Isobutyl Alcohol: A Practical Look at Its Past, Properties, and Prospects
Historical Development
Walking through the chemical history of everyday solvents, isobutyl alcohol has its own track record. Chemists first derived it in the late 1800s while chasing better methods for making paints and varnishes. Back then, the rise of industrialization pushed researchers to stretch beyond traditional alcohols like ethanol or methanol, aiming for compounds that could blend in energy, manufacturing, and even medicine. World War periods pressed factory chemists to look for raw materials that didn’t depend on imported stocks. Synthesizing isobutyl alcohol from petroleum feedstocks or fermentation opened a steady supply, and by the mid-20th century, the alcohol had earned a spot in workshops, labs, and production lines. Industry adoption didn’t happen overnight, but use steadily ramped up as companies discovered its stability, affordability, and versatility.
Product Overview
Modern industry recognizes isobutyl alcohol as a four-carbon, branched-chain alcohol that performs in a wide set of applications. The clear, colorless liquid can be found parked on chemical supply shelves or inside larger drums ready for transport. Unlike some niche reagents, isobutyl alcohol doesn’t demand special conditions to stay shelf-stable. Freight trains and tankers haul the substance around the world, thanks in part to its predictable behavior under normal storage conditions. For paint formulators and resin producers, this alcohol offers just the right mix of volatility and solvency. Those working in agricultural chemistry see isobutyl alcohol as a core part of some pesticides and herbicide formulations. People don’t always realize how much invisible chemistry shapes their daily experience, yet isobutyl alcohol stands as a good example.
Physical & Chemical Properties
Isobutyl alcohol boils close to 107 degrees Celsius, which means it rolls off in vapor at a much lower temperature than water. The faint, sweet odor isn’t overpowering, but it gives the chemical away even at low concentrations. Its density sits just under that of water. Chemists working at the bench appreciate its lower viscosity, which means it pours easily and mixes well. One important trait is its ability to dissolve a broad range of non-polar substances—it unwraps the mystery of how stubborn resins and waxes break down during cleaning or manufacturing. Unlike more reactive alcohols, isobutyl alcohol stands up against slow oxidation but can still catch fire if exposed to an ignition source. Its flash point hovers around 28 to 35 degrees Celsius, so warehouses often store it with care, keeping open flames and sparks at bay.
Technical Specifications & Labeling
Bottles or drums labeled as isobutyl alcohol carry important details: purity, water content, and identification numbers based on standards set by chemical authorities. Most commercial supplies run higher than 99 percent purity. The label usually shows hazard icons—a black flame or exclamation mark—meant to remind users about its flammability and the need for ventilation. Safety Data Sheets explain emergency instructions, symptoms of overexposure, and how to store it away from oxidizing agents. Industrial buyers sometimes request batch-specific analyses, especially if the product ends up in high-value coatings or pharmaceuticals. Consistency matters because impurities can throw off reaction yields and lead to waste. Quality assurance teams—an unsung backbone of safe chemistry—trace every drum back to a documented origin.
Preparation Methods
Original production methods used butyraldehyde as a stepping stone, running it through hydrogenation processes. Today’s factories lean on hydroformylation of propylene, a process that converts a basic chemical cousin of gasoline into isobutyraldehyde, then reduces it with hydrogen gas. The whole sequence needs precise pressure and temperature control. On a smaller scale, fermentation benches have shown you can coax certain yeast species into churning out isobutyl alcohol from sugars, but most commercial tanks run on petrochemicals, not corn or beet juice. Manufacturing always brings trade-offs: renewables offer a greener profile, but scale and price sometimes pull the industry back to tried-and-true refinery methods.
Chemical Reactions & Modifications
Chemists treat isobutyl alcohol as a starting block for a series of reactions—most importantly, esterification. Mixing it with acids in the right ratios produces fruity-smelling isobutyl esters. Fragrance houses capture this chemistry when crafting artificial flavors for candies, sodas, and perfumes. Still, workshops also transform it into isobutyl acetate or oxidize it further to make isobutyric acid. Up the chain, polymer chemists sometimes graft it onto molecules to tweak their flexibility or solubility. All these transformations depend on tight process controls—run things too hot, introduce the wrong catalysts, and side reactions ruin the product. Creativity and caution sit side by side in the chemist’s mind.
Synonyms & Product Names
Walking through chemical catalogs, isobutyl alcohol appears under a stack of names: 2-methyl-1-propanol, isobutanol, or sometimes "IBA." Some older texts list it as "iso-butyl alcohol," with a hyphen, or highlight its structural makeup by calling it "sec-butylcarbinol." Big producers sometimes slap on trade names, usually for blends, though most buyers stick to the common names or IUPAC nomenclature. This diversity in labels means anyone shopping for the chemical needs to double-check—swapping out a single carbon atom or moving a methyl group can land you with the wrong substance entirely. Standardization isn’t just about language—it safeguards quality and trust.
Safety & Operational Standards
Nobody wants to ignore safety when handling isobutyl alcohol. It evaporates quickly, so even a small spill fills a room with fumes that, in high enough doses, can irritate the eyes or throat. Workers suit up in gloves and goggles, and fume hoods run on high during lab work. Storage cabinets stay grounded and free from static sparks. Fire marshals pay particular attention to the flash point; insurance policies often set rules about how much can be stocked in a given area. Regulations from agencies like OSHA, EPA, and the European Chemical Agency guide disposal and emergency responses. So, even for repeated users, the message from seasoned safety officers rings true: keep it ventilated, keep it cool, keep it locked up.
Application Area
Industry leans on isobutyl alcohol in several ways. In paints and coatings, it helps thin formulas for smooth brushwork and faster drying. Adhesives—including some used in woodworking and automotive parts—use it to improve mixing and flow. Agrochemical manufacturers use it as a building block for herbicides and insecticides. Even the pharmaceutical industry taps into isobutyl alcohol as a solvent during drug synthesis or as a part of extraction processes. Flavors and fragrances rely on its esters to create scents found in everything from chewing gum to cleaning agents. In the laboratory, chemists reach for it as a medium in organic synthesis or chromatography. Its wide reach touches construction, art supplies, food additives, and the cleaning aisle.
Research & Development
Research teams pay attention to both sustainable sourcing and new applications. Renewables are gaining ground as biorefinery processes mature—the promise of making isobutyl alcohol from waste biomass or algae is more than a pipe dream. Academic chemists test engineered yeast and bacteria to boost yields without relying on crude oil. Material scientists wonder if modified isobutyl compounds can drop into next-generation resins, plastics, or fuel blends. Some consumer advocates push for clearer toxicity profiles, trying to balance effectiveness with public health. Pharmaceutical researchers experiment with it as a carrier solvent, hoping to refine purification or formulation steps. Every lab breakthrough paves the road toward a greener, stronger, or safer chemical sector.
Toxicity Research
Toxicologists have spent years mapping out how isobutyl alcohol affects people and animals. Short-term exposure usually means headaches, dizziness, or throat irritation—dangers that can worsen without fresh air or protective gear. Swallowing even small amounts causes nausea or central nervous system effects, so it’s no surprise strict limits exist. Researchers set occupational exposure limits based on studies in rats, rabbits, and other models, translating those findings to human workplace safety. Chronic toxicity studies flag potential liver and kidney trouble after repeated high-dose exposures. Consumer watchdogs and regulators stay vigilant, cross-checking the latest animal studies, workplace monitoring data, and real-world case reports. Hazard communication labels keep evolving as new findings refine our understanding.
Future Prospects
Industry leaders and scientists see opportunity in sustainable production. Renewable feedstocks may shrink the carbon footprint if economies of scale take hold. Isobutyl alcohol could find new life in synthetic fuels, especially as transportation looks beyond gasoline and diesel. Its use in advanced polymers or even electronics seems likely if researchers solve purity and compatibility challenges. Environmental advocates want more focus on recycling and closed-loop processes, driving factories to cut emissions and boost efficiency. Educators and policymakers can pitch in by expanding public awareness and updating guidelines as science advances. If research dollars flow to both safety and greener methods, isobutyl alcohol may keep its essential role in commerce while easing strain on the planet.
What are the main uses of Isobutyl Alcohol?
Solvent Power in Industry
Walk through almost any factory using paints or coatings, and there’s a good chance you’ll find isobutyl alcohol somewhere along the line. Its knack for dissolving oils, gums, and resins makes it valuable in formulating lacquers and varnishes. The quick evaporation helps paint layers dry evenly, so surfaces end up smooth and durable. Formulators in the world of industrial coatings trust it because it keeps their recipes balanced and helps products withstand weather and wear.
Fuel Additive and Cleaner
Gasolines and fuels sometimes need a little boost, so manufacturers turn to isobutyl alcohol for its blending properties. With global demand for cleaner combustion and improved fuel efficiency, even small changes matter. Isobutyl alcohol helps fuel combine more completely with air, which can lower carbon monoxide and soot output. Keeping engines running cleaner means less frequent maintenance and improved air quality—something I’ve seen put into practice in fleets trying to cut downtown smog.
Intermediate in Chemical Synthesis
Step into any chemical plant that makes flavors, fragrances, or pharmaceuticals, and isobutyl alcohol’s role becomes clear. It acts as a building block or “intermediate” in the production of esters, such as isobutyl acetate. These esters offer sweet, fruity aromas and show up in everything from candy to perfumes. There’s a science and a bit of artistry to picking the right alcohol for each synthesis, but isobutyl alcohol gets the nod when a robust, mild scent is needed.
Personal Care and Household Products
Shampoos, aftershaves, lotions, and cleaning sprays rely on solvents that won’t irritate skin or mess with the consistency of the blend. Isobutyl alcohol fits the bill. It helps deliver the active ingredients, supports fragrance stability, and never leaves a heavy residue. In my time working with skincare startups, I’ve noticed how formulators reach for isobutyl alcohol to bind everything together while still keeping formulas gentle on the skin.
Extracting and Processing
Isobutyl alcohol has the right qualities for extracting certain elements from natural sources. It helps isolate active components from plants in labs or small-batch operations, pulling out flavors, fragrances, or essential oils without breaking the bank on expensive specialty solvents. This isn’t just about efficiency—it’s about making sure products remain natural and accessible, since costs stay reasonable and processes stay streamlined.
Safety and Environmental Factors
No industrial chemical story would be complete without talking about safety. Isobutyl alcohol is flammable and can cause irritation, so proper handling gear and ventilation matter. It’s not something you want splashed around carelessly. Regulations in the U.S. and Europe set thresholds and procedures for safe use, reflecting lessons learned over decades of industrial practice. Efforts to reduce emissions and improve recycling also shape how much of the chemical ends up in the air or waterways.
Looking Forward
Demand for isobutyl alcohol links tightly to manufacturing, agriculture, and fuel trends. As plant-based production methods grow, some producers seek renewable sources, which could lower dependence on fossil fuels. Better recycling has also started to close loops in chemical supply chains. Everything points toward a future where isobutyl alcohol keeps adapting, finding new uses while addressing the push for greener, safer ingredients.
Is Isobutyl Alcohol safe to handle and what precautions should be taken?
Understanding the Risks of Isobutyl Alcohol
You find isobutyl alcohol in lots of places—paint thinners, some cleaning supplies, even labs and factories. It smells strong and can irritate your nose right away. People see clear liquid and sometimes forget how fast it can cause problems. Isobutyl alcohol can make your skin dry out and crack. Even more serious, it can enter your body through breathing, skin contact, or swallowing, and cause headaches, dizziness, or even make you pass out. Some folks think something sold in common products must be safe, but OSHA and the CDC put isobutyl alcohol near the top of their watch lists because accidents happen so quickly.
Direct Contact and Inhalation: Health Matters
Anyone who’s worked around chemicals for long knows gloves aren’t optional with something like isobutyl alcohol. Just touching it starts to strip oil from your skin. A small spill on bare hands can burn and sting, and with bigger spills, you risk blisters. If you’ve ever worked in a closed space without a mask, that gasping feeling comes fast. Just breathing the vapor makes your eyes sting and your mind fog up. In a busy shop, someone who thinks they’re fine one minute might get dizzy and faint the next because vapors build up before you know it.
Ventilation and Protective Equipment: Key Lessons from the Workplace
No one wants to wear itchy goggles, but after seeing what happens from a splash, you don’t skip them again. Isobutyl alcohol travels through the air, stinging noses and burning lungs. Fans make a huge difference. A warehouse feels fresh with the right air movement, but the wrong space gets suffocating fast. Respirators rated for organic vapor keep lungs clear, and slip-resistant gloves protect against spills that eat away at unprotected skin. It surprises people how easily alcohol seeps through cotton or cheap gloves. Nitrile or neoprene hold up better for longer jobs.
The Fire Hazard: Flammability Nobody Should Forget
Isobutyl alcohol catches fire without much effort—just a spark, a hot bulb, or even static discharge. Stories spread in factories about a careless toss of a rag sparking a blaze. Keeping this chemical away from open flames isn’t just a good idea, it’s vital. Storing it in safe, clearly labeled metal cans saves headaches. Fire extinguishers rated for flammable liquids need to sit close by. At home, never underestimate the risk by using it in the garage near a water heater or space heater.
Accidents and Preparedness: Real World Solutions
Years in field service taught me you never want to scramble for an eyewash station in a panic. Good practice puts water sources and showers right next to where chemicals live. In a pinch, rinsing for 15 minutes saves eyesight. Signage in local languages on every wall reminds everyone what to do. Supervisors who actually train their teams—testing how to handle a spill or get someone to fresh air—build habits that stick, keeping injury rates low.
Clear Labeling, Education, and Making Safety Part of the Routine
Isobutyl alcohol isn’t something you treat as “just another chemical.” Reading the Safety Data Sheet makes every use safer. I’ve seen workplaces get safer by running “stop-and-think” checks before each job. Teaching every new worker—no matter how briefly they plan to stay—sets the standard. At home, keeping chemicals locked away, far from children and pets, cuts down on heartbreaking accidents.
Bottom line: Safety with isobutyl alcohol means respecting its risks and preparing for the worst. Protective gear, fresh air, and smart habits keep everyone safe.What are the physical and chemical properties of Isobutyl Alcohol?
A Familiar Chemical with a Tangible Role
Most people walk right by isobutyl alcohol without even noticing it. This clear liquid pops up in everything from everyday cleaners to industrial coatings and even flavors for food and beverages. It may not have a catchy name, but it has a reputation among folks who work with solvents, fragrances, and synthetic materials.
Physical Features People Notice
You’d probably recognize isobutyl alcohol by its smell before seeing its name on a label. There’s a sharp, slightly fruity scent that lingers in the air, easy to remember once you’ve smelled it in a laboratory or a production plant. As for looks, it’s a colorless liquid, easy to pour and not sticky, sliding right off lab glassware. It boils just above the heat of a cup of tea, right at 108°C. Pour some in a glass, and it’ll evaporate at room temperature before long. Mixing it with water doesn’t go as smoothly; only a little will blend, since it prefers to stick with oils and non-polar mixes. That stubbornness makes it good at drawing out grease and oil, which comes in handy for specialized cleaning or in extracting ingredients from plants.
Chemical Behavior That Shapes Its Uses
Isobutyl alcohol is more than a basic alcohol in a long line of solvents. With four carbon atoms arranged in a way that branches out, it stands apart from its straight-chain cousins. That structure keeps the boiling point low for an alcohol, making it a favorite option when quick evaporation matters, such as in paints and thinners. In the workshop or lab, people count on its moderate toxicity—careful handling required, gloves recommended, but regular use under well-ventilated hoods. It’s flammable—liquids like this don’t need much to catch fire, so safety measures are not optional in facilities or home shops.
Chemically, it reacts just like other alcohols. It forms esters that bring fruity notes to flavor and fragrance applications. Chemists often use it to tweak molecules by swapping or adding groups, giving it a flexible role in many synthetic processes. Its moderate polarity makes it a handy player in cleaning and degreasing tasks, since it can break down oily gunk left behind by machines or cooking, yet doesn’t hang around long after use.
Safety and Impact on Health
Years working around solvents drove home one thing—ventilation and skin protection aren’t negotiable. Isobutyl alcohol gives off vapors that can make someone dizzy, lightheaded, or irritated if allowed to build up. People using it regularly know gloves and masks keep headaches and skin rashes at bay. The Environmental Protection Agency keeps tabs on it because, like most solvents, spills or careless use can seep into soil and water. Responsible handling and disposal have taken bigger priority in labs and plants in recent decades, with more companies switching to closed systems and proper filtration.
What the Future Holds
It’s no secret that chemical safety pushes everyone in this field to look for improvements. Research on greener alternatives or better ventilation systems shows up at trade shows and conferences. Some companies now offer plant-based alcohols that mimic isobutyl alcohol’s properties without the lingering pollution. Even so, isobutyl alcohol holds a firm spot in industry thanks to its efficiency and reliability, so folks in manufacturing, cleaning, or fragrance worlds will keep finding ways to balance performance and safety.
How should Isobutyl Alcohol be stored and transported?
Understanding What’s at Stake
Isobutyl alcohol plays a big role in industries like paint, pharmaceuticals, and flavors. This liquid sits in drums or tanks across factories, and crews move it by road, rail, and ship. People sometimes underestimate its risks because of its everyday uses, but dealing with chemicals like isobutyl alcohol always calls for respect and precision. One slip — a leak, a hot spot, the wrong seal — and you can wind up with hazards that affect workers and communities.
Why Storage Choices Matter
Flash point sits at just 28°C. That means on a warm summer day, vapors can catch fire. If airborne, those vapors feel heavier than air and hug the floor — and without good ventilation, they pool in corners and can ignite with a spark. Metal drums or tanks with grounded fittings work best to take away static charges. My years in a facility showed me: any rusted drum or missing ground wire turns into a safety meeting waiting to happen. Sites that store these drums need dry, cool spaces. If sunlight hits the tanks, temperatures climb, vapor pressure inside rises, and even small leaks get worse.
It pays off to keep isobutyl alcohol away from oxidizers and acids. I once saw two containers stacked too close, and a small drip meant corrosion on seals, which could have stopped us in our tracks if it had gone unnoticed. Pallets go on spill containment trays. Weekly walks through the storage area prevent surprises, and logs tracking container age help keep rotation tight since seals on old drums fail more often.
What Happens on the Road
On the move, isobutyl alcohol shifts from warehouse worry to logistics challenge. Every tanker truck driver knows the DOT hazmat rules, but real safety comes from following them every single mile. Labeling — clear, weatherproof, and with the right UN numbers — stops confusion at docks and during checks. A friend told me about a mixing-up of containers because faded labels wasted half a day and put everyone on edge. You don’t want mystery loads inside a chemical park.
Baffles inside tankers slow liquid surge, giving drivers better control. Well-maintained valves and overfill protection devices prevent costly and dangerous spills during transfer. Emergency kits stay in reach: absorbent pads, goggles, gloves, not because “rules say so,” but because if anything goes wrong, speed matters more than anything.
People Make the Biggest Difference
Training goes further than any sign or checklist. Someone who’s been through a fire drill or leak scenario handles the real thing with calm. I ran drills, and one lesson always came back — everyone forgets the small steps without practice, like closing a vent or signaling for backup. Regular reviews and up-to-date material safety data sheets make sure everyone knows what they’re facing. Forgetting to account for recent regulatory updates on threshold quantities fooled a neighbor down the road into a fine. Regulations shift, and those using yesterday’s protocols invite tomorrow’s headaches.
Better Incentives, Fewer Risks
It is easy to lean on rules alone, but people connect with stories. Sharing local incidents, both mistakes and wins, reinforces habits far better than a laminated poster. Supervisors who reward careful handling and quick problem-spotting find fewer accidents. Investment in double-walled tanks or vapor detectors pays for itself by preventing one major clean-up.
We all owe each other vigilance, from the person placing a drum on the rack to the driver more concerned about their route than the clock. Safety in handling isobutyl alcohol does not come from a checklist; it grows from respecting each step, every time.
What is the difference between Isobutyl Alcohol and other alcohols like n-Butanol or Isopropanol?
Isobutyl Alcohol: Not Just Another Chemical on the Shelf
If you’ve walked through a lab or manufacturing facility, you’ve probably caught a whiff of isobutyl alcohol. This compound shows up in plenty of places, but most people don’t give it a second thought past the sharp, distinctive smell. Isobutyl alcohol stands out from other alcohols like n-butanol and isopropanol mainly because of its chemical structure. That might not sound like a big deal at first, but even small tweaks in a molecule can make a big difference in how it behaves and what it’s good for.
Structure Breathes Life Into Differences
Isobutyl alcohol has four carbon atoms, but they’re arranged in a branched pattern. n-Butanol, with the same number of carbons, lays them out in a straight chain. Isopropanol breaks things up even more, clocking in with three carbons and a branch that gives it a whole different character. Chemists—and anyone who spends time around solvents—know that structure shapes properties. For isobutyl alcohol, this means a lower boiling point than n-butanol, a less harsh smell than isopropanol, and differences in how it mixes or dissolves things.
How Do These Differences Play Out?
Each alcohol lands its punch in unique ways. Isobutyl alcohol crops up in flavors, perfumes, and sometimes as a solvent. In my time working with coatings, I learned that isobutyl alcohol gives more flow and smoother finishes compared to n-butanol, which tends to evaporate slower and gets used where more time is needed for drying. Factories lean on n-butanol for certain adhesives and plasticizers because its straight carbon chain makes it slightly less volatile. Isopropanol, better known as rubbing alcohol, wins the popularity contest for quick-drying sanitizers and cleaning fluids. Its combination of speed, safety, and cleaning power makes it a staple in households and hospitals alike.
Shelving Choices: Safety, Environment, and the Work at Hand
Choosing between these alcohols isn’t just about which label looks best. Isobutyl alcohol gives off moderate fumes but can irritate eyes and skin. n-Butanol stings less, but can build up with prolonged exposure. Isopropanol feels less sticky but packs its own risks if inhaled in large doses. There was a time in a small shop where poor ventilation led to drowsy workers because the wrong solvent got used—underscoring the daily impact of these choices. Companies and home users want options that balance effectiveness with safety and smell, making the familiar rubbing alcohol sometimes more appealing than the strong scents of the others.
Industry and Sustainability: Spilling Into the Future
Alcohols don’t just wash their hands of environmental issues. Isobutyl alcohol has started turning heads as a potential biofuel, since its branching allows engines to use it more efficiently than some other alcohols. n-Butanol also gets interest for the same reason, but harder extraction keeps costs higher. Isopropanol, thanks to its roots in the petrochemical industry, has environmental baggage but still holds a place because it’s so easy to produce.
For companies hunting greener processes, it helps to push for better recycling and sustainable sourcing. At the end of the day, small changes in molecule structure create big choices in labs, factories, and even bathrooms at home. The best pick often hinges on a balance between effectiveness, safety, and responsibility—a lesson anyone who’s worked with chemicals learns sooner or later.
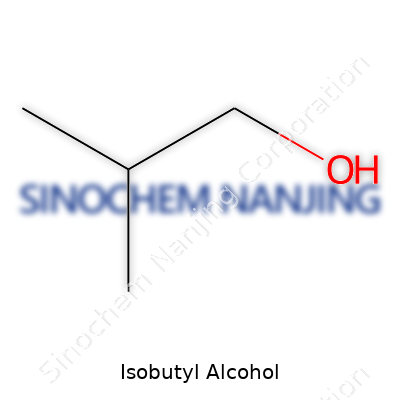
| Names | |
| Preferred IUPAC name | 2-Methylpropan-1-ol |
| Other names |
2-Methyl-1-propanol Isobutanol Isobutylalcohol Isobutanol alcohol 1-Hydroxyisobutane |
| Pronunciation | /ˌaɪsəˈbjuːtɪl ˈæl.kə.hɒl/ |
| Identifiers | |
| CAS Number | 78-83-1 |
| Beilstein Reference | 1360636 |
| ChEBI | CHEBI:16183 |
| ChEMBL | CHEMBL15922 |
| ChemSpider | 5817 |
| DrugBank | DB01924 |
| ECHA InfoCard | ECHA InfoCard: 100.003.958 |
| EC Number | 603-108-00-1 |
| Gmelin Reference | Gmelin Reference: 8363 |
| KEGG | C06510 |
| MeSH | D007561 |
| PubChem CID | 6560 |
| RTECS number | NP9625000 |
| UNII | NR7VL0Q44B |
| UN number | UN1212 |
| Properties | |
| Chemical formula | C4H10O |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless liquid with a characteristic odor |
| Odor | Fruity odor |
| Density | 0.802 g/cm³ |
| Solubility in water | 8.7 g/100 mL (20 °C) |
| log P | 0.83 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | 16.5 |
| Basicity (pKb) | pKb = 15.2 |
| Magnetic susceptibility (χ) | -7.41×10⁻⁶ |
| Refractive index (nD) | 1.399 - 1.402 |
| Viscosity | 3.95 cP (20°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -327.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2675 kJ/mol |
| Pharmacology | |
| ATC code | J02AA08 |
| Hazards | |
| GHS labelling | Warning, H226, H319, H336 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | Flash point: 28°C |
| Autoignition temperature | 426 °C |
| Explosive limits | 1.7% - 10.6% |
| Lethal dose or concentration | LD50 oral rat 2460 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2460 mg/kg (oral, rat) |
| NIOSH | SQ3325000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 1600 ppm |
| Related compounds | |
| Related compounds |
n-Butyl alcohol sec-Butyl alcohol tert-Butyl alcohol |

