The Tangled Story of Isononanoic Acid: Yesterday, Today, and What’s Next
Historical Roots of Isononanoic Acid
Chemistry has a way of sneaking into ordinary life, and isononanoic acid is a good example of that quiet influence. Its story traces back to the mid-20th century, just as the petrochemical industry was realigning what was possible in organic synthesis. Through careful manipulation of hydrocarbons, scientists isolated fatty acids with branching that pushed boundaries in lubricants and plasticizers. As researchers explored isooctanoic and related acids, isononanoic acid emerged out of olefin chemistry. Companies invested in further understanding this compound as motor oils, polymers, and specialty chemicals demanded new levels of stability. The origin isn’t flashy, but it shows real-world curiosity turning petrochemicals into tools for everyday products.
Diving Into the Product
On the shelf, isononanoic acid won’t grab the spotlight. It's usually a colorless liquid, sometimes giving off a mild, slightly pungent odor. Most folks will never handle it directly, even though it’s present in coatings, lubricants, and even some fragrances. The acid carries a branched nine-carbon backbone, which gives it some special tricks — including resistance to oxidation compared to its straight-chain cousin, nonanoic acid. That structure brings important advantages for folks working with oils that can’t afford to turn to goo or solidify during use. It's this backbone that keeps things flowing and stable even as conditions wander above and below room temperature.
Physical & Chemical Properties That Matter
If you spill isononanoic acid on a lab bench, it spreads as a clear liquid. It has a moderate boiling point, running about 240-260°C, with a low freezing point that helps it stay liquid throughout most seasons and industrial setups. Its density hovers close to 0.9 g/cm3, which means it won’t sink like a rock or evaporate in a snap. Water solubility remains minimal, a trait that comes in handy for those crafting hydrophobic coatings or additives. Its acid strength is mild—this isn’t something you’d use to etch metal, but it does play well in chemical modifications. What stands out is the way its branching shifts performance: improved resistance to thermal breakdown, a knack for blending with oils, and a stubbornness against microbial attack.
Technical Details and What the Label Tells You
Labels for isononanoic acid often list its purity, usually topping 95 percent for industrial batches. Key technical specifications focus on acid value, color (sometimes a faint yellow tint), water content, and the presence of related nonanoic acids. The industry worries about impurities because they impact lubricant life, product safety, or paint performance. Chemists—having spent time in both research labs and busy warehouses—know how tiny amounts of a contaminant can leave batches of paint tacky or shorten the life of a synthetic oil. Every bottle, drum, or tanker should ship with numbers that spell out exactly what’s inside, because folks on plant floors don’t have time for surprises.
Putting It Together: How Isononanoic Acid Gets Made
Making isononanoic acid involves a blend of old-fashioned chemistry and modern engineering. Chemists start with higher olefins, often produced through oligomerization or dimerization of smaller alkenes. The backbone takes shape as these react with carbon monoxide and water in a process called hydroformylation, followed by careful oxidation and distillation. This route allows tuning so the end product hits the mark for both branching and chain length. In practice, these reactions need catalysts—often ruthenium or cobalt—and a fair bit of care to control reaction temperatures and pressures. Plant operators must balance efficiency against waste and contamination, relying on years of logbook data and newer process controls for consistency.
Getting Reactive: Chemical Transformations and Use Cases
Isononanoic acid isn't just a finished ingredient—it’s a stepping-stone in bigger chemical pathways. React it with alcohols and you get isononanoate esters, which show up in emollients, synthetic lubricants, and surfactants for cleaners. Heat it with amines and you get amides, sometimes used in specialty coatings and plastic additives. The branching makes each derivative a bit more resistant to breakdown, whether under heat, light, or years of storage. Some researchers use these reactions to tune plasticizers that won’t migrate out of polymers or to make metal soaps that improve pigment flow on industrial paint lines. In my own work, I’ve seen isononanoic derivatives help extend the working life of greases that run at high temperatures—something factory managers care about when bearings start running hot on a summer afternoon.
What’s In a Name? Synonyms & Branding
Depending on where you look, isononanoic acid may turn up under a few synonyms. Call it 7-methyloctanoic acid or 3,5,5-trimethylhexanoic acid, and some specialists will know exactly what you mean. Trade names tend to get assigned by large chemical suppliers or devise their shorthand for internal tracking. These alternate names echo through technical documents, making it important for buyers and lab techs to keep a close eye on batch sheets and specifications—confusion here can mean a costly or dangerous mix-up.
Safety, Health, and Responsible Handling
Anyone who’s worked in a chemical plant knows that no acid is “mild” if it’s splashed in the wrong place. Standard procedures call for skin and eye protection, fume control, and careful storage—usually in tightly sealed containers away from heat and incompatible reactants. Isononanoic acid gives off low corrosivity compared to strong acids like sulfuric, but safety data sheets weigh in on risks of skin and eye irritation. Proper ventilation reduces the chance employees breathe in fumes during bulk transfer. In my experience, minor leaks get caught during routine walks, with teams jumping in fast to rinse and neutralize. Long-term exposure studies suggest it’s not a major health threat under controlled conditions, though repeated skin contact can trigger dermatitis—a reminder that regular training and real-world vigilance matter more than paperwork alone.
Where It Shows Up: Industry and Research Uses
Anytime industrial lubricants or synthetic oils need to push past technical barriers, isononanoic acid sits near the core of those blends. Coating manufacturers add it for its resistance to yellowing and ability to prolong gloss in architectural finishes. In plastics, derivatives help balance flexibility and rigidity, especially in high-value specialty materials. Perfume labs reach for esters that dial down harsh notes and add staying power without overpowering the nose. Some pharmaceutical researchers explore its intermediates for niche applications, though this remains an evolving front. Agricultural chemists sometimes use related esters in adjuvants to help pesticides stick and glide across leaf surfaces. My work intersected with this acid through additives for marine coatings—an environment where salt, sun, and heat expose weaknesses fast, and only robust chemistry holds up across seasons.
Research and What Still Needs Attention
The chemical industry rarely stands still. Teams across academic, private, and government labs keep probing ways to boost yields, cut waste, and uncover new fields for isononanoic derivatives. Green chemistry drives some of these changes, with a push toward renewable feedstocks and milder reaction conditions. Analytical chemists dig deeper into reaction byproducts and trace impurities—lessons learned after production runs that missed spec or field complaints about performance. Application scientists keep nudging the envelope: can isononanoic-based esters hold up to extreme cold, or deliver safer, longer-lasting lubricant films in electric vehicles? Each tweak in preparation or application gets pressure-tested in the real world, not just lab beakers.
Looking Hard at Toxicity and Exposure
Good chemistry does not stop with performance alone. Regulators and independent labs run toxicity studies on isononanoic acid and its derivatives to track health and environmental impacts. Results to date suggest relatively low acute toxicity, but long-term data stay essential. Skin irritation crops up in repeated contact, especially in high-concentration work environments lacking protective gear. Environmental fate has earned its own research: the branched structure slows down microbial breakdown compared to straight-chain acids, raising flags about persistence. European chemical regulations and the American EPA have both pushed industry to monitor run-off and emissions, prompting investments in waste treatment and biodegradable alternatives. Scientists still hunt for greener pathways, both to reduce exposure and to keep breakdown rates in line with environmental needs.
What’s Ahead for Isononanoic Acid
Industry rarely lets a useful chemical gather dust. Isononanoic acid faces a future defined by challenges and opportunity. Growth in synthetic lubricant markets, tougher rules on VOC content, and the shift to electric mobility all ask for more stable, sustainable materials. Researchers chase renewably sourced intermediates and search for catalysts that cut energy use. Regulatory pressures will keep shaping production practices and downstream waste handling. Materials science unlocks further value—improving durability in paints, boosting flexibility in polymers, and extending useful lifetimes in engine oils that now must perform under new stressors. Consumers may never spot isononanoic acid on a label, but its impact will play out across industries eager for smarter, safer, and longer-lasting chemistry.
What is Isononanoic Acid used for?
How Isononanoic Acid Slips into Daily Life
Isononanoic acid might sound like a tongue-twister from a high school chemistry class, but its reach spreads much further than the label suggests. It lands in products people rely on every single day. From personal care shelves to industrial workshops, this compound quietly lends a hand.
Beyond the Lab Bottles
Take a look at those lotions, sunscreens, or creams lining bathroom counters. Formulators rely on isononanoic acid to enhance the way these products feel. Its oil-like texture softens creams, lets them spread smoothly, and helps skin absorb moisture without that heavy, greasy sensation. Dermatologists and chemists both point to it as a game-changer for sensitive skin, since it rarely triggers allergies.
The sunscreen story gets even more interesting. Ultraviolet (UV) filters often need help dissolving evenly in lotions. Isononanoic acid does that job, making sure protection holds up without breaking down too quickly under sunlight. As someone who spends long hours hiking or on the beach, finding a sunblock that feels light but holds steady matters. This ingredient does some heavy lifting to make that possible.
What Keeps Cars and Machines Run Smooth?
It doesn’t stop at beauty routines. Isononanoic acid also finds a second home under the hood of cars and inside heavy machinery. Lubricant manufacturers value it for its stability, especially when engine parts get hot or work under strain. It combines with alcohols and other acids to make synthetic esters, which stand up to high temperatures and don’t break apart easily. Every driver who’s made it through a brutal summer road trip owes a quiet thank-you to these custom-made oils.
Paints and coatings use it, too. Both the finish on car hoods and the walls of homes stay glossy, resist water better, and last longer thanks to modifications made possible by isononanoic acid. A lot of factories building consumer electronics want coatings that stand up to scratches and spills. Without improved acids like this, most products would need extra layers or more frequent replacement.
Why This Matters Now
There’s a real push to trim down the use of harsh chemicals in consumer goods. Isononanoic acid lines up with that trend. Reports from organizations like the National Institutes of Health highlight its low toxicity and steady biodegradability. While not every chemical fits the bill for green chemistry, this one checks more boxes than most—so brands keen on shrinking their environmental footprint have reasons to turn to it.
Price can be a sticking point for some. High demand and specialty production sometimes make the cost climb, hitting smaller companies hardest. But as more suppliers come online and greener production methods gain ground, that trend could shift in a more accessible direction.
Rethinking What Ingredients Matter
After years spent in consumer advocacy, it’s clear that ingredient awareness has never been more important for families and businesses. People ask sharper questions and expect transparency from the brands they trust. Isononanoic acid doesn’t draw headlines, but the role it plays—especially in safer formulations and tougher, longer-lasting coatings—makes a pretty strong case for paying attention to the building blocks behind everyday products.
Speeding up innovation and simplifying regulations around safer chemical production can help new, responsible uses of isononanoic acid break through even faster. Education—both for product developers and everyday shoppers—goes hand in hand with safer, smarter choices.
Is Isononanoic Acid safe for skin or cosmetic use?
What Isononanoic Acid Does in Cosmetics
Isononanoic acid shows up in a surprising number of skincare products, mostly as an emollient or a texture enhancer. Brands often choose it because it helps lotions and creams spread easily without leaving that greasy feeling behind. Anyone who’s struggled with sticky SPF or thick moisturizer can appreciate a formula that feels pleasant on the skin. It’s not one of those trendy natural ingredients, but it’s been around in personal care for decades.
Looking at the Research
Digging into safety reports makes things clearer. The Cosmetic Ingredient Review Expert Panel published a detailed assessment showing isononanoic acid rarely causes irritation or allergic reactions, especially at the concentrations used in typical face and body products. In practice, major brands trust it in everything from sunscreens to makeup removers. There isn’t a pile of complaints or cases piling up in dermatology offices about this fatty acid causing rashes or breakouts. That counts for a lot.
Dermatologists run patch tests before ingredients ever reach store shelves, and most data points to isononanoic acid causing fewer problems than essential oils or some plant extracts do. Results show it does not commonly clog pores. Still, everyone’s skin reacts differently, and there’s always the one-in-a-thousand chance of sensitivity, especially if a person already has eczema or a skin barrier disorder.
Sourcing and Purity Matter
The source and quality of isononanoic acid can’t be ignored. The best skincare brands put effort into traceability and purity testing. Making isononanoic acid often involves petrochemical feedstocks and controlled chemical processes. Any impurities or byproducts could introduce risks if manufacturers cut corners. Reputable companies stick to Good Manufacturing Practice standards so that’s less of a worry when sticking to established products.
Environmental and Health Considerations
Beyond personal use, there’s a bigger conversation about the long-term environmental impact of any synthetic ingredient. While isononanoic acid itself doesn’t hang around in the environment for years like some silicones do, it’s not biodegradable in the way coconut oil or shea butter might be. Some folks care about how products wash down the drain and end up in waterways. Big brands are starting to pay closer attention to supply chain sustainability, which affects decisions on ingredient use.
Who Should Think Twice?
Most people can apply products with isononanoic acid without problems. Even so, folks with very sensitive skin or allergies can never be too careful. Patch testing a product behind the ear before smearing it all over the face prevents surprises. I’ve talked with many people who discovered sensitivities only after switching to a “clean beauty” routine full of unfamiliar ingredients, so nothing beats watching your own skin’s response.
Possible Safer Alternatives
Anyone looking for plant-based options can find plenty of alternatives like squalane or jojoba oil. These offer similar benefits for texture or moisture retention but come from renewable sources. Still, isononanoic acid remains popular because of its stable shelf life, light feel, and proven track record. People with longstanding skin conditions should always check ingredients, bring questions to dermatologists, and not assume plant-based means gentler or safer.
How Informed Choices Help
With so many formulas and new products hitting shelves each year, it pays to read the ingredient list and research any unfamiliar names. Transparency from brands, strong government regulation, and personal experience combine to help consumers make better choices. If a product feels good, does what it promises, and causes no reactions, it’s probably a good fit for your personal routine.
What are the physical and chemical properties of Isononanoic Acid?
Understanding the Substance
Isononanoic acid, bearing the chemical fingerprint C9H18O2, turns up more often than most realize. Either colorless or slightly yellow, it often gives off a distinct, sharp odor. Most folks, unless working directly with chemicals, wouldn't recognize the name, but products on supermarket shelves have likely relied on the stuff at some stage. Its main gig runs in industrial settings, particularly as a raw material for producing plasticizers, lubricants, and special coatings.
Physical Properties Shape Everyday Use
The liquid flows easily at room temperature, so it’s uncomplicated to handle in bulk form. Boiling starts at about 250°C, so ordinary environmental heat doesn’t faze it. As a branched carboxylic acid, it brings a lower freezing point than its straight-chain cousins. That helps companies that want their mixtures to stay liquid in cold places—handy for lubricants and certain antifreeze formulas.
In my chemistry lab days, bottles of Isononanoic acid showed reliable stability in storage. It absorbs slowly through the skin—better than some, worse than others. Accidentally spilling a bit on a glove left a lingering smell, but never caused burns like strong acids. Safety goggles and gloves always made sense here, because repeated exposure could lead to skin irritation.
Chemical Character Lends Versatility
Its chemical backbone carries a carboxylic acid group (–COOH), which means Isononanoic acid can react with alcohols, creating esters. These esters stay in demand for spreading out in engine oils and producing soft, flexible plastics. In the lab, mixing this acid with a base leads to a quick fizz as it neutralizes and produces the corresponding salt. The acid stands up to air and typical chemicals, though concentrated oxidizers can break it down. It doesn’t corrode metal as fiercely as strong mineral acids, so storage tanks outlast their design lives when filled with this stuff.
Water won’t dissolve it completely. After pouring it in, droplets cling together instead of dispersing evenly. That lack of solubility gives manufacturers control when blending it into oil-based mixes, ensuring the acid plays nicely in the hydrophobic (oil-loving) layers instead of disrupting water content.
Why the World Pays Attention
Properties like high boiling point and low reactivity matter directly for safety and longevity, both in the factory and for products people bring home. One example: In automotive lubricants, something resilient and stable keeps engines running smooth, year after year. Food manufacturers don’t turn to Isononanoic acid, owing to its chemical profile, but the markets for cosmetics and personal care see it as a resource for producing ingredients that improve product spreadability and shelf life.
Waste management around this acid needs an eye for detail. While not considered a major environmental risk, improper disposal or large spills could mess with local water sources. Using sealed containers and approved recycling methods minimizes any risk of contamination. Industry training goes a long way—workers who understand both the dangers and benefits of chemicals like this prevent costly accidents and keep the environment cleaner.
Looking Ahead: Smarter Solutions for Processing
As more sectors turn to sustainable production, Isononanoic acid’s chemical profile offers a sweet spot between performance and manageable safety risks. Investing in process improvements—such as advanced capture systems for vapors, safer packaging, and research into less persistent breakdown products—could take its environmental footprint even lower. Sharing chemical knowledge, not hiding it, builds trust and standards that protect both workers and the larger community.
How should Isononanoic Acid be stored and handled?
Understanding the Substance
Isononanoic acid turns up in more places than most folks expect. This fatty acid shows up in plastics, lubricants, and sometimes even in cosmetic products. Ask anyone who’s managed a chemical storeroom and you’ll always hear the same advice about acids: handle them on their own terms. Isononanoic acid arrives with a strong odor and has the potential to irritate skin, eyes, and lungs. It doesn’t play well with open air or heat, and it’ll corrode metals and damage seals that aren’t designed with acids in mind.
Safe Storage Brings Peace of Mind
Store isononanoic acid inside tightly sealed containers, away from any source of moisture or strong oxidizers. Polyethylene and other acid-resistant plastics work well. Metal containers don’t hold up; corrosion ruins the integrity of the vessel, sometimes leading to leaks or dangerous reactions. Keeping the temperature steady, ideally below 30°C, goes a long way in slowing down any chemical breakdown. Find a spot that’s not in direct sunshine—a storeroom tucked away from regular foot traffic works best.
Local fire codes and OSHA guidelines recommend placing acids away from bases and flammable materials. Label each container clearly, both on the side and the top, with the right hazard symbols. Storage shelves should keep containers out of direct line-of-sight from hallways to cut down on accidents. Once, I watched a co-worker stack acids above eye level; one cracked lid and the whole shop carried the stench for days. Lessons like that stick with you.
Careful Handling Makes the Difference
Protection matters most. Gloves—nitrile or neoprene—shield your hands from burns. Safety goggles stop splashes from reaching your eyes. Add a face shield for added safety when decanting or mixing. Some crews prefer lab coats, but work aprons made from acid-proof materials do a better job with splash hazards. Good ventilation keeps fumes from building up and helps folks breathe easier during cleanup.
Open drums or bottles under a fume hood or at least beside an extractor fan. Always pour slowly and steadily. Avoid rushing, since splashing tends to catch even careful handlers off guard. A friend once tried rushing a transfer and ended up spending the afternoon at occupational health. Handling acids isn’t about bravado; small habits prevent big problems.
Spill Response and Waste Management
Spills always bring out the true test of your setup. Keep granular absorbents and neutralizing agents within reach, just outside the storage area. Vinegar won’t do the job—use commercial acid neutralizers instead. Once spilled, isononanoic acid eats through shoes, so boots with chemical resistance should always be nearby. Wipe up residues with plenty of water, but only after neutralizing, since water alone can kick up some heat and put a caustic mess into the drains.
Disposal deserves care. Most waste handlers accept small batches in compatible drums, clearly marked to avoid mix-ups. Training everyone who works near the acid—whether moving boxes or signing forms—sharpens response. Walk-throughs and practice drills don’t just please inspectors; they save hands, lungs, and sometimes lives.
Practical Improvements for the Workplace
Locking storage, daily walkarounds, and keeping written instructions close by each station keep everyone honest and careful. Regular refresher training strengthens the team’s skill, especially for new hires or contractors. Jargon won’t help in a crisis—plain language and bold signs speak the loudest when it counts.
Storing and handling isononanoic acid shouldn’t ever be routine. Bring common sense, solid equipment, and some respect for chemistry’s sharp edges, and you’ll protect the people and property that matter most.
What are the main industries or applications for Isononanoic Acid?
Isononanoic acid pops up in more places than I used to think. After reading dozens of technical sheets and talking with folks in both chemical plants and automotive shops, I started noticing this compound’s role growing over the years. Its main use lands in the world of lubricants. In car maintenance, people rely on motor oils that protect engines and run smoothly at high temperatures. Isononanoic acid helps these lubricants last longer, no matter how much pressure drivers put on engines. I once spoke with an engineer who explained that without certain synthetic acids, a truck’s oil would break down faster, which means more frequent repairs—never a cheap fix.
Car makers aren’t the only ones relying on isononanoic acid. Paints and coatings also grab the benefits. As regulations around volatile organic compounds (VOCs) get tighter, paint chemists keep searching for ingredients that lower emissions but stay tough against weather or regular cleaning. This acid helps bind paint together and stops it from yellowing or peeling—meaning businesses and homeowners don’t repaint as often. If you’ve ever painted a fence only to watch it crack in two winters, you know how annoying poor coatings can be.
The cosmetics industry also likes what isononanoic acid can do. Creams, lotions, and some sunscreens actually use this acid to give products a lighter, smoother feel on the skin. Skin absorption matters—I remember visiting a lab that created “test arms” for lotion trials. Some basic lotions felt sticky after a few hours, but the ones tweaked with specialized acids soaked in cleanly, reducing oiliness and helping other active ingredients reach deeper layers. Safety testing matters, and products using these acids must prove they don’t irritate or clog pores. Real-world results show high demand for skincare that delivers soft, comfortable results—especially as people read more labels and research the ingredients they put on their bodies.
Isononanoic acid also shows up in plasticizers and adhesives. Furniture makers and flooring installers aim for glue that keeps surfaces intact even through long days of sun or heavy furniture moving. For plastics, flexibility and longevity rely on chemicals that hold up under pressure. During one renovation, I saw firsthand what happened when vinyl tiles buckled—clearly, something in the bonding formula fell short. Formulators blend in isononanoic acid to balance toughness and give just enough stretch, so plastics don’t split or turn brittle.
Solutions lie in responsible sourcing and better recycling. Manufacturers can choose suppliers who track feedstocks, limiting environmental harm at production. Engineers and chemists can extend the life of lubricants and plastics using these acids, lowering replacement rates and shrinking waste. Teaching more workers how these chemicals interact could also cut down on workplace accidents and exposure. Regulation only goes so far—people armed with solid training and up-to-date research can judge what works best in specific applications. Instead of always reaching for the newest formula, sometimes the smarter move comes from optimizing proven ingredients, making industries a bit cleaner and more efficient for everyone involved.
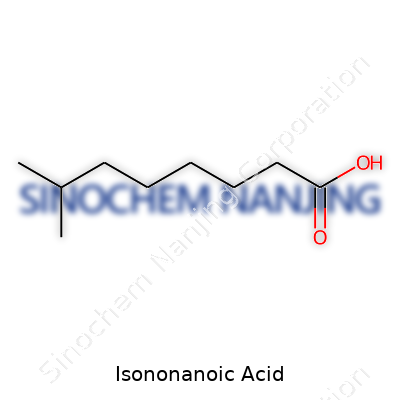
| Names | |
| Preferred IUPAC name | 3,5,5-Trimethylhexanoic acid |
| Other names |
Nonanoic acid, branched isononanoate 3,5,5-Trimethylhexanoic acid C9-branched fatty acid Iso-n-nonanoic acid |
| Pronunciation | /ˌaɪ.soʊ.noʊˈneɪ.ɒɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 26896-18-4 |
| Beilstein Reference | 1208734 |
| ChEBI | CHEBI:30879 |
| ChEMBL | CHEMBL469238 |
| ChemSpider | 61144 |
| DrugBank | DB13810 |
| ECHA InfoCard | 03b1a5c1-10aa-4e2f-868e-19d53fa731d9 |
| EC Number | 204-812-8 |
| Gmelin Reference | 724430 |
| KEGG | C06587 |
| MeSH | D009995 |
| PubChem CID | 8900 |
| RTECS number | RGQ30320N |
| UNII | 4GX5D5N3Y6 |
| UN number | UN3089 |
| CompTox Dashboard (EPA) | DTXSID5020677 |
| Properties | |
| Chemical formula | C9H18O2 |
| Molar mass | 158.24 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | Oily, fatty |
| Density | 0.92 g/cm3 |
| Solubility in water | 0.7 g/L (20 °C) |
| log P | 2.9 |
| Vapor pressure | 0.03 mmHg (20°C) |
| Acidity (pKa) | 4.9 |
| Basicity (pKb) | pKb > 14 |
| Refractive index (nD) | 1.427 |
| Viscosity | 3.5 mPa·s (at 20°C) |
| Dipole moment | 1.686 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 467.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -589.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5726.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements of Isononanoic Acid are: "P280, P305+P351+P338, P310" |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 107 °C |
| Autoignition temperature | 438 °C |
| Explosive limits | Explosive limits: 0.9–5.2% |
| Lethal dose or concentration | LD50 (oral, rat): 3600 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Isononanoic Acid: "3600 mg/kg (rat, oral) |
| NIOSH | Not listed |
| PEL (Permissible) | No OSHA PEL established |
| REL (Recommended) | 200 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Nonanoic acid Isooctanoic acid Neononanoic acid Isodecanoic acid Pelargonic acid |

