Levulinic Acid: Beyond the Laboratory
From Byproduct to Breakthrough: The Story of Levulinic Acid
Levulinic acid often gets mentioned in chemistry circles as a bio-based molecule with plenty of potential. It didn’t achieve this reputation overnight. Its first known synthesis reaches back to the 19th century, when Justus von Liebig observed it as a product of sugar digestion. For decades, researchers saw levulinic acid as a curiosity, an easily overlooked byproduct of carbohydrate breakdown. Nobody rushed to find a use for it, even though it seemed so abundant in plant matter. But as the world started searching for renewable chemicals, levulinic acid moved from the background to a front-row seat. The growing movement toward greener processes and the pressure to cut fossil-fuel use shined a light on this small, versatile molecule.
An Honest Look at Levulinic Acid
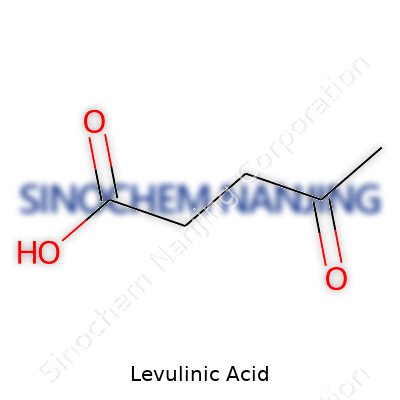
Ask a seasoned chemist what levulinic acid looks like, and there’s no guesswork. Its IUPAC name, 4-oxopentanoic acid, hints at its structure: a five-carbon backbone with both a ketone and a carboxylic acid. Pure levulinic acid pours out as a clear, sometimes pale-yellow liquid. The smell reminds many of burnt sugar or stale vinegar. Its melting point hovers near room temperature, making storage relatively straightforward. Solubility in water and a wide range of organic solvents means it doesn’t create workarounds during lab processes. Its molecular weight—about 116 grams per mole—lets it cross into more applications than bulkier chemicals. Boiling brings a distinct odor, which some find unpleasant, but it evaporates cleanly, which matters when you’re trying to minimize contamination in scale-up work.
Getting from Plant to Acid: How It’s Made
Turning raw plant matter into levulinic acid looks simple on paper, but not every feedstock responds the same way. Sulfuric acid serves as the most common catalyst. Heat it with lignocellulose—say, corn stover or non-food biomass—and you coax out this prized acid as glucose breaks down and dehydrates. The key isn’t just soaking the raw material; controlling reaction temperature, acid concentration, and residence times makes all the difference. There are efforts to switch out harsh sulfuric acid for greener catalysts or even enzyme pathways, as environmental concerns push manufacturers to soften their chemical footprints. Some companies crank up yields by using pressurized reactors, but the basics remain tied to breaking hexose sugars apart. In real-world settings, the process also delivers humins and formic acid—byproducts that call for careful separation and disposal.
Levulinic Acid: Chemical Potential
Many academic labs see levulinic acid as a Swiss Army knife for organic synthesis. Its two functional groups— the keto and carboxylic moieties—react with a range of nucleophiles and electrophiles. Reducing the ketone forms 4-hydroxypentanoic acid; further transformations can build pharmaceutical intermediates, flavor compounds, or biodegradable plastics. If you treat levulinic acid with alcohols under acid catalysis, you can make levulinate esters, often used as solvent components in inks and coatings. Reacting it with amines delivers N-substituted pyrrolidones, which are handy for cleaning and polymer chemistry. And there’s pentanoic acid for food flavors. It does not stop there: the molecule lends itself to hydrogenation, oxidation, halogenation, and condensation reactions, growing in popularity as a platform for new chemical entities. That flexibility means it’s in the crosshairs for anyone seeking to upgrade bio-based feedstocks.
What Do We Call It?
Chemists are notorious for synonyms, and levulinic acid is no different. Besides its common English name, it sometimes goes by “4-oxopentanoic acid,” “keto-valeric acid,” or simply “levulic acid.” These names pop up in research papers and patents. Its salts, known as levulinates, each perform niche functions: sodium levulinate, for example, acts as a preservative in personal care. The range of trade names keeps expanding, but seasoned users stick to the basics, fitting new forms around the needs of each formulation without confusion.
Staying Safe with Levulinic Acid
Anyone handling levulinic acid should read up before pouring it around. Although its toxicity tends to be moderate, the undiluted acid can irritate the skin, eyes, and airways. Accidental releases, especially in concentrated form, prompt prompt cleanups. Workers wear gloves and goggles to keep splashes at bay; good ventilation is common sense in any scale-up facility. European and American guidelines rate it below many other organic acids on the hazard scale, but they still set limits for workplace exposure. Storage in well-labeled, corrosion-resistant containers avoids confusion. It’s less hazardous than strong mineral acids, though not exactly mild. Waste streams need monitoring, as acids can corrode pipes and create local environmental risks if spilled in quantity.
Where Levulinic Acid Ends Up
Levulinic acid crosses boundaries few chemicals can claim. In agriculture, it acts as a building block for herbicides or plant biostimulants. Cosmetics and personal care brands rely on its derivatives for preservation and fragrance stability. Medicine sees it as a precursor for drug candidates, especially those requiring biodegradable scaffolds. The push for sustainable packaging lights up new possibilities: polylevulinic acid and related polymers bring compostability to materials that rarely decompose. Some niche fuels rely on levulinic acid esters for high-octane formulations, though gasoline surrogacy remains a challenge at scale. In the food world, flavors and additives sometimes start from this molecule. Researchers haven’t finished mapping out its full territory, but its presence from lab bench to factory floor shows a reach beyond novelty.
Levulinic Acid in Ongoing Research
Industrial heroes need dedicated minds to unlock every angle, and levulinic acid draws steady attention from biodegradability experts, green chemists, and material engineers. Some research focuses on low-temperature microwave reactors, aiming to push up yields while cutting costs and cutting down waste. Others test deep eutectic solvents or ionic liquids as alternative catalysts, hoping to move away from aggressive acids. The trend turns to valorizing every byproduct for a zero-waste approach: upcycling formic acid or tapping humins for fuel or carbon materials. Genetic engineering already tinkers with bacterial strains designed to ferment sugars directly to levulinic acid, aiming for continuous, low-impact production. The search for new application areas—renewable solvents, advanced plastics, and pharmaceutical intermediates—ties closely to breakthroughs in reaction control and separation science.
Toxicity: The Unanswered Chapter
Despite its history, levulinic acid brings some open questions on safety. It hasn’t shown the harsh toxicity of many petro-derived chemicals, but animal studies reveal that high doses can trigger metabolic imbalance, hinting at effects on kidney function or blood chemistry. Chronic exposure data stay scarce, especially for complex derivatives used in polymers or solvents. Regulatory groups in the US and Europe still want more long-term tests before classifying it as entirely benign. In practical terms, small quantities pass through waste treatment plants without fuss, though high-concentration spills need containment. Nobody calls it a threat like mercury or dioxin, but researchers need robust, multi-year studies to guarantee the safety claims that marketing departments often make.
Looking Down the Road: The Future of Levulinic Acid
Levulinic acid stands on the edge of something bigger. As more industries commit to decarbonization, the appetite for renewable, plant-sourced chemicals grows. Techniques for production continue to improve, aiming for cheaper, cleaner, and larger-scale runs. The future could hold biodegradable plastics fit for everyday use or new classes of safer solvents powered by levulinic acid derivatives. Governments press for greener chemistry, and levulinic acid’s flexibility fits their objectives. For everyone watching the transition away from fossil fuels, this molecule promises progress— but progress won’t come on autopilot. From regulatory safe use to true zero-waste processes, every advance will need more research, tougher scrutiny, and a willingness to innovate. Levulinic acid’s journey reflects the push to match old chemistry with new global needs, making it a molecule worth following as the sustainable future takes shape.
What is Levulinic Acid used for?
What Sets Levulinic Acid Apart
Levulinic acid might not be a household name yet, but it pops up more often than people realize—on ingredient lists, in lab reports, and sometimes in conversations about sustainable chemistry. It’s derived from biomass, like corn stalks or sugarcane, which means the story behind this acid starts on the farm, not a refinery. Growing up with a backyard garden fueled by compost, I learned early on how powerful plant-based solutions can get. These days, manufacturers focus on using levulinic acid in ways that turn agricultural leftovers into real, practical products.
Why Manufacturers Choose Levulinic Acid
I’ve noticed that the move toward plant-based chemicals isn't just about feeling good; it’s about results. People tap into levulinic acid because it acts as a chemical building block for everything from cleaning products to plastics. Big chemical companies look for molecules that break down less harmful stuff. Levulinic acid fits the bill. Instead of relying on petroleum-derived ingredients, companies harness this organic acid to make new types of solvents and plasticizers. These show up in paints, coatings, and certain biodegradable plastics.
During a recent visit to a friend’s green-tech lab, I saw how they use levulinic acid as a starting point for new biofuels. Turns out, the acid helps them convert plant materials into jet fuel and diesel alternatives. This cuts down on fossil fuel demand. According to the U.S. Department of Energy, advanced biofuel projects look to levulinic acid to bridge the gap between renewable resources and heavy-duty transportation needs.
Personal Care and Food Applications
Levulinic acid cropped up on my radar during a search for fragrance-free skincare. Formulators use it to keep lotions and shampoos shelf-stable and fresh longer. In these products, levulinic acid helps stop bad germs from taking over without the harshness of older preservatives. It passes European safety standards in cosmetics, which isn’t something all ingredients can say.
Another area is flavor science. Food technologists sometimes add small amounts of levulinic acid to flavors for a subtle boost, or to help mask bitter notes without adding sugar or salt. This use feels understated, but it shows the flexibility of this molecule.
Challenges and Outlook
Nothing about levulinic acid is magic. Production costs can run high, mainly when the scale is small or the feedstock isn’t reliable. Factories need to invest in new reactors to handle biomass materials, and that means upfront costs. It reminds me of early organic vegetable farming, where every weed and bug was a learning experience. Scaling up takes time and money.
Some companies look to government grants or partnerships with farmers to bring costs down. Investing in better agricultural waste collection and improved processing can push levulinic acid toward price parity with petroleum products. At the same time, more consumer goods companies ask for sustainable and non-toxic options. That steady demand could help tip the scales.
The Way Forward
Levulinic acid shows up as an everyday solution for folks working toward greener chemistry. My experience tinkering with plant waste in my garage compost pile matches what industrial chemists aim to do—a little innovation with what nature already gives. This approach might not solve the world’s problems overnight, but it marks a real shift in chemical manufacturing, one field and one molecule at a time.
Is Levulinic Acid safe for skin and hair care products?
What is Levulinic Acid?
Levulinic acid shows up on the ingredient lists of facial cleansers, hair conditioners, shampoos, and sometimes even sunscreens. Manufacturers love it because it helps adjust pH and makes preservatives work a bit better. It’s a molecule that starts out in plants or wood, turns up after some chemical processing, and ends up blended into creams, gels, and sprays.
Safety Research and Practical Evidence
People ask about safety for good reason—our skin takes the brunt of all kinds of environmental stress, and harsh ingredients can make things worse. Scientific research and expert safety panels have weighed in on levulinic acid, pointing out that most folks won’t have any trouble with it as an ingredient. A review by the Cosmetic Ingredient Review (CIR) panel, which looks at what happens when you use ingredients in real life, backs this up.
In actual day-to-day use, irritation rarely gets flagged. Levulinic acid isn’t sitting at the top of allergen lists. Don’t take my word for it—cosmetic companies that advertise “for sensitive skin” and “fragrance free” formulations frequently use it in place of heavier-duty preservatives, and they stick with it because complaints stay low.
Why People Worry
Many of us know someone who broke out in hives or a rash after using something new, and that shapes how we look at ingredient lists. Some might worry about synthetic origins or the fact that levulinic acid can irritate at high concentrations. In my own household, the only problems we saw came from using way too much product, or slathering on a face mask and not washing it off. Ingredient concentration makes a big difference. In regulated skin and hair care, levulinic acid usually ends up at less than 2%. That’s not enough to cause trouble for healthy, unbroken skin in most cases.
Who Should Take Care?
While most people can use products with levulinic acid with little worry, some exceptions exist. People with eczema, psoriasis, or very reactive skin should test a dollop behind the ear or inside the elbow before rubbing it everywhere. Babies and toddlers have more sensitive skin, so pediatricians might suggest steering clear where possible. But for most adults and teens, the real concern comes with leaving the product on for hours or using it at professional-strength levels, not with typical home use.
Product recalls and documented allergic reactions do not flag levulinic acid as a repeat offender. The Environmental Working Group’s rating system put it in the “low hazard” category—a small green light that matches what experienced dermatologists say. If you’re up for a Google Scholar dive, studies in both Europe and North America find little to no irritation at the levels you see in shampoos and lotions.
Better Choices for Sensitive Skin
If you’re worried or you’ve had trouble with preservatives or acids in the past, look for patch testing kits or sample sizes. Small health food brands and “free from” lines often spell out their entire formula online. Apps like Think Dirty or Yuka let you scan a product, dig up every ingredient, and see how it stacks up.
Where Opportunity Still Exists
Not everyone reacts the same way to skincare, and that’s where clearer labeling helps. Companies can print both the percentage used and the role of levulinic acid (whether it’s a pH adjuster or conditioner) to help customers make smarter choices. If people see honest transparency, trust builds. Dermatologists and pharmacists can step in with evidence-based recommendations, especially in clinics that serve allergy-prone or chronically ill patients. Less confusion leads to less anxiety at the drugstore shelf.
What are the main sources of Levulinic Acid?
Levulinic Acid’s Roots Start in the Field
Levulinic acid, once a rare topic even among chemists, now shows up in headlines about bio-based chemicals. The shift away from fossil resources draws attention to the raw materials that create building blocks for new materials and fuels. Few realize just how close the ties between common farm leftovers and high-tech solutions run.
Corn Stover and Wheat Straw: Harvest Residues Turned Gold
Crop residues stand out as a main source for levulinic acid. After harvest, fields brim with stalks, leaves, and cobs — materials like corn stover or wheat straw. These leftovers don’t only mean waste or bioenergy. They carry a lot of cellulose and hemicellulose, the tough carbohydrates that plants use as their structural backbone. If you’ve ever noticed how difficult it is to break down woody stems or stalks, that’s cellulose at work.
Researchers figured out that with acid treatment and the right temperature, cellulose chains break into smaller sugars like glucose, then down further into levulinic acid. This concept relies on what’s already abundant, avoiding competition with food and tapping into huge, underutilized streams. In the United States, agricultural waste tops 300 million tons a year, so even processing a fraction leaves room for a robust chemical supply without cropland expansion.
Sugarcane Bagasse: Fuel for Brazil, Raw Material Everywhere
Sugar refineries in countries like Brazil generate mountains of bagasse — the fibrous byproduct left after extracting juice from sugarcane. A lot gets burned for energy, but a significant share ends up stockpiled. Bagasse follows the same chemistry as straw or stover: acid treatment, heat, and the result, levulinic acid. The process adds value to what was once scrap, linking the world’s sugar bowl to global bioproduct markets.
Wood Chips and Sawdust: Sawmill Scraps Step Forward
Forests offer another pipeline. Logging operations, sawmills, and wood-based industries produce piles of chips, shavings, and bark. Europe and North America both look to forest waste for more than just pellets and paper. Breaking down lignocellulosic (that is, woody) biomass with strong acids results in the same sugar platform found in crop residues, eventually yielding levulinic acid. There’s comfort in knowing old planks or timber leftovers don’t go straight to landfill; in the right hands, these scraps transform into chemicals that improve everything from pharmaceuticals to fuel additives.
Energy Crops and Algae: Next Generation Hopes
Miscanthus, switchgrass, and other fast-growing energy crops belong in the conversation. They thrive on marginal land, need little upkeep, and pump out large amounts of cellulose each season. They’re less established, but provide a dedicated feedstock for future production. Likewise, some startups and university projects look to algae, which grows fast and doesn’t drain freshwater. For now, algae’s role stays small, but the prospect of using ponds and saltwater, with no direct competition for traditional farmland, makes it worth watching.
Why All This Matters
Levulinic acid doesn’t pop up in daily conversation, but it threads together big questions about sustainability, waste reduction, and renewable industry. Taking value from what we once overlooked — corn stalks, sawdust, cane fiber — means less reliance on petroleum, less landfill, and new economic life for rural areas. Investing in these feedstocks keeps farmers, scientists, and business leaders working side by side. For those living in agricultural regions, this shift brings real opportunities. It’s not a perfect system; collecting and processing residue requires elbow grease and new infrastructure. Partnerships will need to tackle transport, fair pricing, and soil health, making sure that removing leftovers doesn’t damage future crops. Yet the story of levulinic acid—from the field to factory floor—proves there are smarter ways to value what nature gives abundantly.
Does Levulinic Acid have any side effects?
Putting Levulinic Acid Under the Microscope
Levulinic acid comes up a lot in the talk about sustainable chemicals. As more products lean into biodegradable options, this organic acid shows up in cleaners, preservatives, and even personal care lotions. Beyond industry chatter, regular folks are bumping into it on ingredient lists. So, does anything nasty come with all this eco-friendliness?
Digging Into Its Safety Record
Plenty of attention focuses on safety before a substance finds its way into products. A solid base of research tells us that, for most people, levulinic acid acts as a low-toxicity, low-irritation ingredient. EU regulators allow it in rinse-off skin and hair care products under set limits, and US regulatory agencies have not sounded alarm bells. Skin irritation or allergic responses rarely pop up in case studies, and oral toxicity levels appear mild based on animal testing, unless consumed in quantities far above what you'd find in products.
Not Everyone Gets Off Without a Scratch
No chemical works the same in every body. Sensitive skin may react to levulinic acid, especially when exposure repeats or combines with alcohols and other acids. A few users report mild stinging or redness from personal care items. Long-term inhalation studies in factory workers are less plentiful, but for those mixing giant vats of the chemical, extended contact might mean headaches or respiratory irritation. That doesn’t translate to risk in a dab of skin lotion, but it’s not something to ignore in workplaces. Animal feeding trials haven’t flagged it as a carcinogen or a hormone disruptor, but anyone with eczema or chemical allergies should stay watchful about patch testing new products.
Questions About the Environment
The promise of levulinic acid’s biodegradability helped it earn its popularity. So far, environmental monitoring finds very little concern for wildlife or water contamination. Levulinic acid breaks down fast in soil and water, so it doesn’t stack up in the food chain. That’s better than many petrochemicals. Still, no ingredient gets a free pass if used at industrial scale or dumped in bulk. Local spills or leaks—just like with any acid—could disturb aquatic creatures near factories, and that risk shouldn’t be skipped in oversight plans.
Using Lessons From Other Chemicals
Years of trouble with old “wonder chemicals” like parabens or formaldehyde taught a hard lesson: today’s great fix can turn sour if ignored after launch. Modern watchdogs—NGOs, toxicologists, and government agencies—have started constant review cycles for emerging substances like levulinic acid. This strategy, regular independent testing, and real-world monitoring, catch problems before they become crises. Public ingredient transparency helps, too. People can scan an app or check a website to see if something suits their health needs or allergy profile.
Smarter Use, Safer Results
Levulinic acid fits into green chemistry goals but doesn’t erase personal responsibility. Manufacturers valuing safety run their own toxicology checks and adjust use concentrations accordingly. People with a skin condition or known chemical sensitivity should keep product samples small at first. On the industry side, stricter air filtration and worker education make a difference in factories. Community health comes out ahead when local rules cover both in-home and on-the-job exposure.
Room For Watchful Advancement
Basing daily product decisions on real science helps avoid repeating the past. So far, levulinic acid isn’t stacking up big negatives, but open access to new toxicity findings keeps everyone in the loop. If future data raises a flag, clear labeling and honest communication will build trust between product makers and buyers. For now, with eyes open, levulinic acid looks more like a friend than a foe in the shift to cleaner products.
How is Levulinic Acid manufactured or produced?
Behind the Buzz: How Levulinic Acid Gets Made
Levulinic acid isn’t a household name, but it has turned the heads of scientists, manufacturers, and sustainability advocates. It comes from biomass—anything from sawdust to corn stover to sugarcane bagasse. People have been searching for better ways to turn plants into useful chemicals, and making levulinic acid is one of those ways that actually makes sense for the future.
The process starts with plant waste and water. Hydrolysis and acid-catalyzed reactions break down tough fibers and sugars. Most facilities use strong acids, like sulfuric acid, since those get the job done by speeding up the breakdown of cellulose into simple sugars. By cranking up the heat and keeping everything cooked under pressure, this mash transforms. The right conditions turn the original biomass into a soup that contains levulinic acid, plus byproducts like furfural and formic acid.
A big part of the challenge comes from sorting the levulinic acid out of the mix. Acid neutralization, filtration, and distillation all happen in sequence. Facilities recover and clean levulinic acid from the rest, often recycling whatever they can to cut down on waste and keep costs from spiraling. Even with technology pushing hard to improve efficiency, operators still need to keep a close watch on contamination and corrosion caused by strong acids.
Why This Process Matters—And What Needs Work
A few years ago, I saw a demo plant in the Midwest that turned agricultural waste into building blocks like levulinic acid. It struck me how much energy and investment these operations soak up compared to simply burning the leftover stalks. Turning waste into new chemicals is a smarter way forward, especially as oil reserves shrink and demand for non-toxic, biodegradable materials rises.
Current manufacturing costs stop the process from taking over the market. Acid recovery steps turn out more expensive than many people expect, and cleanup leaves behind water streams that need careful handling. The acid itself is valuable, but equipment needs regular upkeep or corrosion will eat it alive. Safety rules stay tight in these plants for good reason.
There’s hope in scaling up. Pilot projects in Europe and North America show that automation and improved recycling of reagents can shave both costs and emissions. Research teams are searching for cheaper acids or even alternatives to traditional catalysts, which could lower barriers for smaller producers. A few startups now pitch “green” versions made using less harsh chemistry, though those haven’t fully proven themselves on an industrial scale yet.
Policy could speed things up. Tax breaks for bio-based chemicals and clear labeling requirements would give levulinic acid a leg up in markets that need safer solvents, new materials, or sustainable plastic ingredients. Honest communication between suppliers, regulators, and buyers should help iron out confusion over purity and environmental benefits.
Looking Ahead
Levulinic acid production stands as a work in progress, not a miracle solution. The ways we make it reflect real choices about sustainability, waste management, and innovation. People keep working on these problems, and it’s worth following their progress—both for the chemistry and for the bigger picture of how we put plant-based materials to work in everyday life.
| Names | |
| Preferred IUPAC name | 4-oxopentanoic acid |
| Other names |
4-oxopentanoic acid gamma-ketovaleric acid 4-oxo-n-valeric acid levulic acid acetylpropionic acid |
| Pronunciation | /ləˈvjuːlɪnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 123-76-2 |
| Beilstein Reference | 605583 |
| ChEBI | CHEBI:28441 |
| ChEMBL | CHEMBL681 |
| ChemSpider | 743 |
| DrugBank | DB08797 |
| ECHA InfoCard | 028-019-00-3 |
| EC Number | 204-624-6 |
| Gmelin Reference | Gmelin Reference: **83057** |
| KEGG | C00417 |
| MeSH | D007985 |
| PubChem CID | Levulinic Acid PubChem CID: `5460243` |
| RTECS number | OZ5075000 |
| UNII | 7K16DKM97A |
| UN number | UN1235 |
| Properties | |
| Chemical formula | C5H8O3 |
| Molar mass | 116.12 g/mol |
| Appearance | Colorless to pale yellow liquid or crystalline solid |
| Odor | Slight odor |
| Density | 1.14 g/cm³ |
| Solubility in water | miscible |
| log P | -0.6 |
| Vapor pressure | 0.49 mmHg (at 25 °C) |
| Acidity (pKa) | 4.59 |
| Basicity (pKb) | pKb ≈ 13.3 |
| Magnetic susceptibility (χ) | -8.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.464 |
| Viscosity | 2.24 mPa·s (25 °C) |
| Dipole moment | 2.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -589.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2212 kJ/mol |
| Pharmacology | |
| ATC code | D11AX10 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 109 °C |
| Autoignition temperature | 215 °C (419 °F; 488 K) |
| Lethal dose or concentration | LD50 Oral Rat 1850 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1850 mg/kg (oral, rat) |
| NIOSH | BW5425000 |
| PEL (Permissible) | 15 mg/m³ |
| REL (Recommended) | 0.05% |
| Related compounds | |
| Related compounds |
Acetone Butyric acid Gamma-Valerolactone Succinic acid Valeric acid |

