Isohexanone: Tracing A Chemical’s Path from Lab Bench to Industry
Historical Development
Isohexanone didn’t just fall out of the sky; it’s a product of modern chemistry’s drive to adapt carbon frameworks for all sorts of uses. In the mid-twentieth century, organic chemists had plenty to do after the war effort fed fresh minds and new funding into science. Focus shifted from the basics—alcohols, ketones, ethers—to the hunt for clever ways to reconfigure them. Isohexanone emerged on that path, through tweaks and trials on larger aliphatic ketones. As collectors of chemical oddities grew more organized, papers started to mention isohexanone as chemists learned to control skeletal branching. The chemical stayed a specialty item, serving as a teaching tool and a touchstone for incremental improvement in synthetic methodology. Eventually, the need for chemical intermediates in flavors, fragrances, and industrial processes gave isohexanone a spot on many laboratory shelves.
Product Overview
This isn’t a blockbuster like ethyl acetate, but isohexanone has carved out a consistent market. The value in this molecule lies in its structure—six carbons, one ketone, with branching that marks it out from straight-chained relatives. That branching changes the game when manufacturers want something that evaporates at a particular rate or interacts differently with other chemicals. Isohexanone often gets picked over more common ketones if a process needs just the right mix of solubility and volatility. Much of the chemical ends up as a solvent or intermediate that smooths out tricky steps in the synthesis of more complex molecules. The applications span specialized coatings, custom flavors, and some niche areas in perfumery. Though not a household name, isohexanone shows up where control and customization matter.
Physical & Chemical Properties
Isohexanone keeps to the conventions of simple organic chemicals: clear liquid at room temperature, distinct smell, and a fair bit of volatility. Its structure—essentially a hexanone with branching off the backbone—pushes its boiling point to the neighborhood of 140-150°C, depending on isomer. Solubility traces a practical line: mixes well with other organics, shrugs off water after the initial splash. Most lab chemists who’ve measured its density or vapor pressure won’t forget its punchy aroma, one not easily mistaken for that of acetone or methyl ethyl ketone. In most industrial settings, it gets handled much like similar ketones, in drums or tanks, moved with the reminders that flammable vapors demand careful attention.
Technical Specifications & Labeling
In the business of chemicals, purity and identification matter. For isohexanone, suppliers usually set standards around GC purity, homogeneity, and water content. Labels follow conventions: danger symbols for flammable liquids, advice for proper ventilation, clear instructions for transfer and disposal. Regulatory agencies set rules for shipping, and importers check that documentation lines up with global standards. Those handling the material on the floor need more than a catalog number; they rely on exact descriptions when blending batches or troubleshooting problems downstream in production.
Preparation Method
Isohexanone doesn’t spring fully formed from a single reaction. Producers typically draw from well-established routes: oxidation of branched alcohols, aldol condensations tuned to favor the right backbone, or careful catalytic manipulations that trim longer molecules down to size. In research labs, chemists often run the risk of byproducts or isomeric mixtures, which means routine purification by distillation or column chromatography. Scale-up for industry raises fresh questions—yield versus cost, waste management, and reaction lifetime all matter to keep the process flowing without surprises. Anyone with a few years in industrial chemistry has watched the same core synthesis play out, tweaked as new catalysts emerge or regulations shift.
Chemical Reactions & Modifications
Isohexanone plays a dual role: useful on its own and as a launching pad for further chemistry. That ketone group draws nucleophiles and serves as a handle for reduction or amination. If the synthetic goal calls for more branching or a longer chain, isohexanone lines up for cross-aldol or Michael reactions. Functional group tolerance represents a big win—unlike more sensitive molecules, isohexanone survives under both acidic and basic conditions. Its modest reactivity compared to aromatic ketones keeps the reactions controllable. Whether tweaking small amounts in research or scaling up for bulk intermediates, chemists appreciate a flexible building block, and isohexanone rarely disappoints on that front.
Synonyms & Product Names
Naming confusion runs deep in organic chemistry, and isohexanone is no exception. Synonyms pile up: 4-methyl-2-pentanone, methyl isobutyl ketone (sometimes in error), or simply “branched hexanone.” Old literature adds even more: isocaproanone, 2-hexanone (in mistaken identity). For procurement, clarity counts; outdated or ambiguous naming can lead to delays, mismatches, or regulatory headaches. Laboratories and suppliers work hard to pin down the correct chemical abstract number and drop all the aliases when the stakes are high.
Safety & Operational Standards
Few people who work in a chemical plant, even for a year or two, take safety lightly after seeing solvent accidents. Isohexanone is no more hazardous than most low-boiling ketones, but flammable vapors, splash risk, and skin contact all add layers of challenge. Correct procedures—grounded transfer lines, proper ventilation, PPE—reduce both major incidents and minor exposures. Upstream, process engineers double-check every valve and joint to stop leaks and overflows. Downstream, waste handling follows the rules set by local and international bodies, separating organics from sewer lines and water supplies. Workers expect annual refreshers on safety protocols, and the best-run firms run regular drills—no one counts on luck when shipping or mixing chemicals at scale.
Application Area
Isohexanone’s main uses come out of its position halfway between volatility and stability. Coating specialists value it as a solvent for resins that demand a touch more solubility than acetone or MEK deliver. Fragrance chemists prize it for intermediate synthesis, where it adds complexity or helps unlock harder-to-achieve aroma notes. Electronics producers sometimes tap isohexanone’s solvency in cleaning agents meant for specific types of flux or residue. In flavors, the ketone structure sometimes makes it into food-grade pathways—though strict regulations guide purity and minimum exposure. Small-scale labs like it for reliability and for the lack of strangeness in handling compared to newer, less-tested chemicals.
Research & Development
Basic research gave us isohexanone, but ongoing R&D still makes room for improvements. In the last decade, lab groups took a fresh look at greener synthesis—less hazardous oxidizers, more efficient catalysts, and solvent-free alternatives to slash waste and cost. Some teams dig into the potential for isohexanone as a novel building block in pharmaceuticals or in biodegradable polymers, but success hinges on turning small-scale wins into commercial reality. Government and corporate funding never seems to stretch far enough to answer every question, forcing chemists to focus on processes that balance safety, scalability, and sustainability. Collaborations between academia and industry speed things up, shaving years off old timelines.
Toxicity Research
Toxicity stands out as a crucial topic in chemical use, especially for anything with a chance of ending up in waterways or in contact with workers. No responsible operator shrugs off gaps in animal studies or long-term exposure data. Early focus stuck with acute effects—headache, skin irritation, dizziness—but regulators and researchers now demand deeper dives into chronic effects, metabolites, and accumulation. Comparison to related ketones shows isohexanone doesn’t stray much from the norm, but stricter risk thresholds drive each new study. The industry answers with fresh assays and honest reporting, building a stronger foundation for occupational safety and environmental stewardship. Updated MSDS sheets, safety audits, and open communication with regulators build trust and keep plants running.
Future Prospects
Looking ahead, isohexanone exists at a crossroads for specialty chemicals. The demand for adaptable solvents and synthetics in energy storage, surface coatings, and custom molecules isn’t fading. Progress will depend partly on more sustainable manufacturing—reduced emissions, lower energy input, tighter recycling of process streams. Advances in catalysis or electrochemical methods could reshuffle cost structures, tipping the balance in favor of isohexanone for niche but demanding markets. At the same time, safety and toxicity data from years of real-world experience help defend its place in the face of growing regulation. In hands-on labs and in bustling plants, people keep finding new uses or smarter ways to make the chemical, nudging it further from obscurity and toward an enduring, if quiet, relevance.
What is Isohexanone used for?
Why Isohexanone Matters
Isohexanone may not be a household term, but it plays a bigger part behind the scenes than most folks realize. This chemical shows up in industries that touch daily life far more than most people would guess. My own work in manufacturing sparked a curiosity about all the ingredients that fuel production. Way back, I noticed how solvents come up again and again on safety meetings, especially when talking about environmental and worker health. Isohexanone has its spot on that list.
Role in Paints and Coatings
Walk into any home improvement store and the shelves stock cans promising a smooth, glossy finish. Many of these products turn to isohexanone as a solvent. Manufacturers choose it because it dissolves ingredients needed for modern paints, making sure color distributes without streaks. Specialty automotive and industrial paints use it to keep finishes durable under stress. I once talked to a body shop owner who said that the right solvent choice avoids headaches, helping paint dry faster and lay down smooth, which matters both for appearance and repair work.
Importance in Adhesives
Ever assemble furniture and notice a glue that holds surprisingly well even after years? Some strong adhesives, especially those meant to perform under heat or pressure, use isohexanone to get their stickiness just right. The chemical helps create the right mix, making sure that the glue doesn’t set too quickly on the assembly line but bonds firmly once applied. During a summer job, I saw workers toss batches due to the wrong consistency, learned then how much hinges on solvent choices.
Pharmaceutical Applications
Production of some medicines—especially those in topical form—sometimes calls for isohexanone. It can help blend different active compounds so the medicine spreads evenly. Drug companies rely on the substance because it evaporates at a controlled rate, meaning leftover residue won’t interfere as much with sensitive compounds. Regulations place limits here, but the role remains clear: get reliable medicines to patients without unwanted by-products.
Environmental and Health Considerations
With any chemical, the health and environmental effects demand attention. Prolonged exposure to isohexanone vapors can cause headaches or irritation. Workers in facilities using the solvent have to use appropriate protective gear. Compliance officers monitor how much goes into wastewater or the air. The chemical industry isn’t perfect—accidents and leaks mean ongoing risk—so safety education and transparent records become crucial tools.
Calls for greener chemicals echo louder every year. I’ve seen small shops try out alternatives with fewer risks, though often these substitutes don’t perform quite the same. Groups such as the Environmental Protection Agency require reporting, pushing for improved engineering controls and better ventilation. No single policy will solve exposure permanently, but tighter regulations, investment in research, and worker training form a real path forward. Until eco-friendly options catch up, smart handling of isohexanone stands as the best shot at protecting both people and landscapes.
What is the chemical formula of Isohexanone?
Isohexanone in the Real World
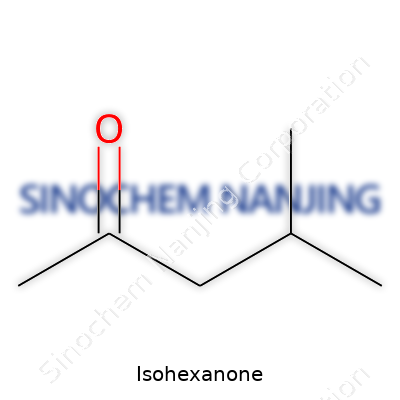
People come across names like “Isohexanone” and wonder what goes into that word. Chemists recognize it instantly as a structural isomer in the hexanone group, but others ask questions about its makeup, its everyday uses, and why accuracy matters in its formula. Isohexanone’s chemical formula is C6H12O, showing six carbons, twelve hydrogens, and one oxygen. The details go deeper because the term “iso” refers to a branch in the carbon chain, not just a plain straight line of atoms. For makers in the chemical industry, this difference affects everything from how the molecule reacts to the smells and volatility it brings into products.
Structure and Sources of Confusion
In university labs, there’s always that one debate about branched versus straight-chain molecules. Isohexanone’s family tree branches off at either the second or third carbon, forming isomers like 4-methyl-2-pentanone, which is often called methyl isobutyl ketone (MIBK). Many students learn to spot the subtle changes in the names, but ISO standards sometimes clash with casual usage. This mix-up spills over into industrial catalogs, academic journals, and even safety sheets, where the wrong name creates risks for everyone handling the substance.
Why the Exact Formula Matters
It’s easy to dismiss chemical names as technicalities. Yet mistaken formulas cause real trouble—fire safety, toxic exposure, and improper waste handling all rely on knowing exactly which molecule is present. Anyone who’s worked with chemicals knows the frustration of looking up a name and finding conflicting answers. Some databases label "Isohexanone" as 2-hexanone, which creates confusion, since 2-hexanone doesn’t have a branched structure. Mislabeling leads people down the wrong safety protocols, especially in storage or transport.
All it takes is one wrong number in a chemical formula to create an emergency. Years ago, I remember seeing a batch of mislabeled solvents nearly cause a chain reaction in a college storeroom. Luckily, a sharp-eyed lab tech caught the error. Since then, I’ve kept a notebook to double check every “iso-,” “sec-,” or “tert-” prefix on bottles. The lesson sticks: chemicals aren’t forgiving when humans get casual with their names or structures.
Importance Beyond the Lab
Outside specialty labs, Isohexanone helps in making fine chemicals and coatings. Safety data and handling rules rest on getting the simple formula right. This has practical implications: workers count on correct hazard ratings and flamibility warnings, and wastewater treatment depends on the molecule’s breakdown products. Even seasoned plant operators admit that misreading a label happens more often than they want to admit, thanks in no small part to inconsistent naming conventions in the supply chain.
Paths Forward for the Industry
Standardization offers one solution. Industry players could agree to reference only the IUPAC name on all labels and safety sheets, reducing the risk of misidentification. Training courses for chemical handling need to stress this habit, because seasoned pros and new workers alike get tripped up by ambiguous shorthand. Online databases should update synonyms regularly and display a clear structural diagram beside every entry. Chemists can be proactive as well, cross-referencing formulas and educating peers, making sure that nobody confuses one ketone for another.
Final Thoughts on Responsibility
Getting the formula right matters far outside academic circles. It’s about workplace safety, environmental responsibility, and public health. From experience, a moment’s diligence reading the small print on labels and formulas pays off in the long run. Everyone, from technicians to chemical engineers, shares the job of dealing carefully with complex organic molecules like Isohexanone—because in chemistry, details always matter.
Is Isohexanone hazardous or toxic?
Understanding Isohexanone in Everyday Settings
Isohexanone pops up in labs and factories all over the world. As a solvent, it makes life easier for folks working in paints, coatings, adhesives, and cleaners. You won’t spot it on a grocery store aisle, but plenty of products rely on it behind the scenes. Some workers cross paths with it every day.
Looking at The Risks
Take a whiff of Isohexanone for the first time, and it’s pretty obvious you’re dealing with a strong chemical. Studies show inhalation at high concentrations irritates the nose and throat. Skin contact usually leads to redness or dryness. I’ve seen this first-hand in industrial shops: gloves and goggles become the unspoken uniform. Accidentally handling it without protection, people tend to regret it.
The bigger concern comes from repeated or heavy exposure. Prolonged breathing of vapors can bring headaches, dizziness, or even trouble with coordination. The U.S. National Institute for Occupational Safety and Health (NIOSH) points out those dangers, especially for workers in closed-in spaces. This isn’t just caution run wild; industries following strict safety protocols usually avoid sick days and ER visits related to solvent fumes.
What The Science Says
Toxicity always depends on dose and duration. On its own, Isohexanone’s acute toxicity isn’t shocking compared to some solvents, but that doesn’t give a free pass. The International Chemical Safety Cards (ICSC) lists possible organ damage with frequent, unprotected exposure. According to the European Chemicals Agency, short-term exposure rarely threatens life, but long-term risks include nervous system effects, especially if someone works daily in poor ventilation or skips personal protection.
It doesn’t take a chemist to see patterns. Flushed faces, headaches, and slowed response times have shown up in case reports where workers handled solvents like Isohexanone. Even diluted forms spread fumes. Some folks might look for signs of carcinogenicity. So far, the available research hasn’t labeled Isohexanone as cancer-causing, but research remains ongoing since chronic effects sometimes take years to uncover.
Practical Steps to Stay Safe
Keeping workers safe means taking the basics seriously. Gloves, goggles, and masks offer real protection, not just a suggestion. I remember a warehouse manager who set up fans and open windows along with regular breaks for anyone handling solvents. He cut down complaints about sore throats and skin irritation just by paying attention to air flow. Safety data sheets recommend storing Isohexanone away from heat and using it in well-ventilated spots, which lines up with what works in practice.
For companies, investing in proper storage, routine air monitoring, and training brings returns far beyond compliance. I’ve seen operations grind to a halt over health and safety claims, but proactive measures keep teams working and morale high. On the personal side, reporting odd symptoms and not shrugging off even mild reactions set the stage for better workplace health.
Towards Better Industry Standards
Chemicals like Isohexanone aren’t likely to disappear from factories soon, but better awareness matters. Making hazard data public, encouraging transparent reporting, and not letting cost-cutting outweigh safety pave the road ahead. We owe it to people getting their hands dirty every day to offer more than just warnings on a label. The most responsible players don’t just follow rules; they rethink and improve safety for everyone down the line.
It’s clear that Isohexanone doesn’t belong on bare skin or inside our lungs, but respecting its real risks keeps a tough job from becoming a dangerous one.
What are the storage and handling recommendations for Isohexanone?
Why Proper Storage Matters
Walking through a chemical storeroom, you notice which drums get extra attention. Isohexanone grabs the spotlight for good reason. Its fumes catch fire easily and, mixed with the wrong chemical, things can go south fast. So, the real question isn’t just about rules and codes—it’s about keeping people, property, and products safe.
Choosing the Right Place
Isohexanone belongs nowhere near heat or open flames. Stashing it in a cool, dry spot pays off. A space with steady airflow keeps leaks or spills from turning dangerous. In my early days handling solvents, I learned that steel drum lids sometimes sweat from temperature swings. A shaded, temperature-controlled locker keeps the container dry, especially when humidity spikes. Real-world storage goes beyond simply ticking the right box; it means checking for leaks and making sure no broken container sneaks past inspection.
Don’t Let Vapors Linger
Nobody wants to walk into a warehouse smelling sharp solvent. Isohexanone vapors settle in low-lying spaces and build up into a hazard. I’ve seen how quickly an overlooked vent or fan stops working—a reminder that mechanical ventilation does more than push air around. Installing exhaust fans close to floor level helps move fumes out of the room, not just around your head. Gauges and sensors can back up the nose test, and using them stops problems before staff feel the effects.
Handling with Care
Spills can mess up a good day fast. Keeping spill kits nearby matters, but training people so they don’t freeze up when something sloshes over makes all the difference. My team learned to use absorbent pads and not reach for water, which only spreads the mess. Chemical gloves, splash-resistant goggles, and a chemical apron hold off splatters. It’s not just about ticking boxes; knowing your gear is fresh and not cracked or brittle saves skin and sight.
Keeping Records Up to Date
People get complacent around old friends, and chemicals you work with every week become background noise. That’s where tracking comes in. Keeping a current log of how much isohexanone comes in and out helps you spot unexpected losses that could point to evaporation, leaks, or even pilfering. Inventory checks once a month catch problems before they turn into headaches with insurance or workplace safety orders.
Fire Prevention Works in Layers
Some think a fire extinguisher parked by the aisle will do the trick. Foam or CO₂ extinguishers stand ready by storage racks, but grounding every metal drum and using non-sparking tools add extra margins of safety. In busy operations, workers sometimes drag pails or pry lids with steel bars, risking static discharge. Wooden pallets and brass tools work better since they don’t build up sparks. In my experience, most incidents come from shortcuts. Taking the extra minute to follow each step means everyone goes home safe.
Looking Ahead
The tools and guidelines for working with isohexanone have been shaped by trial and error. Better labeling, easier emergency wash stations, and regular retraining keep the risk low. Updating standard operating procedures pays off more than fancy equipment upgrades. People adapt quickly if they see why the process matters. Years in the industry taught me that attention to detail isn’t just paperwork; it’s a habit that keeps accidents rare.
Where can I purchase Isohexanone?
The Basics of Buying Isohexanone
Isohexanone falls into a category of chemicals used in different manufacturing and research settings. Some folks in coatings, flavors, or specialty solvents look for this compound, usually because they’ve got a specific project or process that calls for it. Getting your hands on it isn’t the same as picking up aspirin from the corner drugstore. Chemicals like isohexanone require a bit of know-how, a bit of paperwork, and often, a bit of waiting.
The Usual Marketplaces
Most of the time, direct sales go through chemical suppliers. Think of names like Sigma-Aldrich, Fisher Scientific, or ThermoFisher. These companies have built reputations on reliability, safety standards, clear documentation, and verified sourcing. They vet their buyers. If you run a lab, manufacture coatings, or develop pharmaceutical intermediates, chances are, you’ll work with one of these distributors. They want to know that buyers understand the hazards and proper uses that come with chemicals like isohexanone.
What Distributors Need
Before a distributor ships out a chemical, there is a typical process. They’ll request documents—a business license, details about your intended use, and proof of technical capability. They might even want a statement that you won’t misuse or resell the chemical in ways that dodge safety rules. These steps come from a real need to keep dangerous substances from being misused.
Why Regulated Access Exists
Governments worldwide keep a close watch on certain chemicals. Sometimes it feels bureaucratic, but history shows why careful tracking matters. Chemicals leak into the wrong hands. Lax sourcing policies in the past have led to environmental disasters and unsafe working conditions. Those events shape today’s rules in the chemical industry.
In my own experience, buying chemicals for a research lab, patience and paperwork went hand-in-hand. Companies didn’t care if I was in a hurry—they cared about where each drum ended up. One time, a missing MSDS form stalled a whole experiment. If safety slipped, said the supplier rep, they’d rather lose a sale than risk an accident. Years down the line, that attitude sticks with me. Consumer and employee safety come first.
Pricing and Transparency
Isohexanone pricing does not land in a public catalog. Prices swing based on volume, region, and the purity grade. Expect a quote instead of a fixed sticker price. Ask for a certificate of analysis to see exactly what you’ll get. A reputable supplier won’t hesitate to walk through testing or traceability details with you.
Alternatives and Local Laws
Some buyers look for alternatives, especially if isohexanone starts running low or gets expensive. Ask the supplier about structural substitutes that might fit your application. Rules change place to place—some states or countries block certain solvents outright, so local regulations rule the day.
Moving Toward Clearer Pathways
Building a good relationship with a trustworthy distributor pays off. Over time, the process of buying specialized chemicals gets smoother. Push for more transparency in the supply chain and support local compliance. Everyone in the loop stays safer and more informed. If your own line of work calls for these solvents, advocate for responsible sourcing and honest conversations about risks. That’s where real progress happens.
| Names | |
| Preferred IUPAC name | 3-Methylpentan-2-one |
| Pronunciation | /ˌaɪ.soʊˈhɛk.səˌnoʊn/ |
| Identifiers | |
| CAS Number | 4749-02-2 |
| Beilstein Reference | 1900896 |
| ChEBI | CHEBI:89339 |
| ChEMBL | CHEMBL16335 |
| ChemSpider | 17119 |
| DrugBank | DB15903 |
| ECHA InfoCard | DTXSID3058704 |
| EC Number | 255-597-1 |
| Gmelin Reference | 8777 |
| KEGG | C21189 |
| MeSH | D017750 |
| PubChem CID | 12508 |
| RTECS number | KM5075000 |
| UNII | M4R7T977G2 |
| UN number | UN3295 |
| CompTox Dashboard (EPA) | DTXSID5078925 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | sweet |
| Density | 0.801 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.6 hPa (20 °C) |
| Acidity (pKa) | 20.6 |
| Basicity (pKb) | 0.70 |
| Magnetic susceptibility (χ) | -7.72×10⁻⁶ |
| Refractive index (nD) | 1.405 |
| Viscosity | 2.51 mPa·s |
| Dipole moment | 2.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 240.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.3 kJ mol-1 |
| Std enthalpy of combustion (ΔcH⦵298) | -3906.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H336 |
| Precautionary statements | P210, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 1-2-0-NFPA |
| Flash point | 39°C |
| Autoignition temperature | 456°C |
| Explosive limits | Explosive limits: 1%–7% |
| Lethal dose or concentration | LD50 (oral, rat): 7460 mg/kg |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | 500 ppm |

