The Unseen Impact of Isobutyl Propionate: Tracing a Chemical’s Journey
The Roots of Isobutyl Propionate
Looking at the shelves in any lab, most people probably don’t give a second thought to an ester like isobutyl propionate. It didn’t always have a place in the world’s supply chain. Its history tracks closely with industrial growth in the last century, sprouting from efforts to harness the properties of carboxylic acids and alcohols. Chemists have learned volumes since the days of rudimentary solvents and lacquers. In the bustle of chemical innovation after World War II, researchers recognized that reactions between isobutyl alcohol and propionic acid didn’t just yield mundane byproducts, but gave rise to substances with sweet aromas and useful volatility. This time marked a shift, where chemists steered research toward the practical, looking for molecules with a clear edge: pleasant odor, manageable vapors, and compatibility with growing industries.
The Face of Isobutyl Propionate
People outside chemistry circles often ignore what sits behind the names. Isobutyl propionate brings a mildly fruity scent, almost reminiscent of pears. It appears as a colorless liquid, easy to pour, with a low viscosity. Its physical nature gives it an edge in coatings, fragrances, and wide-ranging industrial tasks. On paper, it boils below 150 Celsius, melts far below room temperature, and blends well with standard organics. These properties aren’t just numbers; they shape real-world choice, making this molecule useful in products we all touch—literally and figuratively.
Understanding Reactivity and Modifications
This ester doesn’t just sit there; it reacts in typical ways, especially under strong acid or base. It can get hydrolyzed back into its raw materials or serve as a launching point for new esters or amides. Chemists tinker with it, looking for tweaks that improve evaporation rates or stability. Small changes in chains or ring structures can tune performance, but the practical chemistry always involves a trade-off between what the chemicals want to do and what operators can handle without unnecessary risks. What appeals to a coatings chemist often looks just as good to someone blending perfumes.
Talking Safety and Labels
With every container, there’s more than a barcode and a name. Labels warn about flammability and inhalation. A bottle of isobutyl propionate won’t explode into flames from a gentle touch, but it burns quick if ignited. In concentrated form, the fumes can irritate eyes and lungs, as many volatile organics do. Regulators won’t let this kind of solvent float around unnamed—labelling is precise, with instructions calling for gloves, goggles, and solid ventilation. These aren’t just words on a page; look at workplace injury statistics for solvent-exposed workers and the value of proper warnings becomes crystal clear.
How Chemists Make It
Preparation usually follows the traditional esterification road: react isobutyl alcohol with propionic acid in the presence of a catalytic acid. Water removal pushes the reaction to completion. Operators constantly adjust temperature and acid concentration, watching for the right balance between yield and speed. Much of this work gets automated today, but a good reaction still comes down to steady control, decent raw material, and knowing which byproducts are likely to form. Not surprisingly, process engineers keep searching for greener methods—fewer emissions, safer solvents, steps and catalysts sourced from bio-based feedstocks. This challenge grows as new regulations pressure industries to clean up their acts.
Where the Names Change
On ingredient lists, the same chemical hides behind names like propionic acid isobutyl ester or simply isobutyl propionate. There’s nothing mysterious about synonyms; they reflect different naming conventions, regulatory histories, or the whimsy of industry insiders. Reading a safety data sheet or a fragrance compound summary can feel like a translation task, but it’s just a reminder that language—not chemistry—gets in the way of recognition.
Real-Life Uses
From the world of paints to perfumes, isobutyl propionate holds a spot that many never notice. It dissolves resins in specialty coatings, brings volatility to fast-drying polishes, and adds a subtle fruitiness to fragrances and flavors. Few people ever realize how these seemingly small molecules help balance performance in everything from nail polish to printing inks. Chemists like me spend hours matching properties—solubility, evaporation rate, and odor properties—to precise uses. Pick the wrong ester, and paints smear or dry too slow. Choose well, and performance jumps up, often with no fanfare except a satisfied end-user.
Chasing Answers: Research and Toxicology
Industry’s hunt for safety never really ends, and isobutyl propionate isn’t immune from the microscope. Modern toxicology digs deeper than acute inhalation or skin irritation. Researchers want studies that look at chronic exposure—low-level, daily contact among industrial workers. Early research pointed to classic issues seen with organics: dizziness, headaches at high concentrations, minor skin irritation for those working bare-handed for hours. So far, animal tests don’t show alarms on mutation or carcinogenicity at typical workplace levels, but public pressure for transparency keeps driving the need for longer, more comprehensive studies. Each year, journals add new results on metabolic fate, environmental degradation, and long-term health, all feeding into stricter regulation and better workplace standards.
Looking Ahead
The future for isobutyl propionate won’t follow a single-track path. As industries invest in safer and more sustainable solvents, users see opportunities in using biobased feedstocks or tweaking synthesis to cut waste. Ongoing innovation in coatings and green chemistry research shapes immediate prospects. There’s no doubt regulatory demands will tighten—pushing companies to prove low toxicity, minimal emissions, and high biodegradability. On the R&D side, researchers probe how modified esters might bring new scents to fragrance blends or boost drying efficiency in waterborne coatings. Future improvements rely on collaboration—chemists, engineers, safety professionals working together to ensure that a chemical’s convenience never outweighs its safety or environmental burdens. Nobody wants a repeat of the mistakes seen with legacy solvents. With smart research and open dialogue, isobutyl propionate stands as a case study for how thoughtful innovation can nudge an old compound toward a greener, safer role in our lives.
What is Isobutyl Propionate used for?
The Scent Behind the Scenes
Walk through a department store and you’ll notice a whirlwind of fragrances. Pull the cap off a bottle of perfume, and often, you’re getting a hint of something you probably haven’t heard much about—Isobutyl Propionate. This chemical has a fruity, sweet aroma. People who create scents use it to add a fresh, juicy note to perfumes and body sprays. My own experience working in a small cosmetics shop taught me that buyers expect consistency and appeal in fragrances. Isobutyl Propionate helps make sure the final product smells lively but not overpowering. It’s no secret that customers sniff-test everything from shampoo to cleaning wipes; Isobutyl Propionate plays a big role in shaping the first impression those products leave.
Flavors From the Lab
Take a swig of a flavored sports drink or snack on a piece of candy with hints of pear or apple, and there’s a chance Isobutyl Propionate had a hand in it. Food scientists use it in small amounts as a flavoring agent. There’s a long trail of safety reviews—both the Food and Drug Administration and the Flavor and Extract Manufacturers Association back up its use at low levels. I’ve seen food ingredient labels get more scrutiny lately, with more shoppers looking past “natural” and wanting specifics. Transparency matters, and this chemical—while it sounds complicated—has been around for decades without causing harm when used within industry guidelines.
A Hand in a Cleaner World
Household cleaning products have a tough job: remove dirt while leaving a space that looks and smells fresh. Isobutyl Propionate finds a niche here as a solvent. It helps break down greasy or sticky messes, making sure the cleaner does its job efficiently. It also leaves behind a pleasant scent, which is important as nobody wants ammonia lingering in the air. My friend who manages a janitorial business says workers appreciate solutions that are effective but don’t stink up a room. This is why companies look for ingredients like Isobutyl Propionate, which bring performance and a touch of aroma together.
Industry Knows Its Value
Factories and workshops look to chemicals like this one for more than just pleasant scents. Isobutyl Propionate serves as a solvent in some paints, coatings, and inks. Whether it’s ensuring a glossy finish or keeping ink flowing evenly in a high-speed printer, this ester gets the job done. The people in these industries aren’t chasing the aroma—what they want is reliability. They count on materials that dry at the right speed and leave smooth surfaces. My brother-in-law runs a small printing shop, and he explained that certain inks demand the right solvent blend; without it, jobs run late or end up streaky.
Safe Handling and Solutions for the Future
Proper storage and mindful handling come as standard advice with Isobutyl Propionate. People in factories always wear gloves and work in ventilated rooms—even small-scale users at home can avoid headaches by keeping lids tight and working in open spaces. There’s a growing movement to find greener chemicals. Some startups are developing plant-based alternatives, hoping to give the same results without a heavy footprint. If the transition ramps up, businesses will need training and support to move smoothly. The goal stays the same: deliver effective, safe products, and keep those first impressions bright—whether in the air, in food, or on the factory floor.
Is Isobutyl Propionate safe to use?
Understanding Isobutyl Propionate
Isobutyl propionate pops up in a lot of places you wouldn’t expect. You might notice its sweet, fruity smell in perfumes and flavorings. Companies pick it for paint thinners, nail products, and even as a flavor ingredient. Whenever I start checking a chemical’s safety, I ask: who’s handling it, in what amount, and with how much exposure? Safety isn’t always black-and-white—context matters a lot.
Regulatory Review: What Do the Experts Say?
Regulatory agencies keep a close eye on chemicals that end up near our skin or food. The US Food and Drug Administration gives isobutyl propionate a green light for use as a flavoring agent. The European Chemicals Agency doesn’t list it as hazardous under normal conditions. I trust these bodies because they rely on animal studies, cell tests, and decades of industry observation. These approvals show that isobutyl propionate, in regulated quantities, carries little risk for consumers.
Skin Contact and Smell: Direct Experience
Years spent writing about cosmetic ingredients taught me that some people’s skin reacts more than others. Isobutyl propionate shows low toxicity for skin and eyes in lab tests. I checked for reports of people in nail salons developing allergies or irritation from it but found few complaints. Still, folks with very sensitive skin can have unpredictable responses. Good ventilation in nail bars and gloves for frequent handlers reduce even rare issues.
Breathing It In: Airborne Safety
In factories, workers interact with gallons of solvents each day. Isobutyl propionate can cause headaches and mild dizziness above recommended air limits. I once toured a paint plant, and the smell of solvents hung in the air—nobody wants that headache at home. For products like nail polish, concentrations stay low enough that a quick application won’t cause symptoms. Hobbyists working in enclosed spaces should open a window or use a small fan, just in case.
Environmental Impact and Breakdown
Chemicals that end up in water or soil grab regulators’ attention. Isobutyl propionate breaks down quickly in the environment. Microbes eat it up, and it doesn’t tend to build up in animals or people, based on current research. I care about runoff because pollutants in streams can mess up fish, insects, and the food web. Here, breakdown studies suggest a lower risk to wildlife.
Reducing Risks in Everyday Settings
Choosing personal-care items often means weighing safety against performance. If you use nail polish or a fragrance with isobutyl propionate, you’re exposed to much less than folks filling barrels in a warehouse. Keeping bottles capped, avoiding spills, and letting rooms air out all help. Most local shops follow safety laws, but reading ingredient lists empowers people who want to avoid certain chemicals—the information is there for those who hunt for it.
What Could Go Better?
No one wants chemicals to cause harm, so full transparency and up-to-date research remain important. Some consumer safety groups push for more testing on long-term and combined exposures. Labeling laws can improve, helping sensitive individuals steer clear more easily. Companies take feedback seriously, and many reformulate if problems pop up, keeping pace with new science and consumer questions.
What are the physical properties of Isobutyl Propionate?
Understanding the Basics
Isobutyl propionate falls into the family of organic compounds known as esters. It often shows up in the flavors and fragrance industries, in part because of its pleasant fruity smell. Anyone who spends time in chemistry labs has probably worked with something similar. Its colorless liquid form looks like water at first glance, but its strong, sweet scent gives it away.
Volatility and Odor
People checking out isobutyl propionate right away notice how quickly it evaporates at room temperature. The compound has a relatively low boiling point—around 120 °C. The quick evaporation means it's not a good choice for an environment demanding heavy, lingering solvents, but it fits right in with applications such as perfumes or inks. I remember working in a lab that used isobutyl propionate for a fragrance test. You have to keep containers closed, or the whole area smells like pears and sweet wine in a matter of minutes.
Solubility and Density
Drop isobutyl propionate in water, and nothing much happens because it hardly mixes. The compound feels more comfortable in organic solvents like ethanol, acetone, or toluene. Because of its moderate density—about 0.866 g/mL at 20 °C—it floats on top of water, which creates an extra step for anyone separating mixtures in a lab or industrial process. I made this mistake as a student, expecting fast mixing, but found two neat, distinct layers instead. It sticks with me as a reminder: not everything dissolves in water.
Stability and Handling
Storing isobutyl propionate in a standard chemical container poses little trouble. It shows stability under everyday storage conditions, though it starts breaking down under strong acids or bases. Heat or open flame can trigger its flammable nature since its flash point sits below 35 °C, which seems low for a clear liquid. Many accidents in workshops start this way—assuming every clear liquid is as safe as water leads to trouble.
Practical Applications and Risks
Its combination of volatility and pleasant aroma means isobutyl propionate often finds a spot in personal care products. Paints and coatings rely on its fast drying to speed up the application process. Industries lean on these properties to fine-tune drying times or scent profiles. On the other side of the coin, its easy inhalation and flammable character make it less friendly in a health or safety sense. Prolonged exposure—such as inhaling vapors in a confined workspace—can irritate eyes and lungs. Good ventilation and careful handling matter even with something that smells inviting.
Room for Improvement
Shifting toward greener chemistry feels overdue. Using it in open spaces, recycling leftover solvents, and choosing low-emission production lines all help cut its environmental footprint. Manufacturers might switch to closed-loop systems or better vapor recovery. Labs and small-scale users need clear labeling with hazard warnings, proper spill kits on hand, and well-trained staff handling this compound. Fumes sneak past even folks with plenty of experience, so taking shortcuts rarely pays off.
Final Words
The physical properties of isobutyl propionate shape its usefulness and its challenges. Paying attention to what a substance brings to the table, and where it demands extra care, leads to better outcomes in labs, factories, and homes alike.
How should Isobutyl Propionate be stored?
What’s At Stake With Isobutyl Propionate?
Working around chemicals like Isobutyl Propionate, a common solvent with a sweet, fruity smell, brings responsibilities that reach far beyond getting the job done. Most folks ignore dangers until they watch a spill eat through their shoes or someone grips their eyes in agony after a careless splash. Chemical storage doesn’t excite headlines, but anyone who’s worked with flammable solvents knows what’s at stake if you cut corners.
Why Storage Needs Real Attention
Isobutyl Propionate catches fire without much effort. The flash point sits around 23°C–that’s room temperature for most of us. If you keep it in a warm shop or near sunlight, you’re almost daring it to go up in flames. In cities where summer heat bakes every surface, garages or supply closets get dangerously hot and vapors build up even faster. Let’s not kid ourselves—the risks can pile up fast and one careless mistake can kick off a hazardous situation.
Years ago, a friend’s small lab lost an entire supply room to fire because they left solvents like this in thin plastic jugs near a heater. The fire never made the news, but it sidelined their business for months. These first-hand stories show that safe storage is never just a suggestion.
Containers: Why Material and Labeling Count
Solvents like Isobutyl Propionate belong in containers built for chemical resistance, not whatever’s leftover from last week’s shipment. Metal cans with tight caps hold up well, but not every alloy resists corrosion. Only polyethylene or HDPE containers clearly marked for solvents can stand the test. Clear labeling with chemical name, hazards, and handling directions stops mix-ups. Using any old bottle or hiding solvents in soda bottles practically invites disaster—especially if new staff walk in and don’t know what they’re reaching for.
Cool, Ventilated Storage Stops Build-Up
Dry, cool rooms with steady airflow matter more than fancy equipment. Every warehouse or shop has forgotten corners where heat and vapors stick around. Store Isobutyl Propionate far from ignition sources like pilot lights, tools, or electrical panels. Chemical storage cabinets with self-closing doors and explosion-proof vents keep fumes from accumulating. Even a cheap vent fan and shaded storage brings down the risk noticeably, cutting both temperature and vapor in half. I’ve seen small outfits dodge trouble by turning an old office into a shaded, vented solvent room with minimal expense.
Rules Worth Following
OSHA and local authorities expect detailed chemical storage records—not just to avoid fines, but to give firefighters or cleanup teams real facts in emergencies. Spill kits, fire extinguishers rated for chemical fires, and personal protective gear should stay within arm’s reach. I keep a printed material safety data sheet (MSDS) right on the cabinet, because phones die and wifi goes down during a crisis.
While Isobutyl Propionate does its job in coatings, flavors, and fragrances, it demands respect all the way from shipment to disposal. A bit of up-front diligence makes sure this useful chemical never becomes the reason for an evacuation or an insurance claim. Shortcuts cost far more than a proper storage locker or a bit of extra training.
Is Isobutyl Propionate environmentally friendly?
What Is Isobutyl Propionate?
Isobutyl propionate shows up in everything from perfumes to paints. Its fruity smell often hooks those who work with flavors and fragrances. Factories love it because it dissolves well in other chemicals and boosts the performance of products. Shoppers never see the name on store shelves, but this compound pops up in plenty of everyday items at home and work.
How Does It Behave in the Environment?
A compound's impact rests on what happens after it leaves the bottle. For isobutyl propionate, its quick evaporation means it rarely lingers in liquids or soils. According to data from chemical agencies like the EPA, this compound breaks down in the air after a few hours to a couple of days. Sunlight and oxygen hurry this process along, turning it into less complicated chemicals that don’t stick around. Water pollution experts report that bacteria can finish breaking down this molecule, making it less likely to build up in the food chain.
Still, its sharp vapor doesn’t simply vanish into thin air. People working in poorly ventilated spaces risk breathing in too much, and sensitive lungs notice that right away. Large releases can send enough vapor indoors to cause headaches or coughs. For most people outside of factories or labs, the risk stays low if companies follow safety rules. Wastewater systems also catch a lot of what runs off from industrial sites.
Concerns About Eco-Toxicity
Frogs, fish, and bugs all face risks if too much chemical collects in streams or ponds. Tests show that isobutyl propionate harms smaller aquatic creatures if dumped in large amounts, but this rarely happens with today’s controls in the US or Europe. Regulators pinch its use when spill risk grows. None of the big watchdogs have labeled it persistent, bioaccumulative, or especially toxic at normal levels. Still, any solvent dumped carelessly has a chance to ruin a local ecosystem, so good waste management helps dodge problems.
What Are the Safer Alternatives?
Most manufacturers keep an eye out for greener options. Some solvents now rely on plant-based ingredients that break down faster and don’t threaten fish or soil life. These replacements tend to cost more, but big companies often swallow that expense to meet environmental targets. Biodegradable esters, for instance, have started nudging out old-school solvents in paint and cleaning supply factories. Buying from suppliers who use these choices encourages a shift in the whole supply chain.
What Matters for Consumers and Companies?
Responsible use of chemicals doesn’t just sit with factories. Smart labeling, recycling, and good indoor air control keep homes and workplaces safer. Some stores give buyers products with “low VOC” stamps, an easy way to pick items that release less of chemicals like isobutyl propionate. On the policy side, enforcement of emission limits pushes industries to cut back on the highest-risk uses. As pressure mounts from both regulators and eco-conscious shoppers, the market listens. Companies investing in green chemistry see stronger brand loyalty and a steadier road as rules tighten up.
Room for Improvement
Switching to truly eco-friendly chemicals always takes more than a single law or a catchy ad campaign. Honest, accessible research helps manufacturers, workers, and buyers see the whole picture. It’s not about fear—it’s about clarity. For now, isobutyl propionate looks less scary than many other industrial solvents, but safer and more sustainable options continue to grow. Each of us gets a say, either by what we use or what we buy, nudging the marketplace toward cleaner choices, step by step.
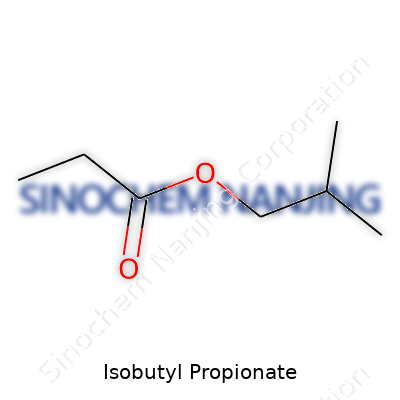
| Names | |
| Preferred IUPAC name | 2-methylpropyl propanoate |
| Other names |
Propanoic acid, isobutyl ester Isobutyl propanoate |
| Pronunciation | /ˌaɪ.səˈbjuː.tɪl prəˈpɪə.neɪt/ |
| Identifiers | |
| CAS Number | 540-42-1 |
| 3D model (JSmol) | `Isobutyl Propionate` JSmol 3D model string: ``` CC(C)COC(=O)C ``` |
| Beilstein Reference | 1722108 |
| ChEBI | CHEBI:8871 |
| ChEMBL | CHEMBL44388 |
| ChemSpider | 12711 |
| DrugBank | DB14624 |
| ECHA InfoCard | 100.011.030 |
| EC Number | 203-690-7 |
| Gmelin Reference | Gmelin Reference: 172827 |
| KEGG | C19601 |
| MeSH | D011971 |
| PubChem CID | 7847 |
| RTECS number | NT9100000 |
| UNII | BII6V8P864 |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DTXSID6020193 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | **116.16 g/mol** |
| Appearance | Clear colorless liquid |
| Odor | fruity |
| Density | 0.862 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 3.9 hPa (20 °C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | '15.37' |
| Magnetic susceptibility (χ) | -7.78×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 0.586 cP (25°C) |
| Dipole moment | 4.47 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 370.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -471.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4616.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 68°C (154°F) |
| Autoignition temperature | 426 °C |
| Explosive limits | Explosive limits: 1.1% - 7.1% |
| Lethal dose or concentration | LD50 (oral, rat): 13,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 7,500 mg/kg |
| NIOSH | SNH65650 |
| PEL (Permissible) | PEL: 250 ppm |
| REL (Recommended) | 75 ppm |
| IDLH (Immediate danger) | 2000 ppm |
| Related compounds | |
| Related compounds |
Propyl propionate Isopropyl propionate Butyl propionate n-Propyl acetate Isobutyl acetate |

