A Close Look at Isophorone: More Than Just a Chemical Solvent
Tracing the Story: Historical Development
Isophorone didn’t show up overnight. Its roots stretch back to early twentieth-century chemistry, where the search for reliable solvents kept labs busy and creative. Back then, the boom in industrial paints, coatings, and adhesives called for compounds that could handle strong resins and deliver consistent performance. Through the decades, isophorone earned a place on factory floors and in R&D labs around the globe. Its journey mirrors broader shifts in chemical manufacturing and environmental concern, as new techniques promised better purity but also raised new questions about safety and sustainability. Early processes felt crude by today’s standards, yet they laid the groundwork for the multi-stage distillation and catalytic methods most producers rely on today. It’s impossible to separate isophorone’s past from the growth of the coatings industry and the growing awareness of workplace hazards.
What Isophorone Really Offers
Isophorone stands out as a solvent, but it does a lot more. It’s a clear, liquid cycloaliphatic ketone with a distinct, sometimes pungent, odor. Users appreciate its ability to dissolve tough polymers, from nitrocellulose to certain resins. They favor isophorone in paints, inks, and adhesives, largely because it brings time to dry and absorbs well in heavy-duty jobs. Unlike many other solvents, designers turn to isophorone when they need a balance of evaporation rate and solvency power. This versatility also sets it apart and helps boost its reputation, even as some applications move toward safer or greener alternatives. Isophorone moves easily between coatings, pesticides, and even intermediate use in chemical syntheses.
Physical and Chemical Profile
The physical and chemical properties of isophorone shape its value on the industrial stage. With a boiling point in the neighborhood of 215°C and a melting point just below freezing, it handles a wide temperature range. It’s less dense than water, and its moderate vapor pressure at room temperature makes storage concerns different from those of lighter ketones like acetone. Isophorone mixes with most organic solvents, which simplifies blending where product consistency matters. The molecule’s cyclohexyl backbone and a single ketone group change how it interacts with light, water, and reactive metals. This mix plays into both its utility and the challenges of limiting workplace exposure.
Technical Specs & Labeling: Why They Matter
Anyone working with isophorone knows labels are more than stickers. They speak to purity, water content, and the precise profile of trace contaminants. Precise specifications protect end-users from failed batches or inconsistent performance. Technical sheets often break out GC-purity levels above 98%, flag water content around 0.1%, and warn of peroxides or aldehyde byproducts. Regulators focus on these details for good reason: impurity levels can change how coatings cure, alter reactivity in syntheses, and affect human exposure. Compliance is not optional—it’s the baseline for doing business in many markets. Labeling also lays out transport and storage considerations, such as flammability ratings and UN numbers, to help technicians manage risks and inventory.
How It’s Made: Preparation Method
True to the ingenuity of industrial chemists, isophorone production lines rely on self-condensation reactions of acetone with alkaline catalysis, followed by fractional distillation. This process transforms a common building block into a specialty ingredient. Over time, facilities have fine-tuned this approach to reduce unwanted side products and recover solvents with higher selectivity. Waste handling, purification, and recovery add complexity at each stage, reflecting tighter regulations and environmental responsibility. Efforts to boost yield and reduce energy draw sit at the center of ongoing process improvements, as does the search for catalysts that permit lower temperatures and faster reactions.
Chemical Reactions and Modifications
Isophorone rarely stays as-is for long in the lab. Its backbone lets chemists perform hydrogenation to make isophorone diols—important for polyurethane manufacture. Under oxidizing conditions, it yields carboxylic acids or other functional groups, expanding its reach in specialty chemicals. The ring system holds up under moderate conditions but shows reactivity at higher temperatures or with concentrated acids, making it useful in targeted synthesis. In agrochemical work, isophorone derivatives support crop-protection chemistry and pesticide development. A structure like this gives versatility but demands careful control to avoid over-reaction or unwanted byproducts.
Name Game: Synonyms and Product Names
Depending on where you source information, isophorone might appear under several guises. Names like 3,5,5-trimethyl-2-cyclohexen-1-one and its various translations crop up in literature and safety data. Trade literature sometimes uses branded names, but the core chemistry doesn’t change. Recognizing technical synonyms avoids confusion between supply chain partners and streamlines regulatory paperwork. This reliance on synonyms reflects not just regulatory requirements across jurisdictions but industrial tradition as well.
Safety and Operational Know-How
Working hands-on with isophorone means paying close attention to safety protocol. Inhalation risks top the list; long hours in poorly ventilated spaces lead to headaches and mucous membrane irritation, and repeated exposure brings more serious risks. PPE guidance isn’t academic—it keeps people healthy. Gloves, goggles, and fume hoods become daily essentials. Fire risk shows up in storage guidelines, and spill response plans get tested by real-world accidents. Training programs tie these details together, reinforcing practical steps over theoretical advice. High-profile accidents from the past push companies to keep checklists updated and to consider engineering controls such as scrubbers and closed handling systems.
Where It’s Used: Application Territory
Ask the coatings and printing industries about isophorone and you’ll get strong opinions. Its power to dissolve tough binders gives it a niche in specialty paints and custom inks. Adhesive producers count on it for performance in high-stress environments. Some pesticide formulations bank on isophorone for its controlled volatility and compatibility with active ingredients. In polymer manufacturing, it operates as a precursor, shaping the properties of advanced synthetic materials. Ongoing efforts to develop waterborne coatings and reduce VOC emissions add pressure, yet isophorone keeps its place for applications where alternatives still fall short. Its persistence reflects more than habit—it’s functionality, performance, and a track record that anchors supply chains around the world.
Emerging Frontiers: Research and Development
R&D teams don’t sit still. Much of today’s research zooms in on reducing environmental impact and discovering greener pathways for isophorone production. Renewable feedstocks, such as bio-based acetone, draw investment and pilot trials in the hope of shrinking fossil fuel demand. Scientists keep pushing for catalysts that allow cleaner conversion and minor byproduct formation. Additionally, new uses arise as polymer chemistry advances and needs shift. Talent crunches and funding cycles slow momentum, but breakthroughs in catalysis or molecular modification could reshape both supply and demand for isophorone within the decade.
Toxicity: Watching the Shadows
Toxicity research digs deeper every year. Inhalation and skin contact top the list of worries—chronic exposure can hit the central nervous system, liver, and kidneys. Animal studies raise flags about long-term health and developmental effects. At the same time, workplace monitoring programs line up with guidance set forth by organizations like OSHA and the EPA. Biomonitoring in high-usage industries offers real-world insights beyond the bench, exposing areas where administrative controls or personal protection keep risk at bay. Substitution sits in the background as pressure from public health advocates and consumer groups grows. As new data come in, product labeling and workplace practice evolve, closing some risks but sometimes opening up new questions.
What’s Next: Looking Ahead
Future prospects of isophorone lie at a crossroads. Regulatory pressure on VOCs, worker safety, and industrial emissions steers innovation both in solvents and in how companies set up chemical processes. Some see promise in biotechnological routes or advanced catalysis to produce isophorone and its derivatives with fewer environmental costs. Developments in coatings, polymers, and agrochemicals could prolong its industrial relevance, but only if researchers solve key health and environmental puzzles. Market forces and regulatory trends will drive changes on the plant floor and in product labs, but isophorone’s adaptability gives it a chance to remain an active part of the chemical conversation for years to come.
What is Isophorone used for?
The Workhorse Chemical Behind the Scenes
I remember walking into a paint factory years ago and catching that harsh, almost peppery smell that lingers in the air. Someone told me that was isophorone. Back then, I didn’t give it much thought, but now, digging deeper into what it actually does, I’m surprised by how much this chemical shapes the things we use every day—without most folks ever knowing its name.
The Backbone of Durable Coatings
Isophorone is a clear liquid mostly made in big chemical plants. If you’ve ever driven over a freshly painted bridge, sat on a new park bench, or spilled nail polish on a café table, you’ve probably benefited from it. Paint and coating companies use isophorone to make products spread smoothly and dry without bumps or cracks. It works as a solvent—kind of like thinning soup so it covers more bread. Builders want their paint to last through storms and sunshine, and isophorone helps toughen up those layers.
Automotive factories rely on it for coatings that can take a beating from salt and sun. Even the aerospace industry turns to it for high-durability finishes. Isophorone helps paints stick better and look good longer.
Tougher Plastics and More Reliable Glues
Some of the toughest plastics owe their resilience to isophorone. Producers use it to make certain polymers—these end up in everything from computer casings to industrial pipes. It has a role in making adhesives, too. I remember fixing my shoes with a heavy-duty glue; turns out, there’s a fair chance isophorone made that glue flexible yet strong. It helps keep the glue workable until you stick things together. Then, once the solvent evaporates, the bond sets fast.
Ink That Stays Put
Printers and graphic shops often count on isophorone to get colors to stick well on labels, posters, and banners. It keeps inks from clumping up or separating in the can. This way, what rolls off the press matches the design on the computer—bright and sharp, not faded or smeared.
Health and Environmental Risks
Before anyone gets too comfortable, it’s important to talk safety. Breathing huge amounts of isophorone vapors can bother your nose, throat, or eyes. Most workers in factories handling chemicals like this know their way around safety goggles and proper ventilation because the long-term health risks are real. The Environmental Protection Agency keeps an eye on it, asking manufacturing plants to limit emissions and keep spills out of waterways. Isophorone doesn’t disappear overnight once it hits soil or water; it can hang around and possibly hurt aquatic life.
Looking Toward Cleaner Solutions
So, what’s next? Some companies now explore greener chemical substitutes—mostly plant-based solvents—for paints and glues that perform just as well. This transition isn’t simple. Traditional methods and supply chains lean heavily on proven chemicals, but the push for healthier air in factories and less pollution in rivers keeps growing. Switching out isophorone will take years, but it’s on the radar for many in the industry, especially as regulations tighten across the globe.
Isophorone won’t make headlines like new gadgets or celebrity trials, but its impact runs all through construction, manufacturing, and design. Next time you see a glossy finish or use a strong glue, remember the chemistry that’s been keeping things together—and the health of workers and the environment that needs a closer watch.
Is Isophorone hazardous to health?
What Isophorone Means for Everyday People
Many of us haven’t heard of isophorone, but some folks living near factories, paint shops, or printing plants might know its sharp smell. It shows up in paints, adhesives, and cleaning products. Years working in the automotive or coatings industries let me notice that certain solvents gave headaches and made people cough. Isophorone ranked among them.
The Experience on the Ground
Walk into a workshop using industrial cleaners and ask for the "good ventilation." There's a reason for those warning labels. Short-term exposure to isophorone’s vapors often leads to eye, nose, or throat irritation. That burning sensation and watery eyes aren’t just an annoyance, they’re a message from your body. In my own painting projects, I learned to respect these signs, as taking shortcuts with protective gear always meant regret.
Looking at the Scientific Facts
Multiple safety agencies study chemical hazards over years. The U.S. Environmental Protection Agency (EPA) and the National Institute for Occupational Safety and Health (NIOSH) recognize isophorone as an irritant. Extended contact can affect the lungs, liver, and kidneys. The International Agency for Research on Cancer has flagged it as possibly carcinogenic. Concerns mostly arise in workplaces, though pollution around certain factories has posed problems for local neighborhoods.
Chronic Exposure Adds Up
Long-term exposure draws the most concern. Folks working without adequate ventilation or skipping gloves and masks add risk year after year. Those headaches, dizziness, and tiredness might not be random. Studies involving workers exposed to high concentrations have noted liver and kidney changes, and animals develop certain cancers at much higher doses.
Neighbors living near waste sites or chemical plants have raised issues about nausea, dizziness, and chemical odors. Living in one industrial town, we had summer days where the air seemed heavier, especially downwind of plants. Some old-timers spoke of nosebleeds and asthma acting up. These stories match what the science says: even if isophorone isn’t as famous as lead or benzene, it deserves attention.
Taking Steps Toward Safety
Preventing exposure should matter both at work and at home. In workplaces, fans and proper masks do more than check a safety box—they protect real people. Ordinary gloves and eye protection show their worth after handling anything labeled with isophorone. Product labels deserve a closer look, not just a quick glance. In homes, good ventilation helps when using any strong-smelling cleaner or adhesive.
Communities near chemical plants need access to information about local air quality. Regular monitoring by city officials can bring peace of mind, or highlight bigger issues needing attention. Public records ought to be easy to find, and families should know who to call if strange odors appear. Over the years, pushing for tougher local regulations has helped some cities force companies to lower their air emissions.
Choosing the Safer Path
Many businesses now consider safer alternatives to harsh solvents. More workers report having safer products available and better tools for cleanup. This shows change happens, slowly but surely, with a mix of science, advocacy, and real conversations about risks. People deserve workplaces and neighborhoods free from hidden health threats. Taking isophorone’s risks seriously brings us closer to that goal.
What are the physical properties of Isophorone?
Understanding Isophorone in Daily Use
I’ve worked around chemicals for much of my career—sometimes in a lab, sometimes in manufacturing. One thing you pick up fast is that the feel and behavior of a chemical matter just as much as its formula. Isophorone, a clear liquid with a unique peppermint-like odor, has made its way into everything from inks to construction materials. You can’t forget its smell—it’s sharp, almost sweet, and definitely not something you mistake for water or alcohol.
Boiling and Melting Points That Shape Handling
Isophorone boils at 215°C. That’s a much higher temperature than most household solvents. So, in an industrial setting, it doesn’t just evaporate like acetone left open on a bench. Instead, it hangs around, which can be good for slow-drying applications like inks or coatings. You don’t see it freezing up in a cold warehouse, either. With a melting point near –8°C, it stays liquid even in chilly climates. Being able to handle it with some ease at room temperature means less risk of clogged pipes or sticky messes in gear lines.
Solubility That Drives Application
One reason isophorone lands in many different products comes down to its solubility. It mixes easily with ether, alcohol, and most other organic solvents, but not with water. This makes it a popular choice in paint and ink manufacturing because it can bring together ingredients that wouldn’t normally mix. Factories like it, too, since workers can count on a predictable performance from batch to batch when they rely on these physical traits.
Why Density and Viscosity Matter
Its density sits right around 0.92 g/cm³ at room temp, so it’s a little lighter than water. That matters when you’re storing and transporting large drums—it won’t weigh down a truck like heavier chemicals. Low viscosity adds another layer of convenience. It pours quickly, flows without much resistance, and cleans up easier than thick, sticky materials. In printing or laminating, this thinness helps coats to spread evenly and cover surfaces without streaks or bubbles.
Volatility and Odor—The Double-Edged Sword
Even though isophorone’s boiling point means it doesn’t flash away, it still releases vapors at room temperature thanks to its vapor pressure. On hot summer days in the shop, you can smell it get stronger. Workers notice it too. Plenty of studies confirm that high levels in the air can irritate eyes and noses. This sharp, distinct scent helps people catch leaks quickly. OSHA and other safety bodies advise air monitoring and strong ventilation in places where it’s used. There’s no way to overstate how important good airflow is when you have a substance that travels by air so easily.
Solutions for Safer Use
Having been around these kinds of chemicals, nothing beats simple practices for keeping people safe. Close lids right after pouring. Use gloves and goggles. Keep storage areas cool. Companies also need good air handling systems, not just cracked windows or a fan in the corner. For anyone cleaning up a spill, absorbent pads and proper disposal matter more than ever now that environmental focus continues to grow. It’s not just good for health; it keeps production moving without surprise shutdowns for leaks or accidents.
Final Thoughts on Handling Isophorone
I’ve seen companies thrive by respecting the quirks of materials like isophorone. By paying attention to physical properties, handling becomes smarter, work moves faster, and the health of everyone involved gets top priority. The way a chemical looks, smells, and flows makes all the difference in safe, efficient production. Staying informed, using solid safety practices, and listening to your senses keep the workplace safe and productive.
How should Isophorone be stored and handled?
What Makes Isophorone a Risky Material?
Ask anyone in a plant or lab about isophorone and a few things come up right away: sharp smell, skin and eye irritation, fire risk. I remember my own early years working with solvents—rules plastered on the wall, gloves that barely fit, goggles that fogged over. Isophorone never got special attention like some compounds, but a single spilled bottle tells you how sneaky it can be. This chemical, used in paints, inks, and adhesives, evaporates quickly and can turn into an inhalation hazard before you know it. Symptoms sneak up, often starting with a headache or dizziness. Data from the US National Library of Medicine point to possible liver and kidney effects with repeated exposure, which makes its storage and handling a frontline safety concern.
Building Smarter Storage Systems
I’ve seen the best results in places where storage rules get real attention. Metal safety cabinets, clearly labeled for flammable liquids, make a big difference. Plastic containers and regular shelving do not cut it—solvent-resistant materials matter, and fire separation isn’t up for debate. The temperature in your storage room should never creep up to the point where you can break a sweat. Data from OSHA shows that chemicals like isophorone, left in hot or direct sunlight, increase vapor pressure and fire risk. A basic thermometer on the wall acts as a first line of defense.
I once walked into a shop where the exhaust fan was barely running and the scent hit you right in the throat. Air movement has to carry fumes out fast and away from workspaces; using a fan that dumps air into the same room betrays a lack of urgency about workers’ lungs. Local building codes and guidance from the National Institute for Occupational Safety and Health (NIOSH) recommend explosion-proof fixtures and grounded containers for transfer, since static sparks like to show up when you least want them.
Everyday Habits and Personal Protective Equipment
People who work with chemicals every day sometimes get casual, and I understand the temptation. But gloves are not all equal—neoprene and butyl rubber guard better than bargain nitrile or latex. Face shields or goggles ought to become part of muscle memory, especially for those who mix or decant solvents regularly. Wash stations belong close to every spot where isophorone shows up. In a pinch, a water bottle can help, but only proper sinks get the job done after accidental splashes.
The key habits don’t change: never eat or drink in storage areas (a lesson reinforced by one too many lunchtime stories gone wrong), and work clothes should never follow you home. Safety Data Sheets (SDS) give a rundown on all recommended PPE, but nothing beats training that walks folks through real-life scenarios. Talking about the time your glove tore and you had to spend an hour at the sink stick with people more than policies printed on the back door.
Spill Response and Waste Disposal
Everyone gets caught off guard eventually. I remember the sting of a small leak spreading on the bench because I left a cap loose. Quick action makes the difference—spill kits with absorbents, neutralizers, and bags for containment belong near every working spot. Updates from the EPA highlight how spills, even small ones, can threaten water quality if not contained and cleaned properly.
Waste should go into sealed, marked containers. Leaving rags or spent absorbent open invites more vapors and possibly, fires. Some states, like California, treat solvent wastes as hazardous the moment they leave your workspace. Calling trained waste handlers keeps you within the law—and the fine print is clear: this isn’t regular trash.
Why Training and Experience Matter
In my experience, the best safety cultures treat chemicals like isophorone with respect, not fear, and demand clear communication. Supervisors model habits, but everyone plays a part. Regular drills and honest talk about mistakes help prevent repeats. Real know-how comes from doing things the right way, every day, not just after a safety inspection. Isophorone can be handled safely, but only if people’s experience gets valued, and every step is backed up by solid science and up-to-date data.
Is Isophorone environmentally safe?
What Isophorone Brings to the Table
Most people haven’t heard of isophorone unless they work with paints, adhesives, or coatings. Chemically speaking, isophorone comes from acetone and plays a big role as a solvent, especially where smooth finishes and flexible formulas help products work better. If you ever watched a painter mix a can of specialty paint, you saw isophorone at work.
Toxicity and Exposure: More Than an Academic Concern
The question isn’t whether isophorone does its job, but what happens while it’s in the air, water, or soil. I’ve handled plenty of solvents as a construction worker, and label warnings never sugarcoat the health side: headaches, nausea, dizziness. The Environmental Protection Agency put isophorone on its watchlist for good reason—animal studies found it causes liver and kidney damage at high doses. While that doesn’t spell instant danger from a single product, any chemical that causes harm with enough exposure can’t just get a “safe” sticker.
Isophorone in the Environment
If you spill isophorone, it doesn’t stick around the way oil does, but it evaporates quickly into the air. Evaporation might sound like a free pass, but it means more solvents getting into the air you breathe. It also seeps into groundwater faster than oil-based stuff, which pushes up the risk for local water supplies. Rivers and lakes near factories using isophorone have recorded higher concentrations, especially after heavy rains wash residues away.
Regulations and Realities
The EPA capped workers’ exposure limits for a reason, but those rules don’t always stop spills in landscaping or construction. In my work, I’ve seen teams dump leftover solvents into drains or onto the ground, usually because nobody enforced better waste handling. A study by the Agency for Toxic Substances and Disease Registry points to bioaccumulation as a big concern, meaning the substance builds up faster than it breaks down. That sticks with me, because no matter how fleeting the smell, the molecules last longer than you think.
Alternatives Carry Their Own Weight
Green chemistry aims to replace harsh solvents with less persistent and less toxic alternatives. Water-based paint thinners have gotten better, but they cost more and sometimes don’t handle big jobs. Solvent recyclers try to recapture fumes and liquids, but not every shop wants to buy the equipment, and small contractors often pick the cheapest option for disposal or cleaning. The push for plant-derived solvents sounds promising, but it takes resources to grow, refine, and ship every gallon, pulling its own environmental strings.
What Steps Make a Difference
Shifting from isophorone toward safer chemicals makes sense, but education needs to be part of the solution. In my experience, most people using solvents don’t read the safety data, or they forget gloves and goggles until after a spill. Stronger enforcement for hazardous waste handling and better access to disposal help more than a new label or ad campaign. Local governments could make it easier for painters and DIYers to drop off leftover chemicals. Researchers have reported that using enclosed mixing systems cuts emissions almost entirely.
Looking Forward
Isophorone holds up demands in manufacturing, but every step—mixing, using, dumping—carries a cost. The more we learn about chemical persistence, the more pressure there is to keep volatile solvents from hitting water and air in the first place. Some new regulations may push companies to make the switch faster, but real change depends on how everyday users handle these products on the ground.
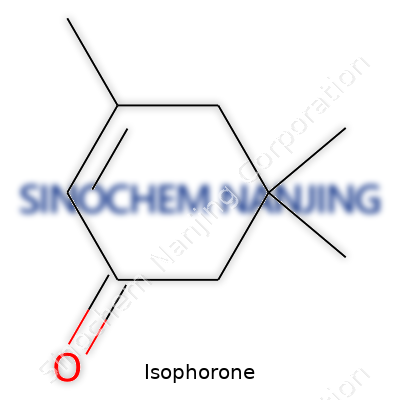
| Names | |
| Preferred IUPAC name | 3,5,5-Trimethylcyclohex-2-en-1-one |
| Other names |
3,5,5-Trimethyl-2-cyclohexen-1-one Isoacetophorone Isoforon |
| Pronunciation | /ˌaɪ.səˈfɔːr.oʊn/ |
| Identifiers | |
| CAS Number | 78-59-1 |
| Beilstein Reference | 0359246 |
| ChEBI | CHEBI:15345 |
| ChEMBL | CHEMBL14235 |
| ChemSpider | 54665 |
| DrugBank | DB08338 |
| ECHA InfoCard | ECHA InfoCard 000026-09-5 |
| EC Number | 201-126-0 |
| Gmelin Reference | 564 |
| KEGG | C06323 |
| MeSH | D010090 |
| PubChem CID | 7909 |
| RTECS number | UF9275000 |
| UNII | 4R9T64A084 |
| UN number | UN1245 |
| Properties | |
| Chemical formula | C9H14O |
| Molar mass | 138.206 g/mol |
| Appearance | Colorless to slightly yellow liquid with a peppermint-like odor |
| Odor | Peppermint-like |
| Density | 0.92 g/cm³ |
| Solubility in water | 8.5 g/L |
| log P | 1.70 |
| Vapor pressure | 0.4 mmHg (20°C) |
| Acidity (pKa) | 7.7 |
| Basicity (pKb) | 5.95 |
| Magnetic susceptibility (χ) | -54.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4755 |
| Viscosity | 2.53 mPa·s (25 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -345.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3934 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D06BX04 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H332, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P314, P321, P330, P337+P313, P362+P364, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 3 2 1 |
| Flash point | 85 °C |
| Autoignition temperature | 444°C |
| Explosive limits | Explosive limits: 0.8–3.8% |
| Lethal dose or concentration | LD50 oral rat 1300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1,450 mg/kg |
| NIOSH | KGA |
| PEL (Permissible) | 25 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Isophorone diisocyanate Isophorone diamine Phorone |

