Mercaptoacetic Acid: Past, Present, and Future of an Unsung Chemical Compound
Historical Development
Mercaptoacetic acid, better known to chemists as thioglycolic acid, has been shaping industries for generations. Born out of early 20th-century curiosity about sulfur-based organics, its discovery marked a turning point in the chemistry world. Back when experimentation formed the backbone of scientific progress, researchers stumbled onto this molecule while tweaking glycolic acid’s structure, trying to understand how sulfur could alter chemical reactivity. Laboratories raced to identify all the possible outcomes, eventually unraveling a compound that changed how we handle hair, metals, and everyday beauty products. Its history reflects the blend of happenstance and relentless tinkering that defines chemistry’s golden age. Much of its industrial momentum kicked off after scientists realized how well it could break down strong keratin bonds, a detail that sent ripples through the world of personal care and beyond. Anyone who has read the early patent filings and chemical journals from the 1940s and 1950s sees the unmistakable rise of thioglycolic acid as a utility player, proving its worth in setting new standards for hair waving and depilation. Its rise wasn’t just luck—it resulted from profound scientific observation, moved by postwar curiosity and the desire to push technical boundaries.
Product Overview
Few chemicals migrate across industries as easily as mercaptoacetic acid. Its structure—a small molecule with a carboxylic acid and a thiol group—makes it especially reactive in controlled settings. Labs love it for its ability to reduce disulfide bonds, making it a staple ingredient in hair perms and depilatories. Factories turn to it for its knack for chelating metals—a feature widely used in electronics, photography, and plastics manufacturing. When brought into formulations, it brings punch, not fluff; results vary depending on concentration and processing, so it feels like a tool rather than a commodity. Product formulations almost always seek a balance, chasing the sweet spot between reactivity and safety, which means chemists pay close attention to tiny tweaks in handling and blending.
Physical & Chemical Properties
This compound comes off as a colorless or slightly yellowish liquid at room temperature. Its sharp, sulfurous odor reminds anyone who has worked with eggs or hot springs that nature and chemistry often rhyme. It dissolves in water well, forms stable solutions, stays active over a wide pH range, and handles temperature swings with a sort of professional indifference. Its dual-function as both a reducing agent and a mild acid means it jumps into action wherever bond-breaking or formation is needed. One reason industry keeps it close is that it holds onto its properties under real-world conditions, and its distinctive characteristics—the foul smell, the high reactivity, the strong affinity for heavy metals—signal its presence unmistakably.
Technical Specifications & Labeling
On any drum or lab bottle, the name mercaptoacetic acid often appears alongside its chemical formula: HSCH2COOH. Concentration matters, and most purchasers demand tight QC standards, typically in the 70–99% range for industrial use. The handling instructions read like a checklist for safety: corrosion-resistant containers, airtight seals, chemical-resistant gloves, and full lab gear. Labeling usually spells out the risks, from skin and respiratory irritation to recommendations for safe storage, since this acid does not forgive carelessness. Regulatory labels reflect global safety standards—the differences between North American, European, and Asian regulations usually come down to labeling nuance rather than substance. In my experience, the strictness in labeling signals not just regulatory compliance but genuine concern for user safety.
Preparation Method
Preparing mercaptoacetic acid in the lab runs like a small but precise production—chloracetic acid reacts with sodium hydrosulfide under controlled conditions, forming the target compound in high yield. Any scale-up to industrial levels brings logistical challenges, especially balancing purity, managing byproducts, and ensuring environmentally sound waste treatment. Smaller labs often prefer purchasing the pre-formed acid for simplicity, but chemical manufacturers fine-tune temperature, pressure, and washing steps to maximize return and minimize contaminants. It’s an old-school process at heart, shaped by decades of tweaking for efficiency and safety. What sets the best producers apart isn’t fancy tech, but an unerring eye for process control and waste minimization.
Chemical Reactions & Modifications
Chemistry’s magic with mercaptoacetic acid lies in its ability to pull double duty. Its thiol group cracks disulfide bonds, making it a favorite for breaking keratin in hair and feather protein in animal by-product processing. It’s also up for esterification and acylation, turning into a range of derivatives for niche applications. Its ability to form chelates with metals made it vital in photographic processing, especially in stabilizing silver halides, and it often appears in technical literature for complex synthesis steps where reducing power or functional versatility are key. The variety of modifications possible shows why chemists look beyond simple reactions—using it opens the door to more creative approaches in organic synthesis and industrial solutions. Every new derivative discovered brings different opportunities, often with tweaks in performance, safety, or environmental impact.
Synonyms & Product Names
Mercaptoacetic acid wears many hats in chemical commerce. To scientists, thioglycolic acid brings the right level of precision, emphasizing both sulfur and glycolic acid heritage. Factories ordering bulk quantities recognize everything from TGA to Acetylmercaptan acid. Each alias hints at its molecular makeup, though the industry generally agrees on CAS Number 68-11-1 to clear up confusion. Multiple trade names pop up in different regions, reflecting local market customs and regulatory requirements. People in the know realize that behind these labels, it’s the same sulfur-infused workhorse, quietly earning its keep in countless processes.
Safety & Operational Standards
Even small spills of mercaptoacetic acid stick out, both for their odor and their risk. Direct skin contact can burn, and inhalation brings irritation and, sometimes, worse. That has turned chemical plants and labs into showcases of safety in action: locked cabinets, continuous ventilation, strict PPE requirements, and a zero-tolerance policy for corner-cutting. Vent hoods, spill kits, and disposal protocols stop incidents before they start. From my own lab days, nothing focuses attention like hearing the pop of a bottle cap and catching that sharp whiff—it’s a never-fail reminder to double-check gloves and eye protection. Compliance isn’t just about rules—it’s about keeping workers and the environment out of harm’s way, with regular audits and emergency drills building a mindset of preparation instead of reaction.
Application Area
Mercaptoacetic acid impacts more sectors than many realize. Personal care and beauty started its boom: perms, relaxers, depilatories—iconic products would be impossible without it. Textile finishing, leathers, and wool scouring operations leverage its ability to break protein bonds, saving time and money in massive operations. The electronics industry pulls it in for etching and complexation, stripping metals or creating patterns that microchips rely on. Photography, once a booming market for silver recovery, relied on it for decades before digital took over. Water treatment plants now employ its chelating ability to help remove heavy metals, making a dent in pollution problems. Even in small-scale biotech, researchers use it to reduce disulfide bonds in proteins, prepping samples for mass spectrometry or electrophoresis. Every industry using it faces trade-offs—reactivity brings efficiency, but also risk, making careful use essential.
Research & Development Trends
Today’s R&D on mercaptoacetic acid pushes both performance and safety. Green chemistry picks at the old processes, searching for ways to reduce emissions and hazardous byproducts. In-house lab reports and academic journals show a steady uptick in efforts to tweak chemical pathways, coaxing the same performance with less environmental baggage. Efforts in encapsulation, blending, and slow-release formulations try to tame its volatility, stretching its applications into areas like drug formulation and targeted therapies. Industry never stops hunting for new derivatives to cut down on handling risks while keeping the chelating and reducing powers intact. Competitive pressures and tightening regulations push teams to find smarter and safer application methods—from low-odor variants to formulations that degrade more easily after use. The scientific literature reads like a roadmap toward a cleaner chemical industry—one that uses the lessons of the past to solve tomorrow’s problems.
Toxicity Research
Toxicologists have yet to give mercaptoacetic acid a totally clean bill of health. Repeated exposure, especially without safeguards, brings real risks—dermatitis, respiratory irritation, and animal studies suggest long-term problems with high doses. These warnings matter: regulatory agencies keep its permissible exposure limits tight, setting guidelines for both industrial and consumer uses. Ongoing studies measure both acute and chronic exposures, led by concerns about workplace safety, environmental persistence, and waste disposal. Modern toxicology zeroes in on how small, repeated exposures stack up over years—a lesson learned from the early history of industrial chemistry, where yesterday’s miracle sometimes became today’s headache. Risk assessments now consider not only the workers handling bulk chemicals, but also the downstream effects as residues end up in water, soil, or recycled materials. Research continues, searching for ways to predict and prevent harm.
Future Prospects
The path forward for mercaptoacetic acid walks a tightrope between utility and caution. Regulatory scrutiny grows year by year, with consumer safety, green chemistry, and waste minimization driving innovation. The market for sustainable chemicals is reshaping expectations—companies look for newer, safer alternatives but still lean on the tried-and-true performance this compound delivers. Research into substitutes often circles back, finding that unique sulfur chemistry is hard to replicate without trade-offs. The coming decades promise a mix of tighter controls and creative new uses, especially as the drive toward environmental responsibility picks up speed. Ultimately, the fate of mercaptoacetic acid rests on chemistry’s ability to innovate, marrying the lessons of a complex past with a future that demands higher standards, safer products, and a lighter ecological footprint. Its story is far from over.
What is mercaptoacetic acid used for?
Hair Care’s Hidden Powerhouse
Mercaptoacetic acid, known in salons as thioglycolic acid, shows up where people change their hair. Walk through any hair salon and you’ll see bottles for perms and straightening solutions. That sharp, sulfur smell often comes from this compound. It breaks the bonds inside hair, making it possible to reshape curls or flatten waves. This gives stylists the power to turn frizzy hair into smooth locks or tight curls into soft waves—something people have chased for decades. The same action that lets hair take on new forms also weakens the strands, so professionals have to watch timing and concentration closely.
Breaking Bonds in Industry and Labs
Beyond hair salons, mercaptoacetic acid helps with more than just beauty. In manufacturing, it works as a strong reducing agent. Leather tanning stands out as one industry that leans on this chemical. Removing hair from hides gets a lot easier when mercaptoacetic acid goes to work. It breaks the keratin structure, allowing clean separation and smoother processing. Without it, many leather products would feel rough or look unfinished.
Science researchers know this chemical for its ability to keep proteins from clumping up. In molecular biology labs, technicians use it to prep samples, especially in protein work. It keeps protein chains separated, making analysis by methods like electrophoresis more reliable. Even small research setups can get better results by adding just a bit of this acid to the mix. High purity and careful measurement matter because the same properties that help untangle proteins can also cause safety issues if misused.
Risks and Realities in Everyday Use
People rarely notice the risks attached to hair treatments. The same chemical that gives curly hair a fresh start also brings skin and eye hazards. Overexposure causes burns or irritates airways. Salons train staff to use gloves, work in ventilated spaces, and keep solutions away from eyes. In my own life, I watched a friend develop skin irritation from home perm kits—proof that at-home safety is just as important as it is in professional spaces. Local regulations control concentration and require clear warnings, but not every shopper reads labels before mixing up a batch.
The environmental side often gets less attention. Rinsing chemicals down the drain adds to wastewater loads, and not all treatment plants neutralize these compounds easily. Communities with lots of salons or leather processing can see local waterways pick up some extra sulfur-laden byproducts. More sustainable practices have started popping up—closed-water recycling, better capture of process liquids, and exploring less harmful alternatives—but these haven’t reached every business.
Looking Toward Safer Solutions
Demand for safer hair treatments and greener industrial processes keeps pushing companies to test new formulas. Some innovators have found milder chemicals for hair reshaping, but few match mercaptoacetic acid’s effectiveness. Encouraging anyone working with it to use proper protection, install good ventilation, and follow disposal regulations makes a difference. Governments and industry groups update safety guidelines as research improves, but old practices hang on, especially in small or home-based operations.
Understanding what’s in a product and the trade-offs behind its use helps both workers and customers make better choices. Checking for certifications, reading ingredient lists, and asking stylists detailed questions about procedures can prevent unwanted side effects. In labs and factories, regular training and investment in safer substitutes limit the risks. The story of mercaptoacetic acid shows how a single chemical connects beauty, science, and environmental responsibility, challenging people to keep safety and sustainability at the center of progress.
Is mercaptoacetic acid hazardous to health?
Understanding the Risks of Mercaptoacetic Acid
Mercaptoacetic acid crops up in hair salons, laboratories, and factories. The sharp smell grabs attention right away. It’s a significant ingredient in chemical hair removal and permanent waving solutions. Chemical workers see it as a tool for synthesizing other substances, too. This chemical doesn’t get much attention outside those circles, but its effects on health matter both for professionals and for the wider community.
Direct Exposure Can’t Be Ignored
Everyday users won’t find mercaptoacetic acid on store shelves, but hair stylists work with it regularly. It irritates the skin, eyes, and respiratory tract. A careless touch or a once-in-a-while splash on ungloved skin leads to burns or blisters. Even something as simple as breathing in vapors can cause coughing and throat pain. Extended or repeated exposure increases risk. Research published by the National Institute for Occupational Safety and Health (NIOSH) points out that repeated skin contact leads to severe dermatitis, and accidental eye splashes could cause long-lasting damage.
What Science Shows About Chronic Hazards
Research teams tracking occupational health risks across Europe and Asia highlight a repeated pattern: People exposed to mercaptoacetic acid in tight work areas deal with higher rates of asthma-like symptoms and persistent coughs. According to toxicology reports from the U.S. Environmental Protection Agency (EPA), this chemical changes protein structure in living tissue, amplifying inflammation. Animal studies suggest risks increase for people who handle it daily without proper protection.
Downstream Impacts in Personal Care
This acid’s role in cosmetic products, especially in the permanent wave and depilatory markets, isn’t just a matter for workers. Residual traces in finished products linger. The Food and Drug Administration flagged accidental overexposure in salon settings, especially with poor ventilation. In my own experience managing a small hair salon, I saw stylists get headaches or itchy skin after handling these treatments for hours.
Environmental Connections
Mercaptoacetic acid doesn’t break down right away. Wastewater from factories or salons sometimes ends up in rivers or soil. Local wildlife can pick up the effects—fish exposed to runoff show abnormal development and stress responses. When that happens, a chemical used on someone’s curls causes problems a long way from the salon chair. Agencies like the EPA advise facilities to treat wastewater carefully and limit discharge for good reason.
Reducing Risk: Simple Steps Matter
Relying on protective gloves, proper goggles, and ventilation makes a clear difference. During a job in a manufacturing facility, I saw a noticeable drop in workplace complaints after management invested in ventilation and set clear policies for glove use. Workers felt empowered to speak up when they needed new safety gear. Industry guidelines line up with this approach—nothing fancy, just solid barriers between chemical and skin, eyes, or lungs.
Health monitoring programs help, too. Regular checks for symptoms catch small problems before they grow into missed work or long-term health issues. Switching formulas when possible or using less hazardous alternatives gives another line of defense. Some salons now seek out hair care systems that skip mercaptoacetic acid entirely, cutting exposure to zero. These decisions recognize that human health should never play second fiddle to convenience.
Industry Has a Role—And So Does Awareness
Manufacturers should keep improving warning labels and instructions, especially since consumers rarely recognize technical terms. Regulatory agencies require accurate hazard data, but everyday awareness goes a long way. Informed decisions start with clear information, not jargon. Building a culture where safety gear is the norm beats luck any day. It boils down to knowledge, preparation, and the stubborn insistence that everyone has the right to a safe workday—no matter the chemical in question.
What are the safety precautions for handling mercaptoacetic acid?
What Makes Mercaptoacetic Acid Risky?
Mercaptoacetic acid, well-known in the world of chemical processing and beauty treatments, packs a punch when it comes to hazards. It’s corrosive, releases a foul sulfur-like odor, and burns skin or eyes on contact. Inhalation can cause headaches or respiratory irritation, and spills eat through clothing faster than bleach. Personally, I saw a lab partner miss a spot of dried acid when cleaning surfaces, only to discover a hole burned straight into the sleeve of his lab coat. This drove home how much care is non-negotiable with this stuff.
Simple Precautions Save Trouble
Direct skin contact leads to fast and deep burns. Heavy-duty nitrile or neoprene gloves shield hands during handling. In my years as a chemistry instructor, I’ve watched regular latex gloves disintegrate—never a fun lesson, but memorable. Always choose gloves rated for corrosive chemicals. Eye protection is next. Goggles with full coverage beat standard safety glasses every time because splashes travel farther than you’d expect. Face shields add extra insurance, especially when measuring out larger volumes.
Clothing makes a difference. Acid-resistant lab coats or aprons act as a last line of defense. Long sleeves protect arms, but those cotton shirts many wear under coats don’t stop acid. I always advise keeping a set of dedicated lab clothes and sturdy closed-toe shoes for chemical work. Spills often land where you don’t look.
Ventilation and Storage
Fresh air is your friend. Fume hoods pull away dangerous vapors, which matters since mercaptoacetic acid stinks and irritates airways almost instantly. Home labs or beauty salons using this chemical should invest in strong exhaust systems. Never work with this acid in small, closed rooms.
Storage often gets ignored, but it’s where trouble brews. Leak-tight containers made of glass or compatible plastic, clearly labeled, help prevent mix-ups. I’ve witnessed containers with ill-fitting caps topple and eat through shelving overnight. Keep acids away from bases, oxidizers, or sources of heat.
Spill Response and Disposal
Accidents happen, even with the best prep. All it takes is one dropped pipette. Quick access to an emergency eye-wash and safety shower matters more than you think—every second counts. Clean a spill with sodium bicarbonate to neutralize before wiping it up. Avoid paper towels without neutralization—otherwise, you’re just spreading acid.
Waste management laws tighten each year. Neutralized mercaptoacetic acid never belongs in a sink. Proper containers, labeled waste, and pickup by certified handlers keep your workplace legal and safe. In my experience, cutting corners on disposal causes more long-term trouble than any single spill.
Building Safer Habits
Constant reminders and hands-on drills make a real difference in reducing incidents. Posting checklists in visible spots, keeping safety equipment unblocked, and building a routine around double-checking container seals pays off. I’ve seen veteran lab techs get caught off guard during a routine transfer, only to be saved by proper shielding and up-to-date first aid. We never outgrow the basics.
How should mercaptoacetic acid be stored?
Why Storage Matters
Mercaptoacetic acid, sometimes called thioglycolic acid, packs a punch with its sharp smell and potent chemical properties. I once got a whiff in an old-school chemistry lab—the memory sticks, and not in a good way. If you’ve spent time around strong acids, the point hits home: proper storage keeps folks healthy, and products reliable.
Understanding the Risks
Let’s talk dangers. This compound causes burns, eats through some metals, and also reacts with strong oxidizers. The fumes, which can sting, give every reason for extra caution. That’s not just speculation—documented accidents have shown people developed lung trouble and skin injuries because of a sloppy approach.
On top of that, mercaptoacetic acid absorbs water from the air, meaning it can break down or turn more dangerous on the shelf if the lid’s left open or the wrong container gets used.
Choosing the Right Container
Forget thin plastic or metals like iron. Any old bottle won't do. My training reinforced that only high-density polyethylene (HDPE) or containers lined with glass handle the acid without corroding. Keep in mind, some labs use PTFE-coated stoppers. These slow down leaks and risk of reaction. Don’t improvise—switching to a soda bottle brings headaches later.
Keep every container tightly closed. Even a little gap draws in humidity, and this chemical loves to react. I recall one story where moisture crept in, corroding shelves and ruining a batch of samples. Labels matter too. If that sticker smears or peels, there’s a real chance someone grabs the wrong thing—so permanent ink, clear warnings, and hazard symbols should be on every bottle.
Setting Up the Safe Zone
Keeping chemicals in their own corners sounds old-fashioned, but it works. Mercaptoacetic acid thrives in cool, dry spots. Never stack it next to bases, oxidizers, or foods. Store away from sunlight—or anywhere that heats up, like next to windows or radiators.
I’ve seen disaster avoided simply by having a shelf for acids only, fitted with a spill tray beneath. Ventilation counts: Do not let those fumes build up. Running a fume hood around the chemical stock area makes all the difference, especially on days when humidity spikes.
Ensuring Personal Safety and Environmental Care
Anybody handling this acid should wear gloves and goggles—always. A splash won’t forgive. Keep emergency eyewash stations and showers within a quick jog of storage. If you spill, neutralize with sodium bicarbonate before wiping up. Never pour leftovers down a standard drain.
For larger stashes, local rules step in. They spell out finer points and penalties for poor storage. Some businesses run annual safety checks, and those walkthroughs have caught leaky caps before they spark bigger issues.
Building Safer Storage Habits
It pays to tackle small issues before they grow. If a container cracks, switch it out immediately. Rotate inventory so nothing sits forgotten at the back, changing over time. Proper storage isn’t a one-time checklist tick; it’s a habit that protects coworkers, customers, and the planet.
A little diligence here saves pain, paperwork, and worry down the line. Respect for chemicals in storage speaks directly to respect for everyone in the building.
What is the chemical formula of mercaptoacetic acid?
Understanding the Basics: More Than Just Symbols
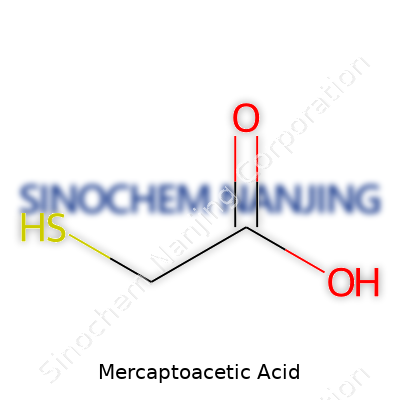
Mercaptoacetic acid goes by another name, thioglycolic acid, and its formula is C2H4O2S. That formula packs more punch than it seems. Beneath those letters and numbers lies a compound that plays a big role across industries and, surprisingly, in personal care routines. You can find it in everything from hair products to industrial cleaners. The formula—two carbons, four hydrogens, two oxygens, one sulfur—shows how its atoms come together to create a powerful chemical tool.
Straight Talk: Why Scientists and Consumers Care
I’ve seen firsthand how this acid stirs up debate. Cosmetic chemists have long relied on mercaptoacetic acid for hair removal creams and permanent wave lotions. Turns out, this compound has a knack for breaking down the disulfide bonds in hair, making it essential for reshaping curls or getting rid of unwanted hair. The strength of its sulfur and the slight, recognizable odor make its presence known, but people keep coming back because it works.
On the industrial side, C2H4O2S takes the spotlight in manufacturing and environmental cleanup. It acts as a chelating agent, grabbing onto heavy metals and helping remove them from wastewater. Its unique structure lets it bond with metals and change their behavior. I once spoke with an engineer from a water treatment plant, and he explained how mercaptoacetic acid made it possible to meet strict regulatory standards. Without it, those treatment facilities would struggle to keep toxic metals out of the supply.
Health and Safety: What Users Need to Know
Every strong chemical comes with risks, and mercaptoacetic acid is no different. Exposure can irritate skin and eyes. That rotten egg smell warns people to be careful—it’s a signal that sulfur is present and not always welcome in large doses. Personal experience with chemical burns taught me to respect warning labels, and this acid deserves the same caution. Professionals who use it regularly gear up with gloves, goggles, and ventilation. Home users handling beauty products should always read directions and avoid shortcuts.
Environmental Impact: Watching Out for More Than Just Ourselves
Mercaptoacetic acid’s formula—small as it is—means nothing escapes scrutiny. Sulfur-containing compounds can harm aquatic life if released in high concentrations. Companies need to control discharge and stick to treatment protocols. Some labs explore biodegradable alternatives, but so far, few options beat the effectiveness of mercaptoacetic acid for breaking chemical bonds and grabbing onto metals. Tight regulations help, but those rules only work if people enforce and follow them. We all share the responsibility, from the factory floor to the bathroom counter.
Looking Ahead: Room for Smarter Solutions
Better safety education, greener chemistry research, and open conversations between scientists, manufacturers, and end users will keep the benefits rolling while keeping risks in check. Chemical formulas like C2H4O2S remind me that science reaches into everyday life in unexpected ways. Learning what’s inside the bottle and how it affects people and the planet makes a difference. Responsible handling, thoughtful use, and persistent innovation open the door to progress—one molecule at a time.
| Names | |
| Preferred IUPAC name | 2-sulfanylacetic acid |
| Other names |
Thioglycolic acid TGA Acetylmercaptan HSCH2COOH |
| Pronunciation | /ˌmɜːrˌkæptoʊəˈsiːtɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 60-35-5 |
| Beilstein Reference | 635542 |
| ChEBI | CHEBI:35557 |
| ChEMBL | CHEMBL1496 |
| ChemSpider | 2159 |
| DrugBank | DB09382 |
| ECHA InfoCard | 100.007.777 |
| EC Number | 200-677-4 |
| Gmelin Reference | 873 |
| KEGG | C00647 |
| MeSH | D017542 |
| PubChem CID | 6267 |
| RTECS number | MC3325000 |
| UNII | DL7Q6ZEW7M |
| UN number | UN2966 |
| CompTox Dashboard (EPA) | DTXSID4020840 |
| Properties | |
| Chemical formula | C2H4O2S |
| Molar mass | 92.12 g/mol |
| Appearance | Colorless liquid with a strong unpleasant odor. |
| Odor | Unpleasant, strong, mercaptan-like |
| Density | 1.32 g/cm³ |
| Solubility in water | miscible |
| log P | -0.31 |
| Vapor pressure | 0.6 mmHg (20°C) |
| Acidity (pKa) | 3.6 |
| Basicity (pKb) | 9.7 |
| Magnetic susceptibility (χ) | -37.1×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.507 |
| Viscosity | 70 mPa·s (25°C) |
| Dipole moment | 1.535 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 86.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -190.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -463.3 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D11AX52 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301, H311, H314 |
| Precautionary statements | P280, P261, P273, P303+P361+P353, P305+P351+P338, P310, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 69 °C |
| Autoignition temperature | 280°C |
| Explosive limits | 4.9–18% |
| Lethal dose or concentration | LD₅₀ Oral Rat 50 mg/kg |
| LD50 (median dose) | LD50 (median dose): 338 mg/kg (oral, rat) |
| NIOSH | K0133 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Mercaptoacetic Acid: 1 ppm |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Thioglycolate Thioglycolic acid Mercaptosuccinic acid Cysteine Dithiothreitol Glutathione Cystamine Dithiobis(succinimidyl propionate) |

