M-Xylene: A Closer Look at Its Story and Impact
Historical Development
M-xylene’s story began in the smoky laboratories of the nineteenth century, back when chemists explored tar distillation with equal parts caution and curiosity. It joined two well-known siblings—o-xylene and p-xylene—as part of the aromatic hydrocarbon family, and all three share roots in coal tar and petroleum refining. A real turning point came with the rise of the petrochemical industry. Production scale jumped, and suddenly, even the most basic household products leaned on the byproducts of xylene. My own memories as a student chemist are filled with the scent of aromatic solvents, reminders that much of what we take for granted today rides on discoveries bound up with substances like m-xylene. Its journey reflects larger industrial shifts, as demand for plastics, adhesives, and resins grew across the globe. The world’s infrastructure owes more than a nod to the evolution of m-xylene processing.
Product Overview
M-xylene stands as a benzene ring holding two methyl groups at the meta positions. That structure gives it versatility many compounds envy. Transparent, colorless, and with a distinct, almost sweet odor, it comes to life not just in factories but in everyday materials most people never pause to consider—foams, films, and coatings. Each batch carries the weight of decades of optimization, and behind every shipment, there’s a network of refining columns, tanks, and careful hands. Xylene as a family often forms a mixture, so m-xylene trades places with its isomers until further purification splits them all apart. This separation process, driven by modern technology, keeps the wheels of industries turning wherever polymers and advanced materials rise to the fore.
Physical & Chemical Properties
M-xylene boils just above 139°C, melts at -47°C, and feels slick, almost oily between the fingers—a courtesy extended only in well-ventilated labs. Its vapor rises readily at room temperature, hinting at why ventilation and personal protective equipment get such attention on the shop floor. Immiscible with water yet dissolving with ease in many organic solvents, its chemical stubbornness and compatibility set it apart. The four extra hydrogen atoms on the ring, compared with benzene, give it just enough reactivity to play well with strong oxidants or halogens. For chemists, these properties aren’t trivia—they guide every choice from reaction setup to waste capture.
Technical Specifications & Labeling
Handling a drum of m-xylene brings more to mind than hazard labels. The numbers matter—CAS registry 108-38-3 marks its family line, but real-world storage keeps technicians measuring purity and scanning for traces of water, sulfur, or other isomeric contaminants. The quality expected for high-purity uses cannot get fudged, because downstream reactions punish even small mistakes. Flammable liquid warnings and codes from global regulators stay posted near work areas. Every bottle in a university stockroom or industrial plant tracks its journey, because a missed step can ripple through quality control, worker safety, and even consumer trust in finished products.
Preparation Method
Still today, most m-xylene emerges as a byproduct of catalytic reforming and toluene disproportionation. Every time a barrel of crude meets the heat and catalysts inside a refinery, there’s a chance some of it ends up in a tank marked xylene. Fractional distillation and crystallization split the mixture down to pure m-xylene, which most often lags behind its better-known sibling p-xylene in production volume. These choices aren’t just economic—they shape the costs and environmental impacts of hundreds of downstream industries. My time in a plant taught me that efficiency in this step makes the difference between profit and waste, and smarter separation tech remains a constant target for improvement.
Chemical Reactions & Modifications
Beyond its simple structure, m-xylene acts as a springboard for new chemicals. Oxidation yields isophthalic acid, a building block for resins and polyester fibers. Bromination, sulfonation, and nitration all unlock new intermediates for dyes, pharmaceuticals, and advanced polymers. The methyl groups fine-tune the reactivity of the aromatic ring, making some substitutions much more practical than with simple benzene. These transformations draw from old-school chemistry and high-throughput automation alike. I recall plenty of failed attempts to coax clean reactions from xylene mixtures back in my research days—proving that even “basic” compounds require sharp technique and deep understanding.
Synonyms & Product Names
M-xylene goes by multiple labels: 1,3-dimethylbenzene, 1,3-xylene, and simply meta-xylene. These names swirl around in scientific literature and on product sheets, sometimes causing confusion among the uninitiated. In production plants and on supply contracts, clarity matters more than clever naming, so the community leans on standard codes and verified grades. In the end, no matter the alias, it’s the properties and purity that pull the most weight in any transaction or application.
Safety & Operational Standards
Working with m-xylene means never getting complacent about safety. Flammable vapors threaten explosions in poorly ventilated spaces, and repeated skin contact brings its own set of health risks. Inhalation at moderate or high concentrations leads to headaches, dizziness, and chronic exposure stirs up both liver and nervous system worries. Emergency showers, fume hoods, and solvent-resistant gloves feel like old friends in any decent lab or factory. Regulations tighten each year to lower allowed exposure limits and sharpen the focus on managing spills and emissions. Forgetting these lessons—especially after a long shift—can leave scars, both physical and environmental. Stories of near misses circulate among workers, serving as real reminders that tight procedures and durable equipment aren’t just “nice to have.”
Application Area
Industrial users pull value from m-xylene in some surprising places. Its main role as a feedstock for isophthalic acid shapes packaging, food containers, and the lightweight bottles carried home after every grocery run. Coatings and adhesives made from m-xylene derivatives keep cars, electronics, and furniture both protected and attractive. Solvents rooted in its chemistry clean paintbrushes, support ink formulation, and flow through electronic parts manufacturing lines. Each step brings high standards for purity and consistency, because customers—consciously or not—demand reliability from the materials that touch their lives. The proliferation of new polymer applications ties tightly to supplies of these hidden aromatic compounds.
Research & Development
Researchers have spent years trying to squeeze more value from m-xylene, push yields higher, and cut waste from industrial processes. Advances in catalytic systems, especially zeolite-based processes, let companies sharpen the separation of isomers and peek into new reaction pathways. Green chemistry challenges push the envelope, seeking out water-saving reaction conditions, renewable feedstocks, and lower emission profiles. One key driver comes from the consumer—rising demand for sustainable plastics and cleaner transport fuels put extra pressure on the industry to innovate. As a chemist with a nose for new methods, I’ve seen the payoff from partnerships between universities and plants investing in real improvements, not just incremental tweaks.
Toxicity Research
M-xylene’s dangers aren’t just theoretical—they show up in both animal testing and stories of workplace accidents. Short-term exposure brings quick symptoms: drowsiness, disorientation, and sometimes loss of coordination. Chronic effects unfold more quietly, affecting the nervous system and raising questions about long-term human health impacts. Epidemiological data lags behind real world exposure, especially in countries with lighter regulation. Researchers still debate safe concentrations, especially since m-xylene often appears in complex chemical mixtures. Progress in analytical toxicology, including sensitive biomarkers and long-term monitoring studies, keeps nudging our understanding forward. Risk management in the real world depends on this foundation, since the best safety plan rests on hard facts, not guesswork or outdated tables.
Future Prospects
Global focus on greener chemistry and tighter regulations will almost certainly pivot the future of m-xylene. Companies already search for alternative raw materials and ways to shrink their environmental footprint. Catalytic advances and closed-loop recycling for solvents could transform the whole supply chain, and any breakthroughs will have broad ripple effects across industries. The pressure to develop bio-based isophthalic acid or more benign solvent systems rises with every new policy draft out of regulatory agencies. From my experience, true shifts often start slow but accelerate as consumer awareness and investment line up. The future of m-xylene sits at the intersection of legacy processes and tomorrow’s sustainability goals, with researchers, operators, and regulators all tugging in the same direction, even if their motivations differ.
What is M-Xylene used for?
What Makes M-Xylene Useful?
M-Xylene doesn’t show up much in daily conversations, but life would look different without it. This clear, sweet-smelling liquid plays a supporting role in dozens of consumer goods and materials. I remember touring a coatings facility and realizing all those glossy finishes trace back in some way to chemistry labs making use of chemicals like M-Xylene.
Industries don’t use M-Xylene alone; they often pull it out of a mixture with its close relatives, O-xylene and P-xylene. Chemists have singled out M-Xylene for certain tasks because it boils at the right temperature and mixes well with other chemicals. Every time I paint a room, I remember the solvents and thinners, many of which rely on compounds like this. M-Xylene certainly runs behind the scenes, but its absence would leave products looking dull and performance falling flat.
Biggest Markets: Paints, Coatings, and Adhesives
The paint cans stacked at hardware stores didn’t get there by chance. Manufacturers want paints that flow easily and dry smooth, and solvents like M-Xylene make that happen. Walk into any new building or recently painted office and you might catch a mild, sharp scent—that’s a clue M-Xylene or its cousins were in the mix.
The same goes for adhesives and sealants. Think about the caulk lining a new bathtub or the glue holding furniture together. Without reliable solvents, those materials would go on too thick, gum up, or struggle to set. M-Xylene’s chemical profile helps break down resins and plastics, making it easier for makers to work with sticky, thick mixtures that become the stuff of everyday repairs and installations.
Making Raw Materials for Plastics
Polymers touch almost everything—cars, gadgets, wrapping on groceries. M-Xylene’s role here is less visible but vital. Chemical engineers rely on it to synthesize isophthalic acid, a building block for PET plastics, specialty polyesters, and certain fibers. This step matters because it influences the toughness and clarity of bottles, food containers, and even some sportswear fabrics.
On the ground, anyone working in plastics knows about bottlenecks in sourcing these intermediates. Producers keep an eye on M-Xylene availability and price, as shortages ripple through to higher grocery bills or production delays for water bottles and food wraps.
Solvent Power in Specialty Industries
M-Xylene stands out for cleaning and degreasing in industrial shops. Watch mechanics wiping engine parts or metalworkers prepping surfaces—industrial solvents do the heavy lifting. M-Xylene’s ability to cut through grease and residue makes shop work faster and surfaces cleaner for further processing or finishing.
Laboratories count on it, too. Analytical labs use it as a reference material and for dissolving stubborn samples. Its unique properties let researchers push deeper into chemical reactions and material testing. Years ago, I watched chemists troubleshoot an analysis by switching to M-Xylene as a solvent, and suddenly their results cleared up. This kind of low-profile, hands-on support holds research together behind the scenes.
Health and Safety Concerns—and Smarter Choices
No story about M-Xylene feels complete without mentioning safety. Prolonged exposure can affect the nervous system, irritate the nose and eyes, and impact the liver. Anyone who’s spent time in paint shops knows to wear gloves and use plenty of ventilation. Most workplaces now teach handling methods that reduce risk, and some are testing greener alternatives wherever possible.
While no magic bullet has replaced M-Xylene in every use, research keeps moving. Companies look at plant-based solvents, advanced recycling, and better air filtration. It’s true that change often means a shift in price or performance, but public demand for safer, cleaner air keeps driving innovation.
M-Xylene rarely grabs headlines, but most products on store shelves depend on it in one way or another. Knowing its backstory helps shoppers and workers alike weigh the cost—not just in dollars, but in health and environment too.
What are the hazards and safety precautions for handling M-Xylene?
M-Xylene: Beyond Just a Chemical
I’ve seen colleagues hesitate before popping open a drum labeled M-Xylene. From a distance, it’s just another solvent, useful in paints, varnishes, and glues. Up close, it’s clear: the risks aren’t printed big enough. M-Xylene affects both those handling it and anyone standing nearby. Exposure often means more than a headache; at higher levels, it bruises the nervous system and leaves workers dizzy, nauseous, or short of breath. Eyes and skin take the brunt next, burning or itching after contact. Absorption doesn’t always need direct skin splash — vapors sneak through cracks in protection, settling into the body without fanfare.
Breathing, Spills, and Day-to-Day Risk
Breathing M-Xylene is a gamble. In small spaces, vapor hangs in the air. Unfiltered masks or open ventilation leave workers with watery eyes and coughs. Years ago, a friend in the coatings industry swapped stories about how fumes spread through poorly sealed rooms and how folks down the hall sometimes tasted bitterness on their tongues. Sometimes, symptoms sneak up—loss of coordination, fatigue, and memory problems surface hours afterward. Blood tests from American and European factories highlight a real pattern: regular, unprotected exposure leads to measurable solvent buildup and organ stress. Skin contact follows close behind, stripping away oils, cracking knuckles, and letting the chemical seep deeper. Clean-up after a small spill grows risky if gloves are thin, or sleeves roll up too far. Steam cleaning methods — hoping the vapor simply disappears — just concentrate danger in the air. That’s a lesson learned the hard way in older shops, which left windows closed tight to save on heating.
Preventing Harm: Getting Back to Basics
Industry guidelines start with simple truths. No open containers. Airflow matters. Using gloves rated for solvents makes a real difference. Nitrile and butyl rubber gloves outlast cotton or ordinary rubber, and goggles catch splash before eyes pay the price. Long sleeves aren’t just for show — any piece of skin missed by gear can burn and flake within hours. Respirators feel cumbersome but save lungs in the long haul, especially fitted models with organic vapor cartridges. I keep a mask by the door; skipping it once left me with a rashy nose and burning throat all morning. In factory settings, exhaust hoods replace fans, and staff train for spills the way people once drilled for fire alarms — every second counts, and confusion multiplies risk.
Routes to Safer Handling and a Healthier Workplace
Some companies have upgraded equipment to closed-system transfer lines, keeping the substance out of open air. I saw a paint shop shave exposure rates in half after switching from buckets to piped delivery. Eye wash stations and chemical showers, once considered expensive extras, quietly cut recovery times after accidents. Training plays the supporting role. Instead of once-a-year safety lectures, seasoned workers mentor new hires, sharing shortcuts that protect both health and the daily workflow. Relying only on warning labels or hoping folks “know better” leaves too much to chance. Now, employers document and label every storage drum, and waste gets disposed of far from work areas — sometimes outside entirely, in containers with ventilated tops. The best efforts come from people who remember what it felt like before — sick days, sore eyes, stained shirts — and who want better for the next round of workers who punch in.
What is the chemical formula and structure of M-Xylene?
Getting to Know M-Xylene
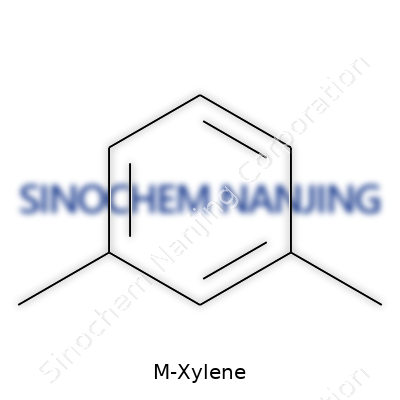
A trip to any paint shop brings the sharp smell of industrial solvents, and m-xylene is one of those familiar compounds lurking behind the scenes. Anybody who’s studied even a little bit of organic chemistry recognizes the formula: C8H10. This molecule plays a larger role in daily life than many realize. Sitting in gasoline, showing up in leather cleaners, sneaking into varnishes, m-xylene's foundation is rather simple. Its chemical structure shows two methyl groups attached to a benzene ring, spaced apart in what chemists call the "meta" position. In plainer English, the three xylene isomers—ortho, meta, and para—differ based on where the methyl groups land. For m-xylene, the groups anchor to positions one and three on the ring, skipping a carbon in between.
Structure in Everyday Chemistry
Drawing out m-xylene on paper shows that classic hexagonal benzene ring, with those two methyl branches attached at a comfortable distance. It’s pretty remarkable that just swapping a methyl from one spot to another in the ring changes so much in behavior and use. For folks who worked in industrial settings, handling these aromatic hydrocarbons just becomes part of daily routine. But even people outside the chemical industry meet m-xylene indirectly. Refineries pump out millions of tons of xylene mixes every year, all riding on how this sturdy carbon ring carries methyl attachments.
Real-World Health and Environmental Impact
One thing stands clear: the safety conversation around m-xylene isn’t just academic. Exposure can cause headaches, dizziness, and irritation. Employees in printing shops or rubber manufacturing sometimes learn this the hard way, especially if their workplaces ignore ventilation. The U.S. Environmental Protection Agency classifies m-xylene as a hazardous air pollutant. Neighborhoods near large plants notice the effects, too. Low-level xylene vapors may leak into the air, prompting community concerns. Groups advocating for environmental well-being push for stricter monitoring of xylene levels in water and air.
From Theory to Solutions: Handling M-Xylene Responsibly
Working with chemicals always comes back to responsibility. Installing effective fume hoods in labs and industrial spaces prevents inhalation. Regular air quality checks keep concentrations below the Occupational Safety and Health Administration’s permissible exposure limits. Companies can train staff, highlighting real symptoms rather than abstract dangers. From experience, simple steps make a world of difference—wearing gloves, keeping containers sealed, and recognizing the strong, sweet odor as a warning sign, not just business as usual.
Some operations switch to less toxic solvents when possible. Water-based cleaners or low-odor alternatives help cut down on exposure risks for both workers and communities. Recycling solvent vapors and using activated carbon filters in exhaust systems also keep more m-xylene out of the environment.
The Chemistry in Context
It’s easy to write off a chemical formula as meaningless outside a textbook, but each compound carries real consequences. In the case of m-xylene, what looks like a simple arrangement of carbons and hydrogens drives larger discussions around industrial safety, environmental health, and everyday awareness. Recognizing that connection, backed by decades of research and firsthand stories, brings meaning to the molecular structure found on a safety data sheet.
What are the storage and handling recommendations for M-Xylene?
Understanding M-Xylene's Hazards
M-Xylene, a common solvent and chemical used in manufacturing paints, coatings, and plastics, brings a set of hazards people can’t afford to overlook. The liquid itself gives off a strong odor and evaporates fairly quickly at room temperature, so even a cracked lid means fumes start to drift through the building. Anyone who’s stored or moved M-Xylene in a job knows it isn’t just about ticking off boxes on a safety list. Proper handling can keep co-workers healthy, equipment safe, and businesses out of regulatory trouble.
Why Proper Storage Shapes Everyday Safety
The drum or container doesn’t do all the heavy lifting. M-Xylene stays stable under normal conditions, but it’s a fire risk if people store it near heat or open flames. Temperatures above 30°C (86°F) raise the pressure in closed containers and push the risk of vapor leaks or even rupture. There’s a reason every good chemical storeroom I’ve seen puts flammable liquids like M-Xylene in steel cabinets, well away from sunlight, hot machinery, or electrical panels. That distance between chemicals and ignition sources is more important than any extra labeling or alarm system.
From my years around plants and warehouses, I’ve seen more incidents caused by carelessness than by equipment failure. People sometimes stack drums too close to walkways or pile them four-high to save floor space. A single slip or a poorly clamped lid means M-Xylene makes its way into the air, and if the storage area lacks proper ventilation, the fumes build up fast. OSHA and similar safety agencies keep pressing for natural or forced ventilation wherever M-Xylene is handled because everyone in the room feels the effects before long: headaches, dizziness, and in higher doses, more severe health effects. Taking the time to keep aisles clear, stacking containers no more than two high, and checking vents does a lot more for human health than just going by the book.
Labeling and Container Choice Count
Labeling isn’t just formality. Emergencies happen, and nobody wants first responders guessing what’s inside a spilled drum or leaking can. Each container needs a sturdy, legible label showing the chemical name and hazard information. Stainless steel or steel drums with full sealing gaskets work best, not repurposed plastic bins with duct tape on the lid. Leaks and chemical reactions with the wrong material do serious damage, both to property and anyone nearby.
Handling and Transfers: The People Factor
Personal protective equipment (PPE) isn’t optional here. You get a splash or vapor cloud, and it stings the eyes, burns the skin, and can irritate the lungs. Workers need to use gloves resistant to organic solvents, chemical goggles, and – in confined or poorly ventilated areas – a respirator suited for organic vapors. Beyond that, hand pumps or drum taps keep spills down during transfers. Pouring straight from a drum or using a jury-rigged siphon only invites disaster. I’ve watched workers skip this step to save a few minutes, only to end up with soaked coveralls and a chemical burn for their trouble.
Grounding and bonding containers before transferring M-Xylene eliminates static electricity that can set off fires. Many folks ignore this step, but that snap of static can ignite vapors even without a visible spark.
Emergency Prep and Real-World Solutions
Accidents still happen, no matter how many layers of protection. Spill kits, designed for solvents like M-Xylene, need to stay within arm’s reach, not locked in a closet across the warehouse. Safety showers and eyewash stations should remain in clear, direct paths, never blocked by stock. Training workers to act immediately, rather than wait for a supervisor, turns a spill from a crisis into just another cleanup drill. Frequent drills and honest conversations about risks matter just as much as written policies.
At the end of the shift, the smartest move always involves respect – for the chemical, the process, and your team. Keeping M-Xylene locked down and dealt with carefully isn’t just regulatory compliance; it’s the backbone of a safer workplace and healthier lives. That’s something nobody wants to take risks with.
Is M-Xylene harmful to human health or the environment?
M-Xylene in Everyday Products
M-xylene shows up in paints, varnishes, adhesives, and certain fuel blends. Most people don’t realize how common it is until a strong, almost sweet smell hits in a freshly painted room or a gas station. Industries count on m-xylene for a lot of modern conveniences, but those conveniences bring a cost that doesn’t get enough attention.
The Science Behind the Risks
Breathing m-xylene vapors can hit pretty fast. At low levels, it irritates the eyes, nose, and throat—nothing life-threatening for a healthy adult in a well-ventilated spot, but enough to notice. Go higher, or stay exposed longer, and headaches, dizziness, and poor coordination start to creep in. Someone working with m-xylene every day—painters, mechanics, or factory staff—carries a heavier burden, especially without protective gear. Skin contact causes dryness and cracking, making daily work uncomfortable and raising the risk for other health issues. Some research points to long-term exposure as damaging the liver and kidneys, and the nervous system can take a hit too. The U.S. EPA and CDC track these effects and push for safety in the workplace and public spaces for good reason.
Environmental Impact Around Us
M-xylene doesn’t stay put. Routine spills, leaks from factories, or improper waste handling let xylene slip into soil and groundwater. From there, it rides into rivers and lakes, disrupting aquatic life. Fish exposed to enough of this chemical show problems growing and even reproducing, throwing local ecosystems off balance. It breaks down in sunlight and air, but not as quick as we’d hope, so build-up happens in places nobody checks until too late. Plants aren’t immune either; too much xylene stunts growth and leaves crops vulnerable to further stress.
Community Action and Solutions
Every step toward managing m-xylene makes a difference. At work, simple habits such as using gloves, goggles, and respirators, and making sure ventilation works keep risky exposure down. At the city or state level, enforcing stricter regulations on disposal and emissions makes companies look for safer alternatives or better recycling practices. Modern refineries use less toxic solvents where possible, and some switch to water-based options to cut down on xylene in products. Still, real change hinges on ordinary people pushing for safer neighborhoods—voting for greener policies, asking companies what’s in their products, and learning what’s under the kitchen sink.
Why the Story Matters
I grew up in a house next to a paint store. Every summer, trucks unloaded drums marked with words I couldn’t pronounce; the air outside clung to my nose. My father always talked about “that chemical smell” and covered the vents with towels. Years later, scientists linked chemicals like m-xylene to long-term environmental and health risks. Science gives clear facts, but real understanding grows from seeing neighborhood kids coughing more, teachers warning about “bad air” days, and learning how a spill upstream can taint drinking water miles away. Calling out harmful chemicals isn’t about fear—it's about common sense and the right to a safe place to live. Every layer of prevention brings fewer health scares and a healthier planet for the next group of kids playing outside my old bedroom window.
| Names | |
| Preferred IUPAC name | 1,3-dimethylbenzene |
| Other names |
1,3-Dimethylbenzene m-Xylol m-Dimethylbenzene 3-Xylene |
| Pronunciation | /ˈɛm ˈzaɪliːn/ |
| Identifiers | |
| CAS Number | 108-38-3 |
| Beilstein Reference | 605605 |
| ChEBI | CHEBI:17233 |
| ChEMBL | CHEMBL14238 |
| ChemSpider | 6497 |
| DrugBank | DB03788 |
| ECHA InfoCard | InfoCard 100.003.891 |
| EC Number | 203-576-3 |
| Gmelin Reference | Gmelin Reference: 778 |
| KEGG | C01407 |
| MeSH | D017916 |
| PubChem CID | 7890 |
| RTECS number | ZE2275000 |
| UNII | FNYL5F0GUU |
| UN number | UN1302 |
| Properties | |
| Chemical formula | C8H10 |
| Molar mass | 106.17 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic odor |
| Density | 0.86 g/cm³ |
| Solubility in water | Immiscible |
| log P | 3.2 |
| Vapor pressure | 6.72 mmHg (25°C) |
| Acidity (pKa) | 34.7 |
| Basicity (pKb) | 12.38 |
| Magnetic susceptibility (χ) | −7.8×10⁻⁶ |
| Refractive index (nD) | 1.497 |
| Viscosity | 0.809 mPa·s (25°C) |
| Dipole moment | 2.36 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 173.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | +8.36 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3893.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB52 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H332 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 25 °C (77 °F) |
| Autoignition temperature | 527 °C (981 °F) |
| Explosive limits | Explosive limits: 1.1–7.0% |
| Lethal dose or concentration | LD50 oral rat 4300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 4,300 mg/kg (oral, rat) |
| NIOSH | NIOSH: ZE2450000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 900 ppm |
| Related compounds | |
| Related compounds |
o-Xylene p-Xylene Ethylbenzene Toluene Benzene |

