Maleic Anhydride: More Than Just a Chemical—A Cornerstone That Shapes Modern Industry
A Journey Through History
A lot of breakthroughs grow from humbling origins, and Maleic Anhydride’s rise traces back more than a hundred years. Early chemists found this compound during the relentless search for new materials to fuel the textile boom and the nascent automotive sector. This white crystalline substance stood out not just because it didn’t dissolve in water but also for what it could become. From the moment its reactivity caught the eyes of researchers, minds set out to transform it into everything from resins to specialty additives. The years swept by, inventions came and went, but Maleic Anhydride kept showing up at key turning points, ready for the next wave of chemical experiments or large-scale industrial processing.
Getting to Know the Material
In its solid state, Maleic Anhydride might be mistaken for common table sugar, with flakes that drift readily into the air. Yet those flakes pack a punch: add heat or a touch of moisture and watch it spring to life, quickly morphing into maleic acid or merging with other chemicals. This transition is vital for the chemical’s biggest roles, whether in the lab or on a production line. Most of us probably never heard of this compound outside a classroom, yet countless daily essentials—paints, car bumpers, water pipes—owe part of their performance to it. It’s not known for being tough on its own, but as an ingredient for producing strong, functional plastics and coatings, it can be a game changer.
Decoding Its Character: Physical and Chemical Traits
Open a jar of Maleic Anhydride and your senses get hit by a sharp, choking odor that means business. That should be the first warning: treat with care. It melts easily, evaporates quickly, and reacts fast to water—just a spill on a humid day can result in a tough cleanup. It rarely stays in its original form for long, preferring instead to connect with other molecules wherever possible. These characteristics open doors for creative chemistry. For example, Maleic Anhydride slips neatly into polymer chains, adjusting flexibility and increasing resistance in ways that straight-up hydrocarbons can’t match. Its double bonds make it a valuable springboard for synthesizing more complex chemicals.
Technical Specs and Labeling Practices
There’s a practical side to handling chemicals that gets overlooked amid all the technical talk. With Maleic Anhydride, trusted suppliers print purity levels, recommended storage, and hazard symbols in bold right on the packaging. Industry standards leave little to interpretation: too high a water content, and the product might clump; too low a purity, and you won’t get predictable outcomes in downstream processes. Batch numbers and production dates keep people accountable in case of recall or performance complaints. These pointers might sound routine, but they underpin the transparency and traceability that customers and regulators demand.
How It’s Made: From Raw Input to Finished Product
These days, production lines start with hydrocarbon sources—mainly n-butane or benzene. Controlled oxidation with catalysts produces Maleic Anhydride vapor, which needs careful cooling and collection before it condenses into usable flakes or pellets. This isn’t just a matter of flipping switches; reaction temperatures soar, and even minor deviations create unwanted byproducts. Large reactors packed with specialized catalysts have replaced small-scale glassware, helping to push yields higher and cut down on waste. There’s an ongoing drive to squeeze every last bit of efficiency from the process while minimizing emissions and chemical spills. Every new tweak to the method must pass through fire—from lab bench successes to real-world plant operations—before entering mainstream practice.
Chemical Chameleon: Reactions and Modifications
Chemists prize Maleic Anhydride for its ability to participate in a broad range of reactions. Press it into service as a precursor for unsaturated polyester resin, and you unlock a suite of fiberglass products, boat hulls, and construction panels. Blend it with alcohols to make plasticizers or tweak it into surfactants for cleaning agents. It reacts easily with amines to produce intermediates for water treatment or pesticide manufacture. The double bond and anhydride ring are ripe for various modifications. The fun part, for those who enjoy pushing boundaries, is in designing molecules that behave differently under stress or can stand up to harsher conditions, with Maleic Anhydride nearly always available as scaffolding.
Other Names in the Field
Walk through textbooks or supplier catalogs and you might find Maleic Anhydride listed under names like toxilic anhydride, cis-butenedioic anhydride, or by its systematic chemical identifiers. The jargon might differ from place to place or industry to industry, soaking up the habits of whoever uses it most. Yet, beneath all those labels, it’s the same reactive core driving new product designs.
Keeping Workplaces Safe
Those sharp odors are more than a nuisance. Breathing in Maleic Anhydride dust or vapor can irritate lungs and eyes, making protective gear mandatory in any workplace dealing with it. Fancy air-filtration systems, gloves, goggles, and strict protocols for handling and storage grew out of years of trial and error. Unplanned releases and poor housekeeping once led to costly cleanups and health scares. Learning from those mistakes, best-practice guidelines—like tight containment, cold storage, and emergency eyewash stations within arm’s reach—are now routine. Failure to follow these doesn’t just hurt productivity; it gets people hurt. Regular training and hands-on supervision build a culture where safe handling becomes second nature.
Where Maleic Anhydride Ends Up
Most of its output disappears into making resins. Unsaturated polyester resin, which forms the backbone for countless reinforced plastics, depends on Maleic Anhydride for its backbone and flexibility. Water-distribution pipes, electrical housings, automotive parts, marine vessels, and storage tanks—many of these carry hidden traces of the compound. Beyond heavy industry, some formulations find their way into agricultural chemicals or specialty surfactants. The diversity of applications grows as new chemistry unlocks more efficient or environmentally friendly options, replacing older, more hazardous additives. In places where safety and performance matter—think wind turbine blades or critical infrastructure—there’s almost always a behind-the-scenes contribution from this unassuming flake.
Pushing Boundaries in Research
University labs and private R&D teams continue to hunt for ways to squeeze more life out of Maleic Anhydride. The search for lighter, stronger composites turns up new copolymers or surface treatments. High-performance adhesives that set faster and hold tighter start with more reactive anhydride blocks. Efforts to use biorenewable feedstocks in its manufacture respond to climate demands and cost pressure from fluctuating oil prices. Chemists still look at Maleic Anhydride as a template for tweaking molecular architecture, exploring fresh routes to release stored energy, transport charge, or resist corrosion. Every so often, breakthroughs spill over into commercial applications, changing the economics for industries as diverse as automotive roofing or electronics encapsulation.
Understanding Toxicity and Health Research
Any chemical as active as Maleic Anhydride gets scrutinized. Health authorities flag inhalation and skin exposure as top risks, with symptoms ranging from mild irritation to severe respiratory issues in sensitive individuals. Animal studies point to inflammatory responses and, in rare contexts, longer-term problems following repeated exposure at high levels. Advances in analytical chemistry now allow detection of parts-per-billion in indoor air or process water, making it possible to police workplace conditions tightly. This progress—combined with ongoing toxicological review—keeps regulators, industry leaders, and labor unions on edge. They keep asking how to lower exposure or swap in less hazardous alternatives for vulnerable workers.
The Road Ahead: Sustainable Chemistry and New Markets
As the world pivots towards sustainable manufacturing, Maleic Anhydride is caught between tradition and innovation. Companies invest in non-fossil feedstocks and cleaner reaction paths to ease pressure from new carbon regulations. Circular economy thinking suggests recycling anhydride-containing polymers or designing products for simpler breakdown at end-of-life. Consumer pressure for safer, lower-impact goods creates both headwinds and opportunities. Many of the smartest minds in green chemistry see Maleic Anhydride as both a challenge and a platform: phase out the worst hazards, drive down run-off and emissions, and deliver the same performance using less energy and less waste. Whether these efforts reshape entire swathes of industry or just fine-tune established formulas, the journey reflects a deeper truth—chemical building blocks like Maleic Anhydride rarely stay static for long, and neither do the people committed to using them responsibly.
What are the main uses of Maleic Anhydride?
Building Modern Plastics
People rarely stop to think about how things like pipes, car bumpers, or even phone cases come to life. Maleic anhydride actually plays a real backstage role here. It goes into making unsaturated polyester resins, which find homes in fiberglass-reinforced plastics. You see these plastics in boats, transport trucks, and storage tanks because they last long, don’t rust, and keep things light. I once worked at a construction site where replacing steel tanks with fiberglass cut a project’s time in half. That speed came straight from materials strengthened by products like maleic anhydride.
Shaping Everyday Chemicals
It surprises a lot of people that ingredients in detergents, water-treatment solutions, and lubricants start out in chemical plants with building blocks like maleic anhydride. Companies blend it into compounds called alkyd resins, which are behind the glossy finish of many paints and varnishes spread onto neighborhood fences or backyard decks. So, basically, items built to weather the elements lean on chemistry rooted in this one ingredient.
Improving Food Storage and Agriculture
The agricultural world keeps finding ways to cut down on spoilage. Fumigants derived from maleic anhydride help control pests in stored grains and produce. Succinic acid, which gets made from maleic anhydride, also ends up in food additives to boost shelf life and keep textures just right. I’ve seen local farmers get extra weeks of storage time, which turned out crucial when weather threatened quick harvests. So, we’re not just talking about far-off factories—these chemistry tweaks ripple right into everyday meals.
Strengthening Roads and Coatings
Think about the rough winters that throw road crews into overdrive. Asphalt additives made with maleic anhydride make pavement last longer by sealing up cracks and dealing better with freeze-thaw cycles. One civil engineer I knew swore by asphalt modified with these additives because it shrank repair budgets along some of the busiest highways. Anyone tired of constant potholes owes a little thanks to tweaks made possible through old-fashioned industrial chemistry.
Boosting the Health Sector
Medical industries don’t escape the reach of this chemical, either. Maleic anhydride acts as a stepping stone for pharmaceutical ingredients. Some medications need special coatings to survive stomach acid or reach the right part of the body, and the chemical links built from maleic anhydride often form the foundation of these protective barriers. Health outcomes rely on tiny changes at the molecule level, and the backbone for some of those changes gets set up thanks to this compound.
The Challenge of Safe & Sustainable Supply
Industries enjoy reliable supplies of maleic anhydride thanks to old production methods, mostly based on fossil fuels. Chemical plants release some air pollutants during production, so researchers look for ways to either recycle emissions or tap into renewable raw materials like plant waste. There’s interest in bio-based routes that could lower the carbon footprint without sacrificing quality, but these methods still cost more and need more development. Real progress will depend on more research, support from policymakers, and a willingness from industries to invest in cleaner options.
What is the chemical formula and molecular weight of Maleic Anhydride?
Getting to Know the Basics
Maleic anhydride stands out in the world of industrial chemicals. Its structure and composition play a large part in shaping what it can do. The chemical formula, C4H2O3, says a lot in just a few letters and numbers. The story behind these elements isn't just academic—there’s some real practicality in knowing what each component brings to the table. That formula means you have four carbon atoms, two hydrogens, and three oxygens, working together in a specific arrangement.
Why Look at Molecular Weight?
The molecular weight of maleic anhydride sits at about 98.06 grams per mole. This bit of information matters whether you’re working in a small lab or a large manufacturing plant. If you’ve ever tried to weigh out the right amount of a chemical for a reaction, you know that even being off by a fraction can throw the outcome. This is the foundation for mixing exact blends or scaling up for bigger batches. Getting the math wrong tends to waste both resources and money.
Real-World Experience
During my own time working with chemical supplies, I ran into plenty of folks who didn’t always double-check values like molecular weight. I saw mix-ups simply because someone rounded a value or rushed to convert it for a batch. The fallout could be anything from having to repeat a process, to facing questions over product purity. Not every mishap was catastrophic, but each one set schedules back and caused extra expenses. Getting fluency in things like C4H2O3 and 98.06 g/mol puts you on solid ground.
Why Accuracy Matters in Everyday Use
Maleic anhydride takes part in manufacturing plastics, coatings, agricultural chemicals, and a range of resins. People count on it for making products tougher, keeping coatings intact, and helping paints stick properly. If somebody measures carelessly, the final products lose quality and can develop weak spots. Some big-name suppliers use advanced sensors to double-check everything, because even a minor miscalculation adds up across tons of material. That solution does come with higher up-front investment, yet over time, precision usually pays for itself with fewer ruined batches and less troubleshooting.
Improving Knowledge and Practice
There’s always room to expand skills, whether it’s in school, a startup, or a big production plant. I’ve found that small changes have a lasting impact. Labels kept clear and updated, calculators and conversion charts within arm’s reach, and regular chances to train with material all help. In a community where knowledge moves quickly, sharing accurate information helps everyone—a true example of experience in action.
Moving Toward Safer, Smarter Chemistry
A fact as basic as the chemical formula C4H2O3 and a number like 98.06 grams per mole form more than just textbook details—they hold up the daily workings of several industries. People make better choices when they know their materials inside and out. Reliable data, regular education, and a respect for accuracy protect both workers and the world outside the lab. Maleic anhydride is one example, but the lesson goes far beyond just one compound.
What safety precautions should be taken when handling Maleic Anhydride?
Understanding the Risks of Maleic Anhydride
Maleic anhydride comes up often in manufacturing plastics, resins, and coatings. It doesn't just sit on a shelf, though; it reacts quickly with water. One little whiff of its pungent odor tells you it’s not something to take lightly. Workers see it irritate skin, eyes, and lungs. Chronic exposure ramps up risks for asthma and other respiratory issues. I’ve seen coworkers experience rashes after a little spill landed on unprotected skin. It’s clear that taking shortcuts with this chemical can spell real trouble.
Protective Gear Makes All the Difference
Good lab safety often starts before you even open a package. Nitrile gloves, long sleeves, and splash-proof goggles have proved reliable in every shop I’ve worked. The fumes can burn your eyes and throat. A face shield and well-fitted respirator aren’t extras—they’re essentials. Filters for organic vapors handle the job, as long as folks replace them before they’re saturated. Chemical-resistant lab coats or aprons stop splashes from seeping through to skin. Closed shoes—never sandals. I’ve seen accidents where just one person ignored shoes, and the results were painful.
Control the Air, Control the Risk
Ventilation matters. Fume hoods and exhaust fans provide a huge line of defense. I always check that fans work before weighing out or mixing maleic anhydride. In one factory, sensors linked to alarms caught vapor spikes before they could hurt anyone. It’s also smart to handle maleic anhydride in smaller amounts where possible, minimizing what might spill or leak if something goes wrong.
Safe Storage Prevents Bigger Problems
Water contact turns maleic anhydride into maleic acid, and that’s a mess if it happens inside a storage area. Storage in tightly sealed containers, kept in dry rooms, stops unwanted reactions. Labeling stays key for avoiding mix-ups; confusion once led a coworker to use the wrong container, with clouds of irritating fumes as a result. Separate maleic anhydride from bases, oxidizers, and moisture sources to avoid dangerous combinations.
Emergency Steps Can Save Health
Spills demand immediate action. I remember drills where we practiced with neutralizing agents and absorbent clay. Fast cleanup keeps everyone safe. In case of contact, eye wash stations and showers should always be nearby and working, not just for looks. Everyone needs a run-through on emergency plans—who to call, what steps to follow. Quick decisions come easier if everyone has clear instructions.
Training and Awareness Go Further Than Rules
Safety posters alone don’t cut it. Peer discussions and shared stories about close calls sink in better than a rulebook. Regular training on handling and emergency response means the information sticks. Supervisors who enforce safety without making it a chore help create a culture of watchfulness. A sense of responsibility to each other keeps eyes sharp for careless habits. Open lines of communication foster honesty; if someone makes a mistake, the team learns from it and adapts.
Looking Ahead
Chemicals like maleic anhydride bring big benefits to manufacturing but only if respect for their risks stays front and center. Personal protective equipment, strong ventilation, careful storage, and ongoing training shape a safer workspace. Every layer of precaution, from double-checking bottles to running emergency drills, matters. When workers look out for themselves and each other, long-term health and safety track record speaks for itself.
How should Maleic Anhydride be stored and transported?
Why Care About Safe Handling?
I’ve seen what goes wrong in plants where chemicals get only basic attention. With maleic anhydride, that relaxed attitude invites trouble. This white, sometimes needle-like solid plays its part in all sorts of products: resins, coatings, additives—vital for industrial routines. Yet it brings real risks. It reacts with water, releases corrosive fumes, and affects the eyes, respiratory system, and skin. Every worker deserves an environment that keeps their health in good shape and avoids environmental trouble.
How to Store It Properly
Walk into a warehouse that cares about safety, and you’ll notice maleic anhydride staying out of damp corners. Industrial containers—often drums or bags lined with polyethylene—do the heavy lifting because they resist chemical attack. The substance hates water. Even normal air moisture can trigger it to clump, cake up, or release vapors nobody wants to breathe. In my own experience, bulk storage usually happens in steel tanks fitted with vents and driers to keep the atmosphere bone-dry.
Temperature makes a difference, too. Maleic anhydride melts at about 52°C. The solid version stays stable when kept cool, away from heat or sunlight. Many facilities space barrels to guarantee airflow. Piling drums close may seem like a space-saving trick, but it traps heat, and one warm day can push the product past its melting point. Nobody wants a surprise pool of chemical underfoot or escaping fumes. At every job I’ve worked, clear labeling and separation from reactive or flammable materials stays non-negotiable. Nitrile gloves, goggles, and masks shouldn’t collect dust on a hook; they need to sit within arm’s reach.
Moving Maleic Anhydride Without Headaches
I’ve seen transport nightmares caused by shortcuts. Good practices start with containers that can’t leak and can take a bump. Most shipments roll out in sealed drums, bags, or, for large orders, special tank trucks. These tanks come fitted with nitrogen blanketing—simply replacing humid air with dry, inert nitrogen stops water from sneaking in. That keeps both people and product safer. Boxes and drums go only in vehicles with stable temperatures, with plenty of anchor points to stop shifting during a sudden stop or sharp turn.
A spill or leak turns into an emergency fast. Regulations demand that drivers know what sits behind them in the cargo. MSDS sheets ride along with every load, and transporters trained in handling chemical leaks reduce panic and guesswork when a drum springs a leak. Contingency plans and easy access to ventilated spaces make all the difference. In my own work, even short hauls included double-checking seals and ensuring spill kits ride in the cab.
Raising Standards—Not Just for the Rulebook
Government rules didn’t pop up by accident. In the US, the Department of Transportation and OSHA offer strict guidelines for packaging, labeling, and record-keeping. European regulators stick to REACH and ADR transport rules. These aren’t annoying hoops to jump through; they reflect disasters that taught us hard lessons. I’ve watched teams grumble at training sessions, then admit relief when procedures kicked in during an incident. Regular checks for corrosion, dented barrels, or leaks matter just as much as fire extinguisher drills or inventory checks. Even budget-minded managers start taking these details seriously after facing a fine—or worse, an accident that injures a colleague.
Shaping a Safer Approach
Warehouses and transport fleets need teamwork to handle maleic anhydride confidently. Smart layouts cut down on errors. Routine inspections flag problems before they turn hazardous. Investment in good personal protective equipment pays off not just in compliance but in peace of mind. Clear communication, straightforward labeling, and an investment in staff training lower the chances of problems. The best-run facilities treat proper storage and transport as everybody’s business, not just the safety manager’s job.
What are the physical and chemical properties of Maleic Anhydride?
Physical Properties: What the Senses Reveal
Maleic anhydride shows up as white, needle-shaped crystals that pack a strong, choking odor—something like the kind of sour tang you smell in some strong acids. If you’ve handled it in a lab, you’ll know the fumes sting the nose and eyes fast. The substance quickly turns powdery or melts into a colorless liquid at fairly low temperatures, around 52.8°C (127°F). While many imagine all white powders dissolve easily, this one breaks that rule; it barely mixes with cold water, but hot water does the trick. When it hits water, a chemical reaction immediately kicks in, releasing heat and turning maleic anhydride into maleic acid. So anyone pouring it close to moisture watches for bubbling and rising steam.
Its vapor feels even more aggressive than the solid. Maleic anhydride vaporizes at 202°C, so in poorly ventilated spaces, the fumes become a hazard quickly. The material works as a solid under normal room temperature, but just a few degrees above that stop point, it liquefies and can even boil if left unchecked. Most people who work with it keep a sharp eye on any spills—contact with skin burns, and breathing in even a whiff brings coughing fits or worse.
Chemical Properties: Reactivity with a Kick
Maleic anhydride behaves as a highly reactive chemical, not a passive bystander. Its biggest trick comes from the anhydride group. Drop it into water, and it doesn’t just dissolve: it jumps into a hydrolysis reaction, quickly forming maleic acid and unleashing heat. I’ve seen beakers go from calm to fizzing in seconds, so handling it in a dry environment always makes sense.
A double bond sits at the heart of its ring structure, which pulls in other reactants with ease. Industries use that trait to their advantage, making resins, plasticizers, and coatings. Maleic anhydride links up with compounds like alcohols and amines in snap reactions, putting it at the center of large-scale chemical synthesis. Reactions with bases lead to salts, and strong oxidizers may trigger even more dramatic shifts in the lab.
Safety and Environmental Insights
Working with maleic anhydride calls for strong gloves and a fume hood. Pure maleic anhydride triggers sneezing, coughing, and burning eyes. If it touches the skin, burns follow. Chronic exposure poses a long-term danger—not just to humans, but to the environment. Spills in water don’t just dilute the chemical; they set off the same hydrolysis reaction as in the lab, potentially harming aquatic life by changing water acidity.
There’s a reason regulations put strict limits on air and water releases from plants that make or use it. Where communities neighbor production sites, proper containment helps prevent the problems that come from these toxic fumes. Management teams keep tight records on storage and training, and you’ll find regular air monitoring in hot production zones.
Alternatives and Solutions
Industry moves to safer alternatives where possible, but for applications like resins and coatings, maleic anhydride still scores points for reactivity and performance. Engineers and chemists look at closed systems and automated handling to curb exposure. Spill kits and training sessions often make the rounds in factories. Plant operators get feedback from health departments and sometimes run drills to keep teams ready for emergencies. These steps make sure the upside of using maleic anhydride comes without leaving health or the environment behind.
| Names | |
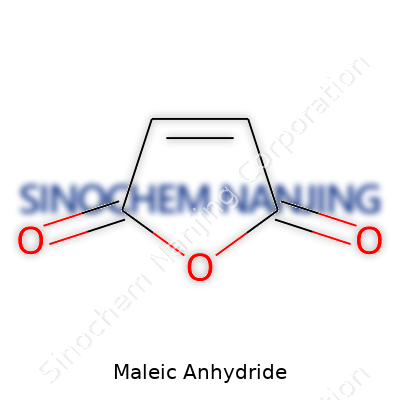
| Preferred IUPAC name | Furan-2,5-dione |
| Other names |
cis-Butenedioic anhydride Dicarboxylic acid anhydride Toxilic anhydride MA 2,5-Furandione |
| Pronunciation | /məˌliːɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | 108-31-6 |
| Beilstein Reference | 1204367 |
| ChEBI | CHEBI:28243 |
| ChEMBL | CHEMBL1402 |
| ChemSpider | 532 |
| DrugBank | DB14405 |
| ECHA InfoCard | 100.003.335 |
| EC Number | 205-015-6 |
| Gmelin Reference | 789 |
| KEGG | C01197 |
| MeSH | D008287 |
| PubChem CID | 11403 |
| RTECS number | OM2100000 |
| UNII | W7M6SJ93ED |
| UN number | 2215 |
| Properties | |
| Chemical formula | C4H2O3 |
| Molar mass | 98.06 g/mol |
| Appearance | White crystalline solid |
| Odor | Pungent choking odor |
| Density | 1.48 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -0.73 |
| Vapor pressure | 0.4 mmHg (25°C) |
| Acidity (pKa) | 1.9 |
| Magnetic susceptibility (χ) | -35.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.554 |
| Viscosity | 2.6 mPa·s (at 25 °C) |
| Dipole moment | 2.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 260.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -462.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1791 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02, GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H334, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P310 |
| NFPA 704 (fire diamond) | 2-2-1-W |
| Flash point | '102 °C (216 °F) - closed cup' |
| Autoignition temperature | 435°C |
| Explosive limits | 1.4–7.1% |
| Lethal dose or concentration | LD50 oral rat 2,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1090 mg/kg |
| NIOSH | BSA:CK6820000 |
| PEL (Permissible) | PEL: 1 ppm (6 mg/m³) |
| REL (Recommended) | 30 mg/m³ |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Maleic acid Fumaric acid Phthalic anhydride Succinic anhydride Citric acid Acrylic acid |

