Isoamyl Isovalerate: A Close Look at an Underappreciated Ester
Historical Development
Isoamyl isovalerate never made front-page news, yet its journey has mirrored the quiet progress of chemical innovation across the last century. Manufactured esters like this one cropped up as chemists explored how nature crafts flavors and fragrances. Isoamyl isovalerate came from the urge to replicate the ripe, juicy notes found in certain fruits, and since then, researchers have revisited it with every leap in both food science and industrial chemistry. Looking back at the patents from the early 1900s, it’s clear that chemical engineers soon appreciated its mild banana and pear scent, steadily rolling out synthetic batches both for testing and for commercial use. Instead of just sticking to natural extraction, labs in Europe and America searched out more controlled, pure ways to synthesize this ester, pushing the envelope for food-grade and fragrance applications.
Product Overview
Isoamyl isovalerate lands in that sweet spot many chemists chase: a compound that quietly serves both sensory and industrial needs. You’ll smell hints of it in everything from candies to chewing gum. Unlike synthetic flavors that shout above the rest, this ester layers subtlety into formulations, letting other notes shine. Labs pick it for its stability and signature aroma, and food technologists rely on it when crafting composite flavors. The same molecule supports cosmetic technicians mixing up perfumed creams, always on the look-out for ingredients that play nice with preservatives and colorants.
Physical & Chemical Properties
Isoamyl isovalerate stands as a clear, colorless liquid at room temperature, giving off a fruity, slightly pungent scent. Its molecular formula—C10H20O2—lets it slip easily between the realms of food chemistry and perfumery. The boiling point hovers above 190 °C, meaning it hangs around in products rather than evaporating at the first hint of warmth. The density fits right within the range for many organic esters, so it doesn’t separate easily from carrier oils or alcohols. Solubility leans more toward organic solvents; water shakes hands with only a small fraction, so most industrial pipelines handle it alongside ethanol or propylene glycol.
Technical Specifications & Labeling
Chemists look for high assay purity to minimize trace byproducts. Reputable suppliers publish gas chromatography data on each batch, giving formulators the confidence that what they’ve bought matches international standards. Food standards agencies require clear labeling, so food technologists check not just for the CAS number but country-of-origin certificates and batch records. In regulated markets, agencies like FDA or EFSA keep a close eye, making sure concentration levels land well below safety thresholds.
Preparation Method
Most commercial batches of isoamyl isovalerate grow out of classic esterification. Mixing isoamyl alcohol with isovaleric acid, plus a touch of acid catalyst—typically sulfuric acid—drives the reaction forward. After a few hours under controlled temperatures, distillation captures the purified ester. Chemists learned that small tweaks in temperature or catalyst loading shift yields, so most producers spend years fine-tuning their set-ups. Protecting purity through each stage means less risk downstream, whether in food, fragrance, or specialty chemicals.
Chemical Reactions & Modifications
Ester bonds handle mild hydrolysis, so when water and a little acid or base cross paths with isoamyl isovalerate, you’ll see a slow breakdown into original alcohol and acid. On the shelf, this matters—producers factor in long-term stability, especially for food products sitting under various storage conditions. As for modifications, chemists sometimes stretch for longer-chain or branched esters by swapping out either starting component, chasing specific aroma profiles, volatility, or physical handling traits.
Synonyms & Product Names
Industry professionals have juggled plenty of names for isoamyl isovalerate: banana oil ester, isopentyl 3-methylbutanoate, and even pear oil ester in some catalogues. Globally, these synonyms pop up on import documents, regulatory filings, and in the fine print of food flavoring databases. Formulators need to cross-reference every alias to avoid mix-ups or regulatory snags, especially when exporting products or submitting for market clearances.
Safety & Operational Standards
The compound hasn't drawn much controversy when used in flavor concentrations that regulators approve. As with most esters, inhalational exposures get the most scrutiny—workers in plants producing or formulating with it wear gloves and splash-resistant eyewear. Ventilation beats out most concerns about accidental vapor build-up, though nobody can afford to ignore chemical handling protocols. Agencies such as OSHA lay out guidelines, but local practices matter just as much; it’s the technicians on the floor who keep these operations humming safely. Toxicologists have noted low acute toxicity by oral and dermal routes for this ester, but rigorous batch testing always runs alongside routine production. Fire risk stays low under proper storage conditions, but emergency responders always have protocols on hand, just in case a spill needs fast neutralization.
Application Areas
Isoamyl isovalerate has found solid ground in the food and beverage sector, acting as a stealthy enhancer in fruit-flavored sodas, candies, and pastries. Its next stop comes in personal care, where it underpins the fruit notes in young-adult perfumes and certain hair products. Some lubricant and plasticizer formulations take advantage of its slip and scent durability, broadening its reach beyond food and fragrance. Every application throws a different challenge at the molecule—from staying stable in acidic drinks to keeping its aroma punchy in body sprays—demanding careful engineering from product developers.
Research & Development
Recent trends in food and fragrance science have nudged researchers to revisit even basic esters like isoamyl isovalerate. Ongoing work explores biotechnological synthesis, where genetically modified yeast or bacteria manufacture esters directly from sugar feedstocks. Success in this line could shrink carbon footprints and foster cleaner, more reliable supply chains, especially as consumer demand for "natural" ingredients outweighs mere synthetic analogues. Academic groups probe the flavor synergy possibilities, vowing to create more sophisticated blends that taste like the real thing, not a laboratory approximation.
Toxicity Research
So far, toxicity profiles for isoamyl isovalerate look benign at levels used in food and consumer products. Studies submitted to regulatory agencies examined both acute and repeat-dose toxicity, covering oral, dermal, and inhalational routes. The compound breaks down rapidly in the body, so it rarely accumulates to dangerous levels. Some research teams have explored allergenic potential in sensitive populations, while toxicologists keep an eye on metabolic pathways to ensure no concerning byproducts sneak into the system. Safety evaluators stress that risk always hinges on dose—every flavor chemist and regulatory body follows strict limits to head off potential problems.
Future Prospects
Isoamyl isovalerate might never claim the limelight, but it holds promise as part of a bigger shift toward sustainable chemistry. Emerging green synthesis platforms—fermentation, enzyme catalysis, and waste valorization—could produce this ester with less environmental impact and tighter quality control. Beyond just taste and smell, researchers eye pharmaceutical excipient applications and specialty material uses, leveraging the ester bond’s reactivity. The next decade could see this compound elevated from under-the-radar helper to a strategic ingredient as new diets, regulatory climates, and consumer values evolve. Whether in a school laboratory or a sprawling chemical plant, isoamyl isovalerate keeps illustrating the subtle art of marrying chemistry with real-world needs, all without drawing too much attention to itself.
What is Isoamyl Isovalerate used for?
A Whiff of Context
Most folks rarely hear about isoamyl isovalerate unless they dive into the small print on a bottle or food label. This compound shows up in places where taste and aroma matter, especially in foods and personal care products. I remember walking through an industrial facility, and the chemists explained how this stuff helps manufacturers give certain foods and products that juicy, fruity punch—without ever slicing a real apricot or banana.
The Flavor Engineer
Isoamyl isovalerate’s claim to fame comes down to its fruity scent and taste. Food companies use it to tweak the flavors of gum, candies, and even non-alcoholic beverages. It pops up in products that need to mimic those naturally sweet smells that trigger memories of childhood treats. Manufacturers pick isoamyl isovalerate when they want to boost flavor without expensive or perishable fruit extracts.
Regulatory bodies like the FDA and EFSA approve it for this purpose based on thorough reviews. Its reputation for being low-risk at usual intake levels means it slides into ingredient lists without much controversy. Compared to other synthetics that might leave a chemical aftertaste, isoamyl isovalerate blends in more smoothly and can create consistent flavors batch after batch.
Perfumes, Scents, and Skin
Anyone who enjoys scented lotions or perfumes indirectly bumps into isoamyl isovalerate. Perfumers use it as part of the fruity note palette in formulations. In a market where customer loyalty hinges on sensory experience, even a faint whiff can set a product apart. Personal care products turn to this ingredient for that tropical, uplifting aroma, offering a simple way to mask less pleasant base scents or create an inviting profile in products that spend all day on the skin.
Safety and Trust in the Supply Chain
Consumer trust hangs on the safety and transparency of chemical usage. Experienced food scientists track usage levels carefully because even compounds recognized as safe can prove irritating if overused. Professional associations and regulatory agencies conduct regular reviews, checking health impacts and reporting on any patterns in adverse reactions. Recent public interest in ingredient transparency means companies have to stay sharp and communicate clearly.
To meet these expectations, I’ve seen industry leaders go beyond the minimum. They source raw materials from long-standing suppliers with good quality records. Batch testing checks every lot for purity, and companies sometimes publish independent lab results. This builds credibility and aligns with the public’s demand for more straight talk about what winds up in their foods and skin care.
Moving Toward Cleaner Labels
A push for fewer ingredients and simpler formulations puts pressure on flavor houses and manufacturers. Instead of hard-to-pronounce chemicals, people ask for recognizable foods and straightforward processes. Companies experimenting with plant extracts or fermentation-based methods aim for the same taste and scent effects as isoamyl isovalerate, just from sources the public finds easier to accept.
Some brands already pilot blends using new biotechnologies. Results show promise—real fruit flavors without spoilage or year-round supply headaches. As demand grows, expect to see more changes behind the scenes, with natural alternatives edging into spaces where synthetic compounds held steady for decades.
Is Isoamyl Isovalerate safe for human consumption?
What Is Isoamyl Isovalerate?
Isoamyl isovalerate shows up on some ingredient lists because it packs a strong fruity, banana-like aroma. If you walk through a candy factory or peel open a stick of bubblegum, chances are you catch this scent. Manufacturers use it to boost flavors or mask unpleasant ones in processed foods, drinks, and even perfumes.
Looking at the Safety of This Compound
Most folks hear a complicated chemical name and pause. I don’t blame them. There’s plenty of reasonable fear about strange-sounding ingredients. Questions start: Who checks this stuff? Am I putting my health at risk? Food safety oversight comes down to more than just a governmental rubber stamp—trust needs transparency.
Reliable oversight counts. For Isoamyl isovalerate, both the US Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) have looked closely. The FDA includes it on the list of flavoring substances “generally recognized as safe” (GRAS) when used in the right amounts. EFSA reviews back this up, citing no evidence of harm at normal levels found in food. These agencies rely on animal studies, metabolism research, and exposure data. So far, there’s been no convincing signal that it causes cancer, disrupts hormones, or leads to allergic reactions in people.
How Much Is Too Much?
Everything people consume carries a question of dose. Even table salt can hurt the body if used recklessly. Regulatory safety shields exist for a reason: flavor companies and food processors can’t just “dump and hope.” Isoamyl isovalerate is present in tiny concentrations—fractions of a milligram per kilogram of food. Toxicity tests have explored far higher doses than anyone gets from eating snacks or sipping drinks; the results show a large safety margin.
What Science Tells Us So Far
Not every food additive stands the test of time. History gives plenty of examples where further research uncovered dangers. Saccharin once looked harmless, then animal studies raised red flags before human studies softened concerns again. That lesson sticks with anyone who writes or researches about food safety: keep asking questions, stay open to new findings.
Reports so far put Isoamyl isovalerate in a low-risk category. I’ve talked with food scientists who monitor flavor safety, and their caution is steady but not alarmist. For those with rare conditions like metabolic disorders, speaking with a doctor about all food additives makes sense. The rest of us can focus concern elsewhere unless new evidence appears.
Trust and Transparency Matter
Seeing the FDA and EFSA base approvals on published research brings a measure of comfort, especially compared to shadowy compounds whose safety data doesn’t see the light of day. Public access to studies grows trust. I’m always glad to see updates made public and clear. Skepticism deserves real answers, but scare stories using complicated names shouldn’t drive panic.
The Path Forward: Staying Informed
Isoamyl isovalerate reflects the broader issue of food science: science needs watchdogs. Regulators, researchers, and consumers share this responsibility. Industry should disclose usage levels and safety data, while agencies should review findings with healthy suspicion and scientific rigor. If the science changes and risks come to light, guidelines can shift—just as they have with other additives in the past.
Right now, eating products flavored with isoamyl isovalerate doesn’t put health at risk under normal conditions. Still, asking what’s in your food and why it’s there stays important for everyone’s long-term well-being.
What are the main applications of Isoamyl Isovalerate?
Flavoring and Fragrance: Everyday Sensory Experiences
Isoamyl isovalerate stands out for the kind of aroma and taste it brings to the table. Its fruity, pleasantly sweet scent shows up in all sorts of products that people use daily. Walk past the bakery aisle, and that hint of ripe fruit sometimes comes from more than just real fruit; companies rely on isoamyl isovalerate to shape the sensory character of candy, chewing gum, and beverages. The public expects authenticity and safety in flavor additives. Regulatory agencies like the FDA and the European Food Safety Authority give the go-ahead to use this compound as a food additive since it meets safety standards, as long as it stays within set limits.
In my own kitchen, I have tinkered with flavorings for homemade liqueurs and desserts. That distinct “banana-like” note comes from esters like isoamyl isovalerate. The fun of adding a few drops of the right essence to boost fruitiness without the hassle of sourcing exotic produce should not go unnoticed; it offers real convenience, given most of us do not have access to tropical fruit year-round.
A Role in Perfumes and Cosmetics
Take a walk into the world of perfumes, body sprays, and soaps. Perfumers need stable ingredients that resist breaking down and mixing poorly with other scents. Isoamyl isovalerate brings a playful touch, sliding into floral, fruity, and even some “green” fragrance compositions. It blends well, so professionals get layered scents that last. Oftentimes, a product’s first impression with consumers comes from smell; using safe, reliable esters that come with a history of non-allergenicity supports hard-earned trust in major brands.
Cosmetic chemists, too, lean on its mild scent and ability to mask harsher odors. I remember a friend who had allergic reactions to certain synthetic fragrances, but tolerated formulas with milder esters like this one. It makes a real-world difference for people with fragrance sensitivity or allergies.
Supporting Pharmaceuticals and Fine Chemicals
Pharmaceuticals use isoamyl isovalerate as a carrier or masking agent in some medicines. Not every pill or liquid has to taste terrible. Flavor masking lets patients, especially kids, take medicine without gagging. Beyond taste, the fact that it plays nicely alongside other ingredients and stays shelf-stable means less worry about products breaking down over time.
Green Chemistry and Industrial Use
Recently, manufacturers began moving away from harsh, high-impact solvents and toward safer, greener alternatives. Isoamyl isovalerate ticks a few important boxes: it comes from non-toxic feedstocks, breaks down easily, and does not linger in the environment. Some companies have started using it in specialty lubricants and as a plasticizer for biodegradable plastics. Choosing minor, safer chemicals like this one lowers the load of hazardous materials used across supply chains. Responsible sourcing and transparency about ingredient safety continue to gain traction as consumer demand rises for sustainable, ethical products.
Challenges and Looking Ahead
The industry does face challenges—scaling up production sustainably, handling supply chain pressures, and managing costs. Sourcing from renewable raw materials and investing in cleaner manufacturing can keep production responsible. Researchers keep looking for ways to synthesize these esters biologically, using fermentation and enzymes to reduce the chemical waste of traditional processes. Companies that openly share their sourcing and safety data will win more trust, especially in food and personal care markets.
What is the flavor or aroma of Isoamyl Isovalerate?
The Everyday Experience of Isoamyl Isovalerate
Open a pack of fruity gum or taste a bonbon loaded with tangy flavor, and chances are isoamyl isovalerate has come into play. The stuff brings its own signature—imagine ripe, yellow Chiquita bananas and a hint of pear. Not many folks stop to break down flavor when biting into a chewy candy, but here, science and daily snacking meet face-to-face.
Where Does the Flavor Take You?
Isoamyl isovalerate, an ester found in certain fruits and a favorite among flavorists, layers sweetness onto almost anything it touches. The sensation lands in the nose first, followed by a creamy, unmistakably “banana” note that lingers. It is more than simple fruitiness, though; there’s a tropical roundness, both playful and familiar, which doesn’t dive into sharp acidity. Every time I’ve encountered it in food labs or kitchen projects, it’s almost like a cheat code for nostalgia—instantly pulling up memories of school lunchboxes or old-fashioned hard candies.
Beneath the Surface
People outside flavor science circles probably don’t know what isoamyl isovalerate smells like alone, unblended. In its natural state, you get an aroma, thick and sweet, closer to overripe banana peels. Older flavor books describe the note as having both a juicy, fruity side and a slightly funky edge—something reminiscent, for some, of sweaty gym socks left to stew a bit. Nature gave us fruit esters like this because they help plants attract animals to spread their seeds. Humans took that cue and folded the molecule into drinks, candies, and even some perfumes.
Why This Matters to Food Makers and Eaters
Choosing the right aroma molecule shapes not just product flavor but also consumer trust. If a banana yogurt doesn’t smell the way our memories expect, folks might turn away before the first bite. Isoamyl isovalerate helps bridge the gap between synthetic and authentic, sketching out a full banana profile without the price—or unpredictability—of real fruit. Its contribution feels subtle, but without it, many snacks and beverages would taste flat or artificial.
Looking Beyond Flavor
For formulators, sourcing pure and food-grade isoamyl isovalerate stands as a non-negotiable. Certifications, lab reports, and supplier track records guide those choices. Safety standards, especially with such a bold aromatic character, need strict attention. Documentation and batch testing cushion against contamination, keeping products both legal and safe to eat. The molecule itself holds GRAS (Generally Recognized As Safe) status by the US FDA, but the lines between “safe” and “satisfying” get clearer only by testing and regular feedback from tasting panels and buyers.
The Search for Better Flavor
Consumers today ask more about what goes into their food than ever before. People want transparency, fewer artificial additives, and profiles that mirror what nature offers. Isoamyl isovalerate—sourced sustainably and blended thoughtfully—can answer those calls. For businesses, highlighting origin, purity, and function strengthens brand integrity in crowded market shelves. For those of us who care about food on both sides of the counter, following not just the flavor but its story takes eating from routine to memorable.
Where can I purchase Isoamyl Isovalerate?
Understanding Isoamyl Isovalerate
Isoamyl Isovalerate doesn't show up in your average grocery store or pharmacy aisle. This compound pops up mostly in the world of flavors and fragrances. It smells a bit like apple or even a touch of pear, which explains why companies add it to certain foods, drinks, and perfumes. Some labs work with it—scientists look at how it interacts with other compounds or how it can safely jazz up consumer products. If your goal involves flavor science, fragrance formulation, or a specialized research project, finding high-purity, well-documented supplies should rank at the top of your checklist.
Where to Buy: Turning to Trusted Suppliers
Good sources for this chemical typically include specialty chemical shops, raw material distributors serving the food or fragrance trade, and laboratory suppliers. Big names in the chemical industry—companies like Sigma-Aldrich, TCI Chemicals, and Fisher Scientific—have thorough catalogs, including safety data sheets, certificates of analysis, and reliable customer support. These suppliers usually focus on professionals and organizations, so expect account verification and minimum purchase requirements.
Sometimes, smaller distributors operate online platforms, offering smaller quantities aimed at hobbyists, researchers, or indie perfumers. Still, customers must check for key things: batch purity, documentation, and whether the vendor willingly answers technical questions. Lack of documentation or reluctance to share safety information should give any shopper reason to look elsewhere.
Regulations and Quality Control
Isoamyl Isovalerate’s use in food and fragrance comes with government rules aimed at keeping consumers safe. In the European Union, REACH and food safety laws spell out requirements for testing and labeling. In the U.S., the FDA and OSHA oversee food additives and workplace safety. Not every vendor keeps up with these rules or provides clear documentation. Here, experience counts: legitimate suppliers back their products with robust testing, batch tracking, and clear usage guidance.
Some buyers cut corners by ordering from online marketplaces or overseas drop-shippers. This brings major risks—counterfeit ingredients, inaccurate labeling, or batches contaminated with unwanted chemicals. These risks go straight to my concern for health and safety in any lab or production environment. My work in science communication taught me plenty about the headaches that result from questionable sourcing—wasted materials, safety scares, and costly disruptions.
Safe Purchasing: Practical Steps and Red Flags
Safe purchasing often starts with identifying suppliers who maintain transparent business practices. Look for suppliers who answer questions about product origin, purity, and compliance. If the supplier skips out on safety data or skips listing the country of manufacture, turn elsewhere. Before placing an order, I always ask for the batch number and COA, then check if third-party test results match up. Never settle for vague answers—reputable sellers stand behind their products.
Some professional groups—like the Society of Flavor Chemists or American Chemical Society—maintain lists of vetted vendors. I recommend starting searches through these resources or directly through established laboratory distributors. If the product is not available domestically, buyers must research the import rules, understand taxes, and factor in shipping times.
The Value of Professional Guidance
My experience suggests talking to industry colleagues or mentors before making a purchase decision. Peer recommendations save time and help sidestep unreliable sellers. Sometimes university labs maintain surplus supplies and can point private-sector buyers in the right direction. The more people share what works, the less often others fall for dubious offers.
Source quality drives product results, especially with chemicals like Isoamyl Isovalerate. Take care in choosing where to buy. Fact-check, ask plenty of questions, and keep your standards high.
| Names | |
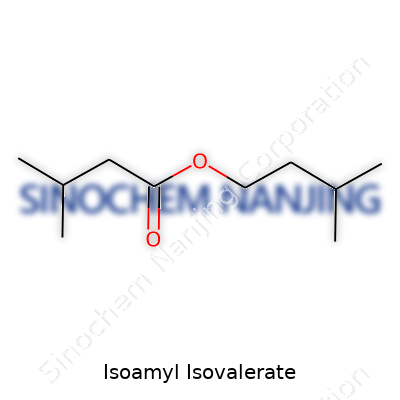
| Preferred IUPAC name | 3-Methylbutyl 3-methylbutanoate |
| Other names |
3-Methylbutyl 3-methylbutanoate Isoamyl 3-methylbutyrate Isopentyl isovalerate Isopentyl 3-methylbutanoate |
| Pronunciation | /ˌaɪ.soʊˈæm.ɪl ˌaɪ.soʊˈvæl.əˌreɪt/ |
| Identifiers | |
| CAS Number | 102-28-3 |
| 3D model (JSmol) | `Isoamyl Isovalerate|JSmol|C1=CC=C(C=C1)C(=O)OCC(C)COC(=O)CC(C)CCC` |
| Beilstein Reference | 2137096 |
| ChEBI | CHEBI:48918 |
| ChEMBL | CHEMBL3184853 |
| ChemSpider | 14194188 |
| DrugBank | DB16544 |
| ECHA InfoCard | 0754E2A2-0E32-49B2-9B7B-6B800AD85F08 |
| EC Number | 208-759-1 |
| Gmelin Reference | 10470 |
| KEGG | C19687 |
| MeSH | D02.241.223.211.700.460 |
| PubChem CID | 8788 |
| RTECS number | NJ3486000 |
| UNII | C38FA97A13 |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | DTXSID10229058 |
| Properties | |
| Chemical formula | C10H20O2 |
| Molar mass | 216.34 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity, apple, sweet, banana |
| Density | 0.868 g/mL at 25 °C(lit.) |
| Solubility in water | Insoluble in water |
| log P | 3.92 |
| Vapor pressure | 0.0735 mmHg (25°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | 12.47 |
| Refractive index (nD) | 1.4150 |
| Viscosity | 3.959 cP (25°C) |
| Dipole moment | 2.55 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 527.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -537.65 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7036.7 kJ/mol |
| Pharmacology | |
| ATC code | **'A16AX16'** |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P280: Wear protective gloves/protective clothing/eye protection/face protection. |
| NFPA 704 (fire diamond) | 1-2-1-0 |
| Flash point | 88°C |
| Autoignition temperature | 403°C |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 5000 mg/kg |
| PEL (Permissible) | PEL for Isoamyl Isovalerate: Not established |
| REL (Recommended) | 10 mg/kg |
| Related compounds | |
| Related compounds |
Isoamyl acetate Isovaleryl chloride Isovaleric acid Isoamyl alcohol Ethyl isovalerate |

