findings and acting on them quickly matters. Workers, business owners, and regulators all shape the safety landscape. Honest discussion, practical solutions, and ongoing education do more to protect health than any label ever will.
What are the safety precautions when handling Methoxyethanol?
Why Methoxyethanol Matters
Methoxyethanol pops up in a lot of industries—paints, inks, electronics, labs. On the surface, it looks like just another solvent, but look a little closer and it can turn dangerous fast. Health agencies, from the EPA to OSHA, flag methoxyethanol for a reason. It gets through skin and lungs, then it travels through the body and starts causing problems for organs and even the central nervous system. I’ve worked around solvents for years, and methoxyethanol has a reputation for sneaking in, even when you thought the air was clear. One slip—wet hands, a cracked glove, a fume hood left off—and you feel it before you know it.
Getting PPE Right Isn’t Optional
Chemicals don’t care if it’s your first day or your fifth year on the job. Methoxyethanol doesn’t give much warning, and symptoms might not show right away. For anyone pouring, mixing, or transferring it, gloves matter a lot. Nitrile or heavy-duty neoprene stand up to methoxyethanol better than basic latex. Your clothes count, too. Lab coats or chemical-resistant coveralls seal off the splashes. Protective eyewear packs a punch—even one drop in the eye stings and risks long-term damage. Respirators aren’t just for industrial settings; in tight or poorly ventilated rooms, organic vapor cartridges make a big difference.
Ventilation Rules Everything
If you’ve ever tasted that sweet odor on your tongue, you’ve already had too much exposure. Open windows and fans alone can’t cut it. Local exhaust, like fume hoods or downdraft tables, pull those vapors away before you breathe them in. It’s easy to leave a cap off a bottle, but every second means more vapors in the air. Alarm systems for volatile chemicals can add a backup layer, especially in enclosed labs or workshops.
Workplace Practices and Habits
Routine saves lives, and it’s not just a formality. I’ve seen coworkers dismiss the “small stuff”—washing hands or wiping down benches—and end up with headaches, nausea, and worse. Keep food and drinks out of chemical areas. Contaminated hands sneak toxins into your mouth, eyes, and nose. Storage makes a huge difference. Seal bottles tight, keep them away from heat, and label everything. Leaking containers left overnight create risky mornings for everyone walking in.
Environmental and Legal Aspects
Dumping methoxyethanol down the drain or in regular trash leads to water and air pollution. Local water treatment can’t clear these things efficiently. Proper disposal usually means a licensed hazardous waste facility. Government fines make mistakes expensive, but the bigger cost shows up in community health and worker trust. Keeping records of use, storage, and incidents doesn’t just check a box. It builds a safer workplace.
Emergency Response: Acting Fast
Accidents happen, even with the best plans. Spills need immediate attention. Absorbent pads and neutralizers speed up cleanup and cut down exposure time. If methoxyethanol gets on skin, rinse with water—don’t stop at a quick wash. Eyes need minutes, not seconds, at the eyewash station. Fresh air helps with inhalation symptoms, but medical help becomes urgent if confusion, difficulty breathing, or strange heart rhythms start up.
Respect Means Protection
Navigating chemicals like methoxyethanol takes more than personal caution; it calls for team awareness, strong routines, and a willingness to stay updated. Regulations shift with new research. Training drills and material safety data sheets keep the knowledge sharp. Every good protocol comes down to keeping people healthy and sending them home okay each shift.
What is the chemical formula of Methoxyethanol?
Why the Formula C3H8O2 Matters Beyond the Lab
Methoxyethanol, with the chemical formula C3H8O2, pops up across manufacturing lines, laboratories, and sometimes in places you wouldn’t expect, like household paint removers. Understanding its formula is useful for anyone handling chemicals, not just people in white coats jotting down reactions. Even something as basic as three carbons, eight hydrogens, and two oxygens tossed together will impact your workplace, health, and maybe even the air you breathe.
Everyday Encounters, Real Impacts
Plenty of people know this liquid as 2-methoxyethanol or methyl cellosolve. Industrial workers and hobbyists run into methoxyethanol in cleaning solutions, inks, some dyes, and coatings. Without knowing that C3H8O2 means methoxyethanol, you miss the warnings on the label, and you miss understanding what your skin, lungs, or environment might be dealing with.
Not long ago, a colleague showed me an ingredient label on an old paint stripper. It didn’t say “methoxyethanol” but listed only the chemical formula. He shrugged, unsure if it was safe. That kind of confusion happens all the time. Recognizing chemical shorthand bridges the gap between safe use and unintentional exposures.
Health and Safety: Facts You Won’t Find on the Label
C3H8O2 sounds harmless. Yet, methoxyethanol absorbs quickly through the skin and travels in the air. Research from the National Institutes of Health has linked it to reproductive issues and nervous system problems after repeat exposure. European regulators classify it as toxic for reproduction and have moved to restrict its use in consumer products.
Anyone working around solvents needs more than rules and gloves. Workplaces matter, but people’s health does, too. A sign on the wall or a safety data sheet can remind you of risks, but experiences leave stronger impressions. I remember a workshop where shop ventilation failed for a day. Every cough and eye itch made the team scramble for answers. Knowledge of that chemical formula on a safety sheet would’ve sent us to find the right respirator sooner.
User Awareness and Practical Solutions
Workers deserve more than fine print at the bottom of bottles. Training helps, but plain-language labels—showing both the common name and formula—make a real difference. Some companies get this right by providing on-the-spot chart guides. Trust builds when people see employers invest in workshops about solvent safety beyond compliance minimums.
On a bigger scale, public disclosure rules push manufacturers to share what’s actually in their products. Groups like the American Industrial Hygiene Association have called for broader education on chemical formulas, so no one stands guessing in the supply closet. Choosing alternatives, like less hazardous glycol ethers, shows up as a smart solution for firms trying to keep both productivity and safety on track.
The Bottom Line for Everyday Choices
Knowing that methoxyethanol’s chemical formula is C3H8O2 means you don’t stay in the dark about what’s flowing through your factory or garage. It’s not just technical trivia; it shapes decisions at the workbench, on a new construction site, and in regulations that protect families and workers alike.
How should Methoxyethanol be stored and disposed of?
Getting Real About Methoxyethanol Risks
Picture a clear liquid with a faint sweet smell, often found in paints, inks, or cleaning agents. That’s methoxyethanol. People might figure a little bottle of this stuff doesn’t do much harm sitting on the shelf, yet one splash or a little too much inhalation can cause headaches, nausea, or even more severe health issues after repeated exposure. Some folks report feeling unusually tired or experiencing skin irritation pretty quickly. There’s evidence methoxyethanol harms bone marrow, reproductive health, and even fetal development. Take a look at statistics from workplace safety reports—serious mishaps happen when folks take shortcuts with chemical safety.
Keeping Methoxyethanol in Its Place
My old workspace stored methoxyethanol in a steel flammable liquids cabinet, far from sunlight and heat sources. A simple fan kept the room ventilated. Nobody wants to open a storage room and find vapor fumes hanging heavy in the air. Leaky containers or shoddy jars never made it past inspection. Glass bottles with tight-fitting caps or high-density polyethylene did the job well. The storage area stood away from acids, oxidizers, or food, and I made sure the labeling shouted out the hazard, not just scribbling “solvent” with a marker. Labels should list content, date, hazards, and emergency spill contacts. We didn’t just toss it on a shelf and call it a day—minimizing clutter kept accidents from turning into emergencies.
Everyday Precaution Beats Regret
People sometimes ask, “What’s the worst that could happen if I just pour a little down the drain?” More than folks realize—methoxyethanol in the water system risks contaminating groundwater and harming aquatic life. Flushing chemicals away like they'll disappear only spreads the problem to neighbors, community water sources, and ecosystems. I’ve seen local fish populations nosedive after a chemical incident.
Responsible Disposal: Not a Bureaucratic Hoop
Rules may feel like red tape to some, but proper disposal stops future trouble cold. Handing off unwanted methoxyethanol to a licensed hazardous waste contractor never felt glamorous. Still, it made a big difference. City and county governments usually organize drop-off events for households, making the process easier for everyone.
Every workplace I’ve been to trained people on spill kits, absorbent materials, and emergency showers. Rags, gloves, and residues went in their own sealable hazardous bags. Landfills and regular dumpsters don’t break down chemicals safely, so only certified facilities handle this job.
Looking for Smarter Solutions
Switching to safer alternatives in products, like using water-based cleaners instead of solvents, also helps. Research and consumer pressure push industries to move away from hazardous chemicals where possible. Still, sometimes methoxyethanol has no drop-in replacement—so proper storage and disposal stay non-negotiable. I always think about not just our crew’s safety, but the kids growing up nearby and the water we all drink. Smart handling practices help communities thrive and protect the natural world for years to come.
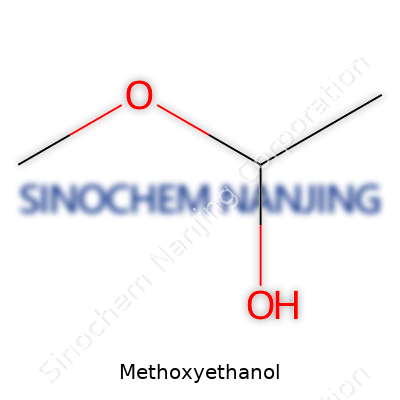
| Names | |
| Preferred IUPAC name | 2-methoxyethan-1-ol |
| Other names |
Ethylene glycol monomethyl ether 2-Methoxyethanol Methyl cellosolve Methyl glycol Cellosolve |
| Pronunciation | /ˌmɛθ.ɒk.siˈɛθ.ə.nɒl/ |
| Identifiers | |
| CAS Number | 109-86-4 |
| 3D model (JSmol) | `/assets/jmol-3d/model.cjson?modelid=4106` |
| Beilstein Reference | 0636997 |
| ChEBI | CHEBI:44562 |
| ChEMBL | CHEMBL17268 |
| ChemSpider | 6761 |
| DrugBank | DB14258 |
| ECHA InfoCard | ECHA InfoCard: 100.003.349 |
| EC Number | 203-713-7 |
| Gmelin Reference | Gm. 1202 |
| KEGG | C01179 |
| MeSH | D008767 |
| PubChem CID | 6567 |
| RTECS number | KL5950000 |
| UNII | KHV6U3R59X |
| UN number | UN1239 |
| Properties | |
| Chemical formula | C3H8O2 |
| Molar mass | 76.09 g/mol |
| Appearance | Colorless liquid |
| Odor | mild pleasant odor |
| Density | 0.966 g/cm3 |
| Solubility in water | miscible |
| log P | -0.77 |
| Vapor pressure | 0.87 mmHg (20°C) |
| Acidity (pKa) | 15.6 |
| Basicity (pKb) | \('pK_b = 14.97'\) |
| Magnetic susceptibility (χ) | -8.09×10⁻⁶ |
| Refractive index (nD) | 1.402 |
| Viscosity | 1.7 mPa·s (at 20 °C) |
| Dipole moment | 1.93 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -368.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1340.7 kJ/mol |
| Pharmacology | |
| ATC code | D01AE15 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H319, H332, H360 |
| Precautionary statements | P201, P210, P260, P264, P270, P280, P301+P312, P303+P361+P353, P304+P340, P308+P313, P312, P330, P337+P313, P362+P364, P405, P403+P233, P501 |
| Flash point | 43 °C |
| Autoignition temperature | 234 °C |
| Explosive limits | 1.8–14.3% |
| Lethal dose or concentration | LD50 Oral Rat 2370 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 2,370 mg/kg |
| NIOSH | KHB61250 |
| PEL (Permissible) | 25 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethanol Methanol Ethylene glycol Diethylene glycol Propylene glycol 2-Ethoxyethanol 2-Butoxyethanol |

