Meglumine: Deep Dive Into Its Story, Properties, and Significance
Historical Development of Meglumine
Meglumine has traveled a long road since early pharmaceutical chemists first started exploring sugar derivatives to solve clinical needs in the mid-20th century. Chemists saw in N-methylglucamine a way to improve the solubility of active drugs and contrast agents, something desperately needed in medical imaging and injectable medicines. Back then, radiologists worked with compounds that were hard on the kidneys and often resulted in patient discomfort due to their low solubility and high toxicity. Meglumine helped medical professionals sidestep some of these hurdles by offering better compatibility and higher water solubility. This opened up a new era for safer and more effective formulations, transforming standards of care in diagnostic procedures.
Product Overview
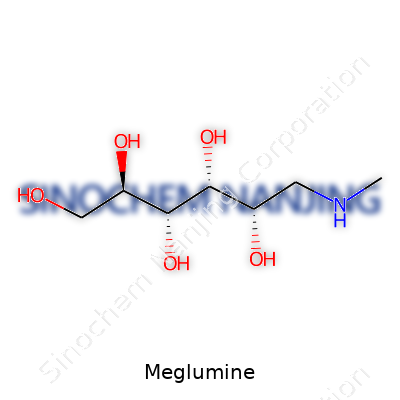
Chemists recognize meglumine as a simple yet powerful compound: N-methyl-D-glucamine or 1-deoxy-1-(methylamino)-D-glucitol. This amino sugar tweaks the properties of many compounds, especially where better solubility in water makes all the difference. Most medical professionals run into meglumine as the salt-forming partner for several imaging agents and antibiotics. Manufacturers, from their side, see its value in pharmaceutical formulations because it steers around harsh pH extremes and supports stable, robust drug delivery. Meglumine’s ability to improve taste or mask bitterness in oral drugs also adds another dimension to its importance in everyday pharmacy work.
Physical & Chemical Properties
Meglumine usually comes as a white, hygroscopic crystalline powder with little to no odor and a vaguely sweet taste. This material dissolves well in water—a solution forms quickly and clearly. Its melting point hovers around 128-132°C. The molecular formula is C7H17NO5, with a molar mass of 195.21 g/mol. Despite its sugar roots, meglumine remains chemically stable under typical storage conditions. Its alcohol and amine functional groups give it flexibility in bonding with diverse drugs, especially acidic molecules. The chemical structure features a six-membered glucamine backbone with a single methyl group on the nitrogen, helping it offer both steric and electronic benefits to pharmaceutical salts.
Technical Specifications & Labeling
Pharma-grade meglumine generally carries a minimum assay of 98.0% by dry basis and comes with tight controls on moisture, heavy metals, and microbial contamination. Labels require the product’s chemical name, batch number, lot number, date of manufacture, and certificate of analysis. Reputable suppliers provide detailed documentation of residual solvents, particle size where relevant, and endotoxin levels for parenteral applications. Regulatory bodies including the USP and EP have set forth specifications that lay out limits for impurities and physical appearance. Packaging typically uses sealed, light-resistant containers, underscoring the need to keep the product dry and uncontaminated throughout the supply chain.
Preparation Method
Meglumine manufacturers start with glucose or sorbitol as a base. Through a reductive amination process, they introduce methylamine to the sugar under controlled conditions, usually applying hydrogen in the presence of a nickel catalyst. This step converts an aldehyde or ketone group on the sugar into its corresponding methylamino structure. Downstream, companies purify the raw product by crystallization and filtration to achieve high purity needed for food and pharmaceutical use. This straightforward chemistry attracts smaller-scale laboratories and big pharma alike, as it allows scalable production and a reliable supply chain.
Chemical Reactions & Modifications
Meglumine’s unique structure allows for salt formation with acids such as sulfonic acids, carboxylic acids, and phosphonic acids, all crucial in pharmaceutical practice. The methylamine group supports further modification, and chemists have explored derivatives for specialized functions, like improved pharmacokinetics or targeted delivery in diagnostic imaging. In synthetic laboratories, meglumine acts as a chiral auxiliary or an intermediate in producing sugar-based polymers. Its ability to form complexes with both organic and inorganic molecules underpins both its versatility and widespread use in modern drug design and materials science.
Synonyms & Product Names
Across industry literature, meglumine appears as N-methylglucamine, 1-deoxy-1-(methylamino)-D-glucitol, methylglucamine, and glycamine. Some manufacturers label it under trade names when incorporated in compound drugs, such as “Meglumine diatrizoate” or “Meglumine iothalamate.” For regulatory filings, the naming stays consistent: its INN remains “meglumine,” which avoids confusion and ensures traceability across drug safety records and supply chains.
Safety & Operational Standards
Workers handling meglumine need to observe standard industrial hygiene: gloves and eye protection prevent skin and eye irritation. Its generally low toxicity profile means the risks are more about dust inhalation or contact than chemical poisoning. Facilities handling pharmaceutical-grade material follow GMP protocols, validated cleaning procedures, and strict environmental controls to avert contamination and degradation. Transportation and storage in sealed drums or bags prevent moisture pickup and maintain chemical stability. Documentation must guarantee full traceability for every step, from manufacturing to final delivery in hospital pharmacies or research labs.
Application Area
Radiology departments use meglumine daily, particularly in contrast media such as diatrizoate and iothalamate salts. Its presence enables the solubility and safety of imaging agents injected into patients for X-ray and CT scans. Certain antiretrovirals, antibiotics, and anti-inflammatories also use meglumine to boost solubility or mask harsh tastes. Nutraceuticals sometimes borrow meglumine for flavor enhancement and stabilization of specific supplements. Beyond medicine, industrial chemists use it in analytical chemistry protocols, serving as a buffering agent or stabilizer in reagent kits for biochemical assays.
Research & Development
Researchers have explored meglumine’s potential well beyond its traditional uses. Ongoing studies look at new ways to deploy meglumine derivatives in drug delivery, targeting, and sustained release. The sugar backbone provides a biocompatible scaffold to attach imaging agents or therapeutics, with chemical engineers seeking next-generation compounds that retain meglumine’s solubility benefits while boosting metabolic stability. Academic and industrial labs continue to push boundaries, leveraging the amine and hydroxyl groups to engineer better molecular interactors, aiming to unlock new diagnostic and pharmaceutical applications.
Toxicity Research
Toxicologists highlight meglumine’s relatively benign safety profile. Acute toxicity in animal studies shows high thresholds before adverse effects kick in. Regulatory safety assessments have led to its inclusion in injectable drugs and oral medications, stipulating limits on residual quantities to safeguard patient health. Longer-term studies suggest low risk for accumulation or organ toxicity, provided dosing guidelines are respected. Cases of allergic reaction or hypersensitivity are rare, well-documented, and usually resolved with supportive care. Pharmacopeias require regular monitoring and batch testing to confirm batches remain below established impurity and endotoxin thresholds to ensure ongoing safety.
Future Prospects
The future looks bright for meglumine, driven by the growing need for advanced pharmaceuticals and cleaner imaging agents. Many researchers believe its role as a solubilizing agent and carrier will stay relevant amid the surge of biologics and targeted therapeutics. Innovations in contrast agent chemistry already depend on meglumine’s versatility. Synthetic chemistry may find new uses in bionanotechnology and smart drug delivery, leveraging sugar-based building blocks like meglumine to bridge technical gaps between performance and patient comfort. Companies and universities pressing ahead with next-generation diagnostics and therapies continue to test meglumine derivatives for new patents and clinical trials, convinced that this compact molecule carries potential far beyond its roots in mid-20th-century chemistry.
What is Meglumine used for?
Meglumine: More Than Just a Chemical Additive
Meglumine rarely makes headlines, but its role in medicine runs deep. Picture yourself getting a scan at the hospital or taking a blood sugar test. Behind many medical procedures, there's a lot of chemistry working in the background. Meglumine represents one of those invisible helpers, quietly improving the reliability of countless diagnostic tools and treatments.
Making Diagnostic Imaging Safer and Smoother
Hospitals depend on clear images to catch disease early. Physicians and nurses place a lot of trust in contrast agents used in CT scans and X-rays. The formulas behind these agents need to balance safety and performance, so patients get the clarity their doctors demand, with the smallest risk possible. Meglumine works in these mixes to increase solubility. That means it helps imaging solutions dissolve better in water, making them flow easier through veins.
By improving how iodine-based contrasts hold together, Meglumine lets a dose work harder without adding stress to a patient's kidneys. Years ago, patients faced more reactions or even allergic problems with contrast agents. I have seen patients leave the medical imaging suite less anxious once their doctors assured them about the advances in these formulas.
Battling Blood Sugar Fluctuations
Many people living with diabetes test their sugar levels often. Each time, a reagent in the test strip interacts with their blood. Meglumine steps in here too, serving as a stabilizer and buffer. This might sound minor, but regular folks and their families depend on each test result to guide serious decisions—when to eat, when to take insulin, how to avoid a dangerous blood sugar crash. Consistent accuracy is essential, not a luxury. Lab tests over the years have confirmed that without compounds like Meglumine, results would jump around too much, defeating their purpose.
Pharmaceutical Solutions Rely on Small Innovations
Making medicines work in the human body isn’t a matter of throwing chemicals together. Drug makers face the tricky job of improving taste, controlling acidity, and making sure the active ingredient reaches the right part of the body. Meglumine works as a pH adjuster and helps stabilize certain formulations. For medicines that need to dissolve in water, it helps make that happen quickly and consistently, which eases swallowing and ensures the body absorbs the medicine as intended.
Looking Out for Patient Well-beingSafety always drives the conversation in healthcare. Tighter regulations now ask drugmakers to justify every additive. Meglumine has shown low toxicity in studies and is considered safe when used in recommended doses, but monitoring remains necessary. Keeping tabs on real-world patient reactions matters more than ever, especially as formulas shift and new uses develop. Open-label clinical trials and patient registries can add a practical layer of vigilance beyond the lab.
Building Trust Through Honesty and Up-to-Date Evidence
Pharmacies and clinics stay busy, and new products appear nonstop. Still, Meglumine shows how even a lesser-known ingredient plays a steady role behind the scenes. Patients benefit when drug and device makers keep lines open with doctors and update their safety knowledge. As a caregiver, I know families feel more secure with clear information. When we can look at the scientific record and ongoing safety checks, trust builds on both sides of the counter.
Is Meglumine safe for human use?
The Real-World Background on Meglumine
Meglumine shows up in places most people never notice. Hospitals mix it into contrast agents for X-rays and scans, chemists use it to make medicines easier to inject, and some drug companies lean on it to help deliver big, water-loving molecules. It’s not a new chemical, either. Since the 1950s, health professionals around the world have worked with it, especially in medicine chest staples like diatrizoate meglumine and some vaccines.
Why Scientists and Doctors Pick Meglumine
One of the big things that pulled me into looking at meglumine’s track record comes down to solubility. Some drugs just refuse to dissolve in water, but meglumine acts like a helper, turning stubborn actives into easy-to-use injections. This gets drugs where they need to be, faster, for patients who don’t have time to wait. Meglumine itself doesn’t provide any direct treatment. It does its job in the background, letting other ingredients shine in medical imaging or therapy.
What Research and Experience Show
Public health agencies and pharmaceutical scientists give meglumine a long, hard look. The U.S. Food and Drug Administration (FDA) has approved several products with meglumine inside, ranging from intravenous contrast solutions to vaccines. Over tens of millions of scans and treatments, data show only rare side effects tied directly to meglumine. Common trouble with contrast agents usually comes from the more reactive iodinated salt, not the meglumine.
I’ve talked to radiologists and pharmacists who trust meglumine-based contrast. In some rare cases, people report headaches, mild nausea, or allergic reactions. Compare that to the risk of undiagnosed tumors or blocked arteries—most medical teams consider the benefits worth the risk. Reports of severe toxicity seem almost non-existent when meglumine is used at medical doses and by trained hands.
What to Watch Out For
It’s not perfect. If someone has serious kidney problems, or if they’re especially sensitive to one of the ingredients, doctors look for other options. Like almost any injectable solution, mistakes can happen: bad mixing, industrial contamination, or dodgy production in places with weak oversight. In my own work, I’ve seen the headaches that come from poor-quality raw materials. Safety also depends on smart storage and handling. If hospitals keep a close eye on their supply chains, risks stay low.
Supporting Safe Use and Transparency
Allergists and patient advocates deserve clear info about every ingredient used in medical treatments. It helps people ask good questions before going in for a scan, or when a new drug is prescribed. There’s also a rising push worldwide for pharma to publish excipient details online, so patients and caregivers aren’t left guessing.
Drugmakers have room to improve. Better batch testing, tough oversight at manufacturing sites, and real-world tracking of side effects build trust. Hospitals can cut down on mistakes by buying from trusted suppliers and keeping up on staff training. If trouble ever pops up with a meglumine product, national and global health organizations share alerts and investigate—making recalls or new rules fast and effective.
The Bottom Line
Meglumine has stood the test of time as a behind-the-scenes player in medicine. In clinics and labs, thousands of professionals rely on it to do a quiet but crucial job, and most patients never notice it’s there. The long record, open data, and careful oversight make it one of the safer supporting ingredients in modern health care. Patients with unique allergies or kidney concerns should share their medical history, but for most people, meglumine stays far from the spotlight—and that’s usually a good sign.
What are the side effects of Meglumine?
What Happens After Using Meglumine?
Meglumine often helps in the world of diagnostic medicine. It forms part of the contrast agents doctors use for X-rays and scans. Folks rarely hear much about it outside the hospital, but for those on the receiving end, its effects can sometimes steal the spotlight from the scans themselves.
The Experiences People Actually Have
Once injected, Meglumine doesn’t fly under anyone’s radar. Some people notice a burning or warm sensation right where it enters the vein. This isn’t surprising. Any substance getting pumped into a vein has the chance to irritate blood vessels or surrounding tissue. With Meglumine, doctors know to watch for this and tell people to say something if it starts feeling pretty uncomfortable.
On top of local effects, some people run into headaches, nausea, or that spinning, lightheaded feeling after Meglumine. Nobody loves sitting through a scan, then having to nurse a throbbing head or a sour stomach. Sometimes, allergic reactions pop up—rash, itching, shortness of breath, or even swelling in tough cases. These reactions push doctors to act fast, bringing in medications to help. It’s worth noting that severe reactions aren’t common, but any medicine running through the bloodstream deserves respect.
Why Do Side Effects Happen at All?
Contrast agents like those containing Meglumine change how the body reacts, at least for a short while. Each person’s immune system steers the ship differently. Some folks create more histamine—a chemical that fuels allergies—after seeing Meglumine. There’s also the fact that contrast agents sometimes draw water out of surrounding tissue or end up irritating blood vessels. That mix gives rise to the side effects people talk about most often.
On rare occasions, kidney function drops after Meglumine hits the bloodstream. This issue mainly crops up in people already dealing with kidney trouble. Studies from large hospitals and health networks keep pointing to underlying health problems—like dehydration or pre-existing kidney disease—making side effects more likely. That’s why before any scan, nurses and doctors pay special attention to health history.
Tackling the Trouble Spots
Walking into a scan room, I’ve always felt more comfortable knowing the staff understand these risks. Honest conversations with the care team make a world of difference. They usually ask about previous allergic reactions, kidney health, and any odd symptoms after past scans. Open, easy-to-understand questions get the ball rolling.
Drinking water before and after scans can help kidneys flush out any contrast agent. Researchers have seen that simple action lower the chances of kidney issues. For those with a track record of allergies, some doctors choose to give medicine before the scan. This extra step helps cut the risk of reactions.
Listening to Personal Experience
My own time dealing with scans sparked a new appreciation for medical teams who explain the process, warn about feelings like warmth or nausea, and lay out what could happen. Not every hospital does it perfectly, so pushing for clear answers and sharing any medical history pays off. Even though most people do fine with Meglumine, staying well-informed and alert keeps people safer before, during, and after a scan.
How is Meglumine administered?
Understanding Meglumine in Simple Terms
Meglumine plays a supporting role in many medical tests, especially in contrast agents for imaging. The most familiar place folks encounter this compound is during radiological procedures, such as CT scans or certain X-rays. Sitting in a waiting room, nervous about what those pictures will show, most people don’t wonder about the liquids that bring those images to life. Yet, the way a contrast agent gets inside the body can shape more than just a test result—it can affect comfort, safety, and even how quickly someone recovers.
Main Ways Doctors Give Meglumine
The most common path for meglumine’s journey into the body is through an intravenous line, often into the patient’s arm. Hospitals have nurses and radiology staff trained to set up these lines with care, checking for allergies and making sure the patient sits steady. Putting the substance straight into the vein gives the fastest, clearest pictures. Having watched patients get ready for scans, there’s a moment of anxiety, but with the right explanation, most folks relax.
Some scans, such as certain urinary tract images, use a different route. Here, the agent flows into the bladder using a catheter. Professionals take steps to keep this as comfortable and sterile as possible. I’ve seen radiologists pause and answer questions, making sure people understand what’s happening and why real-time monitoring helps catch any issues right away.
For gastrointestinal imaging, the compound can travel through the mouth or be administered rectally. Drinking the solution isn’t always a pleasant experience, and anyone who’s been there remembers the odd taste and texture. But compared to older prep methods, many patients handle this way better.
Why Method Matters
How doctors choose to deliver meglumine isn’t decided on a whim. The person’s health, age, and procedure type all factor in. Intravenous use brings speed, letting radiologists snap a perfect sequence of images while the agent courses through blood vessels. Oral or rectal delivery, on the other hand, makes sense when visualizing the digestive tract.
Some people, including children and those with kidney problems, need extra consideration. If a patient’s kidneys can’t clear substances well, doctors might pick another option. In these cases, careful screening and honest discussion matter more than any technical detail.
Safety and Patient Support
Giving contrast agents like meglumine is usually safe, yet allergic reactions, while rare, can occur. Hospitals set clear protocols and educate staff, preparing them for anything from mild itching to rare but severe responses. From my own time shadowing radiology teams, seeing them walk a patient through each step makes a world of difference. Communication isn’t just about comfort—it helps spot problems before they mushroom.
For folks walking into an imaging center, hearing direct, clear explanations goes a long way. Decision-making often involves a quick assessment of risk versus benefit, looking at both medical history and the test’s urgency. At a community level, clinics focus on access to well-trained staff, quality equipment, and patient education materials written in plain language.
Paths for Improvement
Training more professionals on safe administration keeps patients in trusted hands. Encouraging feedback after procedures highlights ways staff can improve bedside manner and reduces anxiety for the next patient. Developing gentler formulations—making solutions less irritating, less likely to trigger reactions—will continue to close the gap between high-tech care and human comfort. As medicine gets more advanced, keeping conversations open between patients, nurses, and doctors remains the quiet foundation for safer outcomes.
Can Meglumine interact with other medications?
Understanding Meglumine’s Role in Medicine
Meglumine doesn’t receive much spotlight outside medical circles, but it plays a big role in helping drugs work better. Often, meglumine gets used as a stabilizer or a solubilizing agent in pharmaceutical products, especially in certain injections and diagnostic agents like radiographic contrasts. Rather than acting as a direct treatment, this compound helps other medicine behave properly in the body. Most folks never hear about it unless they dig through the fine print in a medication insert.
Potential for Interactions: Looking Beyond the Ingredient List
Mixing multiple medications always sparks a bit of worry, especially for people juggling chronic conditions or those with compromised kidneys or liver. Meglumine often travels with other active compounds, and the key concern is how these combinations might change the way drugs move through the body.
Officially, meglumine isn’t seen as a culprit behind dramatic drug interactions on its own. Most available clinical evidence suggests it’s not likely to spark dangerous chemical reactions with common medicines like blood pressure pills, antibiotics, or heart drugs. Even so, many medicines don’t get tested in every possible combination. Mixes can turn up surprises; so can changes in someone’s health status.
Kidney function stands out as a practical concern with any agent that heads through the bloodstream, including meglumine. Some people get intravenous contrast agents that include meglumine during scans, and if their kidneys work slowly, these substances might take longer to clear out. Pentosan polysulfate for bladder pain, as another example, often comes with a meglumine form. Here, the concern revolves around underlying conditions and individual metabolism.
Real-World Issues: Who Should Watch Out?
Patients on multiple drugs for diabetes, heart failure, or autoimmune problems already walk a tightrope. Even so, meglumine rarely becomes the main suspect if medication side effects pop up. More commonly, healthcare teams worry about the main active ingredients. The only way issues get flagged is through careful medication reviews and lab checks.
It’s worth thinking about allergies, too. Some people report reactions after getting radiographic contrast agents. These reactions usually stem from the main contrast component, but doctors sometimes play it safe, considering all ingredients, including meglumine.
From experience, honest conversations between healthcare providers, patients, and pharmacists matter much more than scouring every product insert for meglumine. As a pharmacist, I’ve seen frail patients do fine even on complicated drug regimens, simply because their team catches early warning signs and adjusts medications quickly.
Improving Safety: Taking a Patient-First Approach
Safety starts with medication lists kept up-to-date at every appointment. Patients should always share all prescription and over-the-counter drugs, as well as supplements. Pharmacists double-check for possible overlaps and communicate with prescribers, particularly around procedures using contrast agents.
Manufacturers hold responsibility for clear labeling and making interaction data accessible, so clinicians can make informed decisions. Digital health records with clinical decision support tools lower the chances of oversight.
At the end of the day, drug interactions don’t just depend on chemistry—they depend on teamwork. Clinicians don’t treat ingredient lists; they work with people, and most problems get solved by asking questions, monitoring, and putting the patient’s story at the center of every choice.
| Names | |
| Preferred IUPAC name | (R)-6-(Methylamino)hexane-1,2,3,4,5-pentol |
| Other names |
N-Methylglucamine Methylglucamine Meglucon 4-Deoxy-N-methyl-D-glucamine |
| Pronunciation | /ˈmɛɡljuːmiːn/ |
| Identifiers | |
| CAS Number | 6284-40-8 |
| Beilstein Reference | 3938952 |
| ChEBI | CHEBI:31856 |
| ChEMBL | CHEMBL1201014 |
| ChemSpider | 6737 |
| DrugBank | DB06716 |
| ECHA InfoCard | 13bab29d-7a6b-47b2-b946-9d98c8037b89 |
| EC Number | EC 206-289-5 |
| Gmelin Reference | 9366 |
| KEGG | C01755 |
| MeSH | D008529 |
| PubChem CID | 4206 |
| RTECS number | MB7700000 |
| UNII | 6T57U969ZZ |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | `DTXSID6022756` |
| Properties | |
| Chemical formula | C7H17NO5 |
| Molar mass | 198.24 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.163 g/cm3 |
| Solubility in water | Very soluble in water |
| log P | -2.8 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 8.8 |
| Basicity (pKb) | pKb = 5.3 |
| Magnetic susceptibility (χ) | -63.4×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.457 |
| Viscosity | Viscosity: 20 mPa·s (20°C, 50% aq. sol.) |
| Dipole moment | 7.52 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -421.8 kJ/mol |
| Pharmacology | |
| ATC code | A11GB02 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation |
| GHS labelling | GHS labelling of Meglumine: `"Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008. No GHS hazard pictograms, signal words, hazard statements, or precautionary statements required."` |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "Causes serious eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 166 °C |
| Autoignition temperature | 235 °C |
| Lethal dose or concentration | LD50 (oral, rat): 7500 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5,000 mg/kg (oral, rat) |
| NIOSH | SN2980000 |
| PEL (Permissible) | 50 mg/kg |
| REL (Recommended) | 30 g |
| Related compounds | |
| Related compounds |
Glucamine Sorbitol Mannitol Glucosamine N-Methyl-D-glucamine |

