Lysergic Acid Methyl Ester: A Grounded Look at the Substance
Historical Development
Lysergic Acid Methyl Ester belongs to a family of ergot alkaloids, a group of compounds that once turned entire villages upside-down in centuries past. The roots of these substances stretch back to bread riots blamed on moldy rye—a sign that humans chased answers about these strange compounds long before microscopes came along. Sandoz laboratories in Switzerland became a household name in scientific circles in the twentieth century, thanks to research into ergot derivatives. Their work brought new tools for chemistry and medicine, introducing substances that served both as medical breakthroughs and as cautionary tales. Lysergic Acid Methyl Ester did not rush into the limelight with popular notoriety like LSD, but it stands as a close chemical cousin, woven into the tapestry of chemical innovation that shaped psychiatric and pharmacological research through the mid-1900s.
Product Overview
Lysergic Acid Methyl Ester exists as a lesser-known derivative, riding the coattails of its more famous siblings in the lysergamide family. Its structure provides a bridge between lysergic acid and more complex compounds. Chemists recognize it as an intermediate—neither a mere building block nor an active pharmaceutical agent in its own right. In laboratories, this compound often finds its way onto the shelf as a powder or crystalline solid, set aside for reagent use or as a stepping-stone toward more valuable products. R&D labs value Lysergic Acid Methyl Ester as a chemical landmark, a waypoint in the long journey from natural ergot alkaloids to synthetic medicines that tackle everything from migraines to neuropsychiatric disorders.
Physical & Chemical Properties
Standing in front of a vial of Lysergic Acid Methyl Ester, I see an off-white or beige powder, its grains so fine they drift away with the lightest breath. Crystalline and sometimes waxy to the touch, this compound tends to cling to glassware, leaving a fine residue like powdered sugar. Its molecular formula tells the story of 16 carbon atoms, 17 hydrogens, two nitrogens, and three oxygens, all packed into a fused ring. The melting point floats in the range of 154 to 158 °C. Solubility sits at a crossroads: alcohols and chlorinated solvents invite the ester in, but water keeps it at arm’s length, complicating cleanup. A chemist who has cleaned countless beakers can tell you that basic pH encourages hydrolysis, chopping the methyl group away and sending the compound sliding back toward lysergic acid.
Technical Specifications & Labeling
Working with a bottle of this ester in the lab, the label jumps out with key details: molecular weight, purity percentage, storage temperature, and—always in bold—its schedule of regulatory oversight. Laboratories need to buy from certified suppliers who vouch for a purity level of at least 98 percent, if not higher. A Certificate of Analysis follows every shipment, outlining not only the expected physical data (melting point, appearance) but also analytical signatures: infrared spectra, NMR fingerprints, traces of residual solvents. In regions where the compound crosses legal thresholds, the paperwork swells with documentation for regulatory authorities, batch number tracking, and chain-of-custody logs. Even a moment of carelessness—an unlabeled container or missing batch sheet—invites heavy sanctions.
Preparation Method
Synthesizing Lysergic Acid Methyl Ester rarely starts with a gleaming array of fancy catalysts. The classic route begins with lysergic acid itself, isolated from ergot fungi or generated through semi-synthesis using colchicine or related alkaloids. The acid meets a methylating agent, such as diazomethane or dimethyl sulfate, under cold, controlled conditions. The reaction releases nitrogen gas and a surge of heat—gloves and goggles become a non-negotiable necessity. Skilled chemists keep a close eye on the reaction, since runaway conditions risk side-products that snarl purification efforts. Extraction follows: a careful wash with cold organic solvents, then slow filtration under low light—since bright light chews away at lysergic structures. The crude methyl ester then journeys through column chromatography, losing its tagalongs, until it emerges in relatively pure form. For those who value precision, the whole process demands patience, skill, and respect for both the dangers and the legacy of ergot chemistry.
Chemical Reactions & Modifications
Lysergic Acid Methyl Ester doesn’t exist in a vacuum. The methyl ester group tacked onto the lysergic backbone begs to be swapped under the right conditions. Acidic or basic hydrolysis drops the methyl and tips the compound back toward free lysergic acid, a handy feature for chemists tweaking derivatives. Substitutions on the indole ring open several doors, giving rise to a menagerie of analogs—sometimes only a single atom away from compounds that shaped the cultural landscape of the 1960s. Reductive amination at certain sites transforms this intermediate into entirely new substances with potent biomedical activity. The chemical reactivity rests at the core of its utility, nudging chemists to push boundaries in synthesis, all the while dodging tightrope-walker levels of risk.
Synonyms & Product Names
Ask a supplier or poke through published literature, and Lysergic Acid Methyl Ester goes by a tangle of names: LAME, Methyl Lysergate, and Methyl (8β)-9,10-didehydro-6-methylergoline-8-carboxylate. Sometimes, a simple “lysergic acid methyl ester” on a bottle invites confusion with other esters or lysergamides—an error that trips up stockroom managers and junior chemists alike. This confusing nomenclature forces labs to double-check chemical structures against spectral data, rather than trust a sticker on a bottle. For anyone handling ergot-derived chemicals, precision in names turns into a defensive strategy, not just paperwork.
Safety & Operational Standards
Chemists who spend time with lysergic acid derivatives know the drill: negative air-pressure hoods, gloves that reach to the elbow, dedicated glassware. Lysergic Acid Methyl Ester poses risks—both practical and regulatory. Accidental skin exposure leads to tingling or mild irritation, if not something more dramatic. Fumes, rare but possible, burn the nose and throat, making respirator masks a staple during purification steps. Work areas undergo regular inspections, and fire safety standards get drilled home, thanks to the organic solvents involved in preparation. Waste streams run through hazardous material channels, not down the drain. Regulators step in with demands for locked storage, inventory logs, and spot audits. A single skipped step lands a lab in hot water, risking both reputations and licenses.
Application Area
The reach of Lysergic Acid Methyl Ester stretches across pharmaceutical chemistry, neuropsychological research, and the design of next-generation ergot medicines. While it rarely steps into the world outside the lab, its presence anchors synthetic routes toward drugs that treat cluster headaches, pituitary tumors, or Parkinson’s disease. Scientists leverage its flexible structure, converting it into both active drugs and critical reference standards for forensic analysis. During my own graduate school days, one project revolved around tracking side-products in ergot alkaloid synthesis. Lysergic Acid Methyl Ester surfaced over and over—a reminder that even “intermediate” chemicals leave fingerprints across the entire research pipeline.
Research & Development
Every year, waves of research papers wash over scientific journals, describing new routes to build or use Lysergic Acid Methyl Ester. In chemical research, its importance stems from the methyl ester handhold, an anchor point for connecting up chain modifications. Analytical chemists rely on it as a standard to develop new HPLC and LC-MS methods, vital for tracking residues and breakdown products in pharmaceuticals. Some cutting-edge labs experiment with subtle tweaks to the methyl ester, probing the effects on biological activity and potential receptor affinity. With regulatory agencies tightening rules around recreational lysergamides, researchers lean on this compound as a legal waypoint. Despite restrictions, the compound fuels breakthroughs in synthetic methodology, often under the wary gaze of oversight agencies.
Toxicity Research
Toxicologists face challenges in pinning down the effects of Lysergic Acid Methyl Ester, since few animal models publish clear outcomes. The parent compound, lysergic acid, sports a reputation for vasoconstriction and central nervous system effects. Methylation tweaks absorption and metabolism, sometimes blunting the ability to cross the blood-brain barrier. Initial cell studies suggest low to moderate cytotoxicity, but systemic animal studies remain sparse—largely because the methyl ester stands in as a chemical tool, not an intended drug. For the average researcher, the risks remain hypothetical; caution springs less from established toxicity than from respect for ergot’s notorious side effects and the fine line chemists walk with bioactive compounds. Regulations lag as well: published occupational limits remain vague, so labs hedge their bets by adopting standards intended for the parent compound or better-known analogs.
Future Prospects
Looking forward, the role of Lysergic Acid Methyl Ester in scientific research tracks with the ebb and flow of interest in ergot chemistry. The march toward new migraine and cluster headache treatments comes fueled by synthetic intermediates like this one, with universities and pharma companies investing in more robust, scale-friendly synthesis methods. As the debate around psychedelic-assisted therapies heats up, every derivative of lysergic acid finds itself scrutinized anew. Analytical and forensic labs struggle to keep pace, since designer drugs often pivot away from regulated compounds by swapping just a methyl or ethyl group. As regulations evolve, the need for clear, open-source data on synthesis, handling, and safety only grows. With each revision to chemical registers and pharmaceutical rules, Lysergic Acid Methyl Ester remains relevant—not for its own sake, but as an axis around which the wheel of ergot research spins. Those working with it bear a responsibility; not just to chase new science, but to ensure each breakthrough builds on solid, ethical ground.
What is Lysergic Acid Methyl Ester used for?
Understanding Its Place in Chemistry
Lysergic acid methyl ester doesn’t get the attention LSD or ergot alkaloids receive, but that’s often because much of its life remains tucked away in research labs. Someone working with this compound probably holds a degree in chemistry, possibly a stack of safety gloves, and definitely a healthy respect for what a derivative of lysergic acid can do. It’s not a name you’ll hear in most news cycles, so curiosity about its uses deserves an answer that doesn’t dance around the facts.
Roots in Research
People sometimes think chemicals like this leap straight from a factory into a laboratory just to serve recreational chemistry. That idea ignores the real foundation: exploration in organic synthesis and pharmacology. Lysergic acid methyl ester usually gets filed under “intermediate” by lab techs and researchers, which means it helps create other compounds. This role makes it essential for piecing together molecules that chemists need to study—molecules that range from existing medicines to things not yet known.
Academic circles, especially those focusing on neurobiology or psychopharmacology, use lysergic acid methyl ester to build analogs. These analogs help explore how new compounds interact with serotonin and dopamine receptors. It’s not just about discovering something that gets filed under new drug applications. Scientists track changes on the molecular level, mapping out which tweaks could reduce hallucinations, cut down on side effects, or show promise as a treatment for psychiatric or neurodegenerative disorders.
Connecting the Dots to Public Health
Most people hold a healthy skepticism when they hear about compounds related to LSD, with good reason. Lysergic acid methyl ester shouldn’t raise alarm bells on its own. What does matter is how intermediates like this fit into the bigger picture of drug development. Treatments for migraines, for example, wouldn’t be what they are without past experiments using similar molecules. One of the first drugs in this category owes its existence to research on lysergic acid derivatives.
Sometimes, lawmakers raise questions about regulating precursor chemicals—molecules used to make more restricted substances. That’s a fair concern. Manufacturing control and close watch can help keep illicit labs from using these building blocks. But stricter requirements shouldn’t cut curious research off at the knees. Medical breakthroughs often come from that gray area, where a lot of discovery still waits. Researchers deserve tools and trust, so long as checks and balances hold.
Pathways to Safer Progress
Chemistry doesn’t just operate in a petri dish; it plays out on the policy battlefield too. My view is that smarter oversight strengthens, not stifles, real progress. It’s possible, and important, to keep a lid on illicit use without crushing scientific progress. A solution that works in reality includes controlled access for accredited labs, routine audits, and transparent reporting. These steps don’t just wall off bad actors; they invite the right ones to keep moving science forward.
Looking at lysergic acid methyl ester tells you where discipline and curiosity collide. Every layer of safety, every protocol, every documented result pushes knowledge further while respecting real-world risks. It’s a balancing act between discovery and caution. Most of all, it reminds us that chemistry—done right—keeps doors open for better solutions tomorrow.
Is Lysergic Acid Methyl Ester legal to purchase and possess?
Understanding This Elusive Compound
Conversations about substance legality often leave folks in the weeds. Lysergic Acid Methyl Ester, an obscure chemical cousin to the infamous LSD, grabs curiosity for several reasons. Chemists spot a molecule like this and see a handful of possible uses—research, synthesis, or just plain chemical curiosity. Most people hear “lysergic acid” and picture tie-dye and wild 1960s tales. So, it’s not surprising that questions circle around its legality.
Reading the Laws: Gray Areas and Risks
Lysergic Acid Methyl Ester flies under most mainstream radars. U.S. law classifies “lysergic acid diethylamide” (LSD) as a Schedule I substance, with all the harsh penalties that go with it. Lysergic acid itself, and some of its close relatives, land on various controlled lists too. The real rub comes from the Federal Analogue Act, signed off in 1986. This law says any compound “substantially similar” in structure and effect to a Schedule I or II drug gets treated just as seriously if meant for human use.
That’s a problem for anyone thinking laws need to spell out every single molecule. Lysergic Acid Methyl Ester doesn’t show up on the controlled substances schedule by name. Yet prosecutors and courts look past the chemical name. They ask: Does this stuff act like LSD? Is someone planning to use it, or sell it, for getting high? If so, all bets are off. For research or collecting, intent might seem innocent. Proving that intent, should federal agents ever ask, gets dicey—especially if someone possesses it outside an academic setting.
International Confusion and Global Travel
Traveling or buying from across borders doesn’t clear up the fog. The United Kingdom, for example, casts a wide net through the Misuse of Drugs Act. Australia and parts of Europe rely on analog laws and customs seizure powers. So, ordering a vial of Lysergic Acid Methyl Ester online might be legal in one place and criminal in the next. Some chemical suppliers advertise only to researchers with proof of institutional affiliation, which shows how worried they are about accidentally trafficking in gray-market drugs.
Why Knowing What’s Legal Matters
I think people underestimate how much risk compounds like this bring. Growing up, chemistry sets fascinated me. The shelves at the local library overflowed with tales of self-taught tinkerers. Now, the stakes feel different. Prosecution for buying the wrong chemical can mean years lost, reputations wrecked, families left to pick up the pieces. Beyond criminal law, possession might trigger academic discipline, job loss, or travel bans. Ignorance doesn’t hold up as a defense. Just because a molecule isn't famous doesn’t mean law enforcement doesn’t care.
On the flip side, scientific research can’t move forward if laws block access to every odd molecule. Biomedical discoveries have come from studying obscure lysergic derivatives. Lawmakers and regulators have started conversations about clearer carve-outs for researchers, possibly developing permits or trusted supplier networks. That would protect scientific progress and help prevent accidental criminalization.
Safer Choices and Responsible Curiosity
People can ask suppliers for documentation and clarify intended uses. Legal representation makes a difference if questions crop up. Only buying chemicals from reputable sources, with the right paperwork, cuts the risk of a costly misstep. Knowledge and caution help protect both curiosity and freedom.
What are the potential side effects of Lysergic Acid Methyl Ester?
Psychedelics: Power and Risks
Lysergic Acid Methyl Ester grabs attention because it sits in the same chemical family as LSD, a substance with a long and notorious story. Psychedelics can shake up perception and mood in ways that seem magical or terrifying depending on the person, the dose, or even the day. Back in college, I watched friends test the waters with similar substances, believing that learning about themselves would help them grow. But these trips didn’t always end with enlightenment. Sometimes, they sparked lasting anxiety or left someone feeling disconnected long after the experience faded.
Physical Side Effects Hit Hardest in the Body
The body seldom stays quiet during psychedelic experiences. Researchers note common effects like increased heart rate, higher blood pressure, and sweating. Gastrointestinal problems show up too—a rolling stomach, nausea, maybe vomiting. My own experience tells me that even the strongest-willed users can’t will away a pounding heart or shaky hands. These signs matter most for those with a history of heart problems or compromised immune health. The risk isn’t just discomfort. For some, physical distress can tip over into emergencies, according to studies from the late 2010s examining hospital admissions related to psychedelics.
Mental Effects Demand Respect
Psychedelic trips can feel limitless, but boundaries exist. People report intense hallucinations, confusion, and sometimes, paranoia. The stories my friends carried back from their earliest experiments echo these findings: color and sound would merge or twist, sometimes into beautiful shapes, sometimes into something scary enough to keep them awake at night. These experiences can leave a mark. One research review from 2022 found that temporary psychosis and panic reactions show up often in case reports, especially at higher doses or in people already dealing with mood disorders.
Long-Term Impact Isn’t Fiction
Not everyone shakes off a bad trip. Flashbacks happen, sometimes months after using these compounds. Persistent hallucinations—what experts now call HPPD, or hallucinogen persisting perception disorder—can change color perception or make visual “trails” stick long after the chemical leaves the body. It’s not something many people talk about before experimenting, but the evidence supporting these outcomes keeps growing. Scientists point out that people who start with even mild anxiety or depression face a higher likelihood of trouble sticking around after the experience.
Addressing the Downsides: Risk Reduction and Regulation
No single step solves the problems tied to unsupervised psychedelic use. Honest, science-based education will help more than scare tactics or legal crackdowns. People deserve real information about the risks—not just urban legends told in locker rooms or college dorms. Regulation can encourage safer manufacturing practices, cutting down on accidental overdoses or mystery additives. For people struggling with mental health concerns, screening or supervised use in a clinical setting may be safer. Community conversations about the safe use of psychedelics can change the landscape, moving from secrecy and stigma toward support. With rising curiosity around these chemicals, the tools for handling the fallout need to keep pace with public interest.
How should Lysergic Acid Methyl Ester be stored?
Security Isn't an Afterthought
Handling compounds like lysergic acid methyl ester calls for a good deal of respect. In lab work, respect means more than being careful; it means taking steps so nobody gets careless or curious in a way that lands someone in trouble. Keeping this compound behind a locked door isn't overkill, it’s common sense and a requirement according to basic lab safety manuals. Too many stories start with chemicals “walking off” because someone thought the risk was low. Storage in a steel cabinet, marked for restricted access, helps keep temptation out of reach and research above board.
Cool and Dry Is Not Optional
Most people remember to keep sensitive chemicals away from sunlight, but warmth does just as much damage. In my own experience, storing compounds in a cool room — below 10 degrees Celsius — extends the life of delicate substances. Many forget about the silent role that moisture plays. Even a few days in a humid environment can mess with purity, break down what you’re trying to keep stable, and result in expensive waste. A desiccator or a tightly sealed container with drying agents works as insurance. If you think that's overkill, just ask anyone who’s tried to salvage a ruined batch after a rainy day left seals uninspected.
Containers Matter More Than Labels Let On
Plastics invite trouble here. I’ve seen people cut corners by using plastic vials, only to watch those containers soften or pick up odors over time. Glass remains the right call, especially amber glass if there’s any risk of light exposure. Clean containers, with clear labeling showing content, date, and who put it there, make any future confusion or misstep less likely. If you’re sharing lab space, those detailed labels do more than avoid mix-ups—they help with accountability, and accountability keeps research safe.
Limiting Quantities Protects Everyone
Sitting on a year’s supply of any compound doesn’t make sense if each run only calls for milligrams. Smaller portions stored in separate containers cut risk. If one vial breaks, you haven’t lost the whole stock and cleanup stays manageable. Anyone who’s swept up a full jar of fine powder from a bench will want to avoid repeating the experience.
Legal and Ethical Compliance Isn’t Just Paperwork
Some folks see regulations as only red tape. That's a mistake. Laws around certain compounds exist because bad storage and weak oversight have led to real harm in the past. Checking in with regulatory guidelines means you stand up for safe science. Many universities and research labs have gone through surprise inspections. Staying prepared keeps your research on course and, frankly, keeps the funding coming in.
A Culture of Responsibility Makes Science Work
Getting the basics of storage right depends on the choices made every day in the lab. Setting clear guidelines and making tools for proper storage easy to reach gives everyone fewer chances to slip up. What’s worked for me is routine: a quick check of seals, locks, and labels as part of shutting down at the end of each day. Complacency doesn’t mix well with chemistry. Good storage is what keeps a lab running and what keeps science honest.
Is Lysergic Acid Methyl Ester safe for human consumption?
What Lysergic Acid Methyl Ester Means for Health
Lysergic Acid Methyl Ester, known in some chemistry circles as LAME, traces back to the same family as LSD. The name carries a lot of chemistry baggage, and most folks probably never bump into it—unless their work brings them close to complicated laboratory research. Curiosity about the safety of such compounds comes up because some get mixed up with psychoactive substances, either through history or actual use.
My learning journey taught me that safety with these substances boils down to understanding their background, structure, and proven effects. Unlike aspirin or caffeine, this one doesn’t show up in daily life, medical practice, or even in wellness circles. Lab findings paint a basic picture: information about Lysergic Acid Methyl Ester’s effects in humans remains almost non-existent. Lack of public studies leads to a fog of uncertainty. As a chemical, LAME ends up more discussed in organic synthesis and pharmacology classrooms rather than doctor’s offices or hospitals.
No Room for Guesswork Without Data
Experience tells me that relying on stories or anecdotes for safety isn’t wise. The FDA and health bodies base their advice on trials, long-term evidence, and good data. No published human trials exist for LAME, so we end up with a big blank where answers should be. Toxicity stays uncharted, and no guidelines cover how the liver, heart, or brain reacts. Most information traces back to chemical cousins like LSD, which has its own set of side effects and legal restrictions.
LSD shows us a bit about what can go wrong—think anxiety, confusion, spikes in blood pressure, and the risk of dangerous behavior during intoxication. A molecule only a few atoms away can display very different effects, sometimes safe, sometimes the opposite. In the Lysergic Acid Methyl Ester case, not enough is known to tilt the scale either way. No legitimate medical professional I’ve talked with would ever recommend trying a novel substance, especially one that escaped research, medical regulation, or published safety profiles.
Getting Caught up in Novel Compounds
Every new compound sparks hope in some quarters that it might heal, bring insight, or spark creativity. The history of psychoactives draws in people for a variety of reasons, but the reality hits hard—the jump from test tube curiosity to everyday use can land people in hospital beds or worse if unseen risks pop up. I’ve seen enough cases of people running into trouble after taking unknown substances to caution anyone considering experimentation. In some regions, getting caught with unsanctioned compounds can also mean a run-in with the law.
Reducing Risks: A Smarter Approach
Instead of taking risks with little-known chemicals, I encourage tapping into peer-reviewed research, talking with licensed healthcare pros, and choosing legal and medically approved options for both health and personal exploration. Science moves forward step by step. Volunteers join controlled clinical trials only after lengthy approval stages. That’s the gold standard: safety first, questions later, people protected along the way.
Young people especially often grab onto stories pulled from online forums or social feeds about obscure molecules promising new worlds. As someone who’s seen the downside of such adventures, I’d urge parents, professionals, and communities to talk openly and factually about the difference between chemical facts and internet fiction. Well-being outpaces raw curiosity.
Clear discussion, not hype or half-truths, builds a safer, stronger foundation for people and science.
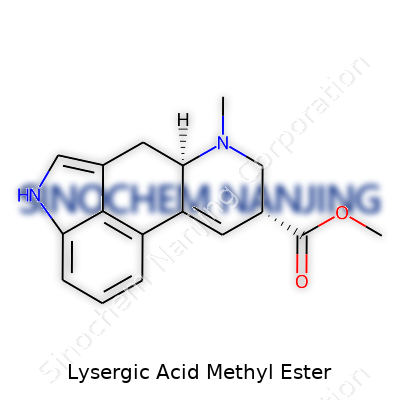
| Names | |
| Preferred IUPAC name | methyl (6aR,9R)-7-methyl-4,6,6a,7,8,9-hexahydroindolo[4,3-fg]quinoline-9-carboxylate |
| Other names |
LSM Lysergic acid methyl ester Lysergamide methyl ester 2D LAMM |
| Pronunciation | /laɪˈsɜːdʒɪk ˈæsɪd ˈmiːθəl ˈɛstər/ |
| Identifiers | |
| CAS Number | **22348-16-7** |
| Beilstein Reference | 153388 |
| ChEBI | CHEBI:156517 |
| ChEMBL | CHEMBL1948114 |
| ChemSpider | 20838688 |
| DrugBank | DB01428 |
| ECHA InfoCard | 13e34020-4651-474b-8b5d-82ab2d425bab |
| EC Number | 1.1.1.25 |
| Gmelin Reference | 3890 |
| KEGG | C06404 |
| MeSH | D049122 |
| PubChem CID | 101999 |
| RTECS number | OJ6825000 |
| UNII | 4R2L11RV6F |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DJ6CT3VC4J |
| Properties | |
| Chemical formula | C17H19NO3 |
| Molar mass | 267.296 g/mol |
| Appearance | White solid |
| Density | 1.1 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.7 |
| Acidity (pKa) | 7.38 |
| Basicity (pKb) | 11.0 |
| Magnetic susceptibility (χ) | -72.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.570 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 372.8 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N01XA02 |
| Hazards | |
| Main hazards | Causes skin and eye irritation. Harmful if swallowed, inhaled, or absorbed through skin. May cause central nervous system effects. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H312, H332, H351 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P305+P351+P338, P310, P321, P362+P364, P405, P501 |
| Flash point | 79.2 °C |
| LD50 (median dose) | LD50: 59 mg/kg (rat, oral) |
| NIOSH | NIOSH: QJ9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 micrograms |
| Related compounds | |
| Related compounds |
Lysergic acid Lysergic acid amide Lysergic acid ethyl ester Lysergic acid hydroxyethylamide Lysergic acid 2,4-dimethylazetidide Isolysergic acid methyl ester N-Methyllysergamide Ergine Lysergic acid diethylamide (LSD) |

