Magnesium Octanoate: From Lab Curiosity to Versatile Workhorse
Historical Development
Magnesium octanoate hasn't always played a starring role in the world of organometallic compounds, but like many specialty chemicals, its story traces back to the drive to solve real problems. Early research delved into fatty acid metal salts, seeking catalysts, stabilizers, and alternatives to harsher chemicals. Magnesium, already favored for its biocompatibility, coupled with octanoic acid—a medium-chain fatty acid—offered a blend of mildness and versatility. Researchers in the late 20th century recognized the compound’s promise, not only in industry but also in certain biological and environmental settings. In my professional experience, looking through decades of applied chemistry literature, magnesium octanoate keeps cropping up in surprising corners, reflecting a pattern: chemists and engineers often revive “old” compounds for new challenges.
Product Overview
Magnesium octanoate can show up as a fine white to off-white powder, or as a waxy solid depending on purity and hydration. It dissolves in many organic solvents and reacts with strong acids, typical of metallic salts bound to organic acids. Its chemical resilience and moderate reactivity underpin its appeal. In practical terms, manufacturers gravitate toward it for its gentle catalytic properties, and because it introduces magnesium—a nutrient element—without harshness. In paints and coatings, it acts as a drying agent. In nutrition, it works as a minor additive. Its place in the chemical toolbox isn’t super-glamorous, but it’s quietly essential where gentler touch or magnesium supplementation is needed.
Physical & Chemical Properties
Anyone working in a lab quickly notices the distinctive smell of medium-chain fatty acids, and octanoate brings that slightly waxy aroma. Magnesium octanoate doesn't pack an elemental punch like heavier metals, but its relatively low toxicity stands out. It's moisture-sensitive, and careless handling in humid conditions leads to clumping or even partial hydrolysis. It melts at a moderate temperature, making it easy to incorporate into manufacturing processes without thermal degradation. Its moderate solubility in both polar and non-polar organic solvents broadens its use—paint shops and processors seldom enjoy fussing with poorly soluble reagents. Chemists remember that it’s more stable than the sodium or potassium salts, which can hydrolyze or clump even faster.
Technical Specifications & Labeling
Regulatory and technical standards push for clear, concise labeling nowadays, especially when a compound appears in consumer-facing products. Specifications commonly mention minimum magnesium content, acid value, water content, and sometimes particle size for specific applications. Labels must warn of combustion risk—octanoates burn, releasing acrid fumes. In my own work in quality control, I always appreciated suppliers who included full traceability: batch numbers, purity grades, and details about residual solvents. Certifications for food applications, like meeting national pharmacopeia standards, have grown in demand as awareness rises about even trace contaminants.
Preparation Method
Making magnesium octanoate is as straightforward as it gets in basic synthesis. Direct neutralization of octanoic acid with magnesium oxide or magnesium hydroxide yields the product, followed by evaporation or solvent removal. Process details matter, especially in controlling particle size and minimizing residual acid. Factories that skimp on purification wind up with off-odors or color changes, which sensitive applications and recalcitrant regulatory auditors catch every time. Problems around reaction exothermicity and solvent management crop up in scale-up, so plant managers invest in good temperature control and waste handling.
Chemical Reactions & Modifications
Magnesium octanoate participates in reactions reminiscent of its carboxylate relatives. It acts as a mild base, sometimes as a catalyst for esterification, and occasionally as a source of magnesium in exchange reactions. Chemists tweak it by substituting other medium-chain acids or blending into multi-metal soaps—balancing drying rates or stability in paint systems. In some emerging technologies, researchers experiment with magnesium carboxylates as feedstocks or cross-linking agents in biodegradable polymers. My own take: the future of these magnesium compounds lies in combining gentle reactivity with sustainability, especially as regulations nip at the heels of heavier metal soaps.
Synonyms & Product Names
Depending on supplier, literature, or country, magnesium octanoate also appears as magnesium caprylate, magnesium octanoic acid salt, and sometimes less formal adaptations. In commerce, “magnesium caprylate” wins out because the food and nutrition industry leans on the caprylic acid naming tradition. This confusion over names has tripped up plenty of purchasing managers, often leading to exasperating delays or miscommunication across borders. It pays to check chemical Abstracts Service (CAS) numbers for certainty, as brand names shift faster than technical standards do.
Safety & Operational Standards
No matter how benign, chemical handling demands respect. Magnesium octanoate’s low acute toxicity makes it less worrisome than many metal salts, but inhalation of dust or prolonged eye contact causes irritation. In the plants I’ve toured, robust local exhaust ventilation and regular spill cleanups keep down allergy and skin complaints. Fire remains a risk—octanoate salts don’t explode, but they feed fires once started. Employees sometimes forget that even mundane chemicals pose hazards in bulk, especially when stored near oxidizers. Food and pharma producers insist on current good manufacturing practices (cGMP), forcing regular reviews of handling procedures and audits of suppliers’ process controls. As magnesium octanoate turns up in new applications, especially in supplements or biomedical research, medical screening for chronic exposure might become a regulatory talking point.
Application Area
Industrial chemists know magnesium octanoate mainly for its role as a drying agent in paints, coatings, and inks, where it speeds up polymer cross-linking without the environmental baggage of heavy metals. In nutritional supplements and food processing, it functions as a mild source of magnesium, bringing the benefits of both magnesium and medium-chain fatty acids. It's used in veterinary formulations, often as a stabilizer, and in pharmaceutical production as a component in magnesium delivery systems. Some water treatment facilities investigate it for its potential to bind unwanted ions, though scale-up remains limited. Environmental pressure to reduce lead, cobalt, and manganese in processes keeps nudging formulators toward magnesium alternatives, making octanoate-based recipes more attractive.
Research & Development
The R&D horizon for magnesium octanoate shines brighter than one might guess, given its old-school roots. Academia and startups alike keep probing its role as a benign catalyst in green synthesis, and as a magnesium supplement in medical research targeting metabolic and neurological health. Biodegradable polymer fields show interest, banking on both its mild reactivity and its positive nutrient impact. Nanotechnology researchers evaluate it as a stabilizer for certain particles and delivery vesicles, though these uses still sit closer to grant proposals than large-scale production. As the push for sustainable specialty chemicals grows, the pace of basic research on magnesium organic salts should only accelerate.
Toxicity Research
Magnesium octanoate’s reputation owes much to thorough animal studies showing low acute and chronic toxicity when used within prescribed limits. It’s not free from risk—sensitive populations or those with impaired magnesium metabolism should avoid extra supplementation. Its breakdown products, mainly octanoic acid and magnesium ions, already show up in trace amounts in common diets. Occupational exposure limits have not been globally harmonized, so some countries press for tighter controls than others. Long-term ecological studies find little persistence in groundwater, yet more work addressing chronic trace ingestion and disruption to aquatic ecosystems would help guide future approvals.
Future Prospects
Magnesium octanoate stands at an interesting crossroads. As regulatory agencies crack down on hazardous metal soaps, and as industries chase after lower-toxicity, biocompatible materials, magnesium octanoate picks up more interest each year. I keep seeing university research projects exploring its use in biodegradable polymers, novel food fortification, and even as a mild plant growth stimulant. There's an opportunity for cleaner synthesis routes with less waste, and broader adoption in food, cosmetic, and biomedical industries as more data supports its safety. This compound may not generate headlines on its own, but in the shifting landscape of specialty chemicals, products with benign profiles and multiple technical roles rarely go out of style. As innovation marches on, magnesium octanoate is likely to surprise everyone with its reach and resilience.
What is Magnesium Octanoate used for?
Digging Into Applications
Plenty of folks may not give magnesium octanoate much thought, but this compound quietly pulls weight across several industries. It's not a household name like table salt, yet it works behind the scenes, keeping products stable and boosting performance. Whenever I step into a factory or visit a lab, there's a good chance I'll bump into something related to this compound in some form or another.
One of the biggest roles for magnesium octanoate shows up in plastics manufacturing. Manufacturers look for ways to control polymerization—the process that turns tiny molecules into the sturdy plastics for packaging, toys, and everyday household items. By using magnesium octanoate as a catalyst, teams can tweak processes to make stronger, longer-lasting plastic products. From a practical perspective, that means less cracking and brittleness in containers or toys I buy for my kids. That sort of reliability matters when you start thinking about waste and consumer frustration.
Helping Paints and Coatings Last Longer
Magnesium octanoate isn't just working in plastics. Every time I pick up a can of paint, there's a good chance that this material played its part. Paints and coatings require additives to help them dry at a predictable pace, and magnesium octanoate gets picked as a drying agent. It helps paints cure more evenly, which cuts down on streaks and bubbling. I've rolled on enough coats over the years to know that smoother drying means less rework and more consistent results on the wall or furniture.
Beyond improving how paint dries, it also strengthens the bond to the surface, so paint jobs last longer before showing wear. Fewer touch-ups over the years means less money out of my pocket and less paint headed to the landfill.
Making Food Production More Efficient
Outside the factory and paint aisle, magnesium octanoate finds its way into food production. Food manufacturers use it as an emulsifier. In simpler terms, it helps mix oil and water together when those ingredients simply don’t want to blend. Think of salad dressings, baked goods, or chocolates where consistency makes a big difference in taste and shelf life. Consistent blending also cuts down on waste from batches that don’t turn out right. The food sector demands clear standards for additives, so only compounds shown to be safe make it into commercial kitchens.
Safety and Sustainability Questions
There are always concerns that tag along with chemical additives. Experiences over the years prove that rigorous testing matters. The right oversight, with real transparency from companies and regulators, makes all the difference. Magnesium octanoate’s safety record comes from both animal and human studies reviewed by food and chemical safety boards. Still, I always encourage open discussion about additive choices, especially for those dealing with allergies or sensitivities.
Sustainability and responsible sourcing should go hand-in-hand with scientific innovation. Producers who focus on minimizing waste and reducing emissions in magnesium octanoate’s manufacturing show leadership. I’ve seen the difference in communities where companies invest in cleaner technology—it pays off in healthier air and water for everyone.
Room for Smarter Solutions
There’s room for improvement. More research into natural alternatives and better recycling systems could lessen environmental pressure down the line. Supporting innovation—not just bigger profits—creates options for safer, more eco-friendly additives. By asking questions and paying attention to labels, everyday people play a part in guiding industries to be both safer and smarter about what they put into products.
Is Magnesium Octanoate safe for human consumption?
Understanding Magnesium Octanoate
Magnesium octanoate doesn't show up in most kitchen cupboards or health food aisles. It’s a magnesium salt connected to caprylic acid, sometimes used in food science and supplements. Folks run into it through preservation, as a minor ingredient in supplements, or as a part of food additives approved for various reasons. The immediate question for many people revolves around one thing: is eating this stuff a risk?
Real-World Evidence and Available Data
Experiences from food chemistry show that many preservatives and additives get scrutinized far beyond what the average apple or orange endures. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) keeps a tight watch on food additives, including magnesium compounds. It's important that magnesium octanoate is distinct from more common magnesium salts like magnesium citrate or magnesium chloride, but it operates on similar metabolic lines—providing magnesium and breaking down to octanoic acid, which the liver processes.
Existing animal studies indicate that both parts—magnesium and caprylic acid—are handled well by the body in reasonable amounts. Magnesium plays a huge part in more than 300 enzyme reactions, heart rhythm, muscle work, and nerve function. Caprylic acid, found in coconut oil and milk, works as an energy source. Too much of either part isn’t wise. High magnesium, especially from supplements, can upset stomachs or, in serious cases, harm kidney function.
You see more additives like magnesium octanoate appearing in ingredient lists as food scientists chase better shelf-life, texture, or flavor. What matters is whether trustworthy authorities have stamped these additives as generally recognized as safe (GRAS). The U.S. Food and Drug Administration (FDA) checks each food additive and provides updates as new science appears. A search on the FDA’s GRAS notices and food additive status for magnesium octanoate specifically doesn’t turn up red flags. In the European Union, EFSA addresses new food ingredients through an even tighter system, prioritizing consumer safety.
Questions About Newer Ingredients
Families and those with allergies or health conditions notice each new chemical added to the food supply. Trace levels of magnesium octanoate in food don’t raise alarms for healthy adults, but folks with kidney issues or people on medications affecting magnesium processing should always talk with their doctors before trying new supplements.
Children, pregnant women, and anyone with chronic disease feel added risk. Extra caution means sticking mostly to whole foods and reading every ingredient label. Processed products often carry a whole string of additives that work together, sometimes in ways that research hasn’t fully tested.
Looking Forward: The Gap in Research
Research on magnesium octanoate’s long-term effect on the gut, allergies, and overall health looks incomplete. Food safety organizations worldwide keep an eye on additives—anything raising a hint of harm usually gets strict re-evaluation. Consumers won’t always spot clear answers, so demanding more transparency from manufacturers makes sense. Asking food companies and supplement brands for current safety studies gives shoppers leverage and assurance.
Paths Toward Reassurance
Most people benefit from a diet focused on fresh, simple meals, but for anyone encountering magnesium octanoate in new formulas, it pays to check sources. Watching for updates from groups like the FDA and EFSA, and reporting any bad reactions to health authorities, keeps the food chain safer. Whenever a batch of synthetic or less common magnesium compounds arrives on the market, it’s important for producers to back it with real research and share results with the public.
Personal experience and professional knowledge agree here—new food chemicals need open discussion, regular testing, and honest labeling. Without that, trust doesn't grow. Magnesium octanoate looks low-risk for most, but attention to individual health differences keeps food choices truly healthy.
What is the chemical formula of Magnesium Octanoate?
What Makes Up Magnesium Octanoate?
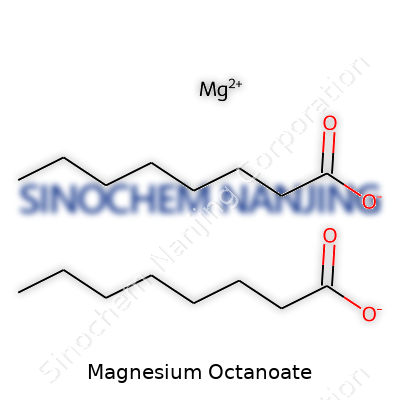
Magnesium octanoate stands out because it connects a mineral—magnesium—with a fatty acid called octanoic acid (caprylic acid). The moment people ask about its chemical formula, they’re looking for more than a string of letters and numbers; they want to know what’s inside. Magnesium usually looks for partners to balance its charges. Octanoic acid, found in coconut oil and even milk fat, links up to form the salt. The final formula: Mg(C7H15COO)2 or, put another way, C16H30MgO4.
Why Does Its Structure Matter?
Taking chemistry at college showed me that the devil is in the details—even simple changes in structure can shift how a chemical behaves in the body or the environment. For magnesium octanoate, that long eight-carbon tail from octanoic acid gives it unique properties. The molecule can dissolve in fats and oils, so it’s different from other magnesium compounds that usually stick with water. This matters for magnesium supplements, cosmetics, and even animal feed.
Plenty of companies reach for this salt because magnesium plays a role in muscle health, nerve function, and enzyme activity. People who struggle to keep magnesium levels up could benefit from a compound that the body absorbs more easily. Research shows that organic salts of magnesium—like magnesium octanoate—tend to pass through cell membranes faster than simple inorganic ones such as magnesium oxide.
Potential Issues and Challenges
Looking into supply chains, consistency jumps out as a problem. The fat end, octanoic acid, often comes from plant oils. Purity can vary. In personal work with supplements, tracing back the origin reveals surprising differences in quality. Contaminants can sneak in if manufacturers don’t pay close attention to sourcing and processing. This stuff matters—nobody wants heavy metals or pesticides in a nutritional product.
Labeling isn’t always clear either. The food and supplement industries sometimes gloss over real chemical differences. Some products say “magnesium” without telling buyers which compound sits inside. That’s where knowing the formula comes in—C16H30MgO4 signals a specific setup with potential benefits for absorption.
Moving Toward Solutions
Better accountability starts with stronger transparency. Labs should publish purity tests and sources for each ingredient, letting customers make informed choices. Personal experience with lab testing for supplements taught me that only a handful of companies share certificates showing what’s really in the bottle.
On the research front, more comparative studies would help decision-makers know whether magnesium octanoate really performs better than other forms in terms of absorption and health impact. Right now, most claims sit on shaky ground, with limited large-scale clinical data.
What to Watch Going Forward
Demand for new forms of minerals keeps rising, especially among those searching for better health. The details in the chemical formula aren’t just for scientists—they help us all understand where our nutrients come from, and why some ingredients feel or work differently. Breaking down labels and formulas lets consumers take charge of what they eat and use. Careful sourcing and full disclosure would raise the bar for everyone.
How should Magnesium Octanoate be stored?
Practical Realities of Storing Magnesium Octanoate
Plenty of folks working with specialty chemicals already know: handling and storing magnesium octanoate isn’t something you shrug off. Having seen spills, ruined material, and tarnished results, it feels personal. It matters on the shop floor just as much as it does in a research facility. If the right steps slip, you deal with more than just wasted product—you face unnecessary hazards and financial losses.
Humidity and Air: The Hidden Threats
Moisture seems harmless until it reacts. Even small changes in the storage area’s humidity can degrade magnesium octanoate over time. Chemical shifts happen fast when the material absorbs water from the air. That pile of expensive raw material now holds less value and possibly brings risk during handling. Anyone who’s opened an improperly sealed drum knows the musty, clumpy result—bad news if purity really matters to your process.
Air exposure can also trigger oxidation. Once magnesium octanoate goes through even minor chemical changes, you lose the consistency needed for pharmaceuticals, lubricants, or specialist synthesis work. Fluctuating temperatures make matters even worse since condensation can sneak past basic seals or ill-fitting lids.
Light and Temperature Issues
Daylight and strong artificial light can break some chemical bonds if you aren’t careful. Sunbeams streaming through a window may look harmless, but over months they kick off slow degradation processes. Keeping magnesium octanoate in a cool, dark space preserves its chemical profile. Growing up around a family business that handled everything from cement to powdered milk, I saw spoiled stock more than once just from neglecting proper lighting or venting.
Most formulations keep well at mild room temperatures. Extreme heat encourages breakdown. A climate-controlled space, even something as simple as a locked steel cabinet in a temperature-stable basement, keeps both sunlight and thermal shifts from ruining inventory.
Safe Packaging and Smart Labeling
Thick, airtight containers make the difference between good chemistry and a supply headache. Storing magnesium octanoate in double-sealed polyethylene or HDPE containers with gasket-fitted lids stops leaks and blocks moisture. I remember patching up cracked containers during one tight month, only to find everything had caked into useless clumps. Investing in the right containers and tight seals reduces the risk and cuts down long-term costs. Always label containers clearly with the chemical name and last inspection date. This sounds obvious but it saved my team more than once during inventory checks.
Separation From Incompatibles
Mixing different chemical types in shared shelves or bins causes mistakes. Keep magnesium octanoate well away from strong acids, bases, or oxidizers. Even a handful of stray granules from other substances can trigger chemical issues—especially in bulk environments or high-turnover labs. Clear zones and physical dividers guide workers so cross-contamination never sneaks in during a rushed moment.
People and Preparedness
No amount of procedure beats actual training. Every person accessing magnesium octanoate needs reminders about gloves, face shields, and mask use, since many people don’t realize the small health risks with repeated skin or lung exposure. A simple checklist by the storage area, paired with monthly refreshers, prevents carelessness. Regular inspection dates matter, too—every bag and drum deserves a look for leaks, label legibility, and the feel of the material inside.
Careful daily effort pays off in fewer wasted supplies, safer workplaces, and long-term savings. Even if it sometimes feels like overkill, the details make the biggest difference.
What industries commonly use Magnesium Octanoate?
The Real Uses Behind This Specialty Ingredient
Magnesium octanoate doesn’t show up in everyday conversation. Most folks won’t spot it on consumer labels, either. Yet it plays a quiet but crucial role in more places than people realize. During my years speaking with manufacturers, formulators, and environmental specialists, I’ve learned how this ingredient shapes results—especially for the coatings, plastics, rubber, and agricultural sectors.
Coatings and Paints: Getting Dry to Perfection
Grow up in an old house and you notice which corners of a room keep paint sticky long after everything should have set. Traditionally, paint chemists have juggled speed, finish, and durability. Alkyd paints rely on “driers” for their quick, reliable cure. Magnesium octanoate is prized here; manufacturers appreciate its steady drying boost without sending off as many fumes as metal-based driers like cobalt or manganese. According to reports from European coatings companies, using this compound helps balance environmental rules and customer expectations for a glossy, smudge-proof finish. Responsible sourcing is starting to matter more in construction, and magnesium fits the bill with a comparatively mild hazard profile and fewer disposal issues. That’s a win for both painters and the families living with the result.
Plasticizers and Polymers: Flexibility Where It Counts
Look at tech or car parts from the last decade, and you’ll see materials that snap back from a squeeze. Plasticizers are behind that. In PVC, magnesium octanoate acts as a stabilizer, helping plastics stay flexible without toxic residues. Industry case studies point out that this helps factories sidestep some of the legacy problems tied to heavy metals. Because magnesium performs the job with less risk, compliance officers and engineers can worry less about regulatory headaches and more about product quality. Many plants also report that workers handle it with standard PPE; there’s less concern about accidental contact compared to lead-based chemicals.
Elastomers and Rubber Goods: Balancing Bounce and Safety
Years spent watching rubber manufacturing lines showed me the balancing act between performance and safety. Car tires, gaskets, hoses—all need a catalyst to cross-link rubber molecules, often under strict time and heat controls. Magnesium octanoate accelerates this process effectively, and manufacturers keep embracing it for consistent product texture, fewer halts in the line, and lower risk of environmental notices. Tire manufacturers want each batch reliable, and the compound helps them deliver.
Agriculture: Feeding Plants, Reducing Runoff
Magnesium’s role extends into fertilizer and crop-protection blends. Large producers explain how octanoate forms deliver magnesium in a way that plants take up efficiently, supporting greener leaves and heartier roots. By choosing this form, modern growers see less waste and less magnesium washing off into waterways. Research shows proper mineral delivery boosts not just yield, but nutrient density in harvested food—a benefit for families and for soil health. Compared with older mineral mixes, magnesium octanoate lowers the risk of fertilizer burn and is often listed in organic and specialty crop production guides as an approved option.
Better Choices, Healthier Workplaces
In all these fields, magnesium octanoate earns trust because it does a tough job without the baggage of older, heavier metals. Consider the rising tide of chemical safety campaigns. Companies and workers gravitate toward tools they trust not to endanger health or compromise performance. Lab managers tell me magnesium-based additives mainstreamed more quickly because of reassuring toxicology results and straightforward handling. Factories set up for this type of chemistry generally see fewer accidents linked to outdated materials. The broader shift to “greener” chemistry keeps putting this ingredient in the spotlight for engineers and buyers alike.
| Names | |
| Preferred IUPAC name | magnesium octanoate |
| Other names |
Magnesium caprylate Magnesium caprylate octanoate Magnesium salt of caprylic acid Octanoic acid magnesium salt |
| Pronunciation | /maɡˌniːziəm ɒkˈteɪnəʊeɪt/ |
| Identifiers | |
| CAS Number | 31169-19-8 |
| Beilstein Reference | 3853086 |
| ChEBI | CHEBI:131334 |
| ChEMBL | CHEMBL3980899 |
| ChemSpider | 13715873 |
| DrugBank | DB14643 |
| ECHA InfoCard | echa.europa.eu/substance-information/-/substanceinfo/100.259.246 |
| EC Number | 208-720-4 |
| Gmelin Reference | Gmelin Reference: "170350 |
| KEGG | C16082 |
| MeSH | D000326 |
| PubChem CID | 162064 |
| RTECS number | OM2625000 |
| UNII | 1P59HP4OQS |
| UN number | UN2813 |
| Properties | |
| Chemical formula | C16H30MgO4 |
| Molar mass | 272.50 g/mol |
| Appearance | White to light yellow powder |
| Odor | characteristic |
| Density | 1.04 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | log P = 4.2 |
| Acidity (pKa) | 5.0 |
| Basicity (pKb) | pKb ≈ 4.8 |
| Magnetic susceptibility (χ) | −13.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.435 |
| Viscosity | Viscosity: 55 mPa·s |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -802.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5592.6 kJ/mol |
| Pharmacology | |
| ATC code | A12CC15 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | Wash thoroughly after handling. Wear protective gloves/eye protection/face protection. IF ON SKIN: Wash with plenty of water. If skin irritation or rash occurs: Get medical advice/attention. Take off contaminated clothing and wash it before reuse. |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | Flash point: >110 °C |
| Autoignition temperature | 335 °C |
| Lethal dose or concentration | LD50 (oral, rat): > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 8100 mg/kg (rat, oral) |
| NIOSH | Not Listed |
| PEL (Permissible) | 50 mg/m³ |
| REL (Recommended) | 300 mg/kg |
| Related compounds | |
| Related compounds |
Magnesium stearate Magnesium laurate Magnesium myristate Magnesium palmitate Magnesium caprylate Magnesium caprate Magnesium oleate |

