M-Toluidine: Looking Beyond the Basics
Historical Perspective
Exploring m-toluidine takes me back to some of the most storied years in organic chemistry. German chemists in the nineteenth century pursued aniline derivatives, searching for compounds that could modify the growing synthetic dye industry. M-toluidine, also known as 3-methylaniline, came about from a combination of scientific curiosity and industry demand. Early researchers recognized the structural relationship between toluidines and aniline compounds, extracting and separating the isomers using methods that seem crude by modern standards. This sort of empirical chemistry often led to breakthroughs almost by trial and error, but over time, more precise distillation and crystallization practices enabled reliable differentiation between the ortho, meta, and para forms. M-toluidine grew popular thanks to its unique chemical reactivity, propelling innovations in dye, pesticide, and pharmaceutical synthesis. In many ways, the development of m-toluidine shows just how interconnected basic chemical research and industrial need have always been.
Product Overview, Physical & Chemical Properties
M-toluidine belongs to a group of organic compounds recognized for a faintly pungent, often fish-like odor, owed to its amine group. Chemically, it carries a methyl group attached to the meta position on the benzene ring, offering a molecular formula of C7H9N. Physical form presents as a colorless to pale yellow liquid, turning brown upon exposure to air due to oxidation—not exactly suited for delicate storage. Boiling at around 200-203°C, its density floats just under that of water. These details might sound technical, but what strikes me most is the way m-toluidine embodies a set of characteristics tailor-made for specific organic syntheses: decent solubility in organic solvents, low solubility in water, and a stable aromatic core flanked by a nucleophilic amine. Its physical and chemical fingerprint explains why both small-scale labs and large chemical plants have kept supplies on hand for generations.
Technical Specifications & Labeling
Industry regulations now enforce tight control over labeling and purity, a far cry from the loosely defined standards once in place. Modern bottles spell out the CAS number (108-44-1) and purity percentages, hostile to contamination that could disrupt subsequent reactions. Safety warnings typically highlight the toxicological risks and necessary precautions. Colorimetric analysis, gas chromatography, and mass spectrometry ensure product consistency before it leaves the factory. These safeguards didn’t appear overnight; they reflect decades of lessons learned from accidents and exposure studies. Compliance with established safety scores underpins confidence that m-toluidine purchased from reputable suppliers will perform according to expectation in research or manufacturing environments.
Preparation Method
Back in university, I followed the electrophilic aromatic substitution routes for synthesizing toluidines in the lab, starting from nitrotoluene and reducing it by catalytic hydrogenation or iron-acid methods. Industrial settings usually upgrade reaction controls, deploying pressurized hydrogen with nickel catalysts to transform m-nitrotoluene with efficiency and scalability in mind. Chemical engineering improvements tackle issues like product separation, catalyst reuse, and environmental discharge. Decades ago, waste streams riddled with residual aromatics could end up in local rivers. Today’s plants work harder to minimize emissions, redirecting spent catalysts into recycling cycles or regulated disposal. People often overlook the invisible progress wrapped up in process chemistry—a simple reduction now walks in step with sustainability commitments.
Chemical Reactions & Modifications
Beyond my own bench-scale experiments, m-toluidine delivers a real lesson in versatility. The ring’s methyl group resists many conditions, but the free amine group opens pathways to acetylation, diazotization, or alkylation. Chemists usually seize on this reactivity to produce azo dyes—linking m-toluidine via diazonium salts to form bright colors for synthetic textile applications. Reacting m-toluidine with various acids generates salts, which affect its solubility or reactivity further. Researchers sometimes modify the methyl group or target selective chlorination or sulfonation on the aromatic ring, diversifying the range of possible compounds. Even within pharmaceuticals, small changes at the amine function or ring positions can yield new drugs or intermediates. What strikes me here is how each molecular tweak opens doors for both new scientific investigation and commercial use.
Synonyms & Product Names
Chemists gave m-toluidine several names across texts and decades. The systematic International Union of Pure and Applied Chemistry (IUPAC) name sticks to 3-methylaniline, offering clarity when cross-checking literature. In commercial catalogs, one may spot 3-amino-toluene, meta-toluidine, or even more old-fashioned terms in European or Asian markets. Some firms abbreviate product listings, using “m-tol” for faster labeling. These variations matter because they can trip up ordering or regulatory paperwork—an error easily fixed by checking the structure or CAS number before shipment. In practice, keeping track of synonyms helps cut down on confusion that otherwise delays research or production schedules.
Safety & Operational Standards
Anyone who has handled m-toluidine knows the chancy nature of aromatic amines. Exposure through skin, inhalation, or accidental ingestion brings risks—headaches, skin irritation, or much worse with repeated contact. Modern labs post hazard labels, stock spill kits, and maintain fume hoods to corral vapors. Regulations in many countries restrict how much may be transported and stored on-site, demanding secondary containment and strict inventory checks. Emergency training drills reinforce what textbooks say: immediate washing in case of spills and swift evacuation during large leaks. Once, chemical plants accepted a greater degree of occupational risk, but the field’s evolved to prioritize both community and worker health. This shift didn’t happen by accident—disease clusters and regulatory pressure motivated real change. Now, even the smallest research group takes no shortcuts, and the entire industry exists in the shadow of safety audits and compliance reviews.
Application Area
Most discussions about m-toluidine focus on dye manufacture, and I’ve watched a host of projects turn to it for both classical and more advanced synthesis. Azo dyes stemming from m-toluidine figure into textile, leather, and even certain ink formulations. The pesticide sector taps its structure to create fungicides or weed control agents; pharmaceutical researchers fashion intermediates that form part of painkillers or other medicines. M-toluidine’s sweet spot lies in its clean aromaticity blended with reactive amine chemistry—qualities that suitors from pigment formulators to fine chemical manufacturers value. Even as new synthetic methods and greener starting materials emerge, m-toluidine’s ease of modification and proven history keep it relevant, especially among industries unwilling to switch away from time-tested base compounds.
Research & Development
In research labs, m-toluidine appears in projects trying out new catalysts or building blocks for organic syntheses that push chemical complexity. Some groups experiment with ring substitutions, aiming for novel photochemical or electronic properties. Environmental assessments look for safer ways to make or degrade m-toluidine, sometimes engineering bacteria capable of bioremediation. A decent number of academic studies focus on the compound’s reactivity under milder conditions or compare it against greener, less hazardous alternatives. Funding agencies show growing interest in finding substitutes or novel applications, but most paint m-toluidine as an essential reference point—a reliable yardstick for measuring progress.
Toxicity Research
Over the years, regulatory bodies and independent scientists have studied the toxicological effects of m-toluidine. Acute exposure can damage blood cells, irritate mucous membranes, and, if mishandled, spark allergic or neurological responses. Chronic exposures have prompted researchers to investigate links with carcinogenicity, though definitive conclusions often collide with confounding factors or inadequate data. Laboratory animal studies show that regular contact can produce methemoglobinemia, a condition where oxygen transport in blood suffers. These findings reinforce why strict industrial hygiene plays a crucial role. The focus now rests on limiting exposure, running regular health checks, and finding alternatives wherever possible. Today’s risk management strategy owes as much to years of research and grim case reports as it does to regulatory muscle.
Future Prospects
Looking ahead, m-toluidine stands at a crossroads. Industrial legacy assures demand in dyes and synthetics for years to come, though green chemistry movements look to shrink aromatic amine use wherever alternatives match performance. Innovations in biocatalysis dangle hope for safer, more efficient production without hazardous intermediates or the same obstructionist byproducts. Pharmaceutical and agricultural chemists scan for next-generation molecules that combine the utility of toluidines with a lower toxicological burden. Environmentalists work on remediation strategies, tackling contamination in soils and waterways from historic and ongoing industrial release. The next wave likely steers toward processes less reliant on aromatic amines overall, yet practical constraints mean m-toluidine’s role won’t vanish overnight. Whether the compound features as a launching pad for molecular design, a cautionary tale for regulatory reform, or the focus of sustainable process innovations, it remains a touchstone in chemistry’s ongoing story of balancing utility and responsibility.
What is M-Toluidine used for?
Looking at M-Toluidine
M-Toluidine has a chemical backbone that rings true for many in the manufacturing and science fields. Many folks, even those outside the lab, may not know what it is, but it plays a quiet role in a wide range of products. The name shows up most often in connection with dyes, but that’s just the beginning.
The Dye Industry’s Go-To Ingredient
M-Toluidine crops up most often in the world of color—making the bright and consistent dyes for textiles. If you’ve ever slipped on a brightly colored t-shirt that held up to laundering, there’s a fair chance M-Toluidine had a hand in it. The chemical helps make “azo dyes,” known for their long-lasting color and strong saturation. Textile producers favor these dyes for their vivid results, to say nothing of the stability they deliver through dozens of wash cycles. As someone who’s watched clothes fade in the sun or after a handful of spins in the washer, it’s easy to see the need for reliable dyes. These are the difference between a shirt bought for one summer and one you wear for several years.
Beyond Color: Pharmaceuticals and Chemicals
Those who’ve spent time in a pharmacy or looked closely at the boxes in their medicine cabinet might never suspect that some medicine molecules begin life as chemicals like M-Toluidine. The pharmaceutical industry counts on it as a starting material for complex medications and ingredients. It shows up in the synthesis of analgesics, local anesthetics, and other medicines. Without reliable sources of precursor chemicals like this, the cost of finished medicines could spike, and research into new treatments might slow down. As someone who has seen the impact of medicine shortages in the community, the benefits of a steady chemical supply chain become clear.
Role in Herbicides and Pesticides
In the fields where crops grow or pests challenge farmers, M-Toluidine plays a subtle but important part. It gives chemical manufacturers a building block for making some herbicides and pesticides. This doesn’t just matter for huge agricultural conglomerates. Small family-run farms also depend on these tools to control weeds and bugs, especially during tough growing seasons. Secure access to critical chemicals translates into better food security and more predictable harvests, and that can keep grocery store prices steadier, even as the weather or insects threaten crops year after year.
Risks and Responsible Handling
Of course, every tool carries risk. M-Toluidine is toxic if handled improperly. Workers exposed to it can face health risks, so proper equipment and good ventilation become non-negotiable in workplaces that use it. Regulation by agencies like OSHA and systems for safe disposal have made a difference. I remember reading case studies about chemical exposure in factories before proper safeguards became widespread; stories like those highlight why training and equipment are so fundamental now. Mistakes cost more than money—they can take lives or harm entire communities downstream.
Looking Ahead: Sustainable Substitutes
Industries benefit from looking for greener alternatives. Chemical firms are investing in research to find dyes and medication precursors that do less harm, both to people handling them and ecosystems. Support for these products grows with public awareness. Consumers and companies both shape safer, cleaner practices by voting with their dollars or decisions. As someone who’s tried to choose low-impact products even when it costs a little more, I see the push for more sustainable chemistry as not just an ideal, but an achievable goal in the coming years.
Facts and Developing News
Research published in journals like the Journal of Hazardous Materials points to the need for better methods to minimize byproducts and worker exposure. The European Chemicals Agency and U.S. EPA regularly review the health and environmental impacts, driving advances in workplace safety. This is a piece of the chemical story that shows up in everyday life, from a favorite t-shirt to a prescription bottle.
What are the safety precautions when handling M-Toluidine?
Why Taking Care with M-Toluidine Matters
M-Toluidine has a reputation for being a reliable tool in chemical labs, factories, and production lines. Its usefulness comes with real risks, though. M-Toluidine can irritate skin, eyes, and airways. Breathe in too much of it, and you might experience headaches or dizziness. Splashes on your skin can cause burning or even longer-term effects if not washed off right away. This chemical can also affect organs like the liver if exposure gets out of hand over time. OSHA groups M-Toluidine with hazardous chemicals, so treating it lightly puts health and safety in jeopardy.
Personal Protection: Gloves, Goggles, and More
Direct contact sets off most accidents. Whenever handling M-Toluidine, proper gloves are the first line of defense. Nitrile or neoprene gloves do a solid job blocking it from your skin. Latex doesn’t hold up as well if the chemical lingers, so it’s smarter to stick with gloves that hold up to organic materials. Splash-proof goggles keep eyes safe. For bigger tasks, add a lab coat or apron made from chemical-resistant material. Never forget to double-check that your gear fits and isn’t worn or punctured.
Ventilation and Air Quality
Strong fumes build up quickly, so keeping the area well-ventilated should always be a priority. Fume hoods and local exhaust systems make a big difference, especially in closed spaces. They pull vapors away from your breathing zone and lower the odds of inhaling something you’ll regret. If you’re working in a place without good ventilation, using a respirator designed for organic vapors adds another level of safety. A trusty air monitor can help keep track of vapor levels and warn you if things start getting risky. Poor ventilation causes problems that are easy to sidestep if you prepare in advance.
Preparing for the Worst: Spills and First Aid
Even seasoned workers make mistakes. Spills need fast action. Spreading an absorbent material (such as activated carbon or commercial spill pads) over the area stops the liquid from moving beyond the affected spot. Always avoid touching the spill with bare hands and wear protection during cleanup. Used pads and rags should go straight into a sealed waste container. Fresh air helps if someone breathes in too much; eyes and skin need plenty of running water right away. Know where to find the eyewash station and safety shower, and practice using them before real emergencies happen. Quick action limits injuries and keeps the workplace running smoothly.
Safe Storage Keeps Trouble in Check
M-Toluidine shouldn’t mix with food, hot surfaces, or strong acids. Use containers with tight lids. Clear labels help everyone working in the area stay aware. Store the chemical in a cool, dry space with little foot traffic and a stable shelf. Lock the cabinet if possible, especially where children or untrained workers might wander in. A chemical inventory and regular checks stop old stock from piling up and lowering the container’s integrity over time.
Training, Habits, and Personal Vigilance
Colleagues depend on each other, so it pays to keep everyone in the loop. Regular training refreshes safety habits and creates a sense of responsibility. If you spot damaged gloves or broken goggles, speak up. Always report suspicious smells, leaks, or unexplained symptoms. Culture goes a long way: people who look out for one another catch mistakes sooner and keep the workplace safer for everyone around. Combining practical safety gear, smart storage, and upfront communication reduces risks and makes working with M-Toluidine much less stressful.
What are the physical and chemical properties of M-Toluidine?
What Is M-Toluidine?
M-Toluidine, also known as 3-methylaniline, often turns up in labs where dyes, chemicals, or pharmaceuticals get made. In simple terms, it’s a colorless to pale yellow liquid. At first sniff, there’s a sharp, almost fish-like smell that lingers—one reason chemists know they’re working with an aromatic amine. This stuff might not look like much, but it has a strong effect on the materials that shape modern life.
Physical Traits with Practical Impact
Pouring M-Toluidine, you’ll notice it’s a bit oily, more so than water. On the temperature scale, it melts just above zero degrees Celsius, solidifying if things get a little too cold. Hit 199 degrees Celsius and it boils, but the vapors are not something to ignore—they sting your nose and might harm your health. The density sits at around 0.99 g/cm³, which means it floats only a bit in water, but don’t let that fool you: it doesn’t dissolve easily in water at all.
On the other hand, mix it with organic solvents like alcohol or ether, and it blends right in. This quality makes it handy in synthetic chemistry labs. In old workshops or dye factories, M-Toluidine usually came out of sealed containers, since contact with air causes it to darken and degrade, thanks to slow but steady oxidation.
Chemical Personality
M-Toluidine’s structure offers both an amino group and a methyl group clinging to a benzene ring. This setup means the molecule isn’t just a bystander—it’s reactive. The amino group pulls in acids, forming salts easily. This action turns M-Toluidine into a building block for colorants, pesticides, and even some medicines.
It’s flammable. If a workplace ignores that, accidents could happen fast. In addition, prolonged exposure to skin often brings rashes and irritation. Breathing in the vapors or letting it absorb through the skin can affect blood, since aromatic amines convert into compounds that damage hemoglobin over time. Fact-based safety procedures go a long way here, like well-ventilated rooms and gloves made of nitrile or neoprene.
Risks and Responsibility
Anyone using M-Toluidine must know its risks. It’s no secret that certain amines link to health problems including cancer, since they can change the way DNA repairs itself. Research has tracked these “bad actors” for years, and regulators enforce strict guidelines, especially in places making dyes or rubber chemicals. It’s not enough to just label a bottle—the right storage, routine air monitoring, and training workers about exposure all offer more protection than leaving things to chance.
Managing M-Toluidine in Industry
Better ventilation and regular safety audits go hand in hand. Spills call for speedy cleanup with absorbent materials resistant to chemical attack. Old habits, like using bare hands or not wearing goggles, contribute to preventable incidents. Safer substitutes slowly enter the market for some applications, but not every process welcomes change easily. Where alternatives don’t yet fit, strict controls and occupational health monitoring play a vital role.
Building a culture where every worker understands the “why” behind safety rules does more than check boxes on a form—it saves lives. Whether making colorants for fabric or isolating chemical intermediates, attention to detail helps keep both workers and the environment safe. People need more than facts; they need the conviction to do things right every day they step near a drum of M-Toluidine.
How should M-Toluidine be stored?
Why Storage Matters for M-Toluidine
M-Toluidine carries a strong reputation among chemists and workers in manufacturing for its role as both a building block and a hazard. Few in labs or plants forget the fishy odor or how easy it is to pick up headaches, even from brief exposure. Safety isn’t a buzzword here—it is essential practice. Over the years, I’ve seen what a neglected storeroom can turn into. Even a small spill can escape notice, only to cause headaches, nausea, and a full-blown chemical incident. This risk alone makes every detail of storage a priority.
Addressing Fire and Health Risks with Proper Containment
M-Toluidine catches fire much quicker than most realize. Its flash point—around 85°C—puts it well below the temperatures found next to a hot motor or sunlight pouring through a window. That’s why it belongs in a well-ventilated, cool, dark spot, away from sources of heat, spark, or static. In real labs I’ve worked in, the chemical sits in dedicated flammable storage cabinets, often painted bright yellow and grounded against static. A common mistake in smaller operations is keeping bottles near solvents or acids. This introduces the risk of dangerous reactions and fires if a container breaks.
Separation Makes a Difference
People sometimes overlook incompatibilities because they focus on the job, not the risk. This chemical reacts with strong oxidizing agents—concentrated nitric or perchloric acid, for instance. These combos can trigger violent chemical reactions. Storing single bottles on separate shelves or sealed bins blocks the chain of disaster. On more than one occasion, I’ve found that storage audits—unannounced, done monthly—make the biggest difference. Teams walking through their setups together catch those old bad habits before they turn into headlines.
Personal Protection: It Starts with How You Store
I’ve heard too many stories about routine handling leading to skin rashes or breathing problems. Sealed, well-labelled containers help, but storage feeds into access. In my own work, only trained staff handled M-Toluidine. Written logs replaced casual access. Having the right gloves, goggles, and lab coats stacked right by the storage site beats scrambling after the fact. Access control isn’t bureaucracy; it draws a line between safe lab work and a visit to occupational health.
Bottles, Ventilation, Cleanliness
Plastic or amber glass bottles with tight seals work best. This chemical isn’t friendly to regular plastic or exposed rubber, so it pays to check containers regularly for leaks, swelling, or fading labels. Cabinets come fitted with proper ventilation, drawing in vapor so it doesn’t build up in the room. I remember an instance where a clogged vent nearly sent fumes out into the general work area. Routine maintenance made all the difference—you smell less, you risk less.
Preparing for the Worst
Spill kits and eye-wash stations sit nearby, not down the hall. Good facilities keep clear copies of the safety data sheet within grabbing distance, right on the cabinet. Training every new staff member or temp worker on those basics pays off. Mistakes come less from the complexity of chemistry and more from forgetting the basics of respect. These simple practices speak louder than any rulebook when something goes wrong—and that’s experience talking.
Is M-Toluidine hazardous or toxic?
Understanding What M-Toluidine Brings to the Table
M-Toluidine shows up in dyes, some resins, rubber products, and laboratory settings. Most folks outside the chemical industry won’t spot it on a label, but for workers in certain factories, it’s a part of daily life. The way m-toluidine smells — kind of fishy, kind of sweet — only hints at its punch. The real problems start long before you can recognize that scent in the first place.
Exposure and Health Effects
Take it from people who have worked in chemical labs: even small exposures to strong organic chemicals like m-toluidine can give you headaches and nausea fast. It gets soaked up through the skin, and inhaling its vapors isn’t any better. Besides immediate irritation to eyes and throat, studies show it can cause cyanosis. Your skin may turn blue from lack of oxygen, since m-toluidine breaks down red blood cells’ ability to carry what your body needs.
The European Chemicals Agency classifies m-toluidine as acutely toxic. The International Agency for Research on Cancer also lists it as possibly carcinogenic to humans. Over time, contact raises the risk of bladder cancer. In workplaces lacking ventilation or personal protective gear, health can spiral quickly.
No Safe Shortcut
Many places try to make it safer through enclosed processes or gloves, but m-toluidine can sneak through latex and even some nitrile gloves. NIOSH recommends butyl rubber as a barrier. People still get exposed by touching work surfaces or contaminated clothes. My own gloves have cracked from chemical wear, making it clear how easily safety routines can break down after hours on the job.
For families living near factories, there’s another worry: accidental releases. M-Toluidine spills have contaminated soil and water in a couple of areas, prompting neighborhood evacuations. The chemical does not break down quickly in natural environments, so what leaks out tends to hang around. It seeps into rivers and well water, where it can stick to sediments for years.
Solutions Worth Chasing
It helps to pay closer attention to process engineering and safety culture. Automation has trimmed down workers’ direct exposure, but old plants don’t always keep up with improvements. Many smaller businesses cut corners because maintenance costs time and money. This creates hidden risks for employees and neighbors alike. As in any workplace, strong training and actual enforcement of safety protocols as the daily routine, not just lip service, truly matter.
Waste treatment needs upgrades as well. Activated carbon filtration captures m-toluidine from both air and water before it gets out the door. Chemical substitution research hasn’t found a dead-simple alternative for every use case, but some industries now look for dyes or curing agents made from safer raw materials.
In the end, talking about m-toluidine isn’t about panicking. It’s about recognizing the problem, facing it head-on, and pressing for better protections for workers and the neighborhoods close by. People’s health shouldn’t be a gamble for the sake of cheap production. Anyone who works around m-toluidine deserves honesty, decent equipment, and respect for the risks staring back from a yellow-green lab bottle.
| Names | |
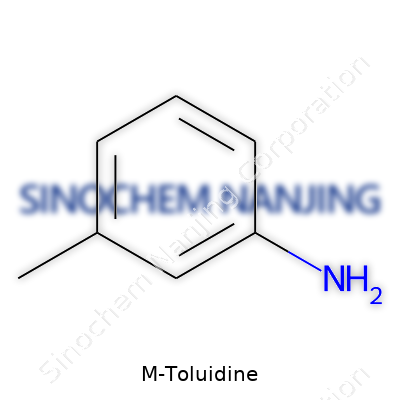
| Preferred IUPAC name | 3-Methylaniline |
| Other names |
3-Methylaniline 3-Aminotoluene m-Toluidine meta-Toluidine 3-Methylbenzenamine |
| Pronunciation | /ɛm təˈluːəˌdiːn/ |
| Identifiers | |
| CAS Number | 108-44-1 |
| Beilstein Reference | 1209242 |
| ChEBI | CHEBI:28491 |
| ChEMBL | CHEMBL14238 |
| ChemSpider | 546 |
| DrugBank | DB11363 |
| ECHA InfoCard | 100.005.392 |
| EC Number | 202-429-0 |
| Gmelin Reference | 827 |
| KEGG | C01407 |
| MeSH | D014413 |
| PubChem CID | 700 |
| RTECS number | BY2975000 |
| UNII | 8I9HQ1XY10 |
| UN number | UN1708 |
| CompTox Dashboard (EPA) | DTXSID1020634 |
| Properties | |
| Chemical formula | C7H9N |
| Molar mass | 107.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Aromatic aniline-like |
| Density | 0.998 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.38 |
| Vapor pressure | 0.86 mmHg (25°C) |
| Acidity (pKa) | 4.80 |
| Basicity (pKb) | 10.65 |
| Magnetic susceptibility (χ) | -7.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.568 |
| Viscosity | 1.47 mPa·s (20 °C) |
| Dipole moment | 1.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.2 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 86.0 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3437 kJ/mol |
| Pharmacology | |
| ATC code | D03AX03 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02, GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H315, H319, H373, H412 |
| Precautionary statements | P264, P270, P301+P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 85 °C |
| Autoignition temperature | 480 °C |
| Explosive limits | 1.3% - 7% |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral rat 350 mg/kg |
| LD50 (median dose) | LD50 (median dose): 650 mg/kg (oral, rat) |
| NIOSH | WI5400000 |
| PEL (Permissible) | 5 ppm |
| REL (Recommended) | 2 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Aniline o-Toluidine p-Toluidine Diphenylmethane Benzoic acid |

