Isoamyl Acetate: More Than Just a Banana Scent
Looking Back: The Long, Fruity Road of Isoamyl Acetate
Isoamyl acetate has one of those stories where science, industry, and culture just keep crossing paths. Discovered in the 19th century, chemists tinkered with the natural compounds behind certain flavors found in fruits. Suddenly, folks realized they could put the unmistakable aroma of ripe bananas into anything from candy to polishing agents. Early production methods used fermentative byproducts from grain processing. Over time, the industrial era needed more efficient processes, leading to direct esterification—the bread and butter of bulk chemical manufacturing. This molecule's journey reflects how chemistry’s progress often unfolds: from the curiosity in academic labs, to the roar of commercial fermenters, and on to everyday consumer products.
Isoamyl Acetate: What Sets It Apart
Isoamyl acetate usually gets noticed for its aroma. Take a whiff of banana-flavored candies or sniff around the varnish aisle and you’ll notice its presence right away. This clear, colorless liquid features a low boiling point and mixes up nicely with most organic solvents but resists water. It slips into both the food and industrial scenes without fuss because of its gentle scent—strong but not overwhelming. That’s not just marketing talk: researchers show humans can detect this ester at concentrations as low as a few parts per billion. For those behind the scenes in labs or factories, that sort of potency is a double-edged sword. Handling the stuff means working out ventilation and storage tricks to keep odors in check.
Getting the Chemistry Right
Chemically speaking, isoamyl acetate belongs to the family of esters formed by marrying an acid and an alcohol—in this case, acetic acid and isoamyl alcohol. Its structure, C7H14O2, brings together five carbons branching off as isoamyl, capped with the familiar banana ester finish. It weighs in at about 130 grams per mole and flashes a boiling point comfortably below water, hovering near 142°C. In the beaker, it remains stable under normal conditions but reacts with strong acids or bases. Shelf lives depend on keeping temperatures steady and avoiding exposure to sunlight or air, as oxygen and UV can break it down over time. Simple, yes, but the details decide shelf safety in warehouses around the world.
Prepping Isoamyl Acetate: Tried-and-True Methods
Old-school chemists often made isoamyl acetate by mixing isoamyl alcohol pulled from fusel oils with acetic acid, then heating the mix in the presence of an acid catalyst. This Fischer esterification still holds up for volume production because it keeps waste down and conversion yields high. Many manufacturing plants recycle leftover alcohol or use clever distillation columns to recover pure product. Researchers also study biotechnological routes—using microbes to nudge alcohol and acid together. These methods might eventually lower the energy bill and reduce unwanted byproducts.
Chemical Moves: Reactions Worth Knowing
Though isoamyl acetate doesn’t pack much drama as a reactant under normal storage, it plays its part in further organic syntheses. It resists most attack unless a strong base swings by, which hydrolyzes it back to the starting alcohol and acid. Nifty modifications crop up in making custom flavorings or prepping analytical reagents. Most chemical companies appreciate its stability: it rarely fouls up process lines with unwanted side reactions.
Other Names, Same Chemical
Pick up a bottle and you might see isoamyl acetate labeled as 3-methylbutyl acetate, banana oil, or isopentyl acetate. Each name hints at its chemical structure or signature odor. The wide list of synonyms arises from decades of use across chemistry, perfumery, food science, and industrial engineering. Keeping track of these synonyms becomes important in regulatory filings, research papers, and import-export records.
Staying Safe: Handling Standards and Lab Wisdom
Handling isoamyl acetate brings up the same basic issues as other volatile organics: flammability, strong odors, and some skin or eye irritation risk. Chemists rely on local legislation for workplace limits—usually, it’s about keeping exposure below certain thresholds to avoid headaches or mucous membrane irritations. Strong ventilation and protective gloves tend to do the trick. In bigger operations, manufacturers lay out clear labeling and usage protocols, flagged by hazard pictograms and warnings against static buildup during bulk transfers. The importance of respecting these standards becomes clear the first time a vapor cloud sets off the fire alarm or brings on a wave of office complaints.
Where Isoamyl Acetate Leaves Its Mark
Think about any product that needs a burst of banana or pear aroma and isoamyl acetate likely played a role. Candy, chewing gum, baked sweets, and even some flavored liquors rely on its punch. Beyond flavors, it acts as a solvent in varnishes, lacquers, and as a carrier in the printing industry. Winemakers sometimes run into it as a fermentation byproduct—good news in small amounts for flavor, not so great if it overwhelms subtler notes. Some manufacturers add it to gasoline as a denaturant. All told, its wide reach wraps back into everyday life, even if most folks have never heard its name.
Pushing the Envelope: Research and Development
Recent studies dig into how microbes can churn out isoamyl acetate from biomass waste, aiming for greener production lines in the future. Computational chemists check out new synthesis catalysts that cut energy needs. Analytical chemists perfect sensors that sniff out trace amounts in food or air for quality control. By using isoamyl acetate as a model system, educators show students real-world links between chemistry and daily experience. New research often asks: can production become cheaper, safer, or cleaner? One project I followed looked at membrane separation technology, hoping to cut down the distillation energy that eats up plant budgets. Results aren’t always headline news, but bit by bit, the industry edges forward.
Toxicity: Respect, Don’t Fear
A lot of folks get nervous at chemical names, but the data on isoamyl acetate shows relatively low toxicity. Acute ingestion or inhalation in high amounts can irritate the digestive or respiratory systems, yet the amounts needed to do serious harm far exceed what consumers taste in candies or drinks. Regulatory agencies place it on lists for careful use in food and fragrance at controlled levels. Chronic long-term data doesn’t highlight significant risks at workplace exposure limits either. Researchers keep tabs on breakdown products and potential allergenicity, especially as more people report sensitivity to food additives. Like many chemicals, risk comes down to sensible handling and respecting boundaries set by decades of toxicological testing.
What’s Ahead for Isoamyl Acetate?
No single chemical can escape change forever. Isoamyl acetate’s future might look different if new green production techniques sweep through chemical manufacturing, based on fermentation advances or even enzyme catalysis. Growing consumer demand for “natural” ingredients also nudges companies toward bio-based sources. On the regulatory side, evolving food safety standards and increasing scrutiny of industrial solvents could reshape the landscape—prompting investment in purification or trace contaminant control. Researchers pitch in by developing faster, cheaper sensing technology to catch off-spec batches before they reach consumers. The story of isoamyl acetate echoes larger themes in science and society: balancing tradition with innovation, managing risks smartly, and keeping the benefits of chemistry open to as many people as possible.
What is isoamyl acetate used for?
The Science Hiding in a Banana's Smell
Anyone who’s peeled a banana has picked up that sweet, fruity aroma. Isoamyl acetate delivers most of that smell, both in bananas and in cheaper candy. Nature does it best, but the lab gets close. In real life, this chemical pops up in places you wouldn’t expect. I spent enough time on factory floors and in college chemistry labs to see how this one chemical crosses industries.
Flavor and Fragrance on a Budget
Isoamyl acetate flavors a lot more than fruit candy. Food makers lean on it when they want something to taste and smell like bananas—bubblegum, ice cream, soft drinks, even some baked goods. The upside of isoamyl acetate is simple. Bananas don’t always cooperate with a factory schedule. Weather hits crops, prices move around. This lab-created version helps keep flavors steady all year, even when storms mess with harvests. The U.S. Food and Drug Administration has checked it out and ruled it as generally safe to eat, so the folks on production lines can keep things humming along.
Beyond Food: Factories, Safety, and the Big Jobs
Isoamyl acetate works far beyond candy. In paint thinner or as a low-toxicity solvent, it holds up strong in industrial settings. Chemists lean on it when they want a cleaner that won't knock everyone out with strong fumes. For me, the real surprise hit in my own work was finding out about its use in leak detection for gas masks. You release a bit of isoamyl acetate; if you catch that banana aroma in your mask, you know there’s a leak. That beats waiting for a dangerous gas to tell you something’s wrong. Safety teams in military or emergency response groups rely on this simple test to protect lives. That’s not flashy science, but it keeps workers safe on tough jobs.
Making Perfume and Spirits Stand Out
Perfumers appreciate isoamyl acetate for its ability to boost fruity notes. It’s a staple for anyone designing new scents in the mid-range price point. Craft distillers and brewers also know this compound. Fermentation can spin out isoamyl acetate in good quantities, bringing fruity notes to whiskeys and brandies—sometimes a plus, sometimes a sign that something’s gone a bit funky. Brewers and distillers keep close tabs on it, because too much can throw off the flavors they’ve worked hard to develop.
Waste Control and Going Green
Industrial use calls for careful handling. Isoamyl acetate carries some risk if people don’t keep to safe storage and ventilation rules. People have become more conscious about chemical hazards, and labs keep it locked down so it doesn’t leak into the air or water supply. Local governments check permits and require proper disposal. One solution gaining momentum: recycling and reusing solvents. Some companies pull spent isoamyl acetate and recover it, cutting back on waste. That’s good for the environment, and it can help companies trim costs.
Looking Ahead
Isoamyl acetate’s role keeps growing between food, safety, scent, and industry. Synthetic flavoring and fragrance chemicals like this one make it easier to produce affordable, consistent goods. Moving forward, companies and labs keep looking for safer, greener ways to use and recycle it, blending reliability with responsibility.
Is isoamyl acetate safe to use in foods?
What’s Behind the Banana Scent?
Pop open a pack of banana-flavored candy, and the aroma you pick up comes from isoamyl acetate. This clear liquid brings a strong, sweet smell, reminding many of ripe fruit or even pear-flavored gum. Food companies add it to candies, baked goods, and even drinks to build flavors we crave. The roots of isoamyl acetate run deep in both natural and artificial processes, where it shows up in everything from real bananas to factory flavors made for mass production.
Digging Into the Science
Everyday folks often wonder: if something smells this strong, is it safe to eat? The U.S. Food and Drug Administration counts isoamyl acetate as “Generally Recognized as Safe” (GRAS) when used in food. Regulators and food safety boards across the world have checked it. There’s history at work—isoamyl acetate has flavored treats since before World War II. Some research has shown that it breaks down quickly in the body. The concentrations showing up in commercial foods are far below any danger zones.
Experience in quality control taught me how labs test food ingredients for safety again and again before products reach shelves. Most cases of food complaints circle back to much larger doses than anything found in flavorings. Even the World Health Organization stepped in, reporting that this compound hasn’t shown lasting harm at the amounts used in foods or drinks.
Concerns About Exposure
No chemical sits completely above scrutiny; some people with sensitive systems say strong flavors upset their stomachs. Chefs and developers rarely see real allergic responses to isoamyl acetate, but mild headaches or queasiness happen to a tiny group if exposed to high vapor levels. These reactions look similar to what happens with other strong scents (like cleaning sprays or nail polish removers). It’s good to remember that people handle aromas in different ways; one sniff rarely fits all.
Regulation and Oversight
Food safety doesn’t run on autopilot. Health agencies track new studies and make updates when needed. For instance, Europe’s food authorities place strict caps on flavoring quantities, requiring labels and reminders for factories. American rules tap into decades of toxicology, too, with the FDA watching closed-loop manufacturing to keep unwanted contaminants out.
Room for Better Practices
Cooks, chemists, and health professionals can do more to teach the public about what goes into their food. Many shoppers skip over label ingredients they don’t recognize, assuming danger. Building trust takes honesty. Training and plain-language food guides keep panic and confusion at bay—nobody wants a return to the days when rumors about “artificial flavors” topped news headlines without evidence.
Sourcing cleaner, well-documented products can feel like a headache, but traceability keeps risk low. Bigger grocery brands now publish sourcing standards for supplies, including for flavor compounds. Companies that welcome third-party checks give shoppers more peace of mind. Cooking with flavorings at home follows the same logic: proper handling and keeping to recommended doses matter.
Looking Forward
Isoamyl acetate has helped build memories—from childhood sweets to bakery treats. Its story shows that flavor science goes hand-in-hand with responsibility. As research continues, keeping honest about what’s in our food brings trust and informed choices to the table.
What does isoamyl acetate smell like?
Banana Candy, Bubble Gum, and Lab Benches
Isoamyl acetate carries a reputation built on the unmistakable scent of bananas. Not the earthy, spotty ones found in the fruit bowl, but the candy version you’ll recognize from childhood sweets and bubble gum. A classroom full of science students uncapping vials of this compound usually bursts out in laughter or surprise. That sweet, playful scent can remind anyone of banana-flavored taffy or those neon-yellow “banana runts.” No other naturally occurring banana aroma hits the senses so directly.
Isoamyl acetate pops up everywhere—somewhere between a lab technician’s bench and a bag of penny candy at the corner store. You’ll find it in fruit, especially banana, but apples and pears make small amounts too. Still, almost all the products clutching that telltale banana scent get their kick from the synthetic form. The candy makers favor it because banana essence extracted from fruit has little strength and costs more to produce.
Why This Odd Scent Shows Up Everywhere
European breweries, winemakers, perfume creators, and even pest control companies all use isoamyl acetate for good reason. Aroma shapes taste. Adding this compound to processed foods and beverages transforms bland into memorable. Commercial bakeries rely on it to jazz up breads, cakes, and even drink mixes. Just a drop or two can turn bland dough into something that hints at bananas or tropical fruit.
Science leans on isoamyl acetate as a marker for testing how well someone smells. In hospitals, doctors rely on it for olfactory assessments. Part of the reason: almost everyone instantly recognizes it—either from fruit, candy, or synthetic fragrances. Our sense of smell draws from personal memories, and that distinctive banana burst delivers a familiar jolt for most people.
Sneaky Uses and Potential Hazards
Beyond the candy shop, this compound slips into liquor and cigarette industries. Pearson’s guide for food scientists notes that some distilleries control their fermentation process to keep just enough isoamyl acetate in their spirits; too much and the drink tastes like bananas, too little and it blends in blandly. In smaller doses, it’s ideal for masking off-flavors, especially in processed foods.
Like many chemicals, getting a strong whiff of isoamyl acetate in a lab—or using it unwisely in industrial spaces—can be rough. Irritation isn’t uncommon if someone gets a high concentration in the air. Workers producing flavors or working in fragrance factories know the headaches or dizziness that can come with careless handling. Regulations cap exposure, and safety data sheets recommend good ventilation with any large-scale use.
Solutions: Balancing Enjoyment and Safety
Flavors spark memories and shape eating habits. Isoamyl acetate can improve foods, help in medical assessments, and even turn a dull drink into something worth savoring. The trick? Clear labeling, strict workplace ventilation, and public awareness. Companies owe transparency about synthetic additives so people can make their own choices. Health and safety training for those who handle concentrated amounts matters just as much.
Building on what’s already known, researchers keep examining potential health impacts. Continuous monitoring, real-time indoor air sensors, and better employee education lessen the risks. If companies stick to these basics and consumers stay informed about what brings that banana flavor to life, enjoying the quirky scent of isoamyl acetate stays sweet—and safe.
Where can I buy isoamyl acetate?
Understanding Isoamyl Acetate’s Role
Isoamyl acetate, often known as “banana oil” for its sweet aroma, has a place in both industrial labs and creative food experiments. It flavors candy and beverages, helps in scenting perfumes, and stands out as a reliable solvent in chemical research. Given its range of uses, people—whether home experimenters, students, or business owners—sometimes wonder where and how to source it safely.
Finding Isoamyl Acetate: More Than a Simple Purchase
Hunting for this compound isn't as simple as picking up sugar at the grocery store. Most household stores won’t have it on their shelves. Chemical supply companies, such as Sigma-Aldrich, Fisher Scientific, and VWR, list isoamyl acetate among their products. These big suppliers deliver quality control, safety information, and clear purity levels, which means you know exactly what you’re getting. For someone working in a lab or creating for the food or fragrance business, the need for a trustworthy source cannot be overemphasized.
Safety, Trust, and Regulations
No one wants to risk harmful impurities or mislabeled products. Regulatory frameworks, including the United States FDA for food grades or the European REACH regulations, keep the bar high for what goes into commercial or laboratory products. In my own research projects in college chemistry, the lab manager would dig deep into certificates of analysis before ordering even small bottles. These steps slowed us down, but they protected us from sloppy work and, more importantly, prevented exposure to unsafe substances.
Safety isn’t just about what comes in the bottle but also about how it’s packed, transported, and delivered. Reputable suppliers carry MSDS sheets, hazard documentation, and offer packaging suited to the chemical’s flammability. They require business documents or proof of research before shipping. Sometimes this feels like bureaucracy in the way, but a little friction protects buyers from risk—accidental or otherwise.
Can Consumers Buy Isoamyl Acetate?
For hobbyists, classroom teachers, or small businesses, some specialty supply shops online handle modest orders. Lab Alley and Science Company in the US, as well as some EU-based chemical vendors, bridge the gap for non-commercial buyers. Still, each customer gets a short list of questions: Why do you want the chemical? Do you have experience handling it? A few times when I ordered reagents for home science, I had to upload my ID and explain the intended use. That effort prevented market misuse and kept me, and anyone else in my home, safer in the process.
Responsible Sourcing and Practical Alternatives
For small-scale needs, food-grade artificial banana flavoring holds some of the same scent and taste molecules. It works in culinary projects, but doesn’t suit chemistry research or industrial cleaning. The lesson here stands clear: Understand your end goal before opening your wallet. If an experiment or recipe really requires the real thing, trust established chemical suppliers. If all you need is the scent of bananas, flavor supply shops offer an easier, safer path.
Real Solutions and Smarter Choices
Sticking with trusted sources always outweighs quick online finds. There’s a reason schools, universities, and businesses stick to vetted suppliers—they minimize risk and guarantee product integrity. Anyone unsure about the process can reach out to professional chemists or industry forums for advice on finding a reputable source. Investing a little time in research always beats cleaning up after a bad chemical buy.
Is isoamyl acetate natural or synthetic?
What Exactly Is Isoamyl Acetate?
Crack open a banana-flavored candy, or take a whiff near a banana-flavored drink, and odds are you’re smelling isoamyl acetate. This compound creates that distinct, sweet aroma often linked with bananas. If you peel a ripened banana, nature is giving you a dose of the same compound. It’s also present in pears and apples, but the banana association sticks, almost like muscle memory for the nose.
Natural or Synthetic: Where Does It Come From?
Straight out of the gate, isoamyl acetate is both natural and synthetic. Some people think a chemical either comes from a lab or from nature; reality has other plans. Yeasts produce this compound during fermentation, so you’ll find traces in beer and even bread. Many fruit-loving microbes in the wild churn it out, too.
Now, climb into the food manufacturing world. Companies can extract isoamyl acetate from plants or fruits, but the supply never matches demand. Most of what flavors candies, ice creams, or sodas gets made in chemical plants. A chemist cooks up isoamyl alcohol with acetic acid, and the process produces isoamyl acetate, purity checked and all. It lands in ingredient lists as “artificial flavor,” even though its makeup mirrors what’s in a real banana.
Why Should Anyone Care Where It Comes From?
Plenty of folks care about what enters their food, makeup, or cleaning products. Some want everything “all natural,” as if nature alone equals health and safety. Others dig into the source because of concerns over allergies, dietary restrictions, or religious practices. From where I stand, reading the origins matters less than understanding the process and impact.
Natural extraction pulls resources out of the ground, needs labor, and creates more waste. Synthetic production may use fossil fuels but skips the harvest, quickens output, and proves more consistent in quality. Lab-made isoamyl acetate doesn’t carry pesticide residues, nor does it cost forests or farmland. Factory control also shakes out microbes or natural impurities that can ride along in botanically sourced flavors.
Safety and Health: Fact Versus Hype
Isoamyl acetate, natural or synthetic, has passed regulatory checks in the U.S., Europe, and plenty of countries in between. The FDA considers it Generally Recognized As Safe (GRAS) for things you eat or drink. Runaway doses, like drinking it straight, would taste vile and sting your nose, but no dessert or soda gets close to that threshold. Still, a few people react to certain flavorings, which is worth keeping in mind. If allergies run in your family, it pays to check labels and stay in the know.
Looking at Better Solutions
Manufacturers constantly seek smarter ways to balance cost, sustainability, and transparency. Some push for green chemistry, using plant waste to make isoamyl acetate with fewer harmful solvents. Others experiment with yeast engineered to produce it in controlled vats, turning biofactories into the new norm. That way, you skip the fossil fuels, avoid stripping fields, and cut down on pollutants. The journey from molecule to market should invite more scrutiny, not less. Clear labeling and better science education can build trust—much needed in an era where “natural” and “synthetic” trigger strong reactions, sometimes without reason.
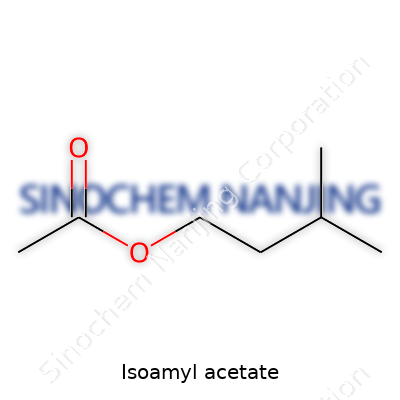
| Names | |
| Preferred IUPAC name | 3-methylbutyl ethanoate |
| Other names |
Isopentyl acetate 3-Methylbutyl acetate Banana oil Pear oil |
| Pronunciation | /ˌaɪsoʊˌæmɪl əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 123-92-2 |
| 3D model (JSmol) | `Isoamyl Acetate JSmol 3D model string`: ``` Isoamyl Acetate CC(C)CCOC(=O)C ``` |
| Beilstein Reference | 1209244 |
| ChEBI | CHEBI:15847 |
| ChEMBL | CHEMBL33071 |
| ChemSpider | 8257 |
| DrugBank | DB11246 |
| ECHA InfoCard | 03b234d1-7666-43b3-967f-5b9e74c73c0b |
| EC Number | 3.1.1.87 |
| Gmelin Reference | 6635 |
| KEGG | C06154 |
| MeSH | D000687 |
| PubChem CID | 31276 |
| RTECS number | AJ3675000 |
| UNII | N8P839M8BY |
| UN number | UN1104 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.18 g/mol |
| Appearance | Colorless liquid with a strong, fruity odor |
| Odor | fruity, banana-like |
| Density | 0.876 g/cm³ |
| Solubility in water | 1.4 g/L (20 °C) |
| log P | 1.98 |
| Vapor pressure | 4 mmHg (20°C) |
| Acidity (pKa) | 7.6 |
| Basicity (pKb) | pKb: 9.97 |
| Magnetic susceptibility (χ) | -7.83×10⁻⁶ |
| Refractive index (nD) | 1.400-1.404 |
| Viscosity | 1.31 mPa·s (20 °C) |
| Dipole moment | 1.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -464.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3527 kJ/mol |
| Pharmacology | |
| ATC code | V03AB05 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P304+P340, P312, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 18 °C (closed cup) |
| Autoignition temperature | 410 °C (770 °F) |
| Explosive limits | 1.1% - 7.5% |
| Lethal dose or concentration | LD50 oral rat 16,600 mg/kg |
| LD50 (median dose) | 6,600 mg/kg (oral, rat) |
| NIOSH | NA0162 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 35 mg/m³ |
| IDLH (Immediate danger) | 1,000 ppm |
| Related compounds | |
| Related compounds |
Ethyl acetate Methyl acetate Propyl acetate Butyl acetate Isobutyl acetate Amyl acetate |

