Itaconic Acid: From Fermentation Beginnings to Promising Industrial Star
A Look Back: The Roots of Itaconic Acid Production
Curiosity and necessity sparked early chemical production booms, and itaconic acid carries a clear legacy from those times. Fungi, notably Aspergillus terreus, brought this substance into the scientific spotlight nearly a century ago. The push to find alternatives to petroleum-based compounds fired researchers’ interest, especially during global disruptions in resource supplies. My own research journey brought me to fermentation labs, where the odor of growing cultures reminded me how resourceful these early chemists were in coaxing products from nature. The humble mold and a sugar substrate became the basis for industrial-scale production long before “green chemistry” became a phrase that showed up in grant proposals and marketing materials.
What Itaconic Acid Brings to the Table
Colored by its industrial genealogy, itaconic acid presents itself as a colorless, crystalline solid at room temperature. Press your nose close, and there’s a faint sharpness—fitting for something so integral in the world of organic acids. Easily soluble in water, it blends well into reaction mixtures, and its stability under normal storage conditions brings some peace to safety managers and production techs. Molecularly, it goes by C5H6O4, and its two carboxylic acid groups paired with a vinyl group open doors to a range of chemical transformations. From an application perspective, these unique sites help make itaconic acid a flexible monomer—ready to form the backbone of polymers, chelating agents, and more.
From Fermentation Tank to Finished Product
Fermentation technology underpins the growth of itaconic acid. In practical terms, setting up a batch means feeding carbohydrates—often glucose—into a vessel, inoculating with a starter fungus, and providing enough oxygen. Within days, the broth acidifies, and the acid emerges from the harvest. Advancements in metabolic engineering and bioprocess optimization have trimmed the cost, lifted yields, and carved a clear spot for itaconic acid as a platform chemical. For those running the reactors or tweaking the recovery steps, the focus turns to filtration and crystallization, which separate pure acid from a soupy mix of byproducts and spent cells.
Digging Deeper: Chemistry, Synonyms, and Labeling Details
Chemists, always searching for shortcuts and solutions, have found that the vinyl group in itaconic acid supports a rich mix of polymerization reactions. Copolymerization with acrylic or methacrylic acid turns out plastics with distinct flexibilities and applications. In the trade, you’ll find itaconic acid sold under names like methylenesuccinic acid or propylene dicarboxylic acid. Packaging regulations and standards direct the labeling, often noting purity, grade, and storage advice. In my time auditing labs and storerooms, missing or mixed-up labeling caused more headaches than most realize. Correct identifiers and hazard cautions prevent confusion on the warehouse shelf.
Safety and Handling: Real Risks, Real Solutions
Handling itaconic acid doesn’t draw the same alarm as strong mineral acids, but respect for any acid goes a long way. Skin and eye irritation pop up in the literature and in safety briefings on the factory floor. Simple gloves, goggles, and proper ventilation go far, and implementation of spill protocols helps head off minor accidents before they become costly. In production, dust generation demands more attention—fine powders can trigger respiratory irritation or even minor explosions under the right conditions. I’ve watched facilities upgrade dust control systems after safety audits highlighted overlooked risks. Training and clearly marked storage play as big a part in preventing injury as any new chemical engineering breakthrough.
Transforming and Modifying the Molecule
Many reactions take advantage of itaconic acid’s active vinyl and carboxylic groups. Hydrogenation can turn it into methyl succinic acid, and amidation chains it to even more complex molecules. I’ve spoken to researchers who see endless promise in tweaking itaconic acid’s backbone to tailor-make additives, plasticizers, or bio-derived surfactants. The push for sustainable materials means chemists get creative, grafting itaconic motifs onto big polymers or using it as a branching point for pharmaceutical precursors. The sheer range of feasible modifications brings flexibility, but it also pushes the need for deeper toxicological studies and consistent regulatory compliance.
The Expanding World of Application
Industries have picked up itaconic acid for diverse roles. Polymeric products—coatings, adhesives, insulating foams—make up a hefty share. Water treatment noticed its chelating power, while the detergent world appreciates how it softens water and tackles mineral deposits. In agriculture, formulators count on its biodegradability and lower toxicity compared to more synthetic ingredients. Bioplastics researchers regard itaconic acid almost as a test case for renewable feedstock impacting product performance and recyclability. Each sector builds out unique requirements, but the underlying goal remains clear: getting the benefits of robust functional groups without leaning on fossil fuels.
Advancing Knowledge: Research, Test Results, and Toxicity
Work continues on how itaconic acid interacts with living systems, and that matters for both worker safety and environmental exposure. Toxicity research suggests low acute toxicity to mammals, but the nuances of chronic exposure and large-scale discharge into waterways still need study. Research labs worldwide drill deeper into its potential as a bio-based platform, experimenting with novel fermentation organisms and greener extraction methods. I’ve heard debate in conference halls between those prioritizing maximum yields and those focused on the cleanest process—both sides want itaconic acid’s promise, just by different routes. The ongoing stream of academic papers and patents reflects a thriving scientific interest in squeezing more value and safety out of every kilogram produced.
Looking Forward: Where Itaconic Acid Could Go Next
Future prospects look bright. The bioplastics industry keeps growing, and regulations pinning down single-use plastics create open lanes for renewable compounds. More chemical manufacturers weigh the trade-offs between price, performance, and environmental impact, and that bumps itaconic acid further up the list for pilot programs and scale-ups. Any disruption in petroleum supply, whether from geopolitics or climate, just adds urgency. Investments in synthetic biology turn up new production strains each year, promising higher output and lower waste. How far itaconic acid will reach depends on this steady interplay of performance, sustainability, and commercial push. From its roots in a flask of fermenting sugar to its place in cutting-edge polymers, itaconic acid shows that centuries-old processes can still carve out new futures.
What is itaconic acid used for?
What is Itaconic Acid?
Itaconic acid comes from fermentation, usually using fungi like Aspergillus terreus, to turn sugar into something new. The funny thing is, most folks have never heard of it, yet a lot of stuff you use, from plastics to detergents, relies on this molecule. For decades, petrochemicals did the heavy lifting, but with fossil fuels getting expensive and the planet heating up, makers started looking for building blocks that grow back or renew instead of run out.
Itaconic Acid in Industry
This molecule steps in as a replacement for things like acrylic or methacrylic acid. That’s because its chemical makeup works for making polymers—think plastics, coatings, adhesives, even some fibers. One of the big wins comes from its double carboxylic groups, helping to toughen up and add flexibility at the same time, which comes in handy for paints and coatings. Companies often choose itaconic acid in water-based paints to cut down on volatile compounds that hurt air quality and cause headaches for families.
You’ll find itaconic acid in the mix for superabsorbent polymers, like those in diapers or sanitary products. The top brands look for high absorbency and gentle touch, and this acid joins the blend, boosting performance while lowering reliance on petroleum.
Environmental Impact and Sustainability
For those tracking eco-friendly trends, itaconic acid checks a lot of boxes. Its production process relies on renewable feedstocks like corn or sugar, meaning less pressure on oil wells. This shift matters to farmers, too, since demand for corn and sugar beet supports agricultural jobs. In the chemical industry, switching even a portion of production over to biobased acids cuts carbon emissions and puts less strain on supply chains tied to global events.
Data shows demand for bio-based polymers will keep rising. In 2023, the global itaconic acid market reached nearly $200 million, and analysts expect consistent growth. Biodegradable plastics and greener detergents stand out as reasons for the shift. As more companies sign on to climate pledges, the need for renewable building blocks keeps climbing.
Room for Improvement and Solutions
Although itaconic acid holds promise, cost still creates pushback. Fermentation can take longer and cost more than old-school chemistry. Labs worldwide work on supercharging the fermentation organisms, using genetic tweaks or process tricks to boost yield. Better fermentation means lower costs, shorter production windows, and, in the long run, broader access. At a community level, education on sustainable materials needs action, so buyers understand why some products may cost more, at least for now.
Another challenge comes from infrastructure. Factories built for oil-based chemicals rarely switch to fermenters without big upfront investments. Government grants or tax credits for sustainable upgrades speed up the transition, as seen in the United States and parts of Europe.
Looking Ahead
The story of itaconic acid shows how tiny molecules can punch above their weight. Science and industry move toward greener choices, and every small switch makes a difference. With creativity and some investment, the future holds hope for more renewable options in everyday items, and itaconic acid leads the charge as one example already making that shift possible.
Is itaconic acid safe for human use?
Breaking Down the Buzz Around Itaconic Acid
Most folks haven’t heard much about itaconic acid, even though companies use it in plenty of products. This organic compound pops up in plastics, resins, and even food additives. Whenever some new chemical slips into things we touch, eat, or wear, people start to wonder about its safety. Authorities and consumers both want straight answers, not just technical talk. Is itaconic acid something that belongs near our food or in our skin creams? Or are we jumping too fast, chasing convenience over caution?
Where It Shows Up
You see itaconic acid on food labels as E353. Manufacturers use it as an acidity regulator or a flavoring agent. It offers a tart taste, much like citric acid. Food safety agencies in the US, Europe, and elsewhere have allowed food makers to use it, but always in very small amounts. Some manufacturers also turn to itaconic acid for biodegradable plastics and synthetic fibers. It crops up in detergents and even personal care products.
What Science Says About Safety
Researchers dove into its safety decades ago. Animal studies looked at how mammals handled itaconic acid. At reasonable concentrations, lab rats and mice didn’t show harmful effects. High doses—doses far bigger than what humans would get—sometimes led to stomach trouble, but not cancer or birth defects. Toxicologists use these kinds of studies to set safe upper limits, often by keeping human exposure a hundred times lower than levels that gave animals problems.
Regulatory bodies haven’t found evidence linking itaconic acid in foods or consumer goods to health issues in people. The European Food Safety Authority and the US Food and Drug Administration have evaluated the compound and set standards to keep exposure minimal. I’ve checked these reports myself, and they’re thorough, spanning many years of real-world and lab testing.
A Natural Part of the Human Body
Surprisingly, your own body makes itaconic acid. White blood cells, when fighting infections, produce small amounts of itaconic acid as part of their defense mechanisms. This gives researchers another avenue to explore its roles and long-term impacts. No connection turned up between natural levels and illness. It appears to be part of our normal metabolism rather than a random contaminant.
What Consumers Should Watch For
Folks with allergies or sensitive guts watch food additives closely, so clear labeling matters. People deserve an easy way to spot ingredients and know what goes into their food or lotion. Industry should keep sharing new research with consumers as product uses change. Adding stronger transparency about manufacture and sourcing also goes a long way toward trust.
Striking a Balance
Modern life runs on chemicals most people can’t pronounce. The goal isn’t to ban every unfamiliar name, but to make sure companies put real safety data ahead of short-term profit. For now, the weight of science says that itaconic acid is safe for human use in the doses we see in foods and consumer goods. That doesn’t mean researchers stop paying attention. Ongoing monitoring, clear labeling, and public access to information shape a fairer, safer marketplace for everyone.
What are the main applications of itaconic acid in industry?
Modern Manufacturing’s Quiet Workhorse
Most people never hear about itaconic acid. So many of the things we touch every day — coatings, adhesives, even diapers — wouldn’t work the same without it. Born from a simple fermenting process, this compound finds roots in old-school methods. Fungi like Aspergillus terreus convert sugars into itaconic acid, which industrial chemists then shape into a range of products that shape our lives.
Powering Biodegradable Plastics
The plastics world faces pressure to clean up its act. Itaconic acid brings a real advantage here: its plant-based origins. Companies blend it into polymers such as polyitaconic acid and copolymers, both of which break down naturally in landfills. Research from the Journal of Cleaner Production has tracked how these plastics degrade more quickly than those based on oil. I worked at a sustainable packaging firm and watched R&D teams swap out old crude-oil-derived resins for those containing itaconic acid. The results spoke for themselves — packages started to compost after a few months, not decades.
Thickening and Binding in Paints and Coatings
A bucket of cheap paint won’t stick to a wall or keep rust at bay for long. Formulators lean on itaconic acid to solve these headaches. The material acts as a comonomer in acrylic resins, a backbone for protective and decorative paints. BASF and other major chemical companies adopted itaconic acid to boost gloss, improve adhesion, and help paint resist cracking. This isn’t just for high-end construction. Even municipal crews see the effects — graffiti-resistant coatings and weather-tough finishes often come from blends containing this bio-acid.
Superabsorbent Polymers That Actually Work
Every parent knows the magic behind modern diapers: the core absorbs liquid and locks it away. That magic traces back in part to itaconic acid. Mixed with acrylic acid, it gives rise to polymers that soak up more than 100 times their weight in water. In the early 2000s, manufacturers struggled with balancing absorbency and skin sensitivity. Research teams added itaconic acid to reduce itchiness and adjust how fast the pad swells. Kids and parents simply saw drier, comfier diapers.
Adhesives and Glues for Greener Choices
Home DIYers and professionals both know a weak glue turns a project into a mess. Adding itaconic acid to adhesives keeps bonds strong, even as humidity and temperature swing. In wood adhesives and pressure-sensitive glues, this ingredient brings better tack and stickiness without relying on fossil ingredients. Big flooring factories introduced these blends to meet stricter emissions rules in the EU, which cut down off-gassing from traditional glues. At home, I’ve noticed fewer headaches after using eco-labeled craft glues with a biopolymer backbone.
Looking Ahead: Challenges and Solutions
Cost and scale slow down widespread adoption. Itaconic acid can still cost more than its oil-derived cousins, tight margins in building and packaging markets matter. Industry and government need to keep investing in fermentation improvements and large-scale biorefineries to trim down production costs. Incentives for using renewable resources, as seen in some bioeconomy strategies, could nudge companies the right way. Universities and manufacturers already team up, pushing fermentation yields with smarter microorganisms. More efficient production will open new doors — and help create everyday products that line up with a sustainable future.
How is itaconic acid produced?
The Story Behind Itaconic Acid
Fermentation sits at the heart of most itaconic acid production. This organic acid isn’t pulled out of rocks or pumped out of wells. Instead, people turn to tiny living factories—mainly fungi from the Aspergillus or Ustilago family. My first time touring a fermentation plant, I noticed the strong earthy smell, the humming tanks, and the quiet, careful monitoring—elements that highlight just how much know-how sits behind every barrel of this acid.
Microbes Do the Heavy Lifting
In most factories, Aspergillus terreus does the actual job. The mold chews through sugar—often glucose or cheap agricultural by-products like molasses. Mix in some key nutrients, a stable temperature, and oxygen, and the fermentation tanks turn that sugar into itaconic acid. Those who grow up around breweries or dairies see the parallel: Control the microbes, get the product. The process involves more than just feeding them sugar. Staff keep a tight grip on pH, oxygen flow, and nutrient levels. Any slip and production drops, or the fungus churns out unwanted by-products instead of itaconic acid.
Getting the Pure Acid Out
The fermentation broth comes out cloudy, full of solids and leftover nutrients. Technicians filter or centrifuge the mess, then use acid precipitation or solvent extraction to draw out the product. The final step often involves crystallization to get pure, white itaconic acid. Each stage takes energy, chemicals, and care—the closer to food or pharmaceutical grade, the higher the standard every worker must meet. Tough environmental rules add an extra layer of work, especially where water treatment and chemical handling matter.
The Facts on Scale and Demand
Right now, most of the world’s itaconic acid goes into plastics, detergents, and paints. It also finds use in water treatment and even textiles. Demand ticks up year after year, in part because this acid comes from renewables. With petrochemical prices jumping around and pressure growing around climate issues, more manufacturers look to fermentation as a stable source. Market research from 2023 puts global production at around 40,000 tons per year. The biggest challenges? Raising yields, cutting costs, and finding new ways to use more types of plant waste instead of just pure sugar.
Hurdles and New Ideas
Getting microbes to spit out more acid seems simple, but the science gets tricky. Researchers tinker with genetics to boost fermentation. Synthetic biology groups even build designer fungi capable of making itaconic acid more cleanly or quickly. At pilot facilities, technicians test corn stover or wheat bran as feedstock, aiming to keep prices down and cut waste. Today’s plants still depend on reliable, clean sugar sources for the bulk of production, but the next big leap could come from upcycling what farms and food factories throw away. That sort of innovation offers an answer to scale and environmental concerns both.
Why It Matters
Itaconic acid production ties together science, agriculture, and green chemistry. Every industry that leans into renewable routes instead of fossil routes helps soften the blow of unstable raw material prices, eases environmental impacts, and puts biotechnology on the map. My own visits to these sites showed people proud of tinkering with microbes and mixing old-school practical skills with cutting-edge lab science. Keeping this momentum matters if chemical manufacturing is going to stay nimble, clean, and competitive.
What are the storage and handling requirements for itaconic acid?
Thinking About Safety
Itaconic acid doesn’t find itself splashed across headlines, but I’ve seen enough in labs and warehouses to know it asks for respect. You crack open a drum and get a faint whiff — a sharp note that reminds you this isn’t just another powder. Anyone handling chemicals appreciates that most industrial acids have two faces: helpful in the right spot, a headache if you get sloppy. In the case of itaconic acid, breathing in the dust or letting it linger on bare skin brings real irritation. Getting proper gloves and a solid dust mask is a no-brainer, but good habits anchor safety: keep workspaces well-ventilated and use eye protection even when you think you’re just scooping “one more time.”
Humidity and Clumping
Itaconic acid grabs at water in the air. I’ve opened an old bag in a damp storeroom more than once and found solid chunks stuck together, making it hard to weigh out. This isn’t some rare disaster — it happens faster than people expect. The smart move is to keep it tightly sealed and dry. Humidity wrecks more than powder flow, too. Over time, unwanted chemical changes creep in. Anyone running a process line counting on clean, consistent acids can lose an entire batch thanks to moisture. Silica gel packs, airtight containers, or climate-controlled rooms protect your stock — not fancy, just effective.
Temperature, Light, and Container Choices
Heat does itaconic acid no favors. I’ve watched temperature swings spoil stashes by speeding up degradation, nudging toward discolored, off-smelling product. Helping it stay stable comes down to storing it cool, away from heaters or direct sunlight, and not letting it bump around near highly reactive chemicals like strong oxidizers and alkalies. polyethylene linings inside steel drums, or extra-thick HDPE containers, hold up well if you’re moving or stacking the stuff. Workers need to label containers clearly, too. I’ve seen mix-ups between “white powders” cause downtime and confusion. Simple labeling stops a week’s work from going sideways.
Fire Hazards and Clean-Up
It’s not the kind of acid that bursts into flame with a spark, but it can still become fuel if a fire takes off nearby. In my time on plant floors, fire drills and spill plans always factored in where the acids sat. Keep extinguishers rated for chemical fires close. If itaconic acid gets loose, dry sweeps work best; avoid using water which turns it into a sticky mess. If skin or eyes get exposed, everyone knows to flush with tons of water and head straight to a safety station.
Training and Procurement
I’ve trained enough new faces to see that confusion breeds accidents. Before handing over so much as a scoop, give clear, simple briefings about what’s inside the barrel, how to store it, and where to find spill response kits. Don’t order more than you can handle — smaller containers limit risk from spills or spoilage. Suppliers can offer batch-specific storage guidelines, but double-check with experienced staff before any chemical hits your shelves.
Keeping Track for the Long Haul
Long-term storage gets overlooked. I make it a habit to rotate stock, track every delivery, and set reminders to check for signs of caking or aging. Visual checks, smell tests, or random sampling all carry weight. Lab tests might catch changes — but noticing a funny color or odd odor early can avoid big problems later. In any workplace, personal responsibility and steady routines shield against surprise disasters — and nothing beats knowing your material, inside and out.
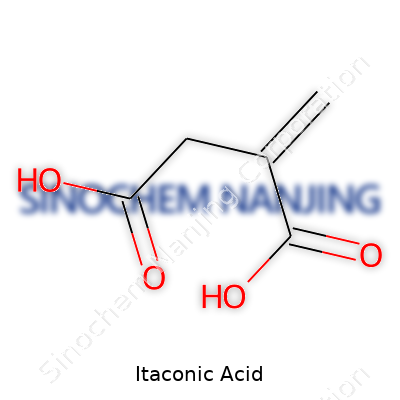
| Names | |
| Preferred IUPAC name | 2-methylidenebutanedioic acid |
| Other names |
Methylene succinic acid Methylene butanedioic acid Propylene dicarboxylic acid Itaconate |
| Pronunciation | /ˌaɪ.təˈkɒn.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 97-65-4 |
| Beilstein Reference | 1905891 |
| ChEBI | CHEBI:36976 |
| ChEMBL | CHEMBL12312 |
| ChemSpider | 52638 |
| DrugBank | DB04452 |
| ECHA InfoCard | 100.003.420 |
| EC Number | EC 211-072-7 |
| Gmelin Reference | 6956 |
| KEGG | C00490 |
| MeSH | D007510 |
| PubChem CID | 5699 |
| RTECS number | NI4370000 |
| UNII | 1U8V939EPU |
| UN number | UN2287 |
| Properties | |
| Chemical formula | C5H6O4 |
| Molar mass | 130.10 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.63 g/cm³ |
| Solubility in water | 78 g/L (20 °C) |
| log P | -1.85 |
| Vapor pressure | 0.4 mmHg (165°C) |
| Acidity (pKa) | 3.83, 5.55 |
| Basicity (pKb) | 3.85 |
| Refractive index (nD) | 1.542 |
| Dipole moment | 1.64 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 276.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -811.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1472.8 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS07,GHS05 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 Health=1, Flammability=2, Instability=0 |
| Flash point | 185 °C |
| Autoignition temperature | 368 °C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 4,500 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Itaconic Acid: Oral, rat: 6400 mg/kg |
| NIOSH | SN 3850000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 6 mg/m³ |
| Related compounds | |
| Related compounds |
Citraconic acid Mesaconic acid Fumaric acid Maleic acid Succinic acid |

