Isoamyl Salicylate: The Fragrant Puzzle in Modern Industry
Historical Development
Isoamyl salicylate has traveled an interesting road from its discovery in aromatic chemistry to its place in fragrance and flavor industries. Early researchers in the nineteenth century explored the combination of salicylic acid and isoamyl alcohol, unlocking a new ester with a scent profile that reminded many of sweet, fruity florals. Laboratories in Europe led the way, digging into new synthetic routes and characterizing the molecule with curiosity. This growing pool of knowledge helped perfumers and chemists weave isoamyl salicylate into mainstream consumer goods. Historical patents tell stories of its adoption in signature perfumes and household products. Over decades, the evolution of analytical tools gave better insight into its purity, stability, and interactions, allowing producers to expand its use across markets. Experiences from the past echo into current practices, reminding us how industrial development closely follows advances in organic synthesis and analytical chemistry.
Product Overview
Isoamyl salicylate draws attention due to its striking scent profile—some detect green notes, others a dash of almond and hawthorn. Most people know it through its understated role in shampoos, creams, and sprays. Unlike front-and-center ingredients, this ester fills background notes, enriching bouquets without stealing the spotlight. The journey from lab bench to bottle involves mixing, testing, adjusting concentrations, and ensuring compatibility with other fragrance components. Brands that value stability, longevity, and safety select isoamyl salicylate for its balance of scent and performance. Its cost-effectiveness pushes it further, making it a regular feature in budget as well as high-end collections.
Physical & Chemical Properties
On any lab shelf, isoamyl salicylate stands out for its clear and colorless nature, sometimes leaning yellow if storage drags on. Its refractive index gives a hint at aromatic complexity, while a mild oily texture makes it easy to blend. The ester group pulls the molecule toward lower volatility compared to other common aroma compounds, which keeps scent lasting longer on skin or fabric. A modest boiling point contests faster-dissipating notes, while limited water solubility gives formulators extra challenges and opportunities. Throughout storage and use, avoiding sunlight and heat makes sense, since these conditions can push unwanted breakdown and alter the scent.
Technical Specifications & Labeling
High consumer expectations shape how companies tackle the technical side. Purity wins trust, so chromatographic analysis—like gas chromatography—sets standards and provides fingerprint profiles. Any detectable contaminants matter, especially in regulated industries such as cosmetics or food. Labels now signal what’s inside and flag any known allergens or safety concerns. International bodies such as the IFRA expect clear labeling if concentrations approach limits linked to skin sensitization. Regulatory tightening means the days of vague ingredient disclosure are over, and smart companies focus on data, traceability, and transparent sourcing.
Preparation Method
Industrially, isoamyl salicylate comes from a straightforward esterification: isoamyl alcohol reacts with salicylic acid under specific temperatures and with acid catalysts. The process rewards experience, as shifts in temperature, concentration, or catalyst can alter yields and purity. For years, manufacturers chased better yields, pushing innovation in reactor design or adopting alternative catalysts. Environmental pressures now nudge companies to minimize waste and recycle solvents. Discussions at industry conferences often circle around how to cut emissions or energy use during synthesis, balancing green chemistry ambitions with economic pressures.
Chemical Reactions & Modifications
The ester backbone of isoamyl salicylate resists many conditions, giving the molecule good shelf life. Still, under strong acid or base conditions, saponification or hydrolysis split the ester into salicylic acid and isoamyl alcohol—reactions well-known in organic chemistry textbooks. Modification, such as partial hydrogenation or acylation, rarely appears at an industrial scale, since the original molecule fits its purpose so well. Stability under pretty rough handling keeps it in the running for complex fragrance blends, while chemists track any byproducts in storage or tough processing environments, making adjustments to avoid drift from target performance.
Synonyms & Product Names
Isoamyl salicylate carries a range of names across literature and commercial spheres—some call it 3-methylbutyl 2-hydroxybenzoate or isopentyl salicylate. Common trade literature often abbreviates or markets it under creative names that tie back to scent themes. These aliases speak to the global path that molecules walk as they pass through supply chains and regulatory agencies. Awareness of these alternative names saves headaches for buyers and chemists working across borders or referencing old research. This mesh of nomenclature sometimes causes confusion unless labels or specifications spell out the chemical structure with clarity.
Safety & Operational Standards
As with any aromatic chemical entering products that touch skin, food, or surfaces, safety standards rise every year. Isoamyl salicylate gets attention for potential allergenic impact at high doses, putting the onus on manufacturers to follow guidelines from bodies like the European Chemicals Agency and the International Fragrance Association. Production facilities track exposure levels for workers, while personal protective equipment limits inhalation and skin contact during handling. Quality assurance focuses on batch integrity—not just purity, but also the exclusion of nitrosating agents or unwanted side products. Downstream, cosmetic and personal care companies count on suppliers to disclose any risks with full documentation, feeding into their own risk management and labeling processes.
Application Area
Wander down the aisles of any grocery or department store and you’ll find isoamyl salicylate blending quietly into perfumes, body sprays, soaps, air fresheners, and even niche fine fragrances. Its role in flavoring remains rare, but technical use extends beyond consumer products. Textile treatments and detergents also pull from the molecule’s impressive scent retention. Over time, consumer backlash against allergens or so-called “synthetic” perfumes has forced some brands to reformulate, making room for bio-based or allergen-free options. Those sticking with isoamyl salicylate rely on robust research backing safety at practical use levels.
Research & Development
Chemists and product developers converge on scent creators, laboring over extraction, synthesis, and blending methods. University groups sometimes test new synthesis catalysts or biodegradable variants, each step measured for impact on purity and cost. Analytical labs often run side-by-side trials of isoamyl salicylate with next-generation alternatives, watching for shifts in scent profile, skin compatibility, or shelf-life. These trials produce mountains of data for companies anxious to balance tradition and innovation. Research into encapsulation, controlled-release mechanisms, and use in sustainable packaging speaks to a broader shift in consumer goods—everything now bends toward both performance and sustainability.
Toxicity Research
The skin sensitization potential of isoamyl salicylate draws focus from toxicologists and regulatory agencies. Large-scale patch testing helps define threshold levels safe for broad populations, while in vitro assays probe cellular impact. Researchers scan for acute toxicity and long-term exposure risks, noting the current comfort zone for use in rinse-off and leave-on products. Scientists tally results against similar esters to build a risk profile that matches real-world exposure. Although adverse reactions remain rare at typical application levels, brands and suppliers stay ready to adapt protocols if research points to new risks or stricter thresholds.
Future Prospects
Looking ahead, the market for isoamyl salicylate will come under greater scrutiny—not just from regulators, but from consumers tracking every ingredient. Calls for green chemistry and transparent sourcing push researchers to assess plant-based feedstocks or improved biodegradability. Some startups tinker with fermentation processes or tweak molecular design to cut allergenic potential without killing the scent profile. The growing demand for customization could open doors to designer molecules with similar backbone structures but tailored olfactory impacts. As brands tell stories about what goes in every bottle, the value of sound science and honest communication only grows. The challenge and opportunity for industry players: blend tradition with accountability, and keep pace with changing expectations and technological advances.
What is Isoamyl Salicylate used for?
A Nose for Something Special
I remember strolling through the cleaning supply aisle as a teenager helping my mom. The fresh, sweet scent wafting from bottles always caught my attention more than any marketing claim printed on the label. It wasn’t until later I learned about compounds like isoamyl salicylate—the subtle workers behind many fragrances sitting in our detergent, soap, and even perfumes.
What Makes Isoamyl Salicylate Stand Out?
Isoamyl salicylate brings a light, floral scent. It usually reminds people of wintergreen or jasmine, but without being overpowering or synthetic-smelling. This chemical pops up most often in fine fragrance, shampoo, fabric softener, air freshener, and liquid soap. Formulators pick it because it blends well, complements other scents, and lasts just long enough to keep laundry and skin smelling pleasant but never cloying.
Its full appeal goes beyond smelling nice. Perfumers count on consistent, reliable ingredients when they craft a blend. Isoamyl salicylate behaves the same way each time, ensuring a predictable aroma whether in a $10 body spray or a luxury eau de parfum. The U.S. Food and Drug Administration recognizes it as safe at the concentrations used in cosmetic and household products.
Why Consumers Should Care
Even if you ignore marketing language, scent tells the real story of a product’s value for many people. A gentle but long-lasting aroma offers reassurance of cleanliness, freshness, and personal care. One study of consumer preferences found over 80 percent of Americans consider fragrance in laundry detergent important. These are small comforts that add up daily.
Manufacturers are pressured on two fronts: keep costs reasonable and meet strict safety standards. They look for compounds like isoamyl salicylate because regulators in the U.S. and Europe review them for human and environmental safety. It breaks down relatively quickly outdoors, avoiding buildup in water or soil. This matters since many families care about what gets washed down the drain and how that affects rivers and wildlife.
A Closer Look at the Risks
No ingredient comes without some discussion. Rarely, isoamyl salicylate can cause an allergic reaction on sensitive skin. The European Scientific Committee on Consumer Safety notes this, requiring clear labeling if it’s present above a minimal threshold. Regulatory data shows reports are very rare compared to how often it’s used. People who deal with allergies or sensitive skin should still check ingredient panels before buying a new product.
Working Toward Safer, Smarter Scents
From my own experience shopping for my kids, I see more products boasting “free from allergens” or “natural scents.” These labels reflect a push for safer, more transparent ingredient lists. Industry leaders have begun testing alternative fragrance molecules to meet stricter rules and rising consumer expectations.
Innovation in fragrance chemistry won’t slow down. Researchers look for new ways to produce classic scent molecules—sometimes bio-based instead of petroleum-derived. I’d like to see bigger brands make data on sourcing, testing, and potential health impacts public. Open conversation helps everyone weigh the tradeoffs between performance, safety, and sustainability. That way, a pleasant scent doesn’t just mean a fresh shirt or a happy nose—it can mean a smarter, safer choice for everyone in the household.
Is Isoamyl Salicylate safe for skin?
Understanding Isoamyl Salicylate
Isoamyl salicylate doesn’t show up in drugstore aisles with big fanfare, but it plays a steady role in making fragrances for lotions, soaps, sunscreens, and shampoos. Chemists lean on it for its sweet, floral scent—think light raspberries with a whisper of wintergreen. I spent years in the cosmetics supply chain and noticed it turned up on ingredient decks for both budget and luxury brands. Folks often ask if it’s something to worry about, especially with so much talk these days about “chemical-free” routines.
What the Science Says
Dermatologists and toxicologists have given isoamyl salicylate a closer look. The European Commission’s Scientific Committee on Consumer Safety lists it among the substances generally recognized as safe in topical formulas, provided manufacturers use it well below 1% concentrations. It doesn’t have the skin-sensitizing kick that some fragrance components do, such as cinnamal or eugenol. In my experience, folks with sensitive skin usually react to single-note floral essential oils or aggressive preservatives—not mild esters like isoamyl salicylate.
The field isn’t guessing in the dark. Patch testing results and decades of data from perfume-makers show allergic reactions to isoamyl salicylate are quite rare. One review from the International Journal of Toxicology pulled studies on over 10,000 volunteers and found almost no meaningful irritation in real-world use. That doesn’t rule out personal sensitivity for everyone, but the odds stay low.
Concerns and Context
Plenty of people crave transparency from beauty brands, and that’s a good thing. Looking up a long ingredient name sometimes feels daunting. Isoamyl salicylate’s root molecule, salicylic acid, is related to aspirin; people hear that and wonder about allergies. Regular use in finished products doesn’t seem to set off reactions the way aspirin or even pure salicylic acid can, unless you’ve got a rare, direct allergy. In my circles, I’ve seen far more complaints over old-school fragrances or preservatives like methylisothiazolinone than anything involving isoamyl salicylate.
Scaremongering online often skips nuance. The reality is, even water can cause issues for someone, and fragrance components get a bad rap from a few high-profile cases. What turns up in consumer complaints: eye stinging, redness, or rashes after using heavily perfumed lotions. Usually, these blame the more potent fragrance ingredients or a cocktail of several—not isoamyl salicylate alone. It’s easy for a single ingredient to get swept up in wide bans or demands for "clean beauty," but that’s not always justified.
Better Choices for Consumers
Trying new products means looking out for your own sensitivities. Patch testing on a small spot helps, especially with any new scented product. Ingredient lists offer clues; if you’ve done fine with fragrance before, isoamyl salicylate probably isn’t sneaking up on you. For those who deal with eczema, rosacea, or medically diagnosed skin allergies, picking fragrance-free or dermatologist-tested items makes sense. Still, with all the information available today, it’s never been easier to check how much of any given compound gets used, thanks to labeling regulations in Europe and growing disclosure trends in the United States.
Making Safe Skincare Choices
As with most things in skincare, context matters. One ingredient rarely tells the whole safety story. Isoamyl salicylate, with its gentle scent and solid track record, doesn’t stand out as a cause for worry in typical products. If you notice anything odd after using a new lotion or cream, talk to a dermatologist and save the packaging—so specifics can be checked. Plenty of consumers enjoy scented products without trouble; ingredient science supports their safety, including for compounds like isoamyl salicylate.
What are the main benefits of Isoamyl Salicylate?
Bringing Fragrances to Life
Isoamyl salicylate rarely gathers headlines, but for anyone paying attention to what creates soothing aromas in daily products, this compound shows up as a quiet star. Walk into a store and pick up a shampoo, body lotion, or even a simple bar of soap. The refreshing blend of floral and balsamic notes often owes some gratitude to isoamyl salicylate. Chemists and perfumers value it for its unique combination: a soft, sweet floral scent that stands out without overpowering other ingredients.
Smooth Compatibility in Everyday Products
Having spent years working with cosmetic and personal care brands, I have seen isoamyl salicylate pop up repeatedly. With its low odor threshold and easy blending alongside dozens of other perfume ingredients, it becomes a sort of unifying force in fragrance design. It’s not just for the perfume industry—this ingredient plays a big role across a whole range of scented products. The consistent quality and adaptability help keep product launches on track, making it easier for companies to ensure each batch smells just right.
Safe for Skin and Better for the Environment
Safety sits at the top of the list for anyone working in skincare and household goods. Extensive research shows isoamyl salicylate rarely causes skin irritation at the levels used in most formulas. Many natural enthusiasts lean towards it since some sources come from plants and its profile fits right in with the shift toward gentle, “green” chemistry. This aligns with findings from the International Fragrance Association (IFRA), outlining safe usage guidelines based on a range of independent studies.
Manufacturers in the fragrance world talk a lot about sustainability. Synthetic alternatives to rare or endangered plant extracts become necessary if we want beautiful scents but also hope to keep rainforests intact. Isoamyl salicylate answers this call. Its production doesn’t damage ecosystems, and it allows perfumers to achieve certain aromas without sourcing rare botanicals.
Cost Savings and Product Stability
For anyone managing costs in the beauty business, ingredients that pull double duty get extra points. Isoamyl salicylate proves robust in a technical sense—it remains stable through storage, doesn’t break down easily under light or heat, and keeps perfume blends from fading quickly. Consumers get lotions and shampoos that smell fresh on the shelf after many months, while companies sidestep expensive reformulations.
Fewer recalls, less waste, and lower raw material costs add up in a competitive industry. Small tweaks with this ingredient often ripple across entire product lines. That means less volatility in supply chains for the big brands and growing room for smaller companies.
Challenges and the Road Ahead
Some pressures remain. Regulatory bodies update guidelines each year, and not every market treats isoamyl salicylate the same way. In Europe, for instance, fragrance allergen labeling rules keep changing. Transparency with consumers gives brands a leg up, especially as buyers want more info about what goes into their bottles.
Research continues into improving how we make ingredients like this one. Methods that minimize waste or energy use hold promise, both for the environment and for wallet-conscious companies. Brands working with third-party certifiers, such as Ecocert or the EU Ecolabel, can turn ingredient choices into clear, trustworthy signals to their customers.
Payoff in Everyday Comforts
Isoamyl salicylate stands out because of how it enhances our daily lives. Every time a new fragrance launches or a favorite sunscreen feels a little more pleasant, most people never think about the chemistry behind the scenes. Yet these small details matter. A more enjoyable sensory experience, safer skin contact, and better environmental outcomes—these keep both businesses and consumers coming back to tried-and-true ingredients.
Does Isoamyl Salicylate have any side effects?
Why People Use Isoamyl Salicylate
Isoamyl Salicylate pops up in everyday life a lot more than most realize. Walk into any pharmacy or supermarket, you’ll find it tucked away in perfumes, lotions, sunscreens, and even some shampoos. It brings a sweet, fruity scent that helps everything smell fresher. Fragrance makers usually chase after ingredients like this one—it gives their blends a special kick.
Getting to Know the Risks
Using scented products on my skin always made me a bit nervous. My first encounter with a lotion containing Isoamyl Salicylate didn’t end well—my skin got red and itchy. Dermatology research shows this isn't so rare. People with sensitive skin, especially children or those with allergies, have reported irritation and mild inflammation after repeated use. The European Cosmetic Ingredient Database lists Isoamyl Salicylate as a known skin sensitizer. In plain words, enough exposure pushes some people’s bodies to react defensively.
Scientists from the American Contact Dermatitis Society studied reactions to common fragrance ingredients. Their findings: Isoamyl Salicylate rarely causes severe allergy, but even so, those with existing allergies should pay attention. Hives, red patches, or swelling can crop up, sometimes several hours after applying a product. The problem is, most packaging buries the ingredient deep in a long list of unpronounceables.
Long-Term Concerns and Regulations
Research from the past few years hasn’t dug up major long-term health risks. Regulatory bodies in Europe, North America, and Asia keep watching for problems, especially since some fragrance chemicals have shown possible links with hormone disruption or other chronic effects. But Isoamyl Salicylate’s record sits clean for now on cancer or hormone interference. Agencies like the European Chemicals Agency update safety reviews often, following new studies as they come in. Still, things can always change if fresh data points in a new direction.
Taking Precautions
I’ve learned to give new body care products a small patch test. Just a dab on the inside of the elbow, and I wait a day. Dermatologists agree that simple steps like this help catch reactions early. People with sensitive skin, eczema, or a history of allergies should go a step further—reading ingredient lists thoroughly. Brands sometimes use other names, like 3-Methylbutyl Salicylate, so a little research goes a long way.
Parents have an extra reason to stay sharp. Children's skin soaks up chemicals faster, so pediatricians advise caution with fragranced lotions or sprays around the very young. Pregnant women also take care, as chemical exposures during pregnancy spark ongoing research.
Looking For Safer Alternatives
The personal care industry moves pretty fast these days. Brands talk more about skin-friendly formulas that ditch risky additives. Botanical options or unscented products cut out a lot of the guesswork for allergy sufferers. Stores are better at labeling products that avoid salicylates and other potential triggers, making it easier for folks to pick safer choices.
Knowing what goes into our skin care and household items matters—those small print ingredients shape comfort and well-being more than many think. My own journey taught me the importance of staying curious, reading labels, and listening to my body. Sometimes the safest bet comes from switching to simpler products and keeping things gentle, especially when skin starts acting up.
Is Isoamyl Salicylate natural or synthetic?
Understanding Where Isoamyl Salicylate Comes From
Isoamyl salicylate always shows up on ingredient lists for perfumes, lotions, and even cleaning products. Its sweet, floral aroma gives a fresh finish, and perfumers rely on it to add both roundness and warmth. The main question pops up again and again: is this compound natural, or does it spring from a beaker in a lab?
Looking at the Source
Nature has ways to deliver isoamyl salicylate. Some plants—like certain primula species—create small amounts during regular metabolic processes. Still, finding enough from natural sources poses a challenge. Synthetic production steps up to offer a practical way to make the material in large batches. Labs blend isoamyl alcohol and salicylic acid, using a process that mirrors what happens in plants, but without the unpredictability of harvest size or growing seasons.
Why Synthetic Matters
Companies pick the synthetic route for a few reasons. Consistency ranks at the top. Harvests vary, soil changes, pest problems, weather shifts—these issues hit crops and change what's available from year to year. Synthetics keep up with demand, delivering a stable supply at lower costs, and allow for adjustments in purity that natural sources simply can’t promise.
Another factor comes from sustainability. Harvesting natural sources sometimes strains wild populations. Synthetic production trims environmental impact, keeping rare plants off endangered lists and lets manufacturers sidestep debates over ethical sourcing.
Safety and Quality Considerations
People want to know if synthetic equals safe. Research from well-known regulatory organizations like the EU Scientific Committee on Consumer Safety points toward agreement: synthetic isoamyl salicylate, when used within recommended limits, does not pose greater risk compared to the natural compound. Its chemical structure matches the natural one, so allergic responses and safety profiles look basically the same.
What changes, though, is the sense of trust. Words like “natural” get leveraged in marketing, often swaying people away from lab-created materials. It helps to remember that nature produces both allergens and toxins, while the lab brings consistency and reliability. As a dad with a child prone to eczema, I’d rather see formulations that have been tested to the last molecule, synthetic or not.
Transparency Helps Guide Choices
Shoppers ask for more honesty from brands on ingredient sourcing. Labels with clear details, and companies explaining their choice to use synthetics (or not), allow folks with sensitivities or strong values to decide for themselves.
Whatever the source, open communication about safety testing, possible allergens, and environmental footprint makes a big difference. Brands and regulatory groups could work together to set standards for labeling so consumers stay better informed.
Looking Toward the Future
Innovation keeps pushing the boundaries between natural and synthetic. Advances in biotechnology may someday allow a middle ground: fermenting yeast or bacteria to produce isoamyl salicylate in controlled environments, offering a “nature-identical” product that benefits both industry and planet.
For now, most isoamyl salicylate on shelves comes from the lab. The synthetic version allows safe, dependable, and more eco-friendly use across a wide range of personal care and household products. Judging value goes beyond the label—people want real information about safety, purity, and impact on both body and environment, no matter the method of production.
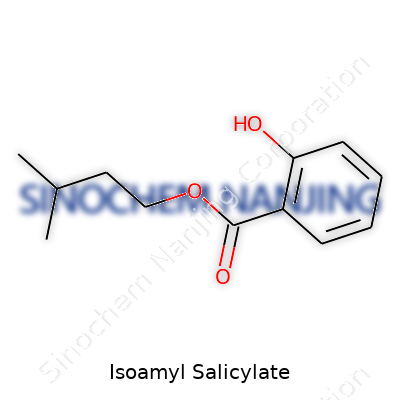
| Names | |
| Preferred IUPAC name | 3-Methylbutyl 2-hydroxybenzoate |
| Other names |
Isoamyl 2-hydroxybenzoate 3-Methylbutyl salicylate Isopentyl salicylate |
| Pronunciation | /ˌaɪ.soʊˈæ.mɪl səˈlɪ.sə.leɪt/ |
| Identifiers | |
| CAS Number | [87-20-7] |
| 3D model (JSmol) | Isoamyl Salicylate JSmol 3D model string: ``` Isoamyl Salicylate InChI=1S/C12H16O3/c1-4-5-8(2)9-15-12(14)10-6-7-11(13)16-10/h6-8H,4-5,9H2,1-3H3 ``` |
| Beilstein Reference | 1327296 |
| ChEBI | CHEBI:77823 |
| ChEMBL | CHEMBL1311317 |
| ChemSpider | 10973 |
| DrugBank | DB14170 |
| ECHA InfoCard | ECHA InfoCard: 100_012_047 |
| EC Number | 246-487-1 |
| Gmelin Reference | 6747 |
| KEGG | C14468 |
| MeSH | D020969 |
| PubChem CID | 68969 |
| RTECS number | WQ2450000 |
| UNII | 8Q1V2V916P |
| UN number | UN3082 |
| CompTox Dashboard (EPA) | DTXSID0020685 |
| Properties | |
| Chemical formula | C12H16O3 |
| Molar mass | 208.25 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | sweet fruity balsamic |
| Density | 1.045 g/cm3 |
| Solubility in water | Insoluble |
| log P | 3.9 |
| Vapor pressure | 0.0012 mmHg (25 °C) |
| Acidity (pKa) | 13.8 |
| Basicity (pKb) | 13.09 |
| Magnetic susceptibility (χ) | -62.0e-6 cm³/mol |
| Refractive index (nD) | 1.5050 |
| Viscosity | 37.8 mPa·s (at 20°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 405.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -489.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7035.4 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H317: May cause an allergic skin reaction. |
| Precautionary statements | Precautionary statements: "P261, P273, P302+P352, P305+P351+P338, P362+P364 |
| Flash point | 104°C |
| Autoignition temperature | > 375 °C |
| Lethal dose or concentration | LD50 (oral, rat): 4,000 mg/kg |
| LD50 (median dose) | > 4.52 g/kg (rat, oral) |
| REL (Recommended) | 1.0% |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Amyl salicylate Menthyl salicylate Phenyl salicylate Benzyl salicylate Methyl salicylate Ethyl salicylate Cyclamen aldehyde |

