Isobutyric Acid: A Closer Look at Its Journey and Place in Modern Industry
Historical Development
The story of isobutyric acid stretches back to chemistry’s early years, when European chemists explored volatility and the world of organic acids. Discovered while investigating the components of butter and fermented products, isobutyric acid quickly became more than just a curiosity. What began as an exploration by pioneers such as Justus von Liebig gained new traction with advances in distillation and identification of carboxylic acids. The industrial revolution made raw materials and purification tools accessible, so laboratories and later factories tapped into the economic potential of chemical building blocks like isobutyric acid. No longer just a byproduct in a bottle on the shelf, it entered the global market, forming a foundation for solvents, flavors, and industrial syntheses.
Product Overview
You only need to walk through a chemical plant or a fragrance manufacturing unit to stumble on isobutyric acid in one form or another. Its recognizable odor—sharp and somewhat sweet at low concentrations—speaks to its duality as both a warning and an asset. Sold mostly as a clear, colorless liquid, isobutyric acid fills a critical niche. Chemists value its solubility and reactivity, while manufacturers use it for everything from making plasticizers to boosting flavors in beverages. Its presence extends beyond obvious industrial boundaries, showing up in pharmaceuticals and even as a feed additive.
Physical and Chemical Properties
Isobutyric acid, or 2-methylpropanoic acid, sports a three-carbon backbone with a methyl group at position two and a carboxyl group on the end. Its boiling point of about 155 degrees Celsius and its sharp, unpleasant smell (much like old sweat socks at strong concentrations) offer practical ways to identify it, though not always pleasant ones. Its miscibility with organic solvents and limited, but not negligible, water solubility make it flexible for formulation across laboratories. The acid’s density and vapor pressure provide clues about storage and use, demanding attention to ventilation and containment, especially in enclosed spaces.
Technical Specifications and Labeling
In practice, nobody can just slap “isobutyric acid” on a container and call it a day. Chemical suppliers define purity grades based on application, whether it’s food, pharma, or general industrial. Regulations like the Globally Harmonized System dictate classification details—corrosive, hazardous, flammable liquids. Labels must spell out hazards, storage advice, and handling cautions, aligning with national safety standards from OSHA, the EU, or local authorities. Chemical purity (often above 98% for laboratory use) determines price and suitability. Impurities, though minor, can become deal-breakers in pharmaceutical synthesis, where even parts-per-million can derail a batch and saddle a company with lost revenue or recalls.
Preparation Methods
Early on, most isobutyric acid was dug out of natural fermentation products, but modern supply chains rely on chemical synthesis. The hydrolysis of isobutyronitrile or the carboxylation of isobutyl chloride with carbon dioxide take top billing. These routes offer consistency and scale, something fermentation can only dream about in today’s manufacturing landscape. As feedstock prices shift, companies may switch between synthetic methods or piggyback on downstream byproducts from other large-volume chemicals. Efficiency, yield, and raw material costs drive research into new catalytic systems, especially as the world gets serious about both reducing greenhouse gas impacts and waste streams.
Chemical Reactions and Modifications
Isobutyric acid doesn’t like to sit idle. Chemists turn it into a litany of derivatives: esters for flavors and fragrances, acid chlorides for pharmaceuticals, amides for more advanced intermediates. The acid’s reactivity lies in the carboxyl group, which opens up paths to polymer building blocks and agrochemical design. Nitration, esterification, and halogenation reactions add layers of complexity while supporting industries from flexible plastics to long-lasting coatings. This versatility, paired with known and reliable reaction mechanisms, keeps isobutyric acid entrenched in labs and plants alike.
Synonyms and Product Names
Like many chemicals, isobutyric acid wears a few hats. You might spot it in literature as 2-methylpropanoic acid or isobutanoic acid. Product listings may abbreviate it as IBA. Although different languages and suppliers play with brand names, the CAS number 79-31-2 puts all debate to rest in scientific circles. For buyers or researchers, cross-referencing synonyms becomes a basic but important practice to avoid disastrous mix-ups, especially when inventory managers deal with thousands of chemicals.
Safety and Operational Standards
You don’t need a catastrophic spill to appreciate the importance of good safety habits. I’ve watched operators in the field, decked out in splash-proof goggles and gloves, always keeping ventilation in mind. Isobutyric acid volatilizes easily, so inhalation risks mix with the danger of skin burns. Occupational exposure limits, emergency shower stations, and proper training save time and money down the road, as well as keep staff from long-term health impacts. Facilities run regular drills in containment and spill control, a routine shaped as much by past horror stories as by compliance inspectors. Comparing lab grade acid with bulk industrial product underlines one thing—safety standards don’t bend, even where purity might.
Application Area
My work in food science introduced me to isobutyric acid’s softer side: small amounts add character to cheese and help mimic natural flavors. Acid esters, particularly isobutyrates, lift fruit flavors and perfume notes. Walk into a livestock operation, though, and you’ll catch it on ingredient lists meant to preserve silage or tweak animal digestion. Its hand in pharmaceutical syntheses runs deeper than most realize, popping up in everything from intermediate reactions to active components. As paint additives, solvent blends, and plasticizer ingredients, it quietly supports sectors as far-reaching as adhesives, blood bags, and even automotive interiors. One chemical, many faces.
Research and Development
R&D hasn’t stood still around isobutyric acid. Universities and chemical companies keep searching for better, cleaner synthesis using renewable sources. Metabolic engineering in bacteria, yeast, or even algae promises sustainable production, provided yields and separation hurdles get solved. The focus extends to downstream derivatives, with companies eager for more efficient reaction conditions, greener solvents, and better catalyst lifetimes. In flavor science, new biosynthetic routes are tested to skirt regulatory hoops and meet “natural flavor” definitions. Patent filings reflect this shift: a steady uptick in enzymatic synthesis, recycling of byproducts, and integration with circular economy models all point to a future less dependent on fossil resources.
Toxicity Research
Any chemical with a sharp smell and reactivity deserves respect. Toxicologists have a long track record with isobutyric acid, charting acute inhalation, dermal, and ingestion risks. Animal studies point to mild to moderate toxicity, causing irritation to skin, eyes, and respiratory tracts. Chronic data remain thinner, but workplace exposure records rarely link isobutyric acid to long-term health conditions in properly managed environments. Still, regulations don’t loosen up; food and feed applications rely on tight maximum limits, with regular monitoring backed up by batch testing and certificate review. On the environmental front, studies find moderate biodegradability and low bioaccumulation, suggesting careful handling at scale rather than outright bans.
Future Prospects
Looking ahead, isobutyric acid stands as a crossroads for old-school industrial chemistry and the new demand for sustainable raw materials. Continued improvements in fermentative production and catalytic refinement fit with trends in green chemistry and circular supply chains. Flavors and fragrances face growing demand for natural labels, offering growth for biosynthetic isobutyric acid as regulators and consumers get more choosy. Specialty plastics and coatings look for new monomers to balance flexibility with environmental responsibility, and isobutyric acid derivatives play a part here. Some companies test its use in newer applications, like electronics or biodegradable polymers, where traditional feedstocks fall short. Everything hinges on efficiency, price, and the growing environmental pressure to turn old petrochemical staples into renewable, low-impact solutions—even for something as once humble as isobutyric acid.
What is Isobutyric Acid used for?
From Harsh Smells to Daily Staples
Anyone who has cracked open a commercial food flavoring bottle probably knows that sharp, almost sour scent drifting up the nose. That’s isobutyric acid—hard to forget once you get a whiff. This strong-smelling compound pops up in places few expect, and sometimes it plays a role in the very experiences that flavor our daily routines or preserve items longer than nature intended.
Food, Feed, and That Tangy Note
Chemists often use isobutyric acid to help create flavors in the food industry. The acid itself rarely lands straight into someone’s recipe, but manufacturers use it—sometimes as a key building block—to make flavoring agents. Ever eaten a snack and wondered how an “artificial cheese” or “buttery” profile tastes so nostalgic or strong? Isobutyric acid often plays a part in these flavoring mixtures.
It doesn’t stop there. Livestock feed sometimes relies on additives like salts of isobutyric acid. These components can help suppress harmful bacteria in stored feed, giving farmers some peace of mind when it comes to their animals’ food safety. As a kid spending summers on a relative’s farm, I remember the importance of preventing mold in the silos. Preservatives like these managed to lower spoilage, and that helped stretch feed supplies through unpredictable weeks.
Making Medicines and Crafting Chemicals
Medication developers need predictable chemical reactions, and isobutyric acid fits that need since it’s a handy “starting block” in pharmaceutical chemistry. It appears in synthesis for certain drugs, like some anti-fungal agents. Knowing this, pharmacists and researchers often pay attention to quality and purity in their source materials. Sourcing a consistent supply matters. Subpar batches can disrupt entire production lines, raising costs and affecting lives. The acid also serves as a “building stone” when chemists build larger or more complex molecules, from perfumes to textiles.
Cleaners, Solvents, and The World of Smell
Isobutyric acid shows up in solvent production too. Solvents help remove that stubborn glue residue from old stickers or clean lab glassware. Despite its sharp scent, the acid’s chemical properties aide in dissolving substances tough to wash off with regular soap and water. A good solvent saves time, prevents mistakes, and keeps workspaces safer.
I once spent a summer patching up gear in a college chemistry storeroom. Without tailored solvents—many containing derivatives related to isobutyric acid—tools wouldn’t come clean, and glassware didn’t stay clear enough for precise experiments. It may seem invisible to most, but the benefits are felt almost everywhere: in clean labs, better-smelling rooms, and in chemical processes that lead to construction materials or simple office supplies.
Risks, Regulations, and Smart Handling
Pungency aside, isobutyric acid can irritate skin and eyes. Chemical producers in the U.S. and Europe treat this seriously, requiring safe handling instructions and protective gear. Government agencies keep tabs on workplace concentrations and waste disposal to protect both workers and the environment. Failing to respect these rules causes trouble—poorly ventilated spaces mean higher risk for headaches or worse health issues down the line.
Plenty of industries still rely on isobutyric acid because alternatives with fewer side effects aren’t always available or cost-effective. Research keeps pushing for safer options wherever possible, but for now, smart usage, tight regulation, and better protective practices give workers, food producers, chemists, and even consumers a fair shot at the benefits with lower risk.
What are the safety precautions when handling Isobutyric Acid?
Understanding the Risks on the Ground
I still remember the sharp, sour smell of isobutyric acid from my college chemistry lab. One small spill turned the air into something you never forget—pungent enough to sting your nose but not immediately threatening unless you ignored it. It’s these real-life experiences that bring home the truth: this chemical isn’t something to shrug off. The stuff can burn your eyes and skin, cause a sore throat, or make you dizzy if you breathe in too much of it.
Why Safety Steps Matter
All it takes is a single misstep to turn a simple transfer or measurement into a trip to the nurse. Isobutyric acid will eat through gloves that aren’t up to the task or vaporize just as you open a poorly sealed bottle. Acid burns heal slow. Breathing it in regularly, even if you don’t notice right away, can add up to long-term health problems. In factories and school labs alike, accidents cost time and money, and more importantly, they threaten the people you work alongside every day. The Centers for Disease Control and Prevention (CDC) points out that even low-level exposure in the air can irritate lungs and trigger asthma. OSHA keeps a close eye on the ways acids like these hurt workers—so much so that safety data sheets (SDS) are required close by at all times.
Practical Steps for Everyday Safety
Good ventilation keeps everyone breathing easy. Fume hoods aren’t just a fancy piece of equipment—a quality fan system actually pulls dangerous vapors away before they get near your lungs. I once tried working with the hood sash open wider than it should have been. I paid for it with watery eyes and a cough that lingered all day.
Gloves, goggles, and lab coats aren’t up for debate. Splash-proof goggles cost less than lunch, and they’ve saved my eyesight more than once. Nitrile gloves form a good barrier, but they don’t work forever. Skin absorbs chemicals. Replace gloves if they show any weak spots, and never touch your face during a procedure. Spills on clothing call for a quick change, not just a wipe-down with a paper towel.
Storage rules protect more than bottles on a shelf. I’ve seen isobutyric acid eat through cheap plastic, so only quality glass or acid-resistant containers make sense. Keep everything labeled with big, clear letters—mistakes in storage can lead to dangerous surprise reactions. No one benefits from guessing which clear liquid sits in a corner flask.
Know Your Exits—And Your First Aid
Emergencies happen so fast you can barely think. Eye wash stations and safety showers are no joke. Practice using them, so your body moves before your brain panics. Exposure to isobutyric acid calls for fast rinsing under cool running water—at least 15 minutes. Medical help comes next, even if it feels minor. Inhalation means fresh air right away. You don’t walk it off: if breathing gets tough, it’s ambulance time.
Building a Culture of Respect for Chemicals
Safety never comes from just reading a handbook. You feel it in the routines: double-checking labels, keeping gloves handy, and having someone trained near you every time you pour even a milliliter. Open conversations about near-misses help everyone get sharper. The Environmental Protection Agency (EPA) urges transparent sharing of hazardous incidents at all levels, knowing that lessons today save injuries tomorrow.
I’ve seen careful attention pay off—not just in accident-free semesters, but in folks leaving work healthy and confident. Isobutyric acid earns respect through experience, consistent training, and smart habits. If you treat it like just another bottle, it will remind you otherwise.
What is the chemical structure and formula of Isobutyric Acid?
Chemical Structure and Formula of Isobutyric Acid
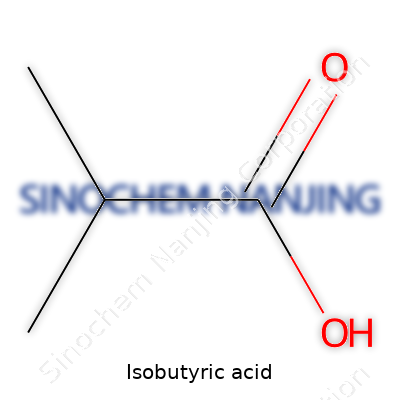
Isobutyric acid goes by the systematic name 2-methylpropanoic acid. Its chemical formula is C4H8O2. This means each molecule carries four carbon atoms, eight hydrogens, and two oxygens. The structure shows a short, branched chain. At the center, three carbon atoms line up. One methyl group sticks out from the middle carbon, giving the “iso” prefix. The molecule ends with a carboxylic acid group (COOH). So, the structural formula looks like this: (CH3)2CHCOOH.
This branching gives isobutyric acid properties that set it apart from its straight-chain cousin, butyric acid. In labs and real-world settings, these small changes in atomic arrangement can create big shifts in a substance’s smell, color, and how it reacts. Isobutyric acid smells like sweaty feet or cheese, a scent not unusual in certain food manufacturing or fermentation setups. But beyond the smell, its simple formula packs a punch in several fields.
Applications and Why Structure Matters
One of the first things that comes to mind with isobutyric acid is flavor science. Cheesemakers, for example, work around the nuanced aromas the acid helps create. The molecule’s branching points make it less acidic than straight-chain alternatives. In practice, I’ve seen labs choose isobutyric acid when they’re hunting for the right touch in artificial flavorings—especially for butter, cheese, or yogurt analogs.
Chemical manufacturers lean on isobutyric acid for synthesizing esters. Esters derived from it often land in fragrance or flavoring formulas because they’re fruity and pleasant—strikingly different from the pure acid’s smell. Get enough chemistry people in a room, and sooner or later someone brings up its use in pharmaceuticals, either as a precursor or as part of an active molecule. The branched structure opens pathways for further modification, flexibility that’s hard to come by in straight-chain compounds.
Health and Environmental Considerations
Even though isobutyric acid pops up in food products, it doesn’t mean safety isn’t a concern. Anyone who’s spent time handling it in a lab will remember its strong, pungent odor—ventilation helps. While it’s not considered exceptionally toxic, direct exposure can irritate skin, eyes, and the respiratory tract. Industry teams working with it typically use gloves and eye protection as a rule. Accidental spills don’t hang around for long; the acid evaporates quickly at room temperature, but properly designed storage prevents unwanted exposure and waste.
Looking at Challenges and Solutions
Production of isobutyric acid often relies on petrochemical sources. Finding renewable or less polluting feedstocks could lower the environmental impact. Certain companies now explore biosynthetic routes using engineered microorganisms to create isobutyric acid from plant-based sugars. These efforts are picking up pace, driven by a mix of cost savings and tighter regulations concerning hazardous emissions.
Safe and efficient transport matters too. Isobutyric acid needs secure containment to prevent leaks, which can affect air quality and pose hazards for workers. Training handling teams in safe transfer and emergency procedures makes a big difference. Sharing best practices among facilities supports a safer workplace and stronger environmental stewardship.
Breaking down the structure and real-world roles of isobutyric acid shows that even a small molecule can leave a large footprint—from the flavor in dairy products, to advances in green chemistry, and the careful steps needed to manage health and safety.
How should Isobutyric Acid be stored?
Thinking About Safety First
Anyone who’s spent a day in a chemical storeroom knows that small oversights turn into big problems, especially with volatile organics like isobutyric acid. Its sharp, sour smell hits your nose the second the cap twists off, hinting at the potent vapor lurking inside. No one who has ever cleaned up a spill wants to see it drip across a workbench twice. It makes you alert to storage details, not out of paranoia, but out of respect for the risks.
Keeping It Cool and Contained
You don’t need a high-tech vault. Isobutyric acid likes simple, proven solutions. A dry, ventilated area keeps it stable. The fewer temperature swings, the better. Heat ramps up vapor release fast, and these fumes irritate eyes and throats long before concentrations get dangerous. Set it away from windows and away from heaters; even regular fluorescent lights shouldn’t sit too close. Airflow does more than keep workers comfortable—good ventilation carries away any fumes that creep past the lid.
Choosing the Right Containers
The acid chews right through some plastics. I’ve watched it cloud a cheap bottle within days. If you don’t want container failures, reach for the glass or high-density polyethylene. Seals matter—a loose cap means a whole room smells like gym socks, and it signals risk for accidental exposure or even combustion. Vapors spread stealthily. Tight caps and honest labeling help everyone know what they’re dealing with.
Respecting Compatibility
Chemicals behave like cranky neighbors. Store isobutyric acid away from oxidizers like nitric acid and active metals. Placing acids near bases—like old ammonia drums—invites trouble. I’ve seen worst-case scenarios described in safety reports, but a few sturdy barriers in the storeroom have saved my colleagues and I from surprise reactions. Don’t trust your luck on this front.
Fire Risk and Spill Cleanup
Flashpoints matter. Isobutyric acid doesn’t wait for a direct flame; even static sparks can do the job in the wrong conditions. Keep it far from ignition sources, and post clear labels about its flammable nature. I don’t trust cheap absorbents—an old, reliable spill kit with pads designed for acids works best. Rags and “just water” will only complicate things. Employees need regular refreshers on cleanup, not just once during orientation. Every time someone practices, the response gets sharper.
Training Makes All the Difference
No fancy system beats a crew who understands the rules and the reasons behind them. OSHA rules back up every point: clear signage, written inventory lists, emergency eyewash stations near storerooms. Take time to walk through protocol once a month, not just after an accident. No one complains about a safety check when it keeps everyone out of the hospital.
Review and Improvement
A few tweaks turn a risky shelf into a safe storage spot—good airflow, proper labeling, solid containers, handling knowledge, and a respect for how strong even a small bottle can be. Pay attention to these steps, and accidents won’t become the day’s story.
What are the potential health effects of exposure to Isobutyric Acid?
Getting Past the Chemistry: What Sort of Trouble Can Isobutyric Acid Cause?
Anyone who’s spent time around certain food plants or chemical warehouses knows the punchy smell that drifts out. Isobutyric acid provides part of that sour, unpleasant scent. It turns up in manufacturing, flavoring, and even some cleaning solutions. Most folks won’t bump into it unless their job connects with labs or production floors, but exposure risk rises if safety steps slip.
The Short-Term Impact: Eyes, Nose, Lungs Pay the Price First
No need for scientific jargon here: isobutyric acid stings. A whiff in the air irritates noses and throats. Too much, and headaches land before you’ve even swallowed your coffee. Workers unlucky enough to spill the liquid on skin report itching, burning, or even blisters. The acid vapor in close quarters gets into the eyes, leaving them red and streaming. These effects show up quickly, so staying alert makes a difference.
Breathing in the acid’s vapor won’t only make you cough. Overexposure leads to chest tightness, shortness of breath, and the kind of raw lungs that drag on for days. Folks with asthma or bronchitis may notice symptoms ramp up quickly. Years ago, I watched a maintenance crew ignore an exhaust fan during a tank transfer. A couple of team members spent the afternoon wheezing just from a few moments of direct exposure.
Longer Haul Risks Deserve Attention
Most cases involve nothing more than a miserable day at work, but routine, unchecked exposure poses unknowns. Studies have not pinned down any cancer links, but scientists recognize that chronic irritation can cause ongoing damage to lungs and skin. Some animal studies hint at liver effects if large doses stick around the body long enough. People with frequent contact need regular monitoring just to rule out creeping health shifts.
Stopping Trouble Before It Starts: Practical Steps That Work
Engineering controls make a difference. Good fume hoods or exhaust fans chop down vapor levels in the air. Simple gloves, eye protection, and a sturdy lab coat build a barrier for workers. Training matters, too. In my experience, nobody wants a chemistry lesson until a rough shift puts someone in the emergency room. Showing crews where hazards lurk and how fast symptoms develop makes protocols stick.
Leaky vessels and sloppy handling stack up the risk. Routine inspection and disciplined storage take extra minutes upfront, saving a load of trouble. Clean-up kits placed wherever acid moves in bulk speed up the response. Pretending spills won’t happen gets people in trouble fast.
What Everyone Should Know
Isobutyric acid doesn’t make headlines. Still, those relying on safe work routines hold the real line between an ordinary day and an incident that rattles an entire team. Calling out every irritation and odd smell feels tedious, but that vigilance pays off. Health authorities suggest quick reporting of symptoms and full medical checks for anyone exposed in quantity. Isobutyric acid is yet another case where preparation and respect for the material prove worth every second and dollar spent.
| Names | |
| Preferred IUPAC name | 2-methylpropanoic acid |
| Other names |
2-Methylpropanoic acid Isobutanoic acid Isobutyrate |
| Pronunciation | /ˌaɪsoʊˈbjuːtɪrɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 79-31-2 |
| 3D model (JSmol) | `Isobutyric Acid JSmol string`: ``` Isobutyric acid CC(C)C(=O)O ``` |
| Beilstein Reference | 2804220 |
| ChEBI | CHEBI:40968 |
| ChEMBL | CHEMBL418182 |
| ChemSpider | 546 |
| DrugBank | DB01998 |
| ECHA InfoCard | 100.003.488 |
| EC Number | 201-148-0 |
| Gmelin Reference | 577 |
| KEGG | C02390 |
| MeSH | D007533 |
| PubChem CID | 6579 |
| RTECS number | NI3325000 |
| UNII | E15L85T59K |
| UN number | UN1214 |
| Properties | |
| Chemical formula | C4H8O2 |
| Molar mass | 88.11 g/mol |
| Appearance | Colorless liquid with a penetrating, unpleasant odor. |
| Odor | Unpleasant, rancid, cheesy |
| Density | 0.962 g/cm³ |
| Solubility in water | 7.5 g/100 mL (20 °C) |
| log P | 0.93 |
| Vapor pressure | 3.1 mmHg (20°C) |
| Acidity (pKa) | 4.86 |
| Basicity (pKb) | pKb ≈ 10.67 |
| Magnetic susceptibility (χ) | -46.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 2.05 mPa·s (at 25 °C) |
| Dipole moment | 1.59 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -389.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −2305.5 kJ/mol |
| Pharmacology | |
| ATC code | A07AB08 |
| Hazards | |
| GHS labelling | **GHS labelling of Isobutyric Acid:** `GHS02, GHS07, GHS05` |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H314, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P270, P273, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P311, P312, P330, P337+P313, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-Acid |
| Flash point | 68 °C (closed cup) |
| Autoignition temperature | 464 °C |
| Explosive limits | 1.7–10% |
| Lethal dose or concentration | LD50 oral rat 2460 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2,946 mg/kg (rat, oral) |
| NIOSH | NM0350000 |
| PEL (Permissible) | PEL: 50 ppm |
| REL (Recommended) | 200 mg/m³ |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
n-Butyric acid Isovaleric acid Propionic acid Valeric acid Acetic acid |

