Isophthalic Acid: More Than a Building Block
Changes Over Time: The Backstory of Isophthalic Acid
Looking back, the story of isophthalic acid starts in the late 1800s, a period when chemistry was cracking open secrets of the benzene ring. Early chemists, searching for new ways to harness coal tar and byproduct streams, stumbled on aromatic acids with three possible arrangements—ortho, meta, and para. Isophthalic acid, the “meta” version, sat in the shadow of its cousin terephthalic acid for years, not because it lacked promise but because the world’s appetite for PET plastics wasn’t on anyone’s radar. As industry moved further into the twentieth century, the need for materials that could take a beating and stay reliable changed the stakes. Isophthalic acid, produced from oxidation of meta-xylene, earned its keep in the polyester boom. As a polymer chemist, I’ve watched how shifts in market demand breathe new life into forgotten molecules. A substance that once filled chemistry books as a curiosity now has a seat at the table when industries seek better resins and coatings.
Getting to Know Isophthalic Acid: Features and Formation
Isophthalic acid doesn’t jump out as exotic. It’s a white, powdery solid at room temperature, with a sharp edge—barely soluble in cold water, a little better in hot, and much more comfortable in organic solvents. Its melting point clocks in above 340°C, a temperature that lets processing lines run with confidence that the compound will hold up under heat. The scent—slightly bitter, a reminder of lab days past—carries a signal of purity. Chemically, we see two carboxylic acid groups sitting at the 1 and 3 positions on the benzene ring. This layout gives isophthalic acid a distinct personality compared to its siblings; it won’t form the same rigid polymers as terephthalic acid, but instead brings flexibility and toughness to a blend. From epoxy resins in pipes to PET bottles for drinks, the world bumps up against this molecule daily.
The Lab and the Line: Making Isophthalic Acid
Most of the isophthalic acid in the world starts out as meta-xylene. Air oxidation in an acetic acid medium, with cobalt or manganese as the catalyst, brings those methyl groups up to full carboxylate status. Over the years, tweaks in catalyst systems have cut energy use and slowed down unwanted side reactions, like burning off valuable feedstocks or generating nonrecoverable byproducts. If you’ve spent time running a reactor, you know the battle with selectivity and yield is never fully over. This process plays by similar rules as the one used for terephthalic acid, but going after the “meta” isomer instead. After the reaction, the team needs to cool things down, filter out the solid acid, and wash it clean of trace metals. It amazes me how a handful of variables—temperature, catalyst loading, oxygen rates—determine whether a multimillion-dollar plant operates profitably or limps along.
Names and Language in the Trade
People in labs and plants toss around other names for this molecule: meta-benzenedicarboxylic acid, 1,3-benzenedicarboxylic acid, or even m-phthalic acid, depending on the chemist’s habit. Sometimes, product lines label it as IPA, which has led to confusion for folks thinking of the alcohol. If you’ve tried tracking shipments, this tangle of names becomes a headache fast—clarity will always beat tradition in these cases.
Staying Safe: Handling and Industry Rules
Isophthalic acid isn’t explosive or highly flammable, yet working with it gets messy if you let fine dust drift through the air. Respiratory irritation is common during handling. In the plant, gloves, dust masks, and air extractors stand between operators and itchy skin or lungs. International safety bodies offer guidelines for workplace exposure, storage, and waste disposal. I’ve seen companies invest heavily in dust collection and personal protective equipment once a routine health survey turned up mild but persistent irritation among staff. Even with modest toxicity, responsible operations make a difference to worker health over long careers. Water management matters too; keeping isophthalic acid out of wastewater streams avoids headaches with permit breeches and environmental agencies.
Beyond Simple Use: Chemical Reactions and Customization
In a chemist’s hands, isophthalic acid acts as a versatile starting point. The carboxyl groups stand ready for reactions: esterification with alcohols, amide formation with amines, the whole set familiar from organic synthesis. Polymers made from isophthalic acid gain resistance to water and chemicals, turning up in anything from polyester resins for boats to plasticizer-free PET films for food packaging. For those developing barrier coatings for cans or pipes, experimenting with isophthalic acid derivatives often leads to better compromise between flexibility and toughness. Creating copolymers with different blends gives physical properties tailored for specific mechanical needs—a trick that polymer researchers use to solve sticky problems involving strength, clarity, and heat resistance.
Places Where Isophthalic Acid Makes a Difference
Think of the hundreds of miles of water pipelines built over the past thirty years. Many survive because of isophthalic acid-based resins that resist corrosion, making failures less common and water losses lower. PET bottles for food and drink use require durability through filling, shipping, and shelf-life. Isophthalic acid shifts polymer properties, improving clarity and impact resistance, allowing for lighter and more attractive bottles. In the electronics and electrical sector, epoxies containing isophthalic acid offer insulation and reliability under thermal stress. Paint producers reach for isophthalic acid to boost the weather resistance of coatings, giving structures and cars longer lives without peeling or chalking. My work with paints taught me that slight tweaks in raw material sourcing ripple through to end-use, and choosing or substituting isophthalic acid often comes after rounds of accelerated weather testing in the real world.
Digging Into Hazards: Toxicity and Environmental Questions
Toxicological research puts isophthalic acid far from the most dangerous industrial chemicals, but modern safety standards demand more than just “not acutely toxic.” Lab animals tolerate high doses, showing few adverse effects at levels way above those encountered in normal use. Still, as regulators push for more data on long-term, low-level exposure—especially with microplastics carrying trace residues—researchers continue to look for subtle risks. Workplace studies haven’t turned up cancer concerns, but reports of mild skin and respiratory irritation keep surfacing in the literature and in worker surveys. Environmental science teams run degradation studies, watching how wastewater treatment and natural processes break down isophthalic acid. Most findings assure us that biodegradation, while not rapid, keeps environmental accumulation low if industrial releases stay within legal limits. I see researchers’ attention now shifting to how these breakdown pathways might intersect with other pollutants—big questions still linger, especially as plastic waste becomes a flashpoint.
The Next Stage: Future Uses and Ongoing Work
Changing consumer habits and climate pressures create a new canvas for isophthalic acid. PET recycling, which once ignored subtle differences between grades, now examines the mix of terephthalic and isophthalic-based bottles. High-end packaging and automotive composites demand strength and barrier properties, and manufacturers experiment with recycled content to meet environmental targets. Green chemistry initiatives, looking to drop petrochemical feedstocks, have worked up pilot plants using lignin- or sugar-derived aromatics as a route to isophthalic acid, but these processes still struggle to match traditional economics. The call for sustainable materials pushes R&D teams to rethink how aromatic acids fit into bio-based plastics and low-carbon supply chains. I’ve watched university groups chase new catalyst systems as they chase both lower energy use and better waste handling in manufacturing. As worldwide demand for lightweight, durable materials grows, isophthalic acid’s role will keep growing, not just as a “filler” but as a molecule with a real impact on design, safety, and environmental performance.
What is isophthalic acid used for?
A Building Block With Real-World Reach
If you’ve ever held a clear water bottle, worked in a kitchen with shiny countertops, or fixed up a boat with fiberglass, you’ve probably come across products touched by isophthalic acid. This chemical offers more than just a complicated name; it acts as one of those behind-the-scenes helpers that improves so many things people use every day.
Plastic Bottles and Packaging
Most folks know PET plastic bottles, but fewer realize that isophthalic acid gives those bottles their clarity and toughness. PET (polyethylene terephthalate) mainly relies on terephthalic acid, but by mixing in isophthalic acid, manufacturers get stronger and clearer bottles. The material resists breaking and cracks, which matters when you drop your reusable water bottle on the sidewalk or expect your milk jug to last in the fridge. These changes don’t just help packaging look better; they also cut down on leaks and spoilage, which helps reduce food waste.
Better Resins for Everyday Durability
Fiberglass boats, car parts, and some home surfaces all count on isophthalic acid for strong, flexible resin. Polyester resins made with this acid fight off moisture and chemicals more than older options. As a result, they last longer out at sea, on the road, or in kitchen countertops that see daily chopping and spills. It’s not about flash but about parts and surfaces that don’t crack under stress.
Paints and Coatings Standing up to The Elements
Makers of industrial and household paints chase coatings that look good and stay put, despite rain, heat, or cleaning. Isophthalic acid steps into this market as a toughener. It helps resins hold pigments and keep corrosion out, so buildings, bridges, and household appliances stay sharp and rust-free for longer. This kind of protection saves money. No one wants to repaint a warehouse every year, and rusted-out structures eat into municipal budgets fast.
What About Health and Safety?
People working in chemical plants handle isophthalic acid in large quantities, so safe handling and air filtration matter. Strict rules kick in at this stage to limit skin contact and inhalation. For most consumers, the biggest concern comes from microplastics—a topic getting much louder. PET bottles using isophthalic acid, when not recycled, often end up in landfills, rivers, and oceans. Small bits slowly break down and make their way up the food chain.
Paths Toward Responsible Use
Recycling stands out as the most immediate answer. Companies and communities benefit by collecting, sorting, and reprocessing PET bottles. Redesigning products so they are easier to recycle also helps. Lab teams poke at ways to create bio-based isophthalic acid, which would shift away from oil and cut the environmental impact. A bigger step comes from using less single-use packaging in the first place. Simple habits—reusing bottles, choosing glass or metal over plastic—pay off in slowing demand for virgin PET.
Everyday Chemistry’s Quiet Role
The impact of isophthalic acid lands quietly, but it makes millions of products stronger and longer lasting. Paying attention to where these materials end up, and looking for ways to cut waste, holds the key to balancing convenience with cleaner rivers, oceans, and communities.
Is isophthalic acid safe to handle?
Getting to Know Isophthalic Acid
Isophthalic acid lands in many factories that make plastics, paints, and some fibers. Checking the label, you see a white, powdery substance. Most folks on the outside will never cross its path, but in the world of chemical manufacturing, it’s everywhere.
A Real-World Look at Risk
Looking at safety data, isophthalic acid brings some risk. Breathing the dust can irritate lungs, nose, and throat. Getting it on your skin sometimes causes redness or swelling. If it finds its way into your eyes, you’ll feel a sting and maybe some burning. Nobody looks forward to that kind of day. Some of the discomforts fade after you leave the space or wash it off, but regular unprotected exposure raises the stakes. I’ve talked to folks in production—most remember one time someone let their mask slip or didn’t wear gloves. The rule: respect the powder, don’t get lazy.
Long-Term Health Concerns
There aren’t signs that isophthalic acid gives you cancer or creates permanent harm at the levels people usually see in a factory. The International Agency for Research on Cancer doesn't list isophthalic acid as a carcinogen. But that doesn’t mean shrugging it off. Repeated mishandling causes chronic irritation, and with enough lapses over a career, you could run into trouble. Respiratory issues or skin conditions never help anybody’s quality of life.
Worker Safety: The Everyday Routine
Factories that get isophthalic acid shipments almost all run tight safety plans. That means respirators, gloves, long sleeves, and goggles—plus good training. People working with chemicals trust simple routines: don’t sweep dust with hands, keep eating and drinking out of the work zone, wash up before lunch, fix leaks as soon as they appear. Management usually carries out air and wipe testing now and then to keep track of any buildup. Turning safety drills into habit keeps the accident rate low. In my experience, personal vigilance sometimes works even better than fancy gear—knowing what could go wrong for real, not just on a sign, sticks in your mind.
The Environmental Footprint
Isophthalic acid ends up in a lot of waste streams, so cleanup matters. Nobody wants it going down the drain and into rivers. It can harm aquatic life if large spills occur, so chemical plants put money into wastewater treatment and spill containment. I’ve seen cases where a sloppy loading dock cleanup led to neighborhood concern. Most towns don’t want extra chemicals in their stormwater runoff. Being careful with handling and disposal isn’t just about workers—it keeps the area around plants healthy too.
Better Solutions and Responsible Choices
Chemists always look for safer alternatives, but for now isophthalic acid remains tough to substitute in certain industries. The solution leans on solid education for workers, reliable protective equipment, and honest reporting of near-misses and issues. Clear labels and instructions help, especially for new team members, and regular audits keep things up to scratch. In my time around manufacturing sites, a culture where people ask questions and stop unsafe behavior before it gets out of hand works the best. Everybody on the floor or in the lab keeps each other safe, and no chemical, big or small, bypasses that teamwork.
What are the chemical properties of isophthalic acid?
What Makes Isophthalic Acid Stand Out
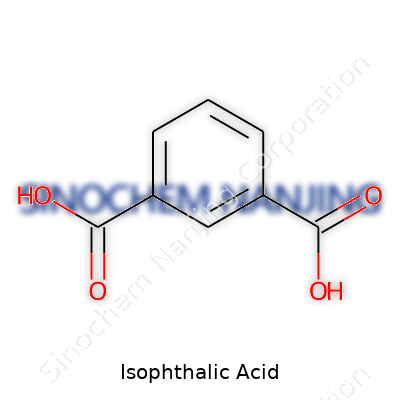
Isophthalic acid represents an important building block for many industrial products. Its formula, C8H6O4, shows it contains a benzene ring with two carboxylic acid groups sitting opposite each other. Anyone who’s spent time studying aromatic chemistry knows that structure really shapes how a compound reacts. These acid groups give it a fairly strong acidic character for an aromatic compound, though it’s not quite as punchy as the more famous benzoic acid. That little tweak in layout goes a long way towards setting its role in polyester, resin, and plastic production.
Acidity and Solubility
If you measure the pKa of isophthalic acid, you’ll find it sits around 3.5 for the first proton, which is a touch weaker than strong mineral acids but enough to participate in neutralization and precipitation reactions. Drop some of this powder into water, and it’s only slightly soluble at room temperature. The real action kicks in with heat – solubility jumps as you raise the temperature, which manufacturers leverage during purification and mixing. Toss it into organic solvents like acetone or alcohol, and you can expect much better dissolution compared to other similar acids.
Reactivity in Everyday Processes
Science students sometimes glaze over carboxylic acids, but in a production line, those acidic groups play a big role in making tough plastics or strong resins. Isophthalic acid reacts easily with alcohols to form esters in the presence of acid catalysts. That’s a workhorse reaction in polyester synthesis — think PET bottles, not just soda containers but countless packaging and textile uses as well. The presence of two acid groups opposite each other in the aromatic ring lets it create linear, rigid structures. These structures mean higher strength and better chemical resistance in resulting plastics compared to simpler alternatives.
Safety and Handling
Anyone handling this stuff should take it seriously. Isophthalic acid powder can irritate your skin, eyes, and lungs. Good lab or factory practice always calls for gloves and safety glasses. Though its immediate toxicity is low, workers in large plants deserve steady ventilation and regular training. It’s easy to overlook proper storage, but keeping isophthalic acid dry and away from incompatible chemicals like strong bases prevents clumping and unwanted reactions.
Role in Environmental Sustainability
Polymers and resins based on isophthalic acid turn up everywhere–from water pipes to food-grade films. Industries use it because those ester linkages hold up against heat, cleaning agents, and sunlight. There’s a challenge, though: breaking down products after their useful life. Research teams keep chasing more efficient recycling and upcycling of polyesters that use this acid. Catalytic depolymerization and more selective hydrolysis processes could change the lifecycle of these plastics, cutting waste and reducing dependency on raw petrochemicals.
Looking to the Future
Chemists continue to explore greener synthesis and improved recovery for isophthalic acid. Using renewable feedstocks rather than fossil-based benzene offers a way for manufacturers to lower their carbon footprint. Lab-scale breakthroughs often face roadblocks at industrial scale, but sustained research commitment can yield cleaner processes year by year.
How is isophthalic acid different from terephthalic acid?
Digging Into the Chemistry
You don’t have to spend years in a lab to see the difference between these two acids. Isophthalic acid and terephthalic acid both start with the same basic building block: the benzene ring, a hexagon made of carbon and hydrogen. Take a closer look and you’ll spot the small shift that changes everything: The carboxyl groups attach to different spots on that ring. In isophthalic acid, they land on carbons one and three; terephthalic acid pairs them on one and four. That small tweak leads to big changes in their behavior.
Why Small Changes Matter for Everyday Plastics
My first stint at a polymer plant showed me how a shift in molecular structure changes the finished product. Anyone who’s ever carried a soft drink in a plastic bottle probably touched something made from terephthalic acid. PET plastic—short for polyethylene terephthalate—relies on that one-four bond to give the chains rigidity and strength. Factories use terephthalic acid to keep bottles from collapsing under pressure.
Now, put isophthalic acid into the mix and the situation changes. The one-three placement bends the plastic chains, making them less likely to line up and pack tightly. Suddenly, plastics get more flexible, less crystalline. This means the final product can fend off water better and can resist chemicals that would normally eat through tougher, but more brittle, alternatives. Water bottles stick with terephthalic acid because nobody wants a soggy bag at the bottom of their backpack. Resin coatings for food cans or tough car paints? That’s where isophthalic acid starts to shine.
Price, Supply, and the Real-World Impact
Factories worldwide lean hard on terephthalic acid, mostly because it’s turned into PET efficiently at a giant scale. It flows through supply chains in every corner of the world. Any dip in crude oil cost or shipping price ripples through everything from snack wrappers to carpet fibers. Isophthalic acid production hits smaller markets, which gives it a higher price tag but also an edge in specialty areas. People in resin production can tweak formulas using isophthalic acid for that premium mix of durability and flexibility.
Environmental Headaches—and Possible Ways Forward
If we talk about what these acids mean for the world outside the lab, we bump into bigger challenges. Both come from petroleum roots, and that drives up concerns about resource use and pollution. In my experience talking with recycling outfits, plastics made from terephthalic acid stand out—they’re easier to recycle. PET bottles cycle through facilities across the globe. Try that with isophthalic acid blends, and the process gets complicated, with coatings and resins often ending up in landfills instead of new products.
Scientists keep searching for greener feedstocks, aiming to shift both acids away from oil. Some startups experiment with plant-based routes. For now, these options cost more and work at a smaller scale, but with demand for recycled materials growing, there’s hope that one day “bioplastic” means more than a buzzword. If manufacturers and policy makers insist on better standards, and if global markets support those choices, maybe one day you’ll grab a drink from a bottle that doesn’t lean so hard on fossil fuels.
What industries commonly use isophthalic acid?
Key Ingredient in Plastics Manufacturing
Ask anyone with a background in materials science, and they’ll tell you that isophthalic acid stands out as an important building block for making plastics tougher. Polyethylene terephthalate, or PET, shows up in food and drink bottles, packaging, and even synthetic fibers. Mixing isophthalic acid into PET production brings an extra punch of strength, keeps bottles from warping with heat, and helps resist stains. Companies run cost-benefit analyses every year and find these improvements are worth it—something I've observed in recycling plants, where stronger PET bottles hold up better through multiple washings.
Vital for Paints and Coatings
Walk into any paint shop and look at the wide range of protective coatings, glues, and paints. Behind the scenes, many rely on resins made with isophthalic acid, especially unsaturated polyester resins. As I found while working with a flooring contractor, these specialty resins keep floors shiny and protect them from chemical spills, abrasive wear, and moisture—important in hospitals and kitchens, places that never get much downtime for repairs. Boats and vehicles, too, benefit from coatings with isophthalic acid. You get fewer cracks from UV rays, more resistance to water, and lower long-term maintenance costs. These features play out in data: marinas regularly send feedback showing hulls last longer where these coatings are used.
Fiberglass-Reinforced Products
Not many people think about what goes into fiberglass pipes, storage tanks, or circuit boards, but isophthalic acid keeps these products reliable. Every time factories pressurize chemical reactors or route hot water through pipes, the polyester resin (made with isophthalic acid) works as an invisible shield. I remember troubleshooting an acid leak in a pulp mill. We traced it back to a tank built with outdated resins. After switching to composites made with isophthalic acid, leaks stopped, and downtime dropped. Plants end up saving money and headaches by choosing the right materials up front.
Emerging Role in Automotive and Construction Sectors
The car industry hunts for lighter, stronger materials, driven by emissions targets and safety goals. Isophthalic acid makes its mark here, appearing in lightweight panels and reinforced bumpers. Construction companies also favor concrete additives based on polycarboxylate, sourced partly from isophthalic acid. These additives improve flow and reduce the need for excess water in concrete mixes. Buildings go up faster, concrete holds up longer, and the environmental impact shrinks. Trade journals report building projects meeting “green” certifications more often now, as designers focus on safer chemicals and better performance—all while keeping costs in check.
Potential Solutions for Safer Use and Environmental Impact
With chemical manufacturing, safety and sustainability should lead the way. Isophthalic acid doesn’t escape scrutiny—plant managers must keep emissions low and treat wastewater with care. Some companies invest in closed-loop processes and advanced filtration, reducing pollution and making recycling more efficient. Academia and industry work together on research to create bio-based alternatives. These efforts point to a future where the industry keeps improving: less waste, fewer hazards, and new materials with the performance users demand.
Trust Built on Proven Performance
Over the years, seeing how reliable these products are in harsh conditions shapes trust across industries. From food packaging to chemical processing to modern bridges, isophthalic acid helps raise the standard for what’s possible—and keeps workers and consumers safer along the way.
| Names | |
| Preferred IUPAC name | benzene-1,3-dicarboxylic acid |
| Other names |
meta-Phthalic acid m-Phthalic acid benzene-1,3-dicarboxylic acid 1,3-benzenedicarboxylic acid |
| Pronunciation | /ˌaɪ.səʊˈfæl.tə.lɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 121-91-5 |
| 3D model (JSmol) | `Isophthalic Acid JSmol 3D model string`: ``` Isophthalic Acid C1=CC=C(C=C1C(=O)O)C(=O)O ``` |
| Beilstein Reference | Beilstein 1722083 |
| ChEBI | CHEBI:30702 |
| ChEMBL | CHEMBL41687 |
| ChemSpider | 17410 |
| DrugBank | DB03826 |
| ECHA InfoCard | 100.013.707 |
| EC Number | 204-506-4 |
| Gmelin Reference | 108011 |
| KEGG | C01882 |
| MeSH | D007553 |
| PubChem CID | 8028 |
| RTECS number | WN5050000 |
| UNII | 3KPG2I2KAP |
| UN number | UN2526 |
| Properties | |
| Chemical formula | C8H6O4 |
| Molar mass | 166.13 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.53 g/cm³ |
| Solubility in water | 0.621 g/L (25 °C) |
| log P | 0.68 |
| Vapor pressure | 0.0000125 mmHg (25 °C) |
| Acidity (pKa) | 3.46 |
| Basicity (pKb) | 3.46 |
| Magnetic susceptibility (χ) | -68.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.635 |
| Dipole moment | 1.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -634.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3226.0 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | ``` GHS07; Warning; H319; P264, P280, P305+P351+P338, P337+P313 ``` |
| Pictograms | GHS07,GHS05 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P260, P264, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362+P364 |
| Flash point | 220°C (closed cup) |
| Autoignition temperature | 685°C (1265°F) |
| Lethal dose or concentration | LD50 oral rat 4900 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat = 6000 mg/kg |
| NIOSH | KWJ0350000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | ISO 14001, OHSAS 18001 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Phthalic acid Terephthalic acid Trimellitic acid Benzene-1,2,3-tricarboxylic acid |

