Isovaleric Acid: A Remarkable History and a Persistent Presence in Science and Industry
Historical Development
People have known about isovaleric acid since the days when early chemists were just starting to map out the rich and wild world of organic compounds. Chemists in the 1800s stumbled upon it during their attempts to isolate and characterize substances responsible for the strong odors coming from both plant and animal sources. Some of the best records show that isovaleric acid stood out for its sharp, sweaty smell, which made it hard to ignore, and just as hard to forget in memory and laboratory notebooks alike. Over the years, the story developed beyond curiosity, leading to a clear identity and growing relevance in research, food flavoring, and synthesis.
Product Overview
At its core, isovaleric acid is a simple branched short-chain fatty acid. It pops up naturally in foods like cheese and in human sweat. Curiosity around this acid ramped up in the 20th century, as people discovered its significant contribution to familiar flavors and aromas, both pleasant and notorious. Pure isovaleric acid is a colorless to pale yellow liquid with a pungent odor—one easily recognized even by people with modest chemistry backgrounds. For manufacturers, the compound serves as a base material for flavoring agents, plasticizers, and certain pharmaceuticals, among a surprising army of niche uses.
Physical & Chemical Properties
With a molecular formula of C5H10O2, isovaleric acid features a branched structure that sets it apart from its straight-chain cousins. Boiling point lands around 175°C, and it has moderate solubility in water. Instead of blending smoothly, it tends to hang around the edges, forming an oily layer and releasing its aroma with even gentle agitation. This volatility can cause headaches for plant workers and researchers, making personal protective equipment an absolute must. Like other fatty acids, the molecule features a carboxyl group at one end but stands out with its isopropyl branch, adding another layer of interest not only for organic chemists but for those working in synthesis, aroma chemistry, and even medicine.
Technical Specifications & Labeling
Labeling for isovaleric acid usually follows global standards, listing it as a corrosive, flammable liquid, demanding careful handling and storage. Container labels highlight its acute toxicity through inhalation and skin contact risks. Reagent and industrial grades often show purity above 98 percent, with trace levels of contaminants like water and related acids. Governments and safety organizations require clear hazard communication for all users, echoing growing standards in chemical safety fostered over decades of hard-learned lessons in industrial history. Not everyone has first-hand experience in the lab with the stuff, but anyone who does quickly adopts caution.
Preparation Method
In a production plant, manufacturers use a few dependable methods to create isovaleric acid. The most common laboratory synthesis involves the hydrolysis of isovaleronitrile, usually under acidic conditions, to release isovaleric acid and ammonia. On an industrial scale, people often rely on oxidation of isopentanol, where a controlled introduction of oxygen triggers the transformation from alcohol to acid. These methods reflect broader trends in chemistry: taking natural reactions that happen in living organisms or in nature and pushing them for yield, safety, and scalability. Each step requires tight control, since runaway reactions with organic acids can create both safety risks and huge losses.
Chemical Reactions & Modifications
Isovaleric acid offers plenty of chemistry for those eager to tinker in the lab. Its carboxylic acid group easily forms esters through reaction with alcohols, making it valuable in the fragrance and flavor industries. These esters, particularly isovaleryl acetate or isovaleryl ethyl ester, have floral and fruity notes, a far more pleasant profile than the starting acid. Researchers use it in acylation reactions, sometimes to modify natural products or create pharmaceutical intermediates. The acid also serves as a building block for synthesizing specialty polymers and surfactants, adapting easily to the demands of modern materials science. To anyone who has run a reaction with a stubborn carboxylic acid, the streamlined branching and volatility present both opportunities and practical obstacles.
Synonyms & Product Names
Chemists and industry folks alike use a long list of names for isovaleric acid. Its most common synonym is 3-methylbutanoic acid, a nod to its position on the carbon chain and methyl branching. Other names, like isopentanoic acid or beta-methylbutyric acid, sometimes show up in the literature or old catalogues. For flavor and fragrance specialists, the code numbers and trade names shuffle from year to year as companies brand and rebrand, but the molecule at the heart of each label remains the same.
Safety & Operational Standards
Safety dominates the conversation whenever people work with isovaleric acid. Despite its small scale in many facilities, the stuff can inflict real harm—corrosive action on skin and eyes, headaches, respiratory irritation, and more if spilled, inhaled, or ingested. Proper air handling, personal protective gear, and rigorous training underpin safe handling. The chemical’s volatility and flammability require all containers to be airtight, stored away from ignition sources, and situated within ventilated chemical storage rooms. Over the years, international bodies tightened rules, drawing on accident reports and the unique risks posed by branched short-chain acids. Even one small incident can set off a rush to improve engineering controls and revise standard operating procedures.
Application Area
Outside the laboratory, isovaleric acid pops up in more industries and products than most people realize. It helps produce snack foods and flavorings, as well as some alcoholic beverages, offering complexity and punch to familiar tastes like cheddar, camembert, and certain hoppy beers. Its esters boost fruity notes in perfumes and lotions. Some pharmaceutical firms turn to isovaleric acid when developing anticonvulsants or intermediates for specialty drugs. Animal nutrition scientists have trialed the molecule as a feed additive, leveraging its natural presence in the rumen and gut ecology. Not every use shines bright in the spotlight, but the sum total results in a steady demand that ripples through specialty chemical supply chains.
Research & Development
Research into isovaleric acid continues on several fronts, with scientists studying its role in both health and disease. In metabolic studies, the compound helps uncover links between bacterial metabolism and human physiology, especially through connections with the gut microbiome. Ongoing work probes how isovaleric acid and its derivatives shift flavor profiles, which matters for both food innovation and authenticity testing. Teams working with synthetic biology have engineered pathways to turn simple sugars into isovaleric acid using genetically modified microbes, pushing the market away from fossil fuels and toward renewable feedstocks. Such work demands patience, multidisciplinary teamwork, and a strong tolerance for frustration, especially when yields dip or competing side reactions swamp the intended outcome.
Toxicity Research
Teams monitoring toxicity keep a well-thumbed file on isovaleric acid, since its role in both industry and health remains complex. Animal testing shows that high doses cause distress, especially in the liver and nervous system, but low concentrations are tolerated in foods and even synthesized naturally inside the body. Researchers track rare metabolic disorders like isovaleric acidemia, where children suffer from a buildup of isovaleric acid, facing seizures and cognitive delays without proper intervention. Guidelines for workplace exposure rest partly on these findings, keeping permissible limits low and flagging the need for tight handling in research and chemical manufacturing settings. No two people react the same way, so regulatory agencies keep a steady eye out for new data and demand rigorous reporting from anyone using or studying the compound.
Future Prospects
Looking down the road, opportunities for isovaleric acid seem to rest in a few big domains: biotechnology, green chemistry, and health research. Bio-based production offers hope for less resource-heavy synthesis, promising better economics and smaller environmental footprints. Some researchers view isovaleric acid as a tool for tracking metabolic health or mapping the diversity of the human microbiome. In flavors and fragrances, regulatory shifts and consumer demand may push companies toward molecules with more transparent origins, nudging more research toward fermentation or enzyme-driven production. Given the complex aroma and stubborn volatility, engineers and formulators will keep tweaking methods for capture, storage, and delivery. The molecule’s reputation—sharp and distinctive—almost guarantees it a continued place at the table, in science as well as industry, for years to come.
What is Isovaleric Acid used for?
More Than Just a Pungent Smell
Isovaleric acid grabs attention, not for glamor but for a sharp, cheesy odor that reminds most people of sweaty socks or certain strong cheeses. Behind that unforgettable aroma hides a chemical with surprising uses. In my own time spent around food production and lab spaces, I’ve seen the double take when someone uncorks a bottle of this stuff. Yet, it finds its way into daily routines more than most folks realize.
Food Flavor and Fragrance
The biggest spotlight shines on flavor. Isovaleric acid plays a lead role in crafting the notes found in blue cheese, soy sauce, and a handful of funky but beloved foods. Chefs and food chemists turn to this compound to dial up that savory punch people crave in certain cheese-based recipes or gourmet creations. In the world of snacks and seasonings, flavorists rely on isovaleric acid to produce a taste people remember.
Beyond savory foods, the same molecule pops up in the world of fragrance. I’ve watched perfume makers harness its punchy scent in small amounts for those who want earthy, leathery notes in colognes. Too much and it overwhelms, but the right dose adds depth. Brands in personal care and household cleaning use it to make products stand out, especially when aiming for unique signature scents.
Industrial and Farm Uses
Farmers see value in isovaleric acid as a feed additive. Ruminant animals, like cows and goats, actually produce it naturally during digestion. Researchers figured out that supplementing animal diets with this acid sometimes helps with rumen health and growth. I grew up around dairy operations; old-timers knew the distinct barnyard aroma hinted at healthy fermentation, something key to a functioning farm.
Production scales up for industrial needs. Manufacturers use isovaleric acid as a starting point to make plasticizers, which keep flexible plastics from cracking or breaking down too quickly. Paints, glues, and coatings often rely on these compounds for stability and durability. That bottle of super glue or bright paint on the wall likely owes part of its flexibility to something no one wants to sniff directly.
Health and Science
Research labs tangle with isovaleric acid in more ways than one. This acid looks like a simple molecule, though its production inside the human body signals certain rare conditions. I remember poring over case studies during biochemistry courses: patients with "isovaleric acidemia" stood out. Their bodies struggled to break down the amino acid leucine, leading to a build-up of this acid and serious health effects if untreated. Science teams developed diagnostics that detect this acid, which now help alert doctors before the condition turns dangerous. The medical field needs to grasp these connections to prevent disability and guide life-saving treatment.
Potential Hazards and Responsible Handling
Long use in labs and factories taught me a lot about respecting isovaleric acid’s hazards. Skin and eye contact burn, and inhaling fumes gives headaches fast. Regulators pay close attention to safe handling and storage because nobody wants an accidental spill in a warehouse or school. Labeling, ventilation, and proper disposal keep people protected, while ongoing research looks for ways to use safer substitutes wherever possible.
For a substance that often stays hidden behind stronger scents and fancier packaging, isovaleric acid plays a big role—from cheeseboards to chemistry labs. Its value comes from both what it adds to our lives and what it reveals about health and safety. Honesty about its upsides and downsides has lasting importance in the real world.
Is Isovaleric Acid safe to handle?
Getting Real with Isovaleric Acid
Bottles labeled “Isovaleric Acid” end up tucked away in chemistry labs, fragrance labs, even some specialty food companies. The moment that cap twists off, though, the air lets everyone know. This stuff packs a punch—like overripe cheese with a sweaty gym sock finish. Even before you read a safety label, your nose warns you.
During my days doing research, I once watched a seasoned tech nearly gag after a waft caught him off guard. That wasn’t a one-off. Smell alerts us right away to take precautions. Still, reputation alone doesn’t answer whether this chemical belongs in the “safe” or “risky” bin.
Health Risks Aren’t Guesswork
There’s little mystery about what isovaleric acid can do if left unchecked. Scientific journals mention it triggers eye, skin, and respiratory irritation at surprisingly lower concentrations than many assume. I’ve witnessed colleagues get rashes just from a splash. One quick slip with an uncapped vial can turn a clean lab into a no-go zone till the air clears. Inhalation? Noses burn, eyes water, throats tighten. That’s just from an accidental whiff, not a long-term or high-volume situation.
It’s not simply “handle with care” and forget. Guidelines published by the National Institute for Occupational Safety and Health (NIOSH) treat exposure seriously, especially repeated or prolonged contact. Over the years, case studies reported headaches and nausea after handling isovaleric acid without proper gloves or mask. The chemical’s strong odor can even lull handlers into thinking odor alone equals danger—fact is, you can be at risk even if the smell isn’t overwhelming after noses adapt.
The Everyday Safety Steps
No chemistry degree is needed to respect what isovaleric acid can do. Gloves—preferably nitrile—should always go on before picking up a bottle. Goggles keep that pesky vapor out of your eyes, even if you’re only pouring a few drops. Working under a fume hood pulls odors and vapors away before they float through the whole room. One time a friend forgot to use a hood; he spent the rest of the afternoon outside, gulping fresh air, face red and eyes streaming.
Spills do happen. Absorbent pads, ventilated space, and knowing exactly where the nearest eyewash is can make the difference. Even on small teams, we made cleanup drills routine, so nobody froze up if a mishap happened. Handwashing doesn’t just “remove contamination”—it can stop days of skin irritation.
Fixing the Culture: More Than Just Rules
Rules alone don’t make a workspace safe. I once watched a new team member, confident after years working with bleach and ammonia, assume isovaleric acid would be much the same as household chemicals. He learned the hard way that the stakes get higher with this acid. Safety training matters not because people can’t read labels, but because real-world habits, like forgetting to double-bag waste or leaving residue on bench tops, create hidden hazards.
Manufacturers sometimes sugarcoat the risks in product sheets, focusing on industrial utility. In reality, those handling any volatile, pungent acid should gear up every time. Everyone appreciates coming home headache-free, with skin unburned and lungs clear. Experience teaches that every shortcut tempts fate. Regular checks, real practice, and open talk about close calls keep teams safer than the thickest manual.
Moving Forward: Safety as a Shared Language
If we’re honest, managing isovaleric acid boils down to treating it with the respect it earns. Smell, sting, rashes—these aren’t just anecdotes. Backed by decades of health studies, they serve as reminders. Safer handling means staying attentive, keeping barriers in place, and making sure every new person gets mentored by those who’ve seen what can go wrong. Not the stuff of headlines, but the reason any lab runs without a hitch at the end of the day.
What are the main applications of Isovaleric Acid?
What Drives Industry Toward Isovaleric Acid?
People tend to notice smells they can’t quite place. Isovaleric acid has one of those unmistakable odors: strong, even cheesy, sometimes compared to old socks. This offbeat trait suggests one interesting truth—it rarely slips under the radar in any process where it shows up. Over time, industries have spotted its value, especially in places you might never expect.
Flavor, Fragrance, and Food
In the world of food technology, flavors matter a lot. Isovaleric acid gets a spot on the list of ingredients, not for its original scent, but as a building block for more appetizing flavors. Chemists tweak it to make esters such as isovaleryl acetate. These esters impersonate apple and fruity notes in candy, gum, baked goods, and beverages. Even the world’s biggest flavor manufacturers lean on isovaleric acid derivatives to round out fruit blends, especially for synthetic flavors where real fruit may fall short due to cost or shelf life. Here’s the thing—I grew up in a household where home-baked goods came together with just a little store-bought magic, and there’s a high chance some of that flavor spark relied on workhorses like isovaleric acid. The compound doesn’t always get applause, but it anchors plenty of the aromas people take for granted.
Animal Health and Feed
Farmers have a long-standing relationship with feed improvement. A cow or pig that eats well tends to grow well, plain and simple. Feed formulators turn to isovaleric acid to make animal feeds more palatable. It also acts as a cue to animals, especially calves, mimicking scents they associate with comfort. There’s research from agricultural colleges that points to improved feed intake and digestion. In many barns, even a small amount added to feed can mean better appetite and smoother transitions during weaning.
Pharmaceuticals and Medical Uses
Isovaleric acid sits right in the mix for drug development and intermediate production. It plays a role in synthesizing medications and supplements, including the vitamin B complex. Some companies use it as a starting-material for anticonvulsant drugs. Lab suppliers keep steady demand to support pilot projects and full-scale production. Trace any bottle of vitamin B12 or specialty anticonvulsants, and a behind-the-scenes process likely leaned on isovaleric acid along the way.
Industrial and Chemical Synthesis
Working in a lab, chemical suppliers teach you: there are no minor chemicals. Isovaleric acid is a critical intermediate for producing plasticizers, additives, and specialty polymers. Chemical engineers seek out compounds like this to help threads and fibers in textiles, coatings, and plastics reach performance specifications for toughness and flexibility. These advances don’t reach headlines but keep factories moving and products durable. From experience, seemingly unremarkable chemicals can make or break a production schedule.
Improving the Approach: Managing Cons and Risks
No one wants strong, unwelcome odors or accidental exposures, so companies handle isovaleric acid with real care. The right engineering controls can cut back emissions, and manufacturers have to stick to environmental guidelines. People interested in sustainability keep pressure on for greener production, and some companies have started to explore bio-based processes. By paying attention to worker safety, refining synthesis, and pushing for renewable sources, industry can tap the benefits without the environmental backlash.
What are the storage requirements for Isovaleric Acid?
What’s in a Smell? The Story Behind Isovaleric Acid
Most folks might not recognize the name isovaleric acid, but the sharp, cheesy odor leaves a lasting impression. A clear liquid in the lab, this compound pops up in everything from flavoring agents and fragrances to organic synthesis. It’s useful, but the real story kicks in with its storage for anyone handling it in industry, research, or logistics. Safety here is not just a checklist—mistakes can sour workspaces, threaten health, and set off headaches nobody needs.
The Real Hazards: More Than Just a Funky Smell
Spilling a little isovaleric acid means more than just plugging your nose. High concentrations bother the skin, eyes, and lungs. Left uncapped, vapors drift fast and irritate like no tomorrow. With flammability rating at the same level as gasoline, storing this stuff in just any cupboard risks fires and chemical burns, especially if heat or open flames show up nearby. Data sheets flag isovaleric acid as corrosive in some situations. I’ve seen firsthand how failing to seal old bottles led to sticky, leaky messes that clogged ductwork and forced lab shutdowns.
No-Nonsense Storage Steps Anyone Can Follow
Glass or high-quality plastic, both with tight-fitting, chemical-resistant lids, outdo ordinary containers in keeping isovaleric acid in check. The sweet spot for storage settles at moderate temperature—preferably below ambient room temp and far from heat sources. Cooling isn’t just about shelf life; it means vapor builds up much slower, keeping the smell down and fire risk lower. Well-marked flammable cabinets, approved for organic acids, mean you’re not cutting corners on regulations. Frequent checks—at least monthly—catch any drips early and keep the workplace clean. Nobody likes surprises where acid pools around the feet.
Keeps Close, Store Alone: Smart Separation
Strong bases, oxidizers, and strong reducing agents have no business bunking next to isovaleric acid. Putting opposites together causes heat, fumes, or worse—reactive fires. In workplace safety training, keeping acids and incompatible chemicals apart ranks above all else. I always liked the three-color system: acids in one, bases in the other, and oxidizers somewhere separate. Easy labels mean nobody guesses what’s in a container during busy shifts.
Ventilation and Emergency Gear: Non-Negotiables
Anyone working with bottles of isovaleric acid knows one whiff can clear a room. Decent ventilation matters more than fancy filters. Storing bottles inside vented cabinets promises the workspace never turns into a sour, eye-watering mess. Spill kits, full-face shields, and gloves hang nearby so cleanup goes smoothly. Sharpening up on spill drills pays off. Fire extinguishers close to storage and a clear path for emergency exits protect teams better than any written policy.
Training and Oversight: Real Safety Starts with People
Straight talk counts in chemical safety. Teams that cut corners with labels or inspections will eventually pay for it. Regular walk-throughs spot cracked lids, faded labels, or sneaky leaks. Training everyone—not just lab techs—puts responsibility where it belongs. Too many accidents happen because a janitor or new staffer didn’t know the risks. In one company I worked for, mandatory safety sessions cut minor incidents in half over two years.
Storage Isn’t Optional—It’s Protection
Having handled dozens of acids over the years, I keep circling back to one thing: clear storage rules do more than tick regulatory boxes. They protect the folks doing the work, the building itself, and even neighbors downwind. Getting it right with isovaleric acid storage means work goes on, the air stays clean, and safety boards stay off your back.
What is the chemical formula of Isovaleric Acid?
Seeing Science in Everyday Life
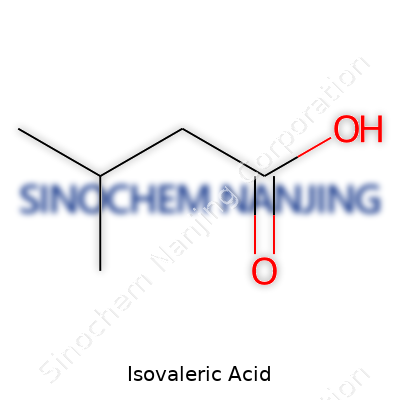
Ask most people about isovaleric acid, and you’ll probably get a puzzled look. Pull out a block of aged cheese or open a sweatsock gym bag, and that unique odor will spark recognition almost instantly. Isovaleric acid, known to chemists by its formula C5H10O2, lurks in the background of so many familiar places. But this small molecule carries a big impact in biology, industry, and even in how the human body works.
A Formula Behind the Funk
C5H10O2 might look like just another sequence of letters and numbers, but each atom in this lineup plays a role. As someone who’s dabbled in both home cheesemaking and backyard composting, that sharp smell has become a kind of signature. Isovaleric acid shows up as a metabolic byproduct in the body, too. Specifically, our muscles and nervous system break down the amino acid leucine, and isovaleric acid is one of the leftovers. People dealing with isovaleric acidemia, a rare metabolic disorder, know this compound on a much deeper level. Their bodies struggle to process leucine, resulting in dangerous buildups. Without medical attention, the effects can turn serious quickly. Like other chemical imbalances, this one doesn’t discriminate—it can hit infants and leave families desperate for answers. Thanks to simple blood and urine tests, most newborn screenings in many parts of the world catch the problem before it spirals.
A Place Beyond the Laboratory
Throughout history, folks have learned to put isovaleric acid’s unique chemistry to work. In the perfume business, it pops up as a building block, joined to other molecules to create more pleasant aromas. For the cheese world, especially hard cheeses like parmesan, the acid adds that distinctive punch. In my own kitchen experiments, trying to mimic those aged, earthy flavors, getting control over fermentation came down to wrangling bacteria that make isovaleric acid. There’s a difference between complex, savory notes and feet-smell disaster, and it all comes back to how much of this chemical grows during the ripening process.
Health and Environment: Connections That Matter
Science isn’t just about names and formulas. It’s about understanding risk, opportunity, and the long tail of even the simplest compound. Isovaleric acid works its way into wastewater from food processing, breweries, and tanneries. If not properly managed, small molecules can become big environmental headaches, particularly in communities where residents already face odor pollution. Some wastewater plants have started using advanced microbial digestion to break down organic acids before they ever reach homes or rivers. Those approaches carry promise, but public oversight and funding lag behind. People who live near factories tell a common story—there’s often little recourse when odors hit hard. Local governments could step up with stronger regulations and more transparent monitoring.
Moving Beyond the Smell
People talk a lot about innovation, but it’s the everyday chemistry like isovaleric acid that shapes health, food, and environment in ways we all feel. As a curious cook and lifelong science nerd, I see these molecules not as villains, but as reminders of how much biology and chemistry intersect. Paying attention at every step—from industrial release to consumer use to medical detection—protects both communities and future generations.
| Names | |
| Preferred IUPAC name | 3-methylbutanoic acid |
| Other names |
3-Methylbutanoic acid Isopentanoic acid Beta-Methylbutyric acid 3-Methylbutyric acid |
| Pronunciation | /ˌaɪ.soʊ.vəˈlɪr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 503-74-2 |
| 3D model (JSmol) | `Isovaleric Acid` JSmol 3D model string: ``` Isovaleric Acid CC(C)CC(=O)O ``` |
| Beilstein Reference | 1306967 |
| ChEBI | CHEBI:28842 |
| ChEMBL | CHEBI:30803 |
| ChemSpider | 6969 |
| DrugBank | DB03426 |
| ECHA InfoCard | 100.003.779 |
| EC Number | 3.2.1.50 |
| Gmelin Reference | 878 |
| KEGG | C00422 |
| MeSH | D007374 |
| PubChem CID | 7900 |
| RTECS number | NC9100000 |
| UNII | 1F8W8C0C7W |
| UN number | UN2348 |
| Properties | |
| Chemical formula | C5H10O2 |
| Molar mass | 102.13 g/mol |
| Appearance | Colorless to pale yellow liquid with a strong, unpleasant odor |
| Odor | Unpleasant, cheesy, rancid |
| Density | 0.93 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 1.39 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 4.84 |
| Basicity (pKb) | pKb: 11.10 |
| Magnetic susceptibility (χ) | -45.3e-6 cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 2.03 mPa·s (25 °C) |
| Dipole moment | 1.689 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 298.15 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -486.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2408.7 kJ/mol |
| Pharmacology | |
| ATC code | A16AX04 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H314 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P330, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-0-A |
| Flash point | 54 °C |
| Autoignition temperature | 451°C |
| Explosive limits | 1.1–7% |
| Lethal dose or concentration | LD50 (oral, rat): 650 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1,150 mg/kg |
| NIOSH | WA0450000 |
| PEL (Permissible) | PEL: 3 mg/m³ |
| REL (Recommended) | Recommended: 0.01% |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Isobutyric acid Valeric acid Pivalic acid Butyric acid 3-Methylbutanoic acid |

