Isopentanol: A Down-to-Earth Look at an Unassuming Alcohol
Historical Development
History doesn't always shine a spotlight on every chemical in a lab. Isopentanol, known in the trade as isoamyl alcohol, tends to appear in the margins. In the late nineteenth century, researchers started to distinguish among the different alcohols from natural fermentation. Isopentanol, plucked from the myriad traces in fusel oils, entered the chemical lexicon as fermentation science grew more refined. Early industrial uses grew out of distilling spirits and then out of chemical manufacturing, as isopentanol became a relatable raw material for chemists searching for something cost-effective and readily available. Over time, it found its way into flavors, fragrances, and even the smoke of campfires scented by burning wood, reminding me of how ubiquitous modest molecules can be, even in places we’d least expect.
Product Overview
Talk about isopentanol, and you’re referencing a five-carbon alcohol, often trailed by its chemical name 3-methyl-1-butanol. It shows up on ingredient labels under a few aliases, but most folks get used to seeing “isoamyl alcohol.” It brings a strong, distinctive odor that companies harness in food flavoring, especially in producing banana and pear notes. Chemists barely blink at its presence in the laboratory since it’s a workhorse solvent, laying a foundation for countless esters, plasticizers, and specialty chemicals, the kind that help glue, paint, and ink perform better in daily life.
Physical & Chemical Properties
If you had a vial of isopentanol in hand, you’d notice a colorless, oily liquid with an unmistakable, pungent aroma, crossing somewhere between burnt sugar and overripe fruit. The boiling point sits at about 131°C, which isn’t high enough to scare off most users, but it does encourage careful handling. It sports a density a bit less than water and dissolves most easily in organic solvents. Pouring it into water would only create layers, a reminder of its moderate polarity. The chemical formula C5H12O sticks in my mind from years of chemistry labs, and its structure—branched but uncluttered—makes it responsive to all sorts of classic transformations.
Technical Specifications & Labeling
Regulations call for precision. Isopentanol comes designated with its CAS number and hazard warnings because its aroma, while memorable, also hints at some risk. Transport and handling require flammable liquid labeling. Companies committed to transparency often provide information on purity, typical assays landing above 98 percent for most lab-grade material. Labels pull in hazard symbols for irritancy and flammability, reminding users not to treat this as a casual ingredient in any setting, even if it once came from distilling fruit brandy.
Preparation Method
The path from raw material to bottled isopentanol gets paved by fermentation or petrochemistry. Traditional routes rely on distilling the byproducts of yeast fermentation; in another context, it gets synthesized from fossil hydrocarbons using methods such as the oxo process on isobutene. Large-scale manufacturing leans on fractionation and purification to boost yields and minimize impurities, since isopentanol often travels to other production lines for even more specialized outputs. Every step in this chain matters—a batch contaminated with heavier fusel oils shifts not only the safety profile but the usability for downstream processes.
Chemical Reactions & Modifications
Ask any organic chemist about isopentanol, and the first thing likely to come up is ester formation. Its reaction with acetic acid produces isoamyl acetate, the compound that evokes the smell of fresh bananas. Isopentanol also reacts well in oxidation, turning into isopentanal or isovaleric acid, which find use in other specialty chemicals. These traits plant it firmly in the toolkit for everyday syntheses, whether in making artificial flavors or ramping up more complex reaction sequences for pharmaceuticals and polymers.
Synonyms & Product Names
Isopentanol goes by several names. Isoamyl alcohol hits the most recognition. Chemists write 3-methyl-1-butanol or 1-hydroxy-3-methylbutane. Sometimes, you’ll see “isopentyl alcohol,” especially in older texts or bulk supply catalogs. No matter how you see it listed, familiarity with its aliases makes life easier when you bounce between industries or shop for supplies on the open market.
Safety & Operational Standards
Every chemical worth its salt asks for thoughtful handling. Isopentanol doesn’t disappoint in this department. Its flammability can’t be understated—one spark near an open bottle could spell trouble in a laboratory or industrial plant. Proper ventilation, grounded storage, and keeping fire extinguishers nearby all factor into daily routines. For skin and eyes, the risk runs beyond mild irritancy; accidental contact causes burning and redness, so gloves and goggles become standard fare. In my own lab time, one spill on an unprotected hand was more than enough to spark better discipline. Regulators flag isopentanol’s risks by requiring labeling and guidance on ventilation and personal protective equipment in both production and university labs.
Application Area
Isopentanol serves up its utility in more places than most folks realize. In the food industry, flavor production relies on this alcohol to make essential esters, like isoamyl acetate and isoamyl butyrate. The solvent qualities land it in paint stripping and ink manufacturing, where it helps dissolve pigments and smooth out inconsistencies. Perfume companies use it to juggle notes and stretch delicate aromas in ways more stable yet less pungent alcohols can’t match. Laboratories reach for isopentanol as part of the classic DNA extraction protocol, separating genetic material with simple, reproducible results. Even in old-school medicine cabinets, you’re likely to find it as a trace component in cooling liniments and antiseptics.
Research & Development
Current research into isopentanol often explores cleaner production, aiming to shift the base from fossil feedstocks to fermentation and bio-based synthesis. Scientists monitor process yields, impurity profiles, and the energy footprint of every step, frequently targeting genetically modified microbes to boost output or reduce byproducts. Drug developers push new reactions, searching for ways to tweak isopentanol and its derivatives into molecules that serve as antiviral, antifungal, or intermediate pharmaceuticals. Even beyond the lab, collaborations with food technologists and perfumers open the door to nuanced flavor creation and fragrance stability, especially as consumers push back against artificial additives and demand greener supply chains.
Toxicity Research
Experience never lets me look the other way when dealing with potential toxicity, and isopentanol shares some of the concerns common to low-molecular-weight alcohols. Short-term exposure triggers headaches, dizziness, and irritation. High concentrations depress the central nervous system. Long-term data remains limited, but research tracks metabolic pathways after ingestion and inhalation, identifying the breakdown products and their potential to cause harm. Studies on animal models flag concerns for organ toxicity at high doses, but the practical risks in occupational settings tend to center on acute exposure and the threat of fire. Some groups push for tighter controls in workplace air, especially where ventilation has limits and workers may encounter repeated exposure, highlighting a wider drive among occupational safety advocates to anticipate risks before they become emergencies.
Future Prospects
Looking ahead, isopentanol stands at a crossroad between legacy chemistry and newer, more sustainable production lines. Bio-based fermentative routes catch the attention of policymakers and green technology developers seeking cleaner inputs for bulk chemicals. Synthetic biology pushes microbes to higher isopentanol yields, offering a lifeline to rural economies sourcing raw materials from agricultural waste instead of oil. In the lab, the molecule’s value as a building block continues to open pathways in pharmaceuticals and specialty polymers. Consumer demand for natural, non-toxic flavors and fragrances keeps industrial chemists invested in extracting purer streams from renewable sources. Regulators and end-users both watch closely, hoping to minimize exposure risks while keeping a vital ingredient in the toolkits of modern manufacturing, science, and everyday life.
What are the main uses of isopentanol?
From the Lab to Industry: The Role of Isopentanol
Isopentanol shows up in places most people wouldn’t expect. This alcohol gets attention mainly from folks working in chemistry, industrial manufacturing, and flavor creation. My first encounter with isopentanol happened during a college organic chemistry course. It hit me as one of those odd-smelling liquids that seemed to pop up in a lot of reactions involving esters and solvents. It stuck in my mind because of that sharp, almost banana-like odor, but what really matters is its flexibility.
Fuel Additives and Solvents
Fuel production and improvement benefit a lot from chemicals people probably never think about. Isopentanol sees widespread use as a fuel additive. Researchers have found that blending small alcohols like isopentanol with gasoline can help engines burn cleaner by increasing the octane rating. This means vehicles can run more effectively and create fewer harmful emissions. Recently, with the push for alternative fuels, chemists keep circling around alcohols like isopentanol for new biofuel projects. Some studies back up that interest—adding isopentanol to fuels sometimes cuts down carbon monoxide output, helping with air quality.
Flavor and Fragrance Creation
People who work with food or scents pay special attention to the natural flavors and aromas produced by chemicals. Isopentanol produces a fruity, slightly pungent scent, so it often lands a spot in fragrances and artificial flavors. For flavor chemists, it acts as a building block. By combining isopentanol with certain acids, they produce esters that mimic the aroma of bananas and apples. It’s hard to imagine a chewing gum or a hard candy aisle without isopentanol lurking in the recipes, although probably no shopper realizes it’s part of their snack.
Industrial Manufacturing
Manufacturing paints, coatings, and plastics leans heavily on good solvents. I’ve talked to a few paint workers who appreciate how isopentanol dissolves both oily and water-based substances. Because its structure lets it mix with a range of compounds, it helps even out the application of paints or coatings. Lab workers often turn to isopentanol for organic synthesis. It does double duty as both a raw material and a solvent, showing up in everything from pharmaceuticals to adhesives.
Health and Laboratory Use
Lab techs value isopentanol for its role in organic extractions, especially when isolating DNA and RNA. Medical researchers often team up isopentanol with other alcohols to break open cells, separating genetic material for analysis. Labs dealing with emergency samples often grab isopentanol for its reliability during extraction, since it keeps proteins away from nucleic acids.
Handling and Safety
Anyone working with isopentanol understands the headaches and safety warnings. Prolonged exposure to vapors leads to irritation and even dizziness. The liquid catches fire easily, so good ventilation and strict storage rules count for a lot. Workers follow safety data sheets and protective gear routines, even in smaller research labs.
Looking Forward
Over time, demand for isopentanol in green chemistry and biofuel creation will likely keep rising. Scientists and manufacturers looking for cleaner, more sustainable solutions tend to return to alcohols like isopentanol because the science points that way. Its combination of solvent ability, flavoring power, and usefulness for research and energy keeps it in the toolkit across fields. If regulations and safety standards keep up, isopentanol will play a bigger part in everyday products and industrial progress.
Is isopentanol toxic or hazardous to health?
Looking at the Risks of Isopentanol
Isopentanol, also called isoamyl alcohol, pops up in a lot of places—for instance, in some industrial manufacturing and even as a byproduct in certain fermentation processes. When a chemical shows up this often, people naturally start asking whether it could harm them. Many folks who handle cleaning or laboratory work have probably caught a whiff of its sharp, strong odor. That smell alone tells you there's something in the air you probably shouldn't inhale for long.
What Science Says About Exposure
A pile of occupational health research points out that isopentanol isn't among the worst chemical threats, but brushing off its risks would be a mistake. According to the U.S. National Institute for Occupational Safety and Health (NIOSH), breathing in high concentrations can leave people dizzy or nauseous. Prolonged exposure to the vapors might also irritate the respiratory tract, bringing headaches, coughing, or even a sore throat.
Skin contact deserves attention, too. Isopentanol tends to dry out the skin, sometimes even causing redness or a mild rash. Most hospital visits connected to handling this compound have involved people who got it on their skin, in their eyes, or inhaled the fumes at close range in a poorly ventilated room.
From my own time in chemical labs, I can say fumes from substances like isopentanol travel farther than most people expect. Someone might open a container in one corner, and soon enough, the odor lingers around the workspace. People skip wearing proper gloves or open windows because they're rushing—and that's when small mistakes cause real problems.
Long-Term Health and the Bigger Picture
The available science indicates isopentanol doesn't build up in the body, and its acute effects clear up once fresh air comes in. So, there’s not much evidence linking it to cancer or serious chronic problems. That’s some relief. But just because a substance doesn’t land you in the hospital right away doesn’t mean it’s harmless.
In the world of occupational safety, it’s easy to become complacent about chemicals like isopentanol because they don’t have the same danger signals as, say, cyanide or mercury. Still, problems add up with repeated careless handling over time. I’ve seen workers develop sensitivities and find themselves coughing or feeling off after months of regular exposure.
Practical Solutions for Safer Workplaces
Even with a “milder” chemical, small steps make a difference. The basics—good ventilation, gloves, safety goggles, and clear labeling—remain the best defense. Companies and labs can protect their teams if they give regular safety refreshers, check equipment, and create a culture where everyone feels fine about speaking up when something seems off.
Isopentanol gives us a reminder: everyday chemicals deserve the same respect as rare or “high-alert” substances. Reading the labels, using protective gear, and respecting safety guidelines all help keep a workday uneventful, which is usually the best outcome of all.
What is the chemical formula and structure of isopentanol?
Looking Closer at the Formula
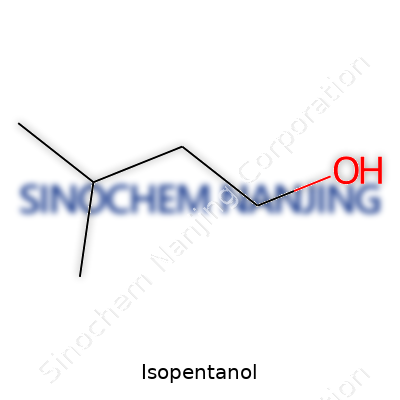
Isopentanol, known by its chemical formula C5H12O, sits among the family of alcohols called amyl alcohols. Its structure reveals five carbon atoms hooked together, with a single hydroxyl group (–OH) attached to a carbon away from the main chain. In a picture, this arrangement takes the branched form of 3-methyl-1-butanol, where four carbons form the main stretch and the fifth links as a side group.
From my college organic chemistry days, drawing isopentanol’s structure meant carefully placing the –OH on the right spot: CH3–CH(CH3)–CH2–CH2–OH. This branch shapes the way isopentanol smells, dissolves, and interacts with other substances.
Why Isopentanol Earns Attention
Isopentanol often appears where people least expect it—inside the aroma of whiskey, in the laboratory, or behind the scenes in manufacturing. Fermentation produces it naturally in trace amounts, contributing fruity or solvent-like notes in alcoholic drinks. Too much can spoil the taste. Distillers monitor its buildup because isopentanol, while present in low concentrations, brings a heady quality and can boost harshness if left unchecked.
On an industrial scale, isopentanol helps make scents and medicinal chemicals. The branched structure lets it form esters—compounds behind flavors and fragrances in food and cosmetics. Imagine the smooth blend in some pear-flavored candies. Isopentanol-based esters often supply a piece of that signature aroma.
In the lab, the molecule becomes a handy solvent, especially for organic syntheses or separating mixtures. Chemists appreciate its balance—enough volatility for quick evaporation, but plenty of strength for dissolving both polar and non-polar compounds. I remember using isopentanol in organic extraction processes to tease out delicate natural products without leaving layers of impurities.
Health and Environmental Notes
The fruity scent of isopentanol floats above a warning sign. Like many alcohols, inhaled vapors irritate eyes and airways; skin exposure causes dryness. High levels bring headaches or dizziness. Industry keeps close tabs on workplace concentrations, following occupational health rules. Good ventilation and protective gloves tend to do the trick.
Disposal and spillage matter too. If poured down the drain, isopentanol can harm aquatic life before breaking down. Careful containment and treatment, often using special biofilters or incinerators, slow the march of pollution.
Building Safer Chemistry and Everyday Awareness
Chemists and manufacturers work on greener synthesis pathways, seeking bio-based sources for isopentanol. Sugar fermentation, guided by custom yeast strains, scales up with fewer chemical byproducts. These biological routes promise a future with lower carbon footprints and reduced hazardous waste.
Personal experience in educational labs serves as a reminder that even simple molecules need respect. Labeled bottles, glove protocols, and chemical hoods aren’t just for show. Mistakes in handling alcohols—spills, splashes, careless sniffing—teach lessons fast and hard. For everyone from brewers to science students, a clear view of isopentanol’s structure, usefulness, and risks leads to smarter, more thoughtful handling in our homes, factories, and research benches.
How should isopentanol be stored and handled safely?
Common Sense Steps Keep Everyone Safer
I’ve watched too many folks cut corners with chemicals, thinking a few quick measures will do the trick. With isopentanol, a liquid used in labs and industry, mistakes carry consequences. Isopentanol carries a strong smell, evaporates fast, catches fire at low temperatures, and runs off easy. It’s not something to treat lightly. I’m sharing what I’ve learned so others stay healthier and out of trouble.
Fire Is the Real Risk People Ignore
Isopentanol lights up with just a tiny spark, even a bit of static or a warm surface. Flash point sits at 49°C, which means a summer day without air conditioning or a slip with equipment might cause a fire you can’t put out fast. Containers and rooms storing this alcohol deserve the same attention as gasoline or acetone. You want metal safety cans designed for flammables, kept away from heaters, sunbeams, and anything that glows or gets hot. Keep it off the concrete, too, since fumes crawl low and can settle where you don’t expect.
Ventilation Means Fresh Air, Always
No one wants their lab or storage room full of isopentanol fumes, since headaches and dizziness come on strong before you realize you’ve breathed too much. Working near a window or open door doesn’t cut it. Fume hoods and well-maintained exhaust fans make the difference. I’ve seen places try to ignore this, then they wonder why everyone’s eyes burn and noses tingle. If the smell hangs around, then air exchange needs cranking up.
Containers: What Actually Works
Plastic can swell and crack if the right type isn’t used. Polyethylene and polypropylene bottles handle isopentanol without trouble. Don’t reach for just any gallon jug or used water bottle sitting around. Use red-colored safety cans or bottles labeled for flammables—clear, tough labeling stops accidents and saves a lot of explaining later. I make sure anyone new to the job knows every container needs both a chemical name and a hazard warning they’ll actually read.
No Substitute for Real PPE
Lab coats, splash-resistant goggles, and chemical gloves (nitrile always beats latex) keep skin and eyes from burning. I always keep a shower and eyewash station in the same room, since “I’ll be quick” never justifies skipping protection. Gloves come off the second anything leaks. If you ever smell isopentanol on your hands, you probably missed a spill or a glove change somewhere.
Planning Ahead: Storing Where Others Work
Fire codes ask for vented, metal cabinets, and there’s a good reason. One leak could fill a closed office or storeroom in minutes. Remote storage sites or locked flammable cabinets keep workers out of harm's way and let people focus on the rest of their tasks. I always choose cabinets placed close to an exit, away from traffic, never anywhere near food or personal items.
Cleaning and Disposal: No Shortcuts
No one likes mopping up spills, but speed really matters. Pouring sand or a spill absorbent powder takes seconds. Shoving contaminated towels in the trash risks fire, so use sealed metal containers and call a disposal service that knows flammable waste. Don’t count on drains or outside dumpsters. Local rules exist for a reason, and fire marshals remember mistakes for a long time.
Good Habits Stop Accidents Long Before They Begin
It takes discipline to check bottles for leaks, review training, and run monthly drills, but the payoff comes in fewer injuries and less downtime. I’ve seen firsthand that folks who build these habits into every day rarely run into trouble, while those relying on luck get burned—sometimes literally. Isopentanol can work for you, not against you, if treated with respect and common sense at every step.
What are the physical properties of isopentanol (boiling point, odor, solubility)?
The Boiling Point: Hot but Not Too Hot
Isopentanol, sometimes found in labs or even as a byproduct in some food fermentations, boils at around 131°C (about 268°F). This temperature means it takes a decent amount of heat before isopentanol starts to vaporize. Anyone who’s ever watched water bubble on the stove can imagine how much longer this alcohol would sit before being lost to the air. This isn’t just an academic curiosity—boiling point affects storage, usage, and even safety in workplaces. If you accidentally let things get too warm, isopentanol can start to evaporate and create fumes you probably don’t want to breathe for long.
The Distinct Odor: Not Your Friendliest Scent
Anyone who’s cracked open a bottle of isopentanol knows it doesn’t have the pleasant notes of lavender or peppermint. Isopentanol carries a sharp, penetrating smell—described in the literature as “fusel oil,” blending earthy, fermented notes with that unmistakable, sometimes stinging, pungency. Years ago, working in a college lab, I learned that a small spill of this stuff could clear a room fast and linger stubbornly in the air or on clothing. In industries handling flavorings or fragrances, that strong smell means ventilation is a top priority. The human nose picks up isopentanol at low concentrations, so even a tiny amount in the air tells you there’s a leak or an open container hanging around.
Mixing with Water: Solubility Challenges
Here’s a fact some folks don’t always expect—dump some isopentanol into a glass of water, and most of it floats to the top. The solubility is pretty low, with about 2.2 grams dissolving per 100 milliliters of water at room temperature. Alcohols closer in structure to ethanol will mix better, but isopentanol’s extra bulk on its carbon chain keeps it more on its own. This makes cleanup trickier and separates it from the group of alcohols we use more freely in hand sanitizers or bartending. For those who work with waste management or chemical production, that low solubility means water treatment plants have to use extra steps to keep it out of supplies intended for drinking or irrigation.
Hazard Awareness in Everyday Settings
Plenty of home distillers or hobbyists run into isopentanol, often without realizing. It shows up sometimes in poorly controlled fermentation, contributing to off-flavors and harsh “bite” in spirits that haven’t been properly purified. On job sites, that odor and the modest boiling point interact with the compound’s toxicity. Skin contact won’t melt gloves off, but inhaling the fumes over time can make people lightheaded or even sick. That’s where basic skills—venting the area, capping containers right away, labeling bottles—keep accidents from happening. Not enough attention gets paid to the basics: open windows, correct storage, and never, ever taste-test unknown alcohols.
Potential Solutions and Smarter Use
Folks in labs and factories rely on training and technical fixes. Local exhaust fans, secure chemical cabinets, and clear signage work far better than waiting for the unmistakable odor as a warning. Waste handlers depend on separation tanks that let isopentanol float up and out before water works its way downstream. Scientists and distillers can rely on vapor control techniques, using temperature control at the core of purifying products. Even in school science labs, clear instructions help prevent confusion—reminding students that not every alcohol dissolves or evaporates like ethanol.
Why It All Matters
Isopentanol sneaks into daily life in more places than you might expect, from distilled drinks to specialized labs. Recognizing the physical side—boiling point, smell, solubility—literally shapes safer workplaces and cleaner products. The history of chemical mishaps, both large and small, proves that knowing your materials isn’t abstract. It changes choices about cleaning, storage, waste, and even what you breathe at work or at home. Experience, practical training, and everyday awareness beat out jargon every time.
| Names | |
| Preferred IUPAC name | 3-Methylbutan-1-ol |
| Other names |
Isoamyl alcohol Isopentyl alcohol 3-Methyl-1-butanol Isopentylol |
| Pronunciation | /ˌaɪ.soʊˈpɛntən.ɒl/ |
| Identifiers | |
| CAS Number | 123-51-3 |
| 3D model (JSmol) | Isopentanol JSmol 3D model string: ``` CC(C)CCO ``` |
| Beilstein Reference | 1718732 |
| ChEBI | CHEBI:28814 |
| ChEMBL | CHEMBL33040 |
| ChemSpider | 6197 |
| DrugBank | DB02236 |
| ECHA InfoCard | 100.120.285 |
| EC Number | 603-006-00-7 |
| Gmelin Reference | 871 |
| KEGG | C00432 |
| MeSH | D013238 |
| PubChem CID | 31260 |
| RTECS number | NT8050000 |
| UNII | 3K9PSM94YW |
| UN number | UN1262 |
| CompTox Dashboard (EPA) | DJMMLYDJJVWZBE-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | C5H12O |
| Molar mass | 88.15 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | Distinctly alcoholic |
| Density | 0.810 g/cm³ |
| Solubility in water | 5.5 g/L (20 °C) |
| log P | 1.51 |
| Vapor pressure | 2.7 mmHg (20 °C) |
| Acidity (pKa) | 16.1 |
| Basicity (pKb) | Isopentanol has a pKb of approximately 5.4 |
| Magnetic susceptibility (χ) | -54.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.405 |
| Viscosity | 3.89 mPa·s (at 20 °C) |
| Dipole moment | 1.791 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 196.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -393.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3320.0 kJ/mol |
| Pharmacology | |
| ATC code | V03AB20 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P403+P235 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 43 °C |
| Autoignition temperature | 398 °C |
| Explosive limits | 1.1–7.5% |
| Lethal dose or concentration | LD50 oral rat 7400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 740 mg/kg (oral, rat) |
| NIOSH | PU1400000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | Chemical protective gloves |
| IDLH (Immediate danger) | IDLH: 1,000 ppm |
| Related compounds | |
| Related compounds |
Pentanol Isobutanol Neopentanol Isopentyl acetate Pentyl acetate Isopentyl nitrite |

